Abstract
Background:
The Surgical Infection Society (SIS) published evidence-based guidelines for the management of intra-abdominal infection (IAI) in 1992, 2002, 2010, and 2017. Here, we present the most recent guideline update based on a systematic review of current literature.
Methods:
The writing group, including current and former members of the SIS Therapeutics and Guidelines Committee and other individuals with content or guideline expertise within the SIS, working with a professional librarian, performed a systematic review using PubMed/Medline, the Cochrane Library, Embase, and Web of Science from 2016 until February 2024. Keyword descriptors combined “surgical site infections” or “intra-abdominal infections” in adults limited to randomized controlled trials, systematic reviews, and meta-analyses. Additional relevant publications not in the initial search but identified during literature review were included. The Grading of Recommendations, Assessment, Development, and Evaluations (GRADE) system was utilized to evaluate the evidence. The strength of each recommendation was rated strong (1) or weak (2). The quality of the evidence was rated high (A), moderate (B), or weak (C). The guideline contains new recommendations and updates to recommendations from previous IAI guideline versions. Final recommendations were developed by an iterative process. All writing group members voted to accept or reject each recommendation.
Results:
This updated evidence-based guideline contains recommendations from the SIS for the treatment of adult patients with IAI. Evidence-based recommendations were developed for antimicrobial agent selection, timing, route of administration, duration, and de-escalation; timing of source control; treatment of specific pathogens; treatment of specific intra-abdominal disease processes; and implementation of hospital-based antimicrobial agent stewardship programs.
Summary:
This document contains the most up-to-date recommendations from the SIS on the prevention and management of IAI in adult patients.
Intra-abdominal infection (IAI) is a ubiquitous, often life-threatening disease affecting millions of patients annually.1–3 Due to its diverse nature and severity, along with modern systems-based approaches to patient care, responsibility for treating IAI may rest with surgeons, infectious diseases physicians, critical care physicians, interventional radiologists, or other healthcare providers. Founded in 1981, the Surgical Infection Society (SIS) follows its mission “to educate healthcare providers and the public about infection in surgical patients, and promote research in the understanding, prevention, and management of surgical infections.” To this end, the SIS produced clinical guidelines for the management of IAI in 1992, 4 with subsequent updates published in 2002,5,6 in 2010 in collaboration with the Infectious Diseases Society of America,7,8 and most recently in 2017. 9 Considering the complex, dynamic landscape of evolving patient demographics and comorbid diseases, increasing prevalence and virulence of resistant microorganisms, and novel antimicrobial agents joining the therapeutic armamentarium combating abdominal pathogens, it remains essential to review periodically and update the IAI guidelines to ensure both clinical relevance and applicability.
In 2022, current and past members of the SIS Therapeutics and Guidelines Committee, along with additional SIS members with subject matter or guideline writing expertise, commenced work on updated IAI guidelines. From the onset, several important distinctions were made between these guidelines and previous iterations. First, this version focuses solely on adults. A separate, subsequent guideline will address pediatric patients. Second, evidence is limited to randomized controlled trials (RCTs,) systematic reviews, and meta-analyses, with the intent of improving consistency and strengthening recommendations with higher quality evidence. Third, this update is not based on preformed clinical questions or topics. Instead, the writing group performed a broad-based literature search of PubMed/Medline, the Cochrane Library, Embase, and Web of Science. Fourth, this update is intended to supplement, not supplant previous guideline iterations. This version, and hopefully future iterations, should be construed as continuations or refinements of previous work. Indeed, many current recommendations are predicated on both new and previously existing literature documented by prior guidelines. Moreover, we include previous recommendations if the literature search failed to provide additional support either for or against them. Lastly, we used Covidence software for literature management and reviews (Covidence Ltd., Melbourne, Australia.) 10
Consistent with previous guidelines, the Grading of Recommendations, Assessment, Development, and Evaluations (GRADE) system serves as the basis for the final recommendations.11–13 The strength of each recommendation is rated strong (1) or weak (2),14,15 and evidence quality is rated high (A), moderate (B), or weak (C). 12 Final recommendations were developed through an iterative process to achieve consensus. All writing group authors voted to accept or reject each recommendation. The primary goal for these recommendations is to facilitate evidence-based treatment decisions for adult patients with IAI. As reflected by weak GRADE recommendations, especially those stemming from lower quality evidence, the risk:benefit ratios for several therapeutic options remain unresolved and would benefit from additional investigation. Conversely, strong recommendations based on high-quality evidence may not suffice when providing care for every clinical circumstance. Consistent with recommendations from other SIS guidelines, none is intended to supersede completely the sound clinical expertise and judgment of the primary patient care team.
There are several important similarities between the scope of this update document and previous versions. First, the term “IAI” refers to complicated IAI (affecting normally sterile peritoneum, mesentery, another organ, retroperitoneum, or abdominal wall) unless otherwise indicated.7,8 Recommendations pertaining to the prevention or management of uncomplicated IAI (confined to a hollow viscus) are classified by a specific disease process, such as acute appendicitis.7,8 Second, certain abdominal disease processes are excluded from this guideline, including primary (spontaneous) bacterial peritonitis, peritoneal dialysis catheter- or other device-related peritonitis, peritonitis in the setting of a solid organ transplant, and gynecologic infections or other infections involving primarily the genitourinary tract. Third, methods of initial diagnosis for IAI are excluded, including preferences for specific laboratory testing or radiologic imaging. Fourth, whereas most recommendations pertain to pharmacologic therapies, there are new recommendations on the timing of performance of source control procedures, and the impact of a source control procedure on subsequent pharmacologic therapy.
Lastly, and perhaps most importantly, recommendations continue to emphasize risk stratification for patients related to illness and infection severity, IAI microbiology, and the likelihood of harboring multi-drug-resistant (MDR), extensively drug-resistant, or pandrug-resistant organisms.
16
Patients treated in hospitals and other healthcare settings, or previously with antimicrobial agent therapy, remain at increased risk for resistant, healthcare-associated intra-abdominal infections (HA-IAI).17,18 Simultaneously, these patients are at risk for a greater variety of invasive pathogens, including staphylococci, enterococci, and Candida spp.17,19 Notwithstanding, antimicrobial agent resistance (AMR) is increasing within community-acquired IAI (CA-IAI).
