Abstract
Background:
This study aims to analyze the risk factors for post-operative pulmonary infection in patients with brain tumors by meta-analysis to provide a reference for its prevention.
Methods:
PubMed, Embase, Web of Science, Cochrane Library, Ovid, and four Chinese databases (CNKI, SinoMed, VIP, and Wanfang databases) were searched for studies covering risk factors of pulmonary infection in patients with brain tumors, limited to the duration from the dates of inception of the respective databases to December 31, 2022. The Newcastle-Ottawa scale was used to assess the evidence. A meta-analysis of the factors affecting the incidence of pulmonary infection was performed using Revman 5.4 software.
Results:
Twelve studies were selected, covering 35,615 patients with brain tumors, among whom pulmonary infection occurred in 1,635 cases with an accumulated incidence of 4.6%, including 38 related risk factors. Meta-analysis results indicated: history of chronic pulmonary disease (odds ratio [OR], 5.74; 95% confidence interval [CI], 1.34–24.51; p = 0.02], diabetes mellitus (OR, 1.58; 95% CI, 1.29–1.95; p < 0.0001), history of cardiovascular disease (OR, 3.97; 95% CI, 2.18–7.24; p < 0.00001), age ≥60 years (OR, 1.55; 95% CI, 1.12–2.15; p = 0.009)], operation time ≥3 hours (OR, 1.03; 95% CI, 1.00–1.05; p = 0.03], Glasgow Coma Scale (GCS) score <13 (OR, 3.5; 95% CI, 1.90–6.46; p < 0.0001), and the American Society of Anesthesiologists classification (ASA) ≥3 (OR, 2.03; 95% CI, 1.68–2.46; p < 0.00001) as independent risk factors.
Conclusions:
History of chronic pulmonary disease, diabetes mellitus, history of cardiovascular disease, age ≥60 years, operation time ≥3 hours, GCS score <13, and the ASA grade ≥3 are independent risk factors for post-operative pulmonary infection in patients with brain tumors, which nursing staff should be aware of.
Brain tumors, also known as intra-cranial tumors or craniocerebral tumors, refer to the nervous system tumors occurring in the craniocerebral cavity, including those originating from the neuroepithelium, peripheral nerves, meninges, and germ cells; tumors of lymphatic and hematopoietic tissues; craniopharyngioma and granulosa cell tumor in the Sella region; and metastatic tumors. 1 The latest global cancer statistics reported 308,000 newly emerging brain tumors and 251,000 deaths in 2020 worldwide. 2 In the United States, the annual incidence of brain and central nervous system tumors is reported to be 10 per 100,000 cases on average. 3 According to cancer statistics in China, 2022, brain tumors accounted for 5.57% of all malignant tumors and 2.91% of the mortality rate of all malignant tumors, ranked as the ninth leading cause of death for malignant tumors. 4
Surgery has been adopted as the primary treatment for brain tumors, 5 which alleviates clinical symptoms to the maximum extent by safely removing the tumor, prolonging patient survival, and allowing sufficient tumor specimens for pathologic diagnosis and molecular genetic testing, where the pulmonary infection is revealed as the most common post-operative complication. 6 Affected by severe illness, central nervous system damage, long-term disorders of consciousness, and bed rest, patients with brain tumors are more likely to exhibit pulmonary infection. 7 Several extensive sample studies have shown that the rate of pulmonary infection after surgery for brain tumors ranges from 0.6% to 18.29%.8–10 Pneumonia pathogens pass through the distal bronchus and alveoli, break through the host's defense mechanisms, and multiply in the lungs to cause invasive damage. 11
Post-operative pulmonary infection may lead to prolonged mechanical ventilation duration and increased hospital length of stay of patients, resulting in ventilator dependence of patients, increasing the difficulty of disease treatment, affecting the prognosis of patients, resulting in increased mortality, and may also increase the number of intensive care unit (ICU) admissions, patient re-hospitalization rate, patient medical costs, and medical resource consumption.12,13 A five-year cohort study in Canada showed that of 4,033 patients, 378 patients with post-operative pulmonary infection had a significantly lower five-year overall survival rate than those without pulmonary infection (62.8% vs. 73.8%). 14 As a result, it important to explore the risk factors of post-operative pulmonary infection in patients with brain tumors.
