Abstract
Background:
Predictive models are necessary to target high-risk populations and provide precision interventions for patients with lung neoplasm who suffer from surgical site infections (SSI).
Patients and Methods:
This case control study included patients with lung neoplasm who underwent minimally invasive surgeries (MIS). Logistic regression was used to generate the prediction model of SSI, and a nomogram was created. A receiver operator characteristic (ROC) curve was used to examine the predictive value of the model.
Results:
A total of 151 patients with SSI were included, and 604 patients were randomly selected among the patients without SSI (ratio 4:1). Male gender (odds ratio [OR], 2.55; 95% confidence interval [CI], 1.57–4.15; p < 0.001), age >60 years (OR, 2.10; 95% CI, 1.29–3.44, p = 0.003), operation time >60 minutes (all categories, p < 0.05), treatments for diabetes mellitus (OR, 2.96; 95% CI, 1.75–4.98l; p < 0.001), and best forced expiratory volume in 1 second (FEV1)/forced vital capacity (FVC; OR, 0.96; 95% CI, 0.94–0.99; p = 0.008) were independently associated with SSI. The model based on these variables showed an area under the curve (AUC) of 0.813 for predicting SSI.
Conclusions:
A nomogram predictive model was successfully established for predicting SSI in patients receiving MIS, with good predictive value.
Lung cancer is the malignant tumor with the highest incidence and mortality rates in China, of which non-small cell lung cancer (NSCLC) accounts for approximately 80%–85%.1–3 With the continuous advances and maturation of the video-assisted thoracoscopic surgery, minimally invasive surgery (MIS) has become the mainstay therapy for early NSCLC.4–7 In comparison with conventional surgical techniques, MIS could reduce pain and compliance after surgery.6,8 In recent years, the application of neoadjuvant chemotherapy 9 and immunotherapy10,11 has expanded the indications for MIS to selected patients with intermediate or advanced lung neoplasms. In spite of this, patients with cancer suffer from immunologic dysfunction and other complications including endocrine dyscrasia, which increases their risk of complications after surgery. Surgical complications can be life-threatening and will increase healthcare costs.
Surgical site infection (SSI) refers to the infection of the surgical incisions, deep organs, or spaces. 12 According to the World Health Organization (WHO), SSI is the most surveyed and frequent type of health-care–associated infections (HAI) in low- and middle-income countries, and remains the second most frequent type of HAI in Europe and the United States. 13 Surgical site infection is also one of the common complications in patients undergoing thoracic surgeries.12,14 It has been established that SSI generally extends hospital stays, increases financial burdens, and even poses a life-threatening risk to patients.15,16 There are a variety of factors that contribute to SSI risk, including microbial, patient-related, and procedure-related factors. 17 In 2018, the WHO provided 29 recommendations covering 23 topics for SSI prevention in the pre-, intra-, and post-operative periods. 13 Although simultaneous application of multiple interventions can reduce SSI incidence in surgical patients, such combined interventions can also waste medical resources, increase medical costs, and elevate the risk of antibiotic resistance.18,19 In light of the increasing complexity and nuance of guidelines for preventing SSI, more studies are urgently needed on prevention of SSI in patients undergoing MIS.
Previous studies demonstrated that risk factor stratification for SSI 20 and active surveillance 21 are effective in early identifying high-risk patients and preventing SSI. Although several studies have already reported the risk factors and predictive measurements for SSI in patients undergoing thoracic surgeries,12,14,16 no risk factor stratification strategies are available specifically for SSI in patients with lung neoplasms undergoing MIS. In addition, the conventional infection assessment methods and indicators are less specific or convenient for SSI after MIS.22,23 Therefore, developing precise, individualized risk prediction models for SSI in patients with lung neoplasms undergoing MIS is urgently necessary.
This study aimed to develop a prediction model for SSI in patient with lung neoplasms after MIS. The results could help assess the risk of SSI and identify patients with high SSI risks early. Consequently, the study tried to identify the subpopulation for precise clinical practices, allowing individualized interventions and treatments to prevent SSI.