20
Therefore, recommendations in these guidelines referring specifically to “higher risk” individuals include those likely to harbor resistant or atypical organisms. In addition, patients are classified as higher risk if they meet the criteria for sepsis or septic shock, have Acute Physiology and Chronic Health Evaluation (APACHE)-II scores
Methods
A writing group was formed in 2022 comprising current and former members of the SIS Therapeutics and Guidelines Committee, other SIS members with content or guideline-writing expertise, and a professional librarian with experience in systematic review development. An initial systematic review was performed using PubMed/Medline, the Cochrane Library, Embase, and Web of Science. Search dates were January 2016 until September 2022. Keyword descriptors combined “surgical site infections (SSIs)” or “intra-abdominal infections” in adults limited to RCTs, systematic reviews, and meta-analyses. Literature reviews, retrospective studies, case reports/series, conference proceedings, and conference abstracts were excluded. Only English language articles and human studies were included (see Supplementary for the search tactic). Before article writing, a follow-up search was performed in February 2024 to include the most recent literature. Additional relevant publications not in the initial search but identified during literature review were also included. The searches identified 5,070 studies representing 3,701 unique studies after de-duplication. Study abstracts were entered into a customized Covidence database for screening. Fifteen reviewers used a two-step screening methodology. Conflicts were resolved by a third author. A total of 3,382 abstracts were considered irrelevant after title and abstract screening. A total of 295 studies then underwent full-text review by two authors. Of those, 220 were deemed irrelevant. A total of 75 studies were included, extracted for data, and reviewed for quality (Fig. 1). Our review followed the Preferred Reporting Items for Systematic reviews and Meta-Analyses (PRISMA) guideline. 20
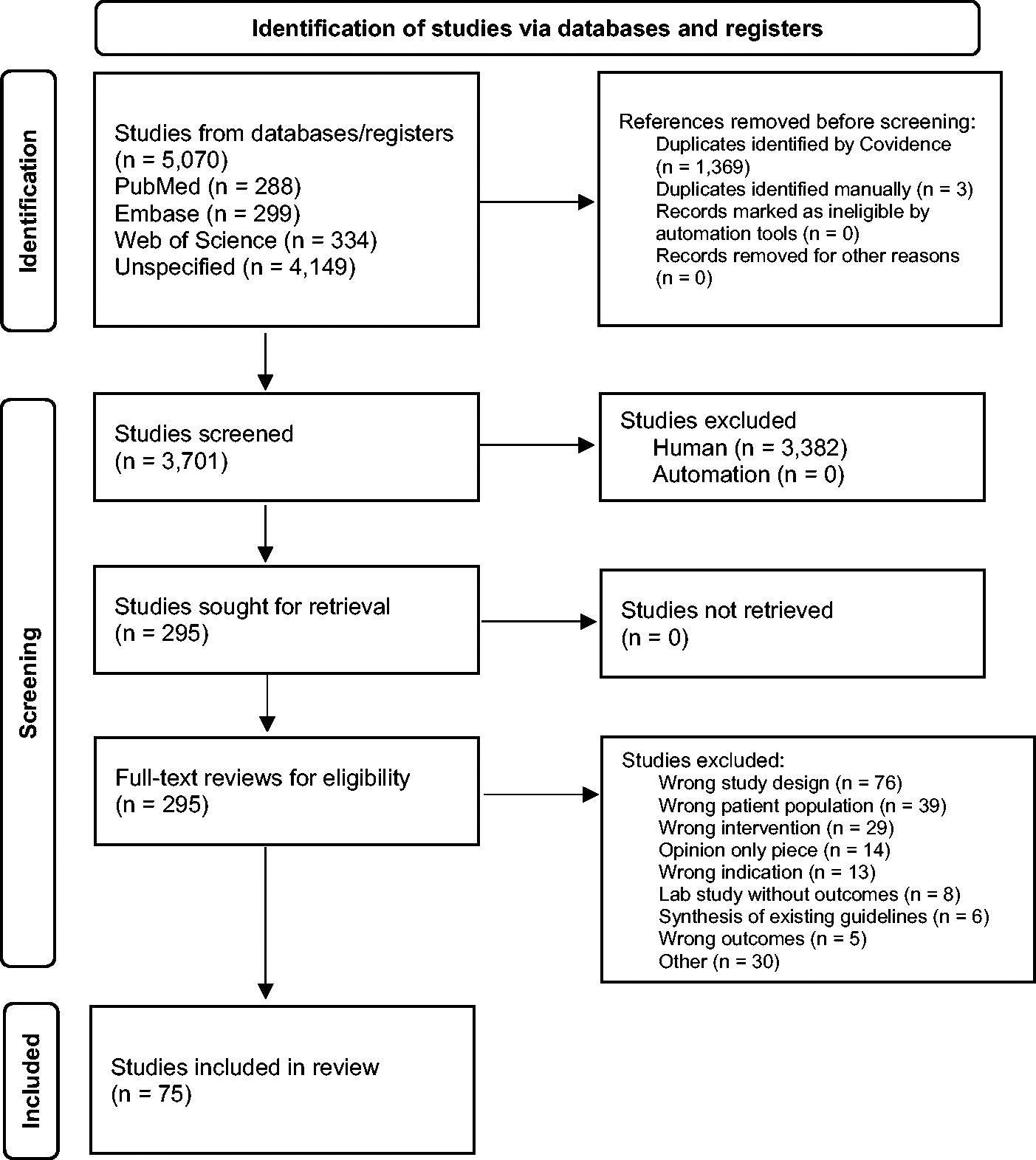
PRISMA flow diagram. 21
Evaluation of the evidence was performed using the GRADE system.11,12 The strength of each recommendation was graded strong (1) or weak (2) (Table 1).13,14 Strong recommendations begin with the phrase “We recommend…,” whereas weak recommendations begin with “We suggest… .” 22 Quality of the evidence was graded high (A), moderate (B), or weak (C) (Table 2). 12 For new recommendations not contained in previous guidelines, GRADE was applied only to evidence in the current literature search. For updates to recommendations from previous IAI guideline versions, GRADE was applied to evidence from the current literature search as well as prior evidence. Final guideline recommendations were developed by an iterative process to achieve consensus. All writing group members voted to accept or reject each recommendation.
Strength of Recommendations
Quality of Evidence
Results
Antimicrobial agent therapy
Aminoglycosides
We recommend against aminoglycoside-based combination regimens for empiric therapy of higher risk patients (Grade 1-A).
This updated recommendation is based on one systematic review, including 69 randomized and quasi-randomized trials, totaling 7,863 patients with sepsis treated with a β-lactam (penicillins, cephalosporins, carbapenems) combined with a β-lactamase inhibitor (BLIC) monotherapy versus β-lactam-aminoglycoside combination therapy. 23 Sepsis-associated infections included urinary tract infections (UTIs,) pneumonia, IAI, skin and soft tissue infection, and infection of unknown origin. Subgroup analyses explored UTI and non-UTIs. The primary outcome was all-cause mortality. Secondary endpoints included treatment failure, superinfections, and adverse events (AEs). Twenty-two trials compared the same β-lactam in both study arms, whereas the remainder compared different regimens, including a broader spectrum drug in the monotherapy arm. In the former, the review found no difference in all-cause mortality between study arms. Comparing β-lactams revealed a trend for lower all-cause mortality with β-lactam monotherapy, and a substantial advantage in terms of clinical failure. Subgroup analyses did not reveal important differences. Compared with combination therapy, β-lactam monotherapy resulted in significantly less nephrotoxicity. All results were classified as low- to very low-quality evidence.
Beta-lactamase inhibitor combinations
Cefoperazone-sulbactam
We recommend cefoperazone-sulbactam for empiric therapy of lower risk patients (Grade 1-B).
This updated recommendation is based on one systematic review and meta-analysis of 12 studies, totaling 1,674 patients with IAIs, post-operative IAIs, and biliary tract infections treated with cefoperazone/sulbactam versus multiple different comparators (clindamycin plus gentamicin; ceftriaxone, ceftazidime, or amikacin plus metronidazole; ciprofloxacin or levofloxacin plus metronidazole; or moxifloxacin). 24 Juxtaposed with comparators, cefoperazone-sulbactam showed a significantly higher clinical efficacy rate (clinical cure or improvement), lower clinical failure rate (need for additional antibiotic agents, abscess drainage, death), higher clinical cure rate, and a higher microbiologic eradication rate (no further culturable material or lack of growth in cultures of drainage fluid.) There was no difference in mortality rate between cefoperazone-sulbactam and comparators.