Until now, multiple clinical studies on the risk factors of post-operative pulmonary infection in patients with brain tumors have been performed domestically and abroad, revealing the possible factors referring to operation time, age, Glasgow Coma Score (GCS), diabetes mellitus, invasive procedures, ventilator use, basic diseases, etc.8,15–17 However, the outcomes and the views among the studies vary. For example, Ren et al. 8 did not believe age was a risk factor for post-operative pulmonary infection in patients with brain tumors, whereas Zhang et al. 18 came to the opposite conclusion. Deng et al. 19 believed the tumor located in the anterior or lateral brainstem was a risk factor, and Longo et al. 15 found a higher infra-tentorial tumor infection rate. As far as we know, no systematic review or meta-analysis has been conducted to summarize post-operative pulmonary infection risk factors in patients with brain tumors. Herein, we carried out a meta-analysis on the risk factors of pulmonary infection in patients with brain tumors to provide the research basis for clinical prevention of pulmonary infection in patients with brain tumors.
Methods
This review was done according to the Preferred Reporting Items for Systematic Reviews and Meta-Analysis (PRISMA) statement (Supplementary Figure S1). The study protocol has been registered under PROSPERO (Ref: CRD42023398999).
Search strategy
Comprehensive research was performed by searching PubMed, Embase, Web of Science, Cochrane Library, Ovid, and the four Chinese databases, including National Knowledge Infrastructure (CNKI), Chinese Biomedical Literatures database (SinoMed), Wanfang Digital Periodicals (WanFang), and Chinese Science and Technology Periodicals (VIP) database, to identify the risk factors for pulmonary infection in patients with brain tumors. The retrieval time was set from the establishment of the database to December 31, 2022. The “brain neoplasms,” “central nervous system neoplasms,” “brain cancers,” “pulmonary infection,” “pneumonia,” “risk factors,” “influence factors,” and “predictor” were used as the search terms, Medical Subject Headings terms, and their combinations in the title/abstract field of the search engines. The languages were limited to English and Chinese. Using PubMed as an example, the specific search strategy is shown in Figure 1.

PubMed search strategy.
Inclusion and exclusion criteria
The inclusion criteria were as follows: population aged over 18 years, who were diagnosed with a brain tumor by computed tomography (CT) and magnetic resonance imaging (MRI) and who have received surgery 5 ; case control studies or cohort studies; the odds ratio values and 95% confidence interval data obtained from multivariable logistic regression analysis can be extracted or converted; the outcome index was a post-operative pulmonary infection (pulmonary infection occurred within 30 days after surgery), covering diagnostic criteria for pulmonary infection in the study; the “Guidelines for the diagnosis and treatment of hospital-acquired pneumonia and ventilator-associated pneumonia in Chinese adults (2018 edition) 11 was used as the diagnostic criteria for pulmonary infection; and the article language was English or Chinese. The exclusion criteria were as follows: duplicate studies; review, systematic review, meeting summary, case reports; literature with incomplete or inconsistent data; lack of full-text literature; the Newcastle-Ottawa scale was lower than seven points.
Data extraction
Two researchers receiving training in evidence-based methodology independently screened the literature and extracted the data according to the inclusion and exclusion criteria. Disagreements on study eligibility were resolved by discussion or consulting another senior researcher until a consensus was reached. The duplication and preliminary screening of the acquired literature were removed using NoteExpress software, and the literature that met the inclusion criteria was re-screened by careful reading to finally determine the literature enrolled. Data of the enrolled studies were extracted, covering title, first author, publication time, country or region, study type, source of sample size, the incidence of pulmonary infection, number of cases in case group and control group, and independent influencing factors, etc.
Quality assessment
The Newcastle-Ottawa scale was utilized to evaluate the methodological quality of each study, 20 which was independently completed by two researchers. Disagreements were resolved via discussion or consulting a third researcher until a consensus was reached. The Newcastle-Ottawa scale involved eight items in three sections with nine scores. covering selection (4 items, 1 point each), comparability (1 item, 2 points in total), and outcome domains (3 items, 1 point each). The literature with scores seven points or more were considered high quality.