Patients and Methods
Study design and patients
The predictive model was developed with data collected from Shanghai Chest Hospital, which had more than 45,000 patients with resected lung neoplasm undergoing MIS from 2018 to 2020. This study was approved by the Ethics Committee of the Shanghai Chest Hospital. The requirement for informed consent was waived by the committee.
The model was developed on observational data from a retrospective cohort using a case-control design. The inclusion criteria were adult patients undergoing MIS for resecting lung neoplasm and with available post-operative 30-day follow-up data. The exclusion criteria were chronic infections, or administration immunosuppression, or converted to an open procedure during MIS, or emergency surgery.
The methods were carried out in accordance with relevant guidelines and regulations. Electronic health record (HER) data from the study center between January 2018 and December 2020 were analyzed. The reporting of the study followed the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) guidelines and adhered to the Transparent Reporting of a Multivariable Prediction Model for Individual Prognosis or Diagnosis (TRIPOD) recommendations.
Identification of cases
Surgical site infection was defined as an infection that occurred within 30 days after the operation and involved the skin and subcutaneous tissue of the incision or the deep soft tissue of the incision or any part of the anatomy other than the incision that was opened or manipulated during an operation. 24 Surgical site infection was defined as the presence of any of the following criteria.: purulent discharge in or exuding from the wound or observed on direct examination; fever >38°C, accompanied by wound pain and tenderness, swelling; imaging diagnosis of pulmonary infection; and positive for bacterial culture.
Surgical site infection occurrence was determined if the patients had an International Statistical Classification of Diseases and Related Health Problems, 10th Revision (ICD-10) diagnosis of J98.402 Pulmonary infection, J86 Empyema, J86.002 Pyothorax associated with fistula, J86.053 Empyema with bronchopleural fistula, J86.054 Empyema with bronchial fistula, J86.951 Acute total empyema with atelectasis, or a mention within a follow-up report of “fever,”” infection,” or “wound pain” using text matching, and then selected patients with inclusion and exclusion criteria.
Identification of controls
A stratified simple random sampling was adopted to select the patients without SSI at 1:4 matched according to the year of admission to the hospital and elimination the non-eligible samples, as the controls for the included SSI patients. Controls were not matched to cases on specific variables to prevent bias and explore all possible relationships.
Potential clinical risk factors and data collection
The data were obtained from the medical records, imaging examination reports, and surgery operative logs, which included general information including gender, age, body mass index (BMI; based on the Asian WHO cutoffs 25 ), and smoking history; comorbidities, including chronic obstructive pulmonary disease (COPD), hypertension, hyperlipidemia, and diabetes mellitus; pulmonary function, including forced expiratory volume in 1 second (FEV1)/forced vital capacity (FVC), expected FEV1/FVC, best FEV1/FVC, FEV1%, expected FEV1%, and best FEV1%; cardiac function, including left ventricular ejection fraction (LVEF); renal function, including estimated glomerular filtration rate (eGFR)); laboratory examination indicators, including C-reactive protein (CRP), leukocytes, erythrocyte sedimentation rate (ESR), neutrophil percentage, alanine aminotransferase (ALT), aspartate aminotransferase (AST), glycated hemoglobin (HbAlc), albumin, procalcitonin (PCT), urinary red blood cell (URBC), and hemoglobin (Hb). The laboratory examination results were categorized into five categories according to the reference range: lower than 2 folds of the lower limit; 2 folds of the lower limit to the lower limit; normal; upper limit to 2 folds of the upper limit; and more than 2 folds of the upper limit. The reference range of the indicators were as follows: CRP, 0.5–10 mg/L; leukocytes, 3.5–9.5 × 10 9 /L; ESR, 0–40 mm/h; neutrophil percentage, 40%–75%; ALT, 9–50 U/L; AST, 15–40 U/L, HbAlc, 4.5%–6.3%; albumin, 35–52 g/L; PCT, 0–0.05 ng/mL; URBC, 0–10/mcL; and Hb, 130–175 g/L. Data on surgery, included pre-operative hospital stay, post-operative hospital stay, operation time, American Society of Anesthesiologists (ASA) grade, and intra-operative blood transfusion; pathologic diagnosis including histologic types, tumor sizes, and metastasis were also extracted.