Imipenem-cilastatin-relebactam
We recommend imipenem-cilastatin-relebactam for empiric therapy (Grade 1-A).
We suggest reserving imipenem-cilastatin-relebactam for higher risk patients due to its broader spectrum antimicrobial agent activity (Grade 2-C).
These new recommendations are based on two double-blind, multi-center RCTs and one systematic review and meta-analysis totaling 398 patients with IAI treated with imipenem-cilastatin-relebactam versus comparator agents.25–27 The first RCT compared two dose regimens of imipenem-cilastatin-relebactam, with imipenem-cilastatin in 351 adult patients with IAI. 25 The primary efficacy endpoint was the proportion of microbiologically evaluable (ME) subjects with a favorable clinical response at discontinuation of intravenous (IV) therapy. Secondary endpoints included clinical response at early and late follow-up, microbiologic response, and global response. The most common diagnoses were complicated appendicitis and cholecystitis. Clinical response rates for both dose regimens were equivalent to imipenem-cilastatin for all time points. There was no difference in safety profiles for either dose regimen compared with imipenem-cilastatin. Of note, there were few non-susceptible organisms identified, which may be already limiting the utility of relebactam.
The second RCT compared imipenem-cilastatin-relebactam with imipenem-cilastatin plus colistin in 47 hospitalized patients with healthcare-associated or ventilator-associated pneumonia, IAI, or complicated UTI caused by imipenem-cilastatin-non-susceptible pathogens. 27 The primary endpoint was a favorable clinical response in the modified microbiologic intention-to-treat (mMITT) population. Secondary endpoints included clinical response, all-cause mortality, and treatment-emergent nephrotoxicity. The clinical response rate was similar between groups. Compared with imipenem-cilastatin plus colistin, there were significantly fewer serious AEs and drug-related AEs and less nephrotoxicity in the imipenem-cilastatin-relebactam group. Of note, there were only four patients with complicated IAI, two in each treatment group, which limits the findings.
The systematic review and meta-analysis reference includes the above studies plus two additional studies of patients with UTIs or pneumonia. 27 The meta-analysis found that imipenem-cilastatin-relebactam has a similar efficacy and safety profile to comparators when treating IAI or other severe bacterial infections.
Ceftolozane-tazobactam
We recommend ceftolozane-tazobactam plus metronidazole for empiric therapy (Grade 1-A).
We suggest reserving ceftolozane-tazobactam for higher risk patients, including those with resistant Pseudomonas aeruginosa infections (Grade 2-C).
We suggest ceftolozane-tazobactam for empiric therapy of patients at risk for infection with extended-spectrum β-lactamase (ESBL)-producing Enterobacterales (Grade 2-B).
These updated recommendations are based on three systematic reviews and meta-analyses, one meta-analysis, one phase 3, double-blind, multi-center RCT, and one subgroup analysis of a phase 3 double-blind RCT, totaling more than 1,300 patients with IAI treated with ceftolozane-tazobactam plus metronidazole versus meropenem, ceftolozane-tazobactam monotherapy versus combination therapy, or ceftolozane-tazobactam versus colistin with or without an aminoglycoside.28–31 The RCT and systematic reviews and meta-analyses report ceftolozane-tazobactam plus metronidazole as non-inferior to meropenem. There was no difference in treatment-related AEs, serious AEs, tolerability, or mortality between groups. The subgroup analysis reported equivalent efficacy between ceftolozane-tazobactam plus metronidazole and meropenem when P. aeruginosa infection was suspected or confirmed. The systematic review and meta-analysis analyzing ceftolozane-tazobactam for use in patients with severe ESBL-producing Enterobacterales infections is at risk for bias due to the inclusion of only a single retrospective study with small sample sizes.
Ceftazidime-avibactam
We recommend ceftazidime-avibactam plus metronidazole for empiric therapy (Grade 1-A).
We suggest reserving ceftazidime-avibactam for higher risk patients because of its broader spectrum antimicrobial agent activity (Grade 2-C).
We recommend ceftazidime-avibactam plus metronidazole for empiric therapy of patients at risk for infection with ESBL-producing Enterobacterales (Grade 1-B).
These updated recommendations are based on one phase 3, double-blind, multi-center RCT, two systematic reviews and meta-analyses, two meta-analyses, one systematic review and network meta-analysis, one integrated analysis, and one pooled analysis of five phase 3 multi-center RCTs, totaling more than 3,700 patients with IAI treated with ceftazidime-avibactam plus metronidazole versus meropenem or the best available therapy, including imipenem-cilastatin, doripenem, colistin, tigecycline, or combination treatments.32–39 All studies reported ceftazidime-avibactam plus metronidazole as non-inferior to meropenem or the best available therapy. There were no differences in safety between any groups. One systematic review and meta-analysis and the pooled analysis of five phase 3 trials reported equivalent efficacy between ceftazidime-avibactam plus metronidazole and meropenem in IAIs caused by ESBL-producing Enterobacterales. The pooled analysis reported equivalent efficacy between ceftazidime-avibactam plus metronidazole versus meropenem for AmpC-producing gram-negative infections, but a systematic review and meta-analysis could not provide conclusive recommendations for this type of infection.
Glycylcyclines
Eravacycline
We recommend eravacycline for empiric therapy (Grade 1-A).
We suggest reserving eravacycline for higher risk patients due to its broader spectrum antimicrobial agent activity (Grade 2-C).
These new recommendations are based on two double-blind RCTs, four meta-analyses, and two systematic reviews and meta-analyses, totaling 1,080 patients with IAI treated with eravacycline versus comparator agents, including ertapenem or meropenem.40–47 The first RCT is a phase 3 non-inferiority trial that evaluated the efficacy and safety of eravacycline versus ertapenem in 541 adult hospitalized patients with IAI requiring surgical or percutaneous intervention. 46 The primary outcome was a clinical response at the test-of-cure (TOC) visit in the microbiologic intention-to-treat (microITT) population, and secondary endpoints included clinical responses in the modified intent-to-treat (mITT) and clinically evaluable (CE) populations. The study found no differences in clinical cure between any of the treatment groups. Most patients in the microITT population underwent open surgical procedures. The most common reasons for treatment failure were unplanned surgical or drainage procedure and initiation of rescue antimicrobial agent therapy. Of note, there were more treatment-emergent AEs in the eravacycline group, but the number of severe or life-threatening AEs was similar between groups.
The second non-inferiority RCT evaluated the efficacy and safety of eravacycline versus meropenem in 400 adult hospitalized patients with IAI requiring surgical or percutaneous intervention. 47 The primary objective was to demonstrate statistical non-inferiority in clinical cure rates at the TOC visit in the microITT population. Secondary endpoints included clinical and microbiologic responses for the microITT, mITT, CE, and ME populations at end-of-treatment, TOC, and follow-up visits. The study found no difference in clinical cure between any of the treatment groups. As above, the most common reasons for treatment failure were unplanned surgical or drainage procedure and initiation of rescue antimicrobial agent therapy.
The systematic reviews and meta-analyses or meta-analyses of eravacycline include the above RCTs plus a third phase 2, double-blind RCT published in 2014 comparing two dose regimens (1.5 mg/kg q24h, 1.0 mg/kg q12h) of eravacycline with ertapenem in 139 adult hospitalized patients with IAI requiring surgical or percutaneous intervention. 48 The primary efficacy endpoint was clinical response in the ME population at the TOC visit. The study found no difference in clinical success between any of the treatment groups. There were no differences in treatment-emergent AEs between groups. Overall, the systemic reviews and meta-analyses found similar clinical efficacy of eravacycline versus comparators.