Statistical analyses
Meta-analysis was performed on the RevMan5.4 software. The odds ratio was used as the effect analysis statistic with its 95% confidence interval provided. The heterogeneity between the enrolled studies was analyzed by the χ 2 test (α = 0.1), and I2 determined the heterogeneity. That p > 0.1 and I2 ≤ 50 indicated that the heterogeneity between studies was small, and the fixed effect model was applied for meta-analysis. Instead, the random effects model was applied with statistical heterogeneity existing between the studies. Sensitivity analysis was used to explore the source of heterogeneity. By modulating the effect model, sensitivity analysis for the included literature was performed to evaluate the stability of the combined results. Descriptive analysis was performed to explore the influencing factors of inappropriate meta-analysis. If the number of original studies were 10 or more, the funnel plot was taken to determine the existence of publication bias. A p value <0.05 was considered statistically significant.
Results
Search results
A total of 2,880 articles were retrieved, including 1,591 in English and 1,289 in Chinese. After eliminating duplicate literature, 2,310 articles remained. After reading the title and abstract, the remaining 29 articles were preliminarily screened, and the full text was searched and read. Further exclusion of unable to extract data (5 articles), irrelevant literature (11 articles), and unable to obtain full text (1 article), 12 articles were finally included, including three Chinese articles8,18,21 and nine English articles.6,10,11,15,19–23 The search results are shown in Figure 2.
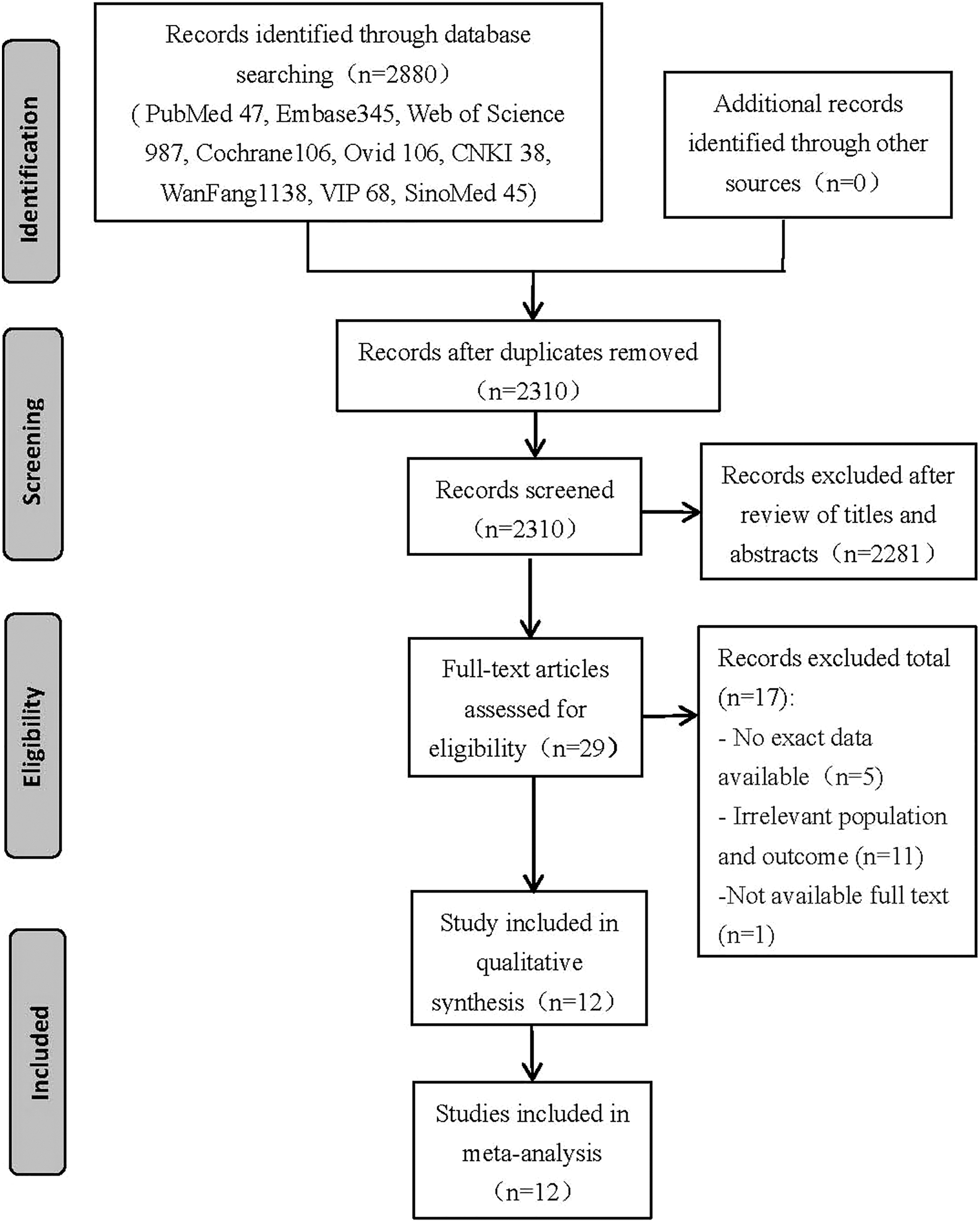
Literature search diagram.