Statistical analysis
The Shapiro-Wilk test was used to test the continuous variables for normal distribution. Normally distributed variables are expressed as mean ± standard deviation (SD), whereas skewed variables are expressed as the median (interquartile range, IQR). The unpaired two-tailed Student t-tests and Mann-Whitney U tests were applied to examine difference for normally and non-normally distributed continuous variables, respectively. The categorical data were described as frequencies (percentages) and compared using the χ 2 test or Fisher exact test. Missing or incomplete data were considered missing data. Logistic regression was used to explore the risk factors of SSI. All variables with statistical significance in the univariable analyses were included in the multivariable logistic regression analysis. A nomogram was designed based on the results of the multivariable analysis. The receiver operator characteristic (ROC) curve was used to assess the predictive value of the final model. The area under the curve (AUC), sensitivity, specificity, positive predictive value (PPV), negative predictive value (NPV), and accuracy were recorded. SPSS Statistics, version 22 (IBM Corp, Armonk, NY) was used for the analyses. A p value of <0.05 was considered statistically significant.
Results
Baseline characteristics
A total of 151 SSIs were diagnosed and identified as cases in this study, and 604 patients without SSIs (1:4) were selected as controls. Among the 755 cases and controls, there were 44.9% males and 55.1% females, and patients with SSI had a higher proportion of males than controls (74.2% vs. 37.6%). A total of 239 cases (31.7%) were estimated as overweight (no significant difference between cases and controls; p = 0.698). There was a higher proportion of patients who used tobacco in SSI cases and controls (14.1% vs. 7.6%; p = 0.021). Surgical site infection cases showed a higher proportion of >60 years of age (72.8% vs. 47.5%; p < 0.0001) and treatments for diabetes mellitus (33.1% vs. 11.3%; p < 0.0001) than controls. Concerning disease progression, patients with SSI showed higher proportions of squamous carcinoma (21.9% vs. 5.3%; p < 0.0001), tumor
Patient Characteristics
SSI = surgical site infection; COPD = chronic obstructive pulmonary disease; ASA = American Society of Anesthesiologists.
Based on the World Health Organization cutoffs for Asians. 25
Benign lung tumors include inflammatory nodule, hamartoma, tuberculoma, etc. Other pathologic types include large cell lung neoplasms (n = 2), small cell lung neoplasms (n = 2), mixed (n = 10), unclear (n = 28), and metastatic lesion (n = 6).
Table 2 presents the laboratory and cardiopulmonary examinations. Compared with the control group, the SSI group showed lower ESR (p = 0.012), lower albumin (p = 0.011), lower FEV1/FVC (p = 0.001), lower predicted FEV1/FVC (p < 0.0001), lower best FEV1/FVC (p < 0.001), lower FEV1% (p = 0.003), and lower eGFR (p < 0.0001).
Laboratory Examination Results and Cardiopulmonary Function
SSI = surgical site infection; CRP = C-reactive protein; ESR = erythrocyte sedimentation rate; ALT = alanine aminotransferase; AST = aspartate aminotransferase; HbAlc = glycated hemoglobin; PCT = procalcitonin; URBC = urinary red blood cell; Hb: hemoglobin; FEV1 = forced expiratory volume in 1 second; FVC = forced vital capacity; LVEF = left ventricular ejection fraction; eGFR = estimated glomerular filtration rate; IQR = interquartile range.