Tigecycline
We recommend against tigecycline for empiric therapy of low- and high-risk patients (Grade 1-B).
We suggest reserving tigecycline for patients with resistant infections or as part of a combination regimen when no therapeutic alternative exists (Grade 2-B).
These updated recommendations are based on an open-label, single-center RCT, one systematic review and meta-analysis of four retrospective studies, and one systematic review and network meta-analysis, totaling 14,479 patients with IAI treated with tigecycline versus meropenem, standard- versus high-dose tigecycline with or without concomitant antimicrobial agent administration, or tigecycline versus multiple other regimens, including cefepime plus metronidazole, ceftolozane-tazobactam, carbapenems, fluoroquinolones, and BLIC monotherapies or combination therapy with metronidazole.49–51 Whereas the RCT and systematic review and meta-analysis report tigecycline as non-inferior to meropenem or no difference between high- and standard-dosing, respectively, these studies have small sample sizes or are observational with high risk of bias. The systematic review and network meta-analysis reports significantly higher risk of all-cause mortality with tigecycline versus cefepime plus metronidazole, and significantly higher risk of serious AEs with tigecycline versus imipenem-cilastatin.
Carbapenems
Biapenem
We suggest biapenem for empiric therapy (Grade 2-B).
We suggest reserving biapenem for higher risk patients because of its broader spectrum antimicrobial agent activity (Grade 2-C).
These new recommendations are based on one meta-analysis of eight RCTs totaling 1,685 patients, but only one multi-center study of 118 patients with IAI, treated with biapenem versus imipenem-cilastatin.52,53 There was no difference in clinical success (complete resolution or substantial improvement in symptoms) or mean microbiologic success (eradication or presumed eradication of pathogens between biapenem and imipenem-cilastatin.) There was no difference in drug-related AEs or numbers of withdrawn patients due to AEs between biapenem and imipenem-cilastatin. Compared with imipenem-cilastatin, biapenem had significantly higher numbers of laboratory AEs possibly or probably related to treatment.
Doripenem
We recommend doripenem for empiric therapy (Grade 1-A).
We suggest reserving doripenem for higher risk patients because of its broader spectrum antimicrobial agent activity (Grade 2-C).
These updated recommendations are based on one systematic review and meta-analysis of eight RCTs totaling 3,499 patients, but only one phase 3, double-blind, multi-center non-inferiority RCT of 476 patients with IAIs, treated with doripenem versus meropenem.54,55 The primary outcome was clinical success rate, and secondary outcomes were microbiologic eradication rate and risk of AEs. Compared with meropenem, doripenem had similar clinical success and microbiologic eradication rates and treatment-emergent AEs.
Fluoroquinolones
We recommend ciprofloxacin plus metronidazole for empiric therapy of lower risk patients (Grade 1-A).
We suggest moxifloxacin for empiric therapy of lower risk patients (Grade 2-A).
These updated recommendations are based on one meta-analysis, including seven multi-center RCTs (six double-blind, one open-label), totaling 4,125 patients treated with fluoroquinolone-based (ciprofloxacin, moxifloxacin) versus β-lactam-based (piperacillin/tazobactam, ceftriaxone plus metronidazole, ertapenem, imipenem-cilastatin) antibiotic agent regimens for IAI. 56 The primary outcomes were treatment success at the TOC visit and all-cause mortality in the CE populations. Secondary endpoints were treatment success in the ITT and ME populations and incidence of treatment-related AEs, severe AEs, and withdrawal due to AEs. There was no difference in treatment success in the CE population between fluoroquinolone- and β-lactam-based regimens. There were no differences between groups in treatment success in the ITT or ME population, nor were there differences in all-cause mortality, treatment-related AEs, severe AEs, or withdrawal due to AEs. By subgroup analysis, moxifloxacin was significantly less effective versus β-lactam-based regimens in the ITT, CE, and ME populations. Moxifloxacin was less effective than β-lactam-based regimens in patients with complicated appendicitis.
Glycopeptides
Vancomycin
We recommend against vancomycin for empiric therapy of low- and high-risk patients (Grade 1-B).
This new recommendation is based on one post hoc analysis of an open-label multi-center RCT, totaling 518 patients with IAI treated with short- versus long-course antibiotic agents after adequate source control.57,58 This analysis selected patients receiving piperacillin-tazobactam with or without a carbapenem, and then allotted them into two groups based on vancomycin administration or not during the study period. The primary endpoint was a composite metric of SSI, recurrent IAI, and death assessed at 30 days. There were no major differences in baseline demographics, co-morbid diseases, or source control procedures between groups. Compared with subjects not receiving vancomycin (n = 234), multi-variable analysis of 110 patients who received vancomycin showed no difference in composite outcome, despite significantly higher APACHE-II scores, prolonged length of stay, and longer antibiotic agent courses. Despite these differences, vancomycin therapy did not independently predict the composite outcome. Of note, significantly more subjects in the vancomycin group received piperacillin-tazobactam, whereas significantly more patients without vancomycin administration received ertapenem. The vancomycin group had significantly higher rates of HA infections.
Metronidazole
We recommend metronidazole as the preferred anti-anaerobic agent in combination regimens for empiric therapy of low- and high-risk patients (Grade 1-A).
This updated recommendation is based on one systematic review and meta-analysis, including eight studies (five double-blind RCTs, three open-label RCTs,), totaling more than 1,100 patients treated with metronidazole combination therapies (cefepime, cefotaxime, ceftazidime/avibactam, ceftolozane/tazobactam, cefuroxime, or ciprofloxacin) versus a carbapenem (imipenem-cilastatin, meropenem) for IAI. 59 The primary outcomes were clinical success and microbiologic eradication. Secondary endpoints were all-cause mortality and drug-related AEs. For clinical success, there was no difference between metronidazole combination therapy versus a carbapenem. There was no difference in microbiologic eradication, drug-related AEs, or all-cause mortality between groups. Of note, after removal of three studies with high risk of bias across at least two assessment domains, metronidazole combination therapy had significantly increased clinical success compared with carbapenem alone.
Polymyxins
Polymyxin B
We suggest polymyxin B for empiric therapy of higher risk patients with MDR infection, including P. aeruginosa, Acinetobacter baumannii complex, Klebsiella pneumoniae, Escherichia coli, and Enterobacter spp. (Grade 2-B).
This updated recommendation is based on one review and meta-analysis of 27 studies (five prospective, 28 retrospective, one cross-sectional, two retrospective case series, seven case reports) totaling more than 2,900 patients, of which 18 studies specifically identified patients with IAIs treated with IV polymyxin B either as mono- or combination therapy for MDR gram-negative bacterial infections. 60 The outcomes were effectiveness, safety, overall mortality, and nephrotoxicity. Compared with literature reports of patients treated with IV colistin, patients treated with IV polymyxin B have similar mortality rates. Nephrotoxicity of polymyxin B may be less severe compared with colistin. 61
Anti-fungal therapy
We suggest against empiric anti-fungal therapy in lower risk patients (Grade 2-B).