General characteristics of the included studies
Twelve studies were included in the meta-analysis, involving eight case control studies and four cohort studies covering 35,615 patients, among whom 4.6% (1,635) developed post-operative pulmonary infection. All enrolled studies were published from 2012 to 2022, and most after 2019. The general characteristics and methodological quality evaluation results and demographic table of the included studies are listed in Tables 1–3.
Characteristics of the Included Studies
1 = number of sputum aspiration times per day; 2 = tracheotomy time; 3 = Glasgow Coma Scale (GCS) score <13; 4 = history of chronic pulmonary disease; 5 = invasive operation; 6 = operation time ≥3 h; 7. age ≥60 years; 8 = intraoperative infusion volume ≥3,000 mL; 9 = endotracheal tube; 10 = history of chronic pulmonary disease; 11 = pulmonary imaging abnormalities; 12 = epilepsy; 13 = systemic inflammatory response index (SIRI); 14 = tumor size; 15 = tumor located in the anterior or lateral brainstem; 16 = estimated blood loss; 17 = gender; 18 = self-care ability; 19 = diabetes mellitus; 20 = dyspnea; 21 = chronic steroid use; 22 = pre-operative white blood cell (WBC) count >2; 23 = American Society of Anesthesiologists (ASA) classification ≥3; 24 = emergent case; 25 = infra-tentorial; 26 = body mass index (BMI) ≥40 kg/m2; 27 = tumor recurrence; 28 = skull base tumors; 29 = blood transfusion; 30 = red blood cell distribution width-standard deviation (RDW-SD); 31 = neutrophil to lymphocyte ratio (NLR); 32 = Koos grade of vestibular schwannoma (VS); 33 = Karnofsky performance score (KPS); 34 = high lactate dehydrogenase (LDH); 35 = mechanical ventilation time; 36 = extraction time of tracheal catheter; 37 = stroke; 38 = anesthesia time.
Quality Assessment of Included Studies
NOS = Newcastle-Ottawa scale.