Concerning the clinical processes between groups, patients with SSI had a longer pre-operative (6.0 vs. 4.0 days; p = 0.0003) and post-operative hospital stay (median, 14.0 vs. 4.0 days; p < 0.0001), longer operation time (p < 0.0001), higher frequency of blood transfusion (19.2% vs. 0.2%; p < 0.0001) compared with the controls.
Univariable and multivariable logistic regression analysis for SSI
Multivariable model included 25 variables that were significant in univariable analysis (with p < 0.15; gender, age, pathologic type, tumor size, metastasis, COPD, hypertension, hyperlipidemia, medication for diabetes mellitus, CRP, leukocytes, ESR, neutrophil percentage, ALT, FEV1/FVC, predicted FEV1/FVC, best FEV1/FVC, FEV1%, predicted FEV1%, best FEV1%, pre-operative hospital stay, operation time, ASA, and intra-operative blood infusion), and five of them remained by backward regression in multivariable analysis (Table 3).
Multivariable Analysis of SSI
SSI = surgical site infection; OR = odds ratio; CI = confidence interval; FEV1 = forced expiratory volume in 1 second; FVC = forced vital capacity.
The multivariable logistic regression analysis showed that the male gender (OR, 2.55; 95% CI, 1.57–4.15; p < 0.001), age >60 years (OR, 2.10; 95% CI, 1.29–3.44; p = 0.003), operation time >60 minutes (all categories, p < 0.05), diabetes mellitus (OR, 2.96; 95% CI, 1.75–4.98; p < 0.001), and best FEV1/FVC (OR, 0.96; 95% CI, 0.94–0.99; p = 0.008) were independently predictors for SSI. The study also conducted a sensitivity analysis to assess the model performance. The results showed an AUC of 0.813, a sensitivity of 76%, a specificity of 73%, a PPV of 0.408, an NPV of 0.924, and an accuracy of 73% for SSI prediction (Fig. 1A). The AUCs of models for predicting SSI were 0.8153, 0.7838, and 0.7313 for patients who received lobectomy (Fig. 1B), wedge resection (Fig. 1C), and segmentectomy (Fig. 1D), respectively. Based on the regression model, Figure 2 illustrates a nomogram for calculating SSI risk for patient undergoing MIS. The total point, which was the sum of each predictor point, ranged from 50 to 240. The point indicated a 5%–90% possibility getting SSI.
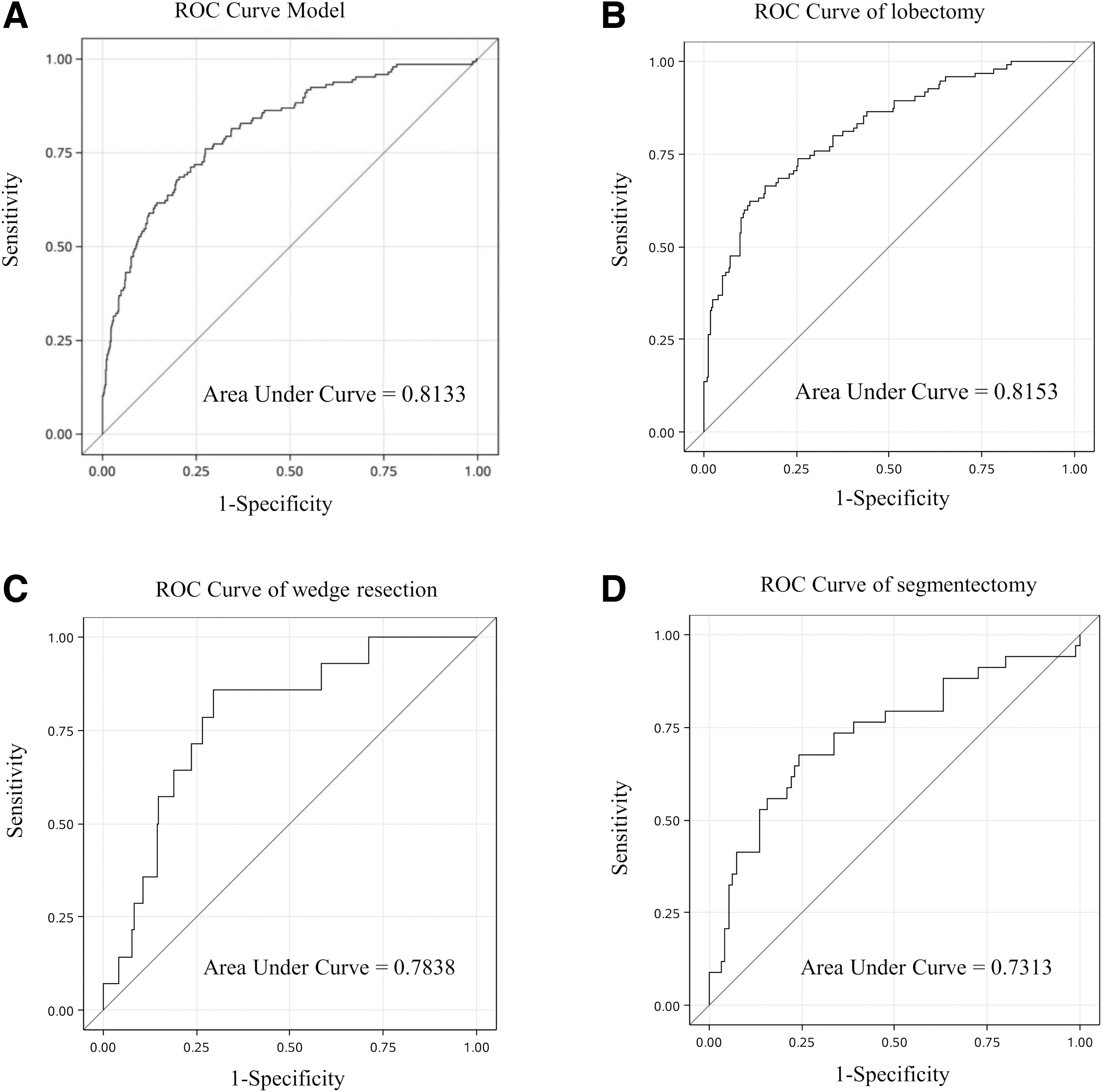
Receiver operating characteristics (ROC) curve of the model for (
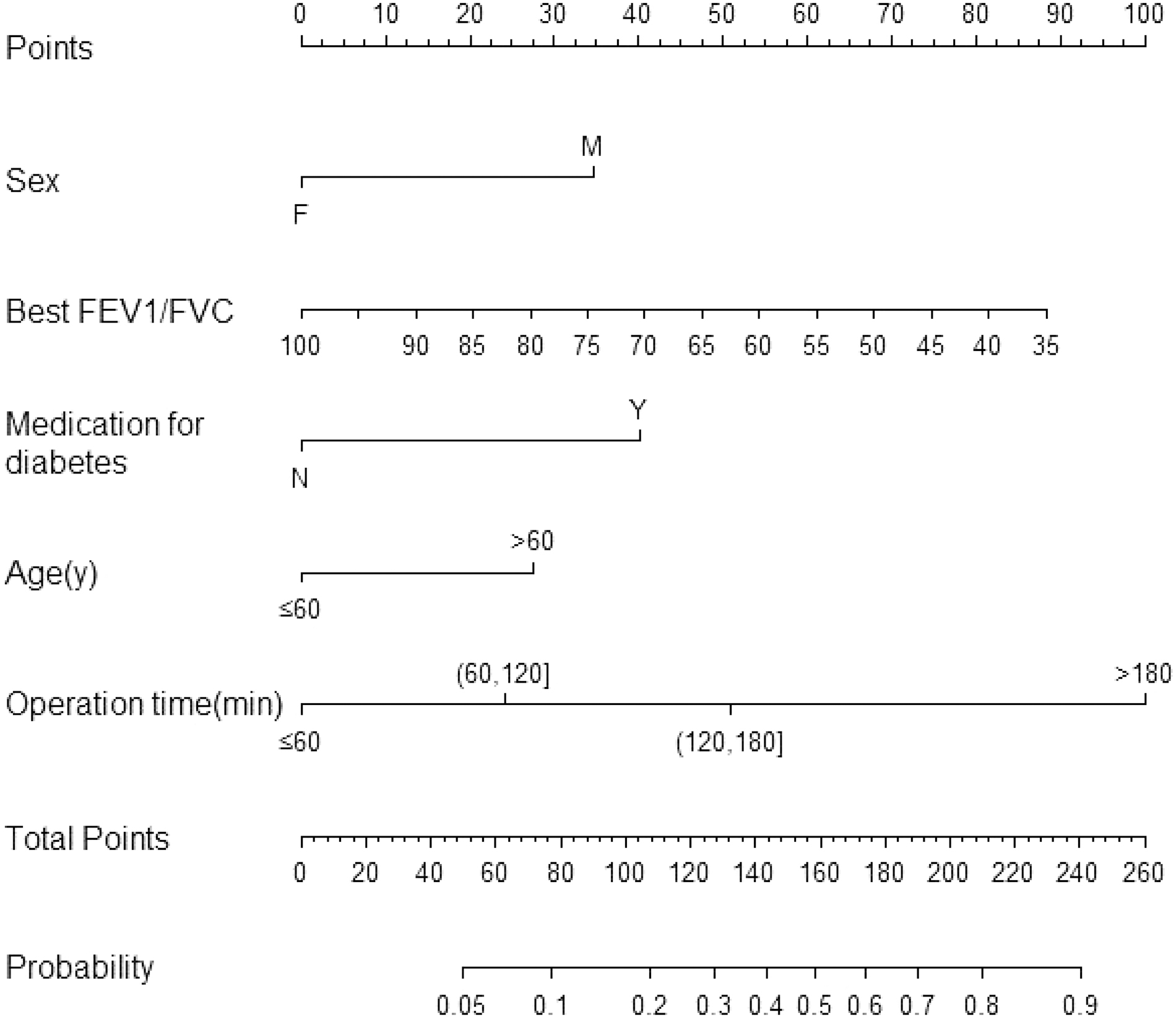
Nomogram for determining the risk of surgical site infection (SSI) after lung minimally invasive surgery (MIS).
Discussion
Our study was able to successfully develop the SSI risk model with predictive SSI after MIS, using commonly available Electronic Health Record (EHR) data. The model used data elements available as structured data fields within the EHR, commonly collected in multiple practice settings.
According to global data announced by WHO in 2018, the overall SSI rate was 1.9% around 2010. 13 A decrease in the SSI rate has been observed because of global prevention efforts and surveillance since 2010. The SSI rate differed in different types of surgical procedures: higher for procedures in cardiac and gynecologic surgeries and lower for laminectomy and knee prosthesis. 13 Previous studies have reported the SSI rate among all-type thoracic surgeries around 6%, but ranged from 2% in video-assisted thoracic surgery to 7% in open thoracic surgical procedures.14,26–27 A retrospective observational study based on national surveillance data in Japan reported rates of SSI around 1.2% video-assisted thoracic surgeries. 28 Because of prodigious growth of thoracic surgeries, the absolute quantity of SSI would be concerning. There were at least 76% that could benefit from early recognition according to our findings.