This updated recommendation is based on one systematic review and meta-analysis, including six RCTs (four single-center, two multi-center) in the review and four in the meta-analysis, totaling 1,067 patients with IAI treated with untargeted anti-fungal therapy (azoles, echinocandins, polyenes, allylamines, or nucleoside analogues) in any dose, timing, or duration versus placebo or no treatment. 62 The primary outcome was all-cause short-term mortality (≤ 90 d, including ICU and in-hospital). Secondary endpoints were long-term mortality, AEs, duration of mechanical ventilation, days free of mechanical ventilation, need for kidney replacement therapy, days free of kidney replacement therapy, duration of vasopressor/inotrope support, days free of vasopressor/inotrope support, emergence of AMR at longest follow-up, emergence of fungi not susceptible to the anti-fungal agent, ICU length of stay, hospital length of stay, and quality of life at longest follow-up. The most-administered anti-fungal agent was fluconazole, followed by micafungin and nystatin. There was no difference in short-term mortality between untargeted anti-fungal therapy versus comparators. There were no differences in secondary endpoints between untargeted anti-fungal therapy versus comparators. Of note, all, but one trial, were assessed as having high risk of bias secondary to unclear allocation concealment or financial bias.
Anidulafungin
We recommend anidulafungin for empiric therapy of higher risk patients with infections from Candida spp. (Grade 1-B).
This updated recommendation is based on one pooled analysis of five prospective studies (one comparative, four open-label), totaling 79 adult surgical patients with microbiologically confirmed Candida IAI, treated with IV anidulafungin, with possible switching to an oral azole after 5–10 days of IV therapy. 63 The primary outcome was the global response rate (GRR) at the conclusion of IV treatment, and secondary endpoints were GRR at the end of therapy, all-cause mortality at days 14 and 28, and safety. The most common pathogens were C. albicans and C. glabrata. The GRR of anidulafungin was similar to a previous trial comparing the drug with fluconazole for invasive candidiasis. Anidulafungin was well-tolerated with only mild-to-moderate AEs.
Anti-enterococcal therapy
We recommend against empiric therapy targeting Enterococcus spp. in lower risk patients with CA-IAI (Grade 1-B).
We suggest empiric therapy targeting Enterococcus spp. in higher risk patients (Grade 2-B).
These updated recommendations are based on one post hoc analysis of an open-label, multi-center RCT and one systematic review and meta-analysis, including 23 RCTs and 13 observational studies.58,64,65 The post hoc analysis included 50 patients (among 518) with isolation of Enterococcus spp. treated with short- versus long-term antibiotic agent courses after adequate surgical source control. Analyses were conducted to determine whether isolation of Enterococcus is an independent predictor of SSI, recurrent IAI, or death. The systematic review and meta-analysis evaluated whether empiric enterococcal antibiotic agent coverage for patients with IAI improves treatment success or reduces AEs or mortality. The post hoc analysis demonstrated similar baseline demographics, antibiotic agent utilization, infection characteristics (CA- vs. HA), and origin of infection between groups. Clinical outcomes were not statistically different between Enterococcus and no Enterococcus groups, and isolation of Enterococcus did not independently predict the incidence of the composite outcome. The systematic review and meta-analysis found that anti-enterococcal regimes provide no improvement in treatment success compared with control regimens. There were similar mortality rates and AEs in both groups. Of note, most patients were low-risk with CA-IAI, and both HA-IAI and ICU admission were risk factors for enterococcal infection, which suggests that high-risk CA-IAI and HA-IAI may benefit from empiric anti-enterococcal coverage.
Specific Disease States
Hepatopancreatobiliary malignant disease
We recommend targeted, perioperative antibiotic agent prophylaxis based on a positive preoperative bile culture in patients undergoing surgical procedures for hepatopancreatobiliary malignant disease (Grade 1-A).
This new recommendation is based on an open-label, single-center, parallel-group RCT and one systematic review and meta-analysis, including seven studies (one RCT, four prospective, and two retrospective comparative), totaling 849 patients undergoing a hepatopancreatobiliary surgical procedure or specifically pancreaticoduodenectomy, respectively.66,67 Regimens included targeted (organisms prevalent in bile or present by culture) prophylactic antibiotic agents (third- or fourth-generation cephalosporin, piperacillin or piperacillin-tazobactam, piperacillin-tazobactam plus vancomycin) versus standard prophylaxis (first- or second-generation cephalosporin). The primary outcomes were rates of SSI and their microbiology profiles. Baseline demographics were similar between groups. Compared with standard prophylaxis, targeted prophylaxis significantly reduced the rate of SSIs. Targeted antibiotic agent prophylaxis significantly reduced both incisional and organ-space SSIs. Of note, whereas Enterococcus spp. were the bacteria isolated most frequently, prophylaxis regimens lacking anti-enterococcal activity demonstrated efficacy.
This new recommendation is also based on one pragmatic, open-label, multi-center, phase 3 RCT of 778 patients undergoing open pancreaticoduodenectomy who received piperacillin-tazobactam versus cefoxitin for perioperative antimicrobial agent prophylaxis (up to 24 h post-operatively.)
68
Subjects were stratified by the presence of a preoperative biliary stent. The primary outcome was the development of SSI within 30 days. Secondary endpoints included 30-day mortality, development of “clinically relevant” post-operative pancreatic fistula, and sepsis. Baseline demographics were similar between groups. Compared with cefoxitin prophylaxis, piperacillin-tazobactam significantly decreased the incidences of SSI at 30 days, pancreatic fistula, sepsis, and mortality.
We suggest antibiotic agent prophylaxis for at least 24 hours in patients undergoing pancreatoduodenectomy with positive bile cultures following preoperative biliary drainage to prevent organ-space SSI (Grade 2-C).
This new recommendation is based on one systematic review and meta-analysis, including eight studies (two RCTs, six observational), totaling 1,170 patients receiving perioperative (24 h) (second- or third-generation cephalosporin) versus prolonged (>24 h) antibiotic agent (piperacillin-tazobactam, gentamicin, ticarcillin-clavulanic acid, or cefuroxime plus metronidazole) prophylaxis after pancreaticoduodenectomy, with or without preoperative biliary drainage. 69 The primary outcome was rate of organ/space SSI. Secondary endpoints were rates of superficial incisional SSI, post-operative pancreatic fistula and bacteremia, hospital length of stay; and bile culture results. Overall, baseline demographics were similar between groups. The duration of prolonged antibiotic agent prophylaxis ranged from 2 to 10 days post-operatively. Compared with perioperative prophylaxis, prolonged antibiotic agent prophylaxis resulted in similar rates of organ/space SSI. However, for patients undergoing preoperative biliary drainage, prolonged antibiotic agent prophylaxis significantly decreased organ/space SSI rates. There were no differences in superficial incisional SSI rates from preoperative biliary drainage. Of note, optimal antibiotic agent durations remain unclear.
Acute appendicitis
Non-operative management of uncomplicated appendicitis
We recommend oral moxifloxacin for empiric therapy of lower risk patients undergoing non-operative management of acute uncomplicated appendicitis (Grade 1-B).
This new recommendation is based on an open-label, multi-center, non-inferiority RCT of 599 patients with acute uncomplicated appendicitis managed non-operatively and administered either oral moxifloxacin for seven days or IV ertapenem for two days followed by oral levofloxacin and metronidazole for five days. 70 The primary outcome was treatment success (≥65%,) defined as hospital discharge without a surgical procedure and no recurrent appendicitis during a one-year follow-up, and if oral antibiotic agents alone were non-inferior to IV plus oral antibiotic agents. The study groups showed similar treatment success (70.2% vs. 73.8%,), and thus, oral moxifloxacin demonstrated non-inferiority versus IV plus oral antibiotic agents.