Demographic Table of Pooled Patients
Meta-analysis results
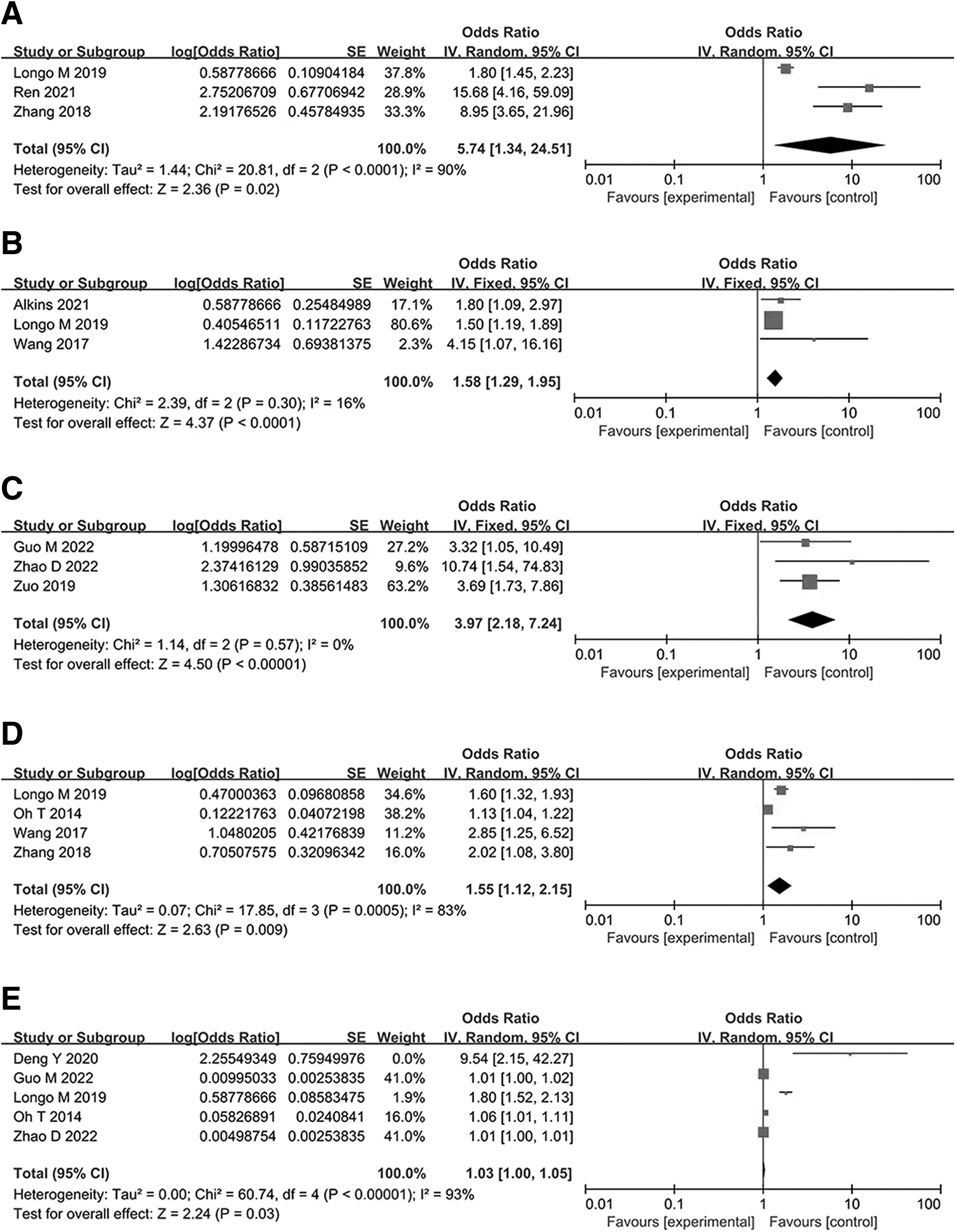
To avoid results bias, the influencing factors mentioned in only one study were excluded, and those in two or more studies were combined. Meta-analysis indicated the history of chronic pulmonary disease, diabetes mellitus, history of cardiovascular disease, age ≥60 years, operation time ≥3 hours, GCS score <13, and the ASA grade ≥3 as the independent risk factors for post-operative pulmonary infection in patients with brain tumors.
History of chronic pulmonary disease
Three studies8,15,18 reported the relation between the history of chronic pulmonary disease and pulmonary infection after brain tumor surgery, and there was obvious heterogeneity among the studies (p < 0.0001; I2 = 90%). Therefore, the random effects model was used for analysis. The results showed that history of chronic pulmonary disease is a risk factor for pulmonary infection in patients with brain tumors (odds ratio [OR], 5.74; 95% confidence interval [CI], 1.34–4.51; p = 0.02). Forest plot is shown in Figure 3A.
Diabetes mellitus
The relation between diabetes mellitus and pulmonary infection was reported in three studies,15,24,25 displaying acceptable statistical heterogeneity (p = 0.3; I2 = 16%). As a result, the fixed-effects model was used for analysis, which revealed diabetes mellitus as a risk factor (OR, 1.58; 95% CI, 1.29–1.95; p < 0.0001). Forest plot is shown in Figure 3B.
History of cardiovascular disease
The relation between the history of cardiovascular disease and pulmonary infection was reported in three studies,6,10,23 without heterogeneity (p = 0.57; I2 = 0%). The fixed effects model was applied, which revealed the history of cardiovascular disease as a risk factor (OR, 3.97; 95% CI, 2.18–7.24; p < 0.00001). Forest plot is shown in Figure 3C.