However, the disease burden of SSI might be underestimated for the following reasons. First, it is possible that some infections occurred after discharge, which were not recorded by institutions. Another concern is about the widely use of antibiotic agents before and after MIS, which would lead to negative bacterial cultures when diagnosing SSI.29,30 Since 2016, WHO guidelines have focused on preventing SSI in patients of any age undergoing any surgical procedure. The forefront of these measures is antimicrobial prophylaxis. Nevertheless, given their generality, these measures would pose problems. For example, applying antimicrobial-coated sutures before the surgery has now been used intensively in pre-operative patients without indications prophylactically or remedially. Empirically, the clinicians tend to extend the medication course or give special-use antibiotic agents for longer periods until discharge to prevent SSI. However, it would lead to possible side effects such as the emergence of resistant organisms with antibiotic exposure. 31
Under these circumstances, risk factor stratification strategies could be a useful evidence-based guide for identifying high-, mid-, and low-risk populations. Therefore, an accurate prediction model screening for potentially high-risk populations and intervention subjects is needed. Identifying such a set of criteria could provide a reference for precision medication and intervention in the clinic, preventing and reducing the occurrence of adverse drug reactions or antibiotic resistance. A major strength of the model developed here is that it is based on routine clinical and laboratory parameters and no special or specific test is required. Reliable sources from examination and medication, compared with a retrospective investigation, could provide much more efficient and accurate predictors for real-time prediction in clinical practice. For example, the authors selected “medication for diabetes” as an indicator for diabetes mellitus.
Surgical site infection can significantly affect patient outcomes, but no risk factor stratification strategies are available specifically for SSI in patients with lung neoplasms undergoing MIS. Therefore, this study aimed to develop a risk prediction model for SSI after MIS for lung neoplasm. Predictors of the model were based mostly on inspection and medication information. The present study suggested that a model based on male gender, age >60 years, operation time >60 minutes, treatments for diabetes mellitus, and best FEV1/FVC were independently associated with SSI (AUC = 0.813). The traditional risk factors for SSI included advanced age, diabetes mellitus, and operation time 32 support the present study. Indeed, advanced age is associated with comorbidities, frailty, decreased immune functions, and decreased repair functions, all of which can contribute to SSI development.33,34
Diabetes mellitus is a condition associated with hyperglycemia, low-grade inflammation, white blood cell dysfunction, and increased risk of infections in general, although the exact mechanisms remain unclear.35,36 Operation time is usually associated with more complex procedures and a higher surgical trauma. 37 On the other hand, there are still debates on the association between gender and post-operative infection,38–40 and the differences in risks might depend upon the type of surgery. 38 Interestingly, the present study suggested that decreased lung function (FEV1/FVC) was a risk factor of post-operative infection. Despite the absence of previous findings supporting these results, we speculate that the respiratory functions of the patients could reflect their disease severity, which could influence the post-operative outcome of the patients.
There were several limitations to this study. First, the study was a case control design because of the low rate of SSI among MIS, leading to missing some irregular examination data such as diffusing capacity of the lung for carbon monoxide (D
Conclusions
In conclusion, this study explored factors associated with SSI in patients with lung neoplasm undergoing MIS. A prediction model was established, based on male gender, age >60 years, operation time >60 minutes, treatments for diabetes mellitus, and best FEV1/FVC, of which the AUC was 0.813. A prospective cohort study is ongoing to validate the prediction model.
Footnotes
Authors' Contributions
Study concepts: Yuejia Cheng, Yong Chen, Hou. Study design: Hou. Data acquisition: Yuejia Cheng, Yong Chen, Wen. Data analysis and interpretation: Yong Chen. Manuscript preparation: Yuejia Cheng, Yong Chen. Manuscript review: Hou. Manuscript editing: Yu, Wen. Quality control of data and algorithms: Yu. Formal analysis: Dai. Supervision: Dai, Zheng. Validation: Zheng.
Funding Information
This study was funded by Xuhui District Artificial Intelligence Medical Hospital Local Cooperation Project of 2021 (2021-010); Shanghai Shenkang Hospital Development Center clinical Science and Technology Innovation Project of 2020 (SHDC22020202); Shanghai Shenkang Hospital Development Center Medical Quality safety and Medical service project of 2021 (SHDC12021624).
Author Disclosure Statement
All authors declare that they have no competing interests.