Complicated appendicitis
We recommend substitution of oral for IV antibiotic agents to complete a short course (24 h) of therapy in lower risk patients with acute complicated appendicitis undergoing adequate source control by laparoscopic appendectomy (Grade 1-B).
This new recommendation is based on an open-label, single-center RCT of 104 patients with acute complicated appendicitis (gangrenous, perforated, peri-appendicular abscess) who received 24 hours of oral versus IV antibiotic agent therapy after laparoscopic appendectomy.
71
The primary outcome was 30-day post-operative complications (Comprehensive Complication Index, CCI), and the secondary outcome was hospital length of stay. Baseline demographics and disease severity score were similar between groups. All patients received amoxicillin plus clavulanic acid. Compared with IV antibiotic agent administration, oral antibiotic agent treatment resulted in no significant differences in 30-day complications, median CCI, or hospital length of stay. Of note, 74% of patients had gangrenous appendicitis versus 16% with perforation and local free fluid, 9.6% with regional abscess, and none with diffuse peritonitis. Considering that definitions of complicated appendicitis differ between studies, this recommendation may not apply to patient populations that are not enriched with gangrenous appendicitis.
We suggest a carbapenem for empiric therapy of patients undergoing non-operative management of acute appendicitis (Grade 2-B).
This new recommendation is based on one systematic review and network meta-analysis, including nine RCTs and 12 observational studies, totaling 4,551 adult or pediatric patients with acute appendicitis receiving either antibiotic agent therapy or antibiotic agent prophylaxis plus appendectomy as initial therapy.
72
The network meta-analysis compared with results indirectly between different antibiotic agent regimens utilizing the surgical procedure as a common comparator. Antibiotic agent regimens include carbapenems, cephalosporins, and BLICs. The primary outcome was one-year treatment success, and secondary outcomes were initial treatment success and treatment-related AEs. The surgical procedure had a significantly higher one-year treatment success compared with cephalosporins or BLICs, but not carbapenems. Compared with the surgical procedure, carbapenems were associated with fewer treatment-related AEs.
We suggest limiting post-operative antibiotic agents to 24-48 hours for low- and high-risk patients with adequate source control for complicated appendicitis undergoing appendectomy (Grade 2-B).
This new recommendation is based on two RCTs. The first is a pragmatic, open-label, multi-center, non-inferiority RCT totaling 1,066 patients with complicated appendicitis (necrosis, perforation, or abscess, as assessed intra-operatively) receiving two- versus five days of IV post-operative antibiotic agents (cefuroxime or ceftriaxone plus metronidazole) after appendectomy. 73 The primary outcome was a composite endpoint of infectious complications (intra-abdominal abscess and SSI) and mortality within 90 days after the surgical procedure. Secondary endpoints were duration of post-operative antibiotic agents, rates of intra-abdominal abscess and SSIs, all post-operative complications, adverse effects to antibiotic agents, restart of antibiotic agents, hospital readmission, surgical or radiologic reinterventions, hospital length of stay, type and number of post-operative imaging studies, and cost. Baseline demographics were similar between groups. There was neither a difference in the primary outcome between groups, nor in complications or reinterventions. Adverse effects of antibiotic agents were significantly lower in the two-day group. Hospital readmission was significantly more frequent in the two-day group. Of note, patients receiving open or laparoscopic converted to open appendectomy randomized to the two-day group were at higher risk of infection complications.
The second is an open-label, single-center RCT of 80 patients with complicated appendicitis (gangrenous, perforated, peri-appendicular abscess) treated with short (24 h)- versus extended (> 24 h)-duration antibiotic agent courses after appendectomy. 74 Primary outcomes included composite post-operative complications and CCI. Secondary outcome was hospital length of stay. Patient demographics and disease severity were similar between groups. The rates of complications were 17.9% and 29.3% in the 24-hour and extended groups, respectively. Compared with subjects in the extended antibiotic agent group, those receiving 24-hour antibiotic agent courses had significantly shorter hospital lengths of stay.
Chronic appendicitis
We suggest against antibiotic agent prophylaxis in lower risk patients undergoing elective laparoscopic appendectomy for chronic appendicitis (Grade 2-B).
This new recommendation is based on one double-blind, single-center RCT, totaling 106 patients treated with preoperative (30 min before surgical procedure) IV antibiotic agents (cefuroxime or clindamycin) versus physiologic saline undergoing laparoscopic appendectomy for chronic appendicitis. 75 The primary outcome was SSI. Secondary outcomes were post-operative complications, time to return of gastrointestinal function, and resolution of primary abdominal pain symptoms. Baseline demographics were similar between groups. There were no differences in the primary or secondary outcomes between antibiotic agent- or physiologic saline-treated controls. Of note, 94 patients were diagnosed pathologically as chronic appendicitis after a surgical procedure, whereas the remaining patients had acute uncomplicated appendicitis or a mucocele. Moreover, the cohorts were relatively young (mean ages, 34–37 yrs) with most patients having no medical co-morbidities, suggesting that these findings may not apply to higher risk individuals.
Abdominal trauma
We suggest ertapenem for antibiotic agent prophylaxis in low- and high-risk patients undergoing laparotomy for traumatic injury (Grade 2-B).
This new recommendation is based on one pre/post protocol implementation, single-center study of 302 patients undergoing exploratory laparotomy for trauma at a level I trauma center. 76 The pre-protocol cohort received antibiotic agents according to surgeon preference. The post-protocol group received ertapenem (1 g) before incision. Patients with carbapenem allergy or anaphylaxis to a β-lactam antibiotic agent received alternative agents at the surgeon’s discretion. Post-operative antibiotic agents were discouraged, but if given, limited to 24 hours except in cases of delayed fascial closure. The primary endpoint was the number of patients with infection directly related to abdominal trauma within 30 days of laparotomy. Secondary endpoints included the number of patients with an incisional SSI, IAI, or any other type of infection at 30 or 90 days after laparotomy. Compared with pre-protocol, protocol implementation with ertapenem significantly decreased infections related to abdominal trauma. Multi-variable logistic regression analysis showed that treatment during the post-intervention period was significantly associated with this reduction, and ertapenem was significantly associated with this reduction in trauma-related infections. Of note, ertapenem usage increased significantly from 2% of pre-intervention patients to 54% of post-intervention patients. Usage of any cephalosporin decreased significantly from 84% in the pre-intervention group to 35% in the post-implementation cohort.
Duration of antibiotic agent therapy
We recommend limiting antimicrobial agent therapy to four days for low- and high-risk patients with source control achieved via a percutaneous drainage procedure (Grade 1-B).
This new recommendation is based on one post hoc analysis of an open-label, multi-center RCT of 129 patients with IAIs who underwent percutaneous drainage versus surgical procedure for source control.58,77 There was no difference in outcome (recurrent IAI, Clostridioides difficile infection, hospital days) between shorter and longer durations of antimicrobial agent therapy.
We recommend limiting antimicrobial agent therapy to four days after achieving source control in high-risk patients with sepsis (Grade 1-B).