Age ≥60 years
Four studies15,18,22,25 examined the effect of age on pulmonary infection, with heterogeneity (p = 0.0005; I2 = 83%). The random effects model was used, which indicated age ≥60 years as a risk factor for pulmonary infection in patients with brain tumors (OR, 1.55; 95% CI, (1.12–2.15; p = 0.009. Forest plot is shown in Figure 3D.
Operation time ≥3 hours
Five studies with obvious heterogeneity (p < 0.00001; I2 = 93%) were included.10,15,19,22,23 Sensitivity analysis indicated no change in heterogeneity, and the random effects model was therefore used for meta-analysis, which indicated the significantly long operation time in patients with pulmonary infection compared with those without infection (OR, 1.03; 95% CI, 1.00–1.05; p = 0.03]. Forest plot is shown in Figure 3E.
Glasgow Coma score <13 and ASA grade ≥3
Two studies8,14 without heterogeneity (p = 0.71; I2 = 0%) investigated whether a GCS score <13 exerted an effect on pulmonary infection. The fixed effects model was applied, revealing a GCS score <13 as a risk factor for pulmonary infection in patients with brain tumors (OR, 3.5; 95% CI, 1.90–6.46; p < 0.0001]. Two studies15,26 reported the relation between the ASA grade ≥3 and pulmonary infection, with acceptable statistical heterogeneity (p = 0.18; I2 = 45%). The fixed effects model was applied, which indicated ASA grade ≥3 as a risk factor (OR, 2.03; 95% CI, 1.68–2.46; p < 0.00001).
Descriptive analysis results
According to the studies enrolled, one study 8 indicated the daily number of sputum aspirations, tracheotomy time, and invasive operation to exert an impact on post-operative pulmonary infection in patients with brain tumors. The data 18 indicated a higher risk of pulmonary infection associated with brain tumor patients with intra-operative infusion volume ≥3,000 mL and tracheal catheter retention after surgery. One study 23 reported the pulmonary imaging abnormalities and epilepsy as risk factors for post-operative pulmonary infection in patients with brain tumors. One study 26 demonstrated the association of patients with high systemic inflammatory response index (SIRI) with higher risk of lung infection. One study 14 pointed out that tumor size and tumor location in the anterior or lateral brain stem were risk factors for post-operative lung infection. Another study 10 indicated gender, self-care ability, dyspnea, long-term steroid use, pre-operative white blood cell (WBC) count ≥12 × 109/L, emergent case, infra-tentorial tumor, and body mass index ≥40 kg/m2 as risk factors; One study 6 demonstrated tumor recurrence, skull base tumor, blood transfusion, red blood cell distribution width-standard deviation (RDW-SD) and neutrophil to lymphocyte ratio (NLR) as risk factors for pulmonary infection. Another study 25 reported the Koos grade of vestibular schwannoma (VS) as a risk factor for pulmonary infection. One study 21 reported the positive correlation of Karnofsky performance score (KPS), lactate dehydrogenase (LDH), and mechanical ventilation duration with the occurrence of pulmonary infection after brain tumor surgery. Another study 10 indicated the extraction time of tracheal catheter and anesthesia time as risk factors. Another study 24 reported the stroke as a risk factor for pulmonary infection after brain tumor surgery. Considering the report from a single study without enough data for meta-analysis, the description was qualitative.
Sensitivity analysis and publication bias detection
The fixed effects model and the random effects model, respectively, were used to estimate the combined odds ratio values of the risk factors with high heterogeneity, which gave similar results, indicating the essential reliability of the present study. The Cochrane Handbook for Systematic Reviews 5.1 27 suggests that conducting publication bias analysis by funnel plot will achieve higher meaning when the number of studies is not <10. In our study, the number of studies involved in risk factors was <10, so there may be publication bias, and publication bias analysis was not conducted.