This new recommendation is based on one post hoc analysis of an open-label, multi-center RCT of 112 patients with IAI who met systemic inflammatory response syndrome criteria for sepsis (temperature <36°C or >38°C and white blood cell count <4,000 cells/mm3 or >12,000 cells/mm3) treated with short- versus long-course antibiotic agents after adequate source control.58,78 The analysis showed no difference in incidence of SSI, recurrent IAI, extra-abdominal infection, C. difficile infection, number of hospital days, or mortality between groups.
We recommend limiting antimicrobial agent therapy to four days after achieving source control in higher risk patients at increased risk of complications from diabetes mellitus, obesity, or higher illness severity (Grade 1-B).
This new recommendation is based on one post hoc analysis of an open-label, multi-center RCT of 152 obese patients, 64 diabetic patients, and 83 patients with higher illness severity (APACHE-II), with IAI treated with short- versus long-course antibiotic agents after adequate source control.58,79 The analysis showed similar incidences of and days to SSI, recurrent IAIs, extra-abdominal infection, and C. difficile infection between treatment groups. There was no difference in mortality between groups.
We suggest limiting antimicrobial agent therapy to eight days in critically ill patients after achieving adequate source control of IAI (Grade 2-B).
This new recommendation is based on an open-label, multi-center RCT and one systematic review and meta-analysis including that trial and one additional retrospective cohort trial, totaling 489 critically ill patients with post-operative IAIs treated with antibiotic agent therapy for eight- versus 15 days or five- versus 10 days, respectively.80–82 Compared with longer antibiotic agent courses, subjects receiving shorter antibiotic agent courses had similar outcomes, including mortality, ICU and hospital length of stay, emergence of MDR bacteria, or reoperation rate. Patients treated for eight days had a significantly higher median number of antibiotic agent-free days.
De-escalation of antibiotic agent therapy
We suggest de-escalation of antibiotic agent therapy in ICU patients with HA-IAI (Grade 2-B).
We suggest maintaining anaerobic coverage with de-escalation of antibiotic agent therapy (Grade 2-C).
These updated recommendations are based on one prospective, observational, single-center study, totaling 206 patients with HA-IAI admitted to the ICU with retrospective chart analysis of the de-escalation process. 83 Peritoneal fluid samples were collected during surgical procedures, and patients with negative (no growth) microbiologic samples were excluded. Empiric therapy usually combined piperacillin-tazobactam or imipenem-cilastatin with amikacin and vancomycin, with inclusion of anti-fungal therapy (fluconazole) based on risk factors. Definitive antimicrobial agent therapy was based on identification and susceptibility testing. De-escalation involved withdrawal of one agent, narrowing spectrum of activity, or switching from combination- to monotherapy. Patients without de-escalation underwent maintenance of empiric treatment or escalation (defined as addition of or switch to a new broad-spectrum agent, or upgrade to a broader spectrum β-lactam agent.) De-escalation occurred in 53% of patients (n = 110). Cultures of E. faecium, non-fermenting gram-negative bacilli, and MDR strains were less prevalent in the de-escalation group. There was no clinical difference at day seven between patients undergoing de-escalation versus not. Adequate empiric therapy and empiric use of vancomycin, carbapenems, and aminoglycosides were significant determinants of de-escalation by multi-variable analysis. The presence of non-fermenting gram-negative bacilli and MDR strains was a significant risk factors for failure to de-escalate. There was no emergence of MDR organisms following de-escalation in patients who underwent reoperation. Of note, there was no assessment of the quality of initial surgical source control.
Timing of source control procedures
We recommend undertaking source control within 12 hours in lower risk patients (Grade 1-B).
We recommend undertaking source control within six hours in higher risk patients with associated septic shock (Grade 1-B).
These updated recommendations are based on two reports. The first is a systematic review and meta-analysis, including nine observational (two prospective cohort, seven retrospective cohort) studies, totaling 3,373 patients with IAI (acute diverticulitis, gastrointestinal perforation, complicated appendicitis, or peritonitis caused by other hollow organs), treated with surgical operation. 84 Study groups were assigned based on timing of surgical intervention (before or after 12 h). The primary outcome was mortality. Secondary endpoints were hospital length of stay and procedure-related complications. Compared with the delayed intervention group, early surgical exploration resulted in significantly lower mortality and hospital length of stay. Subgroup analysis showed significantly higher survival with early exploration within six hours. For patients with septic shock due to gastrointestinal perforation, survival decreased with operative delay and was zero with delays >six hours. The risk of procedure-related complications was significantly lower with early surgical exploration. Early surgical exploration was associated with significantly shorter hospital length of stay. Of note, although all trials are of high quality, there is no mention of funding sources.
The second report is an observational cohort study from a planned secondary analysis of a cluster-randomized, multi-center RCT of 4,792 adult patients with sepsis admitted to the ICU with at least one new organ dysfunction related to the underlying infection (IAI, respiratory, urogenital, soft tissue/wound/bone, or other/unknown). 85 The primary variables were timing of antimicrobial agent therapy and delay of surgical source control during the first 48 hours after sepsis onset. The primary endpoint was 28-day mortality. Secondary outcomes included hospital length of stay of survivors, progression to septic shock, and success of surgical source control. Delay in surgical source control significantly increased the risk of death in patients with septic shock. Of note, patients who received surgical source control within the first six hours more often had CA-IAI compared with patients who received delayed therapy.
Antimicrobial agent stewardship
We recommend initiating antimicrobial agent therapy within an hour in higher risk patients with sepsis (Grade-1B).
This updated recommendation is also based on the above observational cohort study from a planned secondary analysis of a cluster-randomized, multi-center RCT of 4,792 adult patients with sepsis admitted to the ICU with at least one new organ dysfunction related to the underlying infection (IAI, respiratory, urogenital, soft tissue/wound/bone, or other/unknown).
85
The primary variables were timing of antimicrobial agent therapy and delay of surgical source control during the first 48 hours after sepsis onset. The primary endpoint was 28-day mortality. Secondary outcomes included hospital length of stay of survivors, progression to septic shock, and success of surgical source control. The analysis showed that delays in antimicrobial agent treatment >6 hours significantly increased mortality. Delays in antimicrobial agent therapy significantly increased risk of progression to septic shock. Of note, patients who received antimicrobial agent therapy within the first hour more often had CA-IAI compared with patients receiving delayed therapy.
We recommend implementation of empiric antimicrobial agent therapy protocols to improve antimicrobial agent stewardship (Grade 1-B).
This new recommendation is based on two prospective cohort studies. The first is an observational single-center trial totaling 310 patients with CA- or HA-IAI. 86 Subjects were studied to evaluate compliance with an empiric, initial antimicrobial agent therapy protocol. Patients with medical causes of IAI (not requiring surgical intervention,) post-operative IAI, and acute traumatic perforations <6 hours were excluded. The primary endpoint was protocol compliance. Non-compliance was defined as failure due to illness severity, having risk factors for MDR bacteria, or β-lactam allergy. Appropriateness of therapy was determined by antimicrobial agent susceptibility testing. Secondary endpoints included ICU admission, ICU and hospital length of stay, and in-hospital mortality. Overall, 52.3% of empiric initial antibiotic agent therapy complied with the protocol. Appropriateness of empiric therapy against isolated bacteria was 80%. Protocol non-compliance was associated with higher mortality and higher rates of re-laparotomy, hemodynamic failure, post-operative pneumonia, prolonged mechanical ventilation, ICU stay, and hospital stay. By multi-variable logistic regression analysis, non-compliance with the protocol was independently associated with mortality.