Discussion
History of chronic pulmonary disease
The present study indicated a 5.74 times greater risk of developing a pulmonary infection in patients with brain tumors with a history of chronic pulmonary disease compared with those without, which is consistent with previous findings. A study by Daming et al. 9 based on data from the American College of Surgeons National Surgical Quality Improvement Program (ACS-NSQIP) database demonstrated a higher tendency to suffer from post-operative infection in patients with a history of chronic lung diseases because of the reduced airway clearance, increased airway inflammation, and reduced lung compliance, also supported by Lee et al. 28 For patients undergoing surgery, the traumatic stress response elicited by surgical trauma will further lead to the decline of immune function and the weakened resistance of patients, making it easy for the invasion and colonization of pathogenic bacteria, and thus promoting the risk of pulmonary infection. 29 Therefore, clinical medical staff is should actively encourage patients to undergo lung rehabilitation training, such as lip contraction breathing by lung rehabilitation education, to improve the lung function. 30
Diabetes mellitus
Diabetes mellitus is considered a risk factor for post-operative pulmonary infection. An earlier study reported a 6% to 10% increase in the risk of pulmonary infection with every 1 mmol/L increase in blood sugar. 31 The mechanism may be the inhibited expression of TLR2 and TLR4 in monocytes of patients with diabetes that damages the immune function of patients. In addition, the hypoperfusion and hypoxia environment caused by diabetic autonomic neuropathy and peripheral vascular disease will also affect the aggregation and attachment of immune cells, reducing the anti-infection ability of the body and increasing the risk of post-operative infection.32,33 Chronic hyperglycemia leads to the increased concentration of liquid glucose on the surface of the airway, which drives the multiplication of bacteria. Altered cell function, bacterial proliferation, and vascular permeability promote the risk of pulmonary infection.34,35
Poor control of blood sugar or the prolonged duration of diabetes mellitus may aggravate the susceptibility to pneumonia after surgery. Therefore, it is essential to strengthen the pre-operative control of blood glucose in patients with brain tumors complicated by diabetes mellitus. However, fasting or random blood glucose is often used in clinical work for pre-operative assessment and regulation, and it is easy to be affected by many factors such as diet, exercise, pain, traumatic stress, and other transient interference, which is difficult to reflect the blood glucose control level for a period of time, affecting the correct assessment of pre-operative risk and prognosis. 36
Hemoglobin A1c (HbA1c) is a continuous, slow, and irreversible non-enzymatic glycation product of glucose and hemoglobin in the blood. As a gold standard reflecting the blood glucose control level, HbA1c can reflect the blood glucose control level of patients in the past two to three months. 37 Pre-operative elevated HbA1c, as a marker of poor glycemic control, is associated with increased peri-operative risk in patients with diabetes, and patients with HbA1c >6.5% have a higher risk of post-operative pulmonary infection than those with HbA1c <6.5%. 38 Pre-operative assessment of patient's HbA1c was not analyzed in detail in the original study included in this article. In future clinical studies, more attention should be paid to pre-operative changes in patient's HbA1c to assess blood glucose control.
History of cardiovascular disease
Consistent with the previous reports, the pooled results of the present study suggest the history of cardiovascular disease as a risk factor for post-operative pulmonary infection. It may trigger or aggravate the instability of hemodynamics, resulting in pulmonary congestion, edema, and infection. 39 Studies have evidenced the high risk of developing severe post-operative complications in patients with meningioma who have hypertension or are taking cardiac drugs. 6 It is suggested that the cardiovascular system status of patients should be evaluated carefully before surgery for brain tumors. 40
Age
Xiang et al. 41 carried out a large single-center retrospective study and found age >60 years as an independent risk factor for post-operative pulmonary infection. Kunisaki et al. 42 reported the different incidences of post-operative pulmonary infection between patients >75 years old and those between 45 and 65 years of age (13.3% and 6.3%). Similarly, Yamada et al. 43 reported a higher incidence of post-operative pulmonary infection in patients over 85 years old compared with those between 75 and 85 years old (16.7% and 3.3%). These findings are in accordance with the present study, in which the risk of post-operative pulmonary infection is increased with patient age. This could be explained by the decline in respiratory system function, swallowing, and immune function with age in elderly patients with brain tumors, which reduces the resistance to bacterial invasion and poses a higher risk of post-operative lung infection. 25 Therefore, effective measures should be taken to prevent pulmonary infection in elderly patients with brain tumors.
Operation time
The higher risk of post-operative vomiting induced by the longer operation time may increase the incidence of post-operative aspiration pneumonia in patients with brain tumors, especially for those in a post-operative coma and impaired swallowing function.44–46 In addition, prolonged operation time also represents longer tracheal intubation time, which will elevate the risk of post-operative pulmonary infection. 47 In the present study, the incidence of post-operative pulmonary infection was 1.03 times in patients with operation time of three hours or more compared with those less than three hours. Therefore, it is suggested to identify the high-risk patients to shorten the operation duration so as to reduce the incidence of pulmonary infection after brain tumors, especially for patients with large tumors in the anterior or lateral brainstem. 19
Glasgow Coma Score <13 and ASA grade ≥3
Lower GCS score is associated with a higher risk of post-operative pulmonary infection in patients with brain tumors, which is consistent with previous studies.48,49 On the one hand, it may be related to the inhibited gag reflex induced by coma that leads to aspiration pneumonia. On the other hand, these patients generally require mechanical ventilation, which may damage the airway and aggravate the possibility of ventilator-associated pneumonia. 50 By conducting a large single-center observational cohort study, Lioutas et al. 51 found that mechanical ventilation increased the risk of pulmonary infection by more than four times. Endotracheal intubation makes the lower respiratory tract exposed to the outside world, followed by difficulty in cleaning the oral cavity. As a result, the colonizing bacteria multiply in the oropharynx, and the secretions containing colonizing bacteria penetrate the lower respiratory tract through the gap between the air sac and the air tube wall, leading to pulmonary infection. In addition, our meta-analysis also demonstrated ASA grade ≥3 as a critical risk factor for pulmonary infection after brain tumor surgery.
As an effective method to evaluate the health status and surgical risk of patients, ASA classification could contribute to predicting peri-operative risk. The higher ASA grade is associated with the worse general condition of patients and a higher tendency to develop pulmonary infection.52,53 However, considering the limited amount of studies related to the correlation between these two risk factors, with only two studies included in the meta-analysis for each factor, large-sample, multicenter studies are still required to verify this conclusion.
Limitations
To the best of our knowledge, this is the first meta-analysis of the risk factors for pulmonary infection in patients with brain tumors. However, there are some limitations. First, the studies on the risk factors of pulmonary infection after brain tumor surgery domestically and abroad are limited, so included articles are limited, which makes it insufficient to elucidate the risk factors of pulmonary infection after brain tumor surgery. Second, the risk factors involved in the included studies were numerous but scattered; some risk factors are reported in a few studies, so meta-consolidation cannot be performed, only descriptive analysis. Third, some factors of the meta-analysis were mentioned in only two studies; the authenticity and reliability of the results remain to be discussed. Among the included studies, few were cohort studies, accompanied by the small sample sizes of some included studies. Therefore, more prospective cohort studies with rigorous designs and large sample sizes are recommended to explore the risk factors of post-operative pulmonary infection further in patients undergoing brain tumor surgery in future research.
Conclusions
In summary, this study demonstrated that history of chronic pulmonary disease, diabetes mellitus, cardiovascular disease, age ≥60 years, operation time lasting three or more hours, GCS score <13, and ASA grade ≥3 were independent risk factors for post-operative pulmonary infection in patients with brain tumors. The results can serve as a reference for clinical medical staff to strengthen the evaluation and management of patients undergoing brain tumor surgery and take targeted and effective intervention as early as possible so as to avoid the incidence of post-operative pulmonary infection in patients with brain tumors and improve the clinical outcome. At the same time, because of the limited number of articles included in this study, some risk factors may exert a particular impact on the research results. Future large-sample, multicenter, high-quality research recommended.
Footnotes
Acknowledgments
The authors thank the Guangxi Medical and Health Appropriate Technology Development and Application Project for funding this paper.
Authors' Contributions
Conceptualization (lead): Lan. Conceptualization (supporting): Lei. Formal analysis (lead): Lan. Writing—original draft (lead): Lan. Writing—original draft (supporting): Lei. Writing—review and editing (equal): Lan, Yan Wei, Zhu, Y. Zhang, S. Zhang, Mo, Lei. Software (lead): Yan Wei, Zhu, Y. Zhang, S. Zhang. Methodology (lead): D. Wei.
Funding Information
This work was supported by Guangxi Medical and Health Appropriate Technology Development and application project (grant number: S2020104).
Author Disclosure Statement
This manuscript has no potential conflict of interest to disclose. Because this is a systematic review and meta-analysis, ethical approval and consent to participate are not applicable.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