The second report is a prospective, interventional, multi-center trial totaling 4,439 surgical patients receiving antibiotic agents ≥ 7 days for IAI, skin/soft tissue infections, or UTIs to evaluate a nationwide stewardship intervention aimed to reduce antibiotic agent treatment duration in surgical patients to <7 days. 87 The stewardship program provided clinical recommendations regarding the microbiologic appropriateness of treatment and use of broad-spectrum agents. The primary endpoint was the proportion of patients receiving >7 days of antimicrobial agents. The most utilized antimicrobial agents included piperacillin-tazobactam, metronidazole, amoxicillin-clavulanic acid, meropenem, ceftriaxone, and ciprofloxacin. Overall, the infectious focus was considered controlled in 59.9% of patients, and 91.5% of recommendations made by stewardship teams were accepted by the treating physicians. The study reported a significant decrease in the percentage of prolonged antibiotic agent treatments during the study period, from 15.0% to 11.4%.
Prior guideline recommendations
Considering that this updated guideline is not based on preformed clinical questions or topics, and limits available evidence to RCTs, systematic reviews, and meta-analyses, there is no new additional evidence to support or refute many prior recommendations. As this work is intended to supplement, rather than supplant, previous guideline iterations, members of the working group voted to include prior (2017) strong (Class 1), high-to-moderate quality (Level A or B) recommendations in the current version (Table 3).
Non-Updated Prior Guideline Recommendations
Adapted from Mazuski et al. 9 In the interim, microbial phylogenetic taxonomy has changed. Where Enterobacteriaceae are mentioned in this table (previously an order, now a family,) the order Enterobacterales should be substituted. See text for abbreviations.
Discussion
For more than 30 years, the SIS has provided healthcare providers with up-to-date, scientifically validated, clinically relevant guidelines for the prevention and treatment of IAI. This iteration contains recommendations for the care of adult patients with IAI, based on an expansive, systematic literature review with rating of content and quality by members of the SIS Therapeutics and Guidelines Committee and additional SIS members with extensive content or writing expertise pertaining to surgical infections. This version continues where the previous guideline ended by validating and updating prior IAI recommendations while also generating new or updated recommendations based on the current scientific literature. Hopefully, adoption of these evidence-based recommendations will facilitate clinical decision making by healthcare providers who manage adult patients with IAI.
The 2017 guideline iteration made multiple recommendations for future investigations into the management of IAI. 9 These included options and timing for source control, introduction of new anti-infective agents to counter increasing AMR, further reductions in duration of antimicrobial agent therapy, and other approaches to improve antimicrobial agent stewardship. 9 In this latest update, we provide guidance for these important patient care situations. For example, a new recommendation states that patients undergoing percutaneous source control for IAI can receive shorter durations of antibiotic agents akin to individuals managed operatively. An updated recommendation states that source control procedures should occur even earlier than suggested previously. There are multiple new anti-infective agents for the management of IAI, including recommendations for eravacycline, imipenem-cilastatin-relebactam, or biapenem for empiric therapy; and updated, strengthened recommendations for cefoperazone-sulbactam, ceftazidime-avibactam, ceftolozane-tazobactam, and doripenem. The guideline includes recommendations for reducing the durations of antibiotic agent administration or eliminating antibiotic agents altogether under specific circumstances, including with adequate source control for complicated appendicitis or patients undergoing laparoscopic appendectomy for chronic appendicitis. Lastly, this update recommends implementing empiric antimicrobial agent therapy protocols to improve antimicrobial agent stewardship.
Following core principles of antimicrobial agent stewardship, this latest update provides recommendations to start antimicrobial agent therapy within an hour in patients with sepsis, de-escalate antibiotic agent therapy in ICU patients with HA-IAI, and consider oral antibiotic agents equivalent to IV forms for treatment of complicated appendicitis. We continue to recommend against empiric aminoglycoside- or vancomycin-based regimens, anti-fungal therapy in low-risk individuals, or therapy targeting Enterococcus spp. in low-risk adults with CA-IAI. Post hoc analyses of the STOP-IT trial are consistent with the original study findings that shorter antibiotic agent durations are equivalent to longer courses in patients with sepsis, co-morbid diseases, or those undergoing percutaneous source control procedures. By contrast, recommendations for the management of hepatopancreatobiliary malignant disease call for broader-spectrum perioperative prophylactic agents targeting preoperative bile cultures, often necessitating third- or fourth-generation cephalosporins or piperacillin-tazobactam, and a longer antimicrobial agent prophylaxis (at least 24 h) in patients undergoing pancreatoduodenectomy if bile cultures are positive following preoperative biliary drainage.
Non-operative management of uncomplicated acute appendicitis has garnered attention recently, especially during the coronavirus disease-2019 (COVID-19) pandemic.88,89 Whereas the merits of non-operative management versus the surgical procedure are beyond the scope of this guideline, here we provide a recommendation that empiric therapy with oral moxifloxacin shows equivalent treatment success with IV plus oral regimens when treating acute uncomplicated appendicitis without a surgical procedure. Interestingly, a systematic review and network meta-analysis of patients with acute appendicitis undergoing non-operative management or appendectomy found that the surgical procedure had a significantly higher one-year treatment success compared with β-lactams, but not carbapenems. 71 Moreover, carbapenems were associated with fewer treatment-related complications. 71 Whether oral agents such as moxifloxacin are equivalent or superior to an IV carbapenem remains unknown.
Whereas this latest guideline provides timely updates, questions remain for several patient care scenarios highlighted initially back in 2017 or earlier. 9 For example, tools for predicting treatment failure remain elusive. Whereas there is an updated recommendation on the duration of antibiotic agent therapy when caring for patients with increased risk of complications from medical co-morbidities (e.g., diabetes mellitus, obesity), there are no new data regarding antibiotic agent dosing for those individuals. Similarly, there are no additional data regarding optimization of pharmacodynamics, such as with prolonged infusions. Lastly, there is a dearth of high-quality prospective studies including critically ill surgical patients. Thus, recommendations persist for more aggressive empiric therapies in these individuals, thereby discounting the importance of source control for surgical outcomes, and despite the increasing risks of AMR.
In summary, here we provide the most current recommendations from the SIS on the prevention and management of IAI in adult patients. These recommendations are a continuation and refinement of previous work from the SIS and its membership. Whereas important advances have occurred over the intervening years, several important unanswered questions remain from the last guideline iteration that hopefully will prompt further clinical and translational research. It may be necessary to use new research tools or approaches, such as artificial intelligence or machine learning, to solve these more intractable clinical challenges. Or it is possible that a previously unidentified field of scientific inquiry holds the key to further advances. Notwithstanding, as demonstrated over more than 30 years of guideline development, the unwavering dedication and perseverance of surgeons, nurses, and other healthcare providers caring for patients with IAI will prove essential to improving future patient care outcomes.
Footnotes
Authors’ Contributions
Conception and design: Huston, Barie, Forrester. Data analysis: all authors. Data interpretation: all authors. Drafting the article: Huston, Barie, Forrester. Critically revising the article: all authors. Final approval: all authors.
Author Disclosure Statement
E.P.D. has consulted for Ferring Pharmaceuticals and Qiagen. R.W.S has consulted for AbbVie and Shionogi. The remaining authors declare no financial interests.
Funding Information
No funding was received for this article.
