Abstract
Abstract
Background:
A systematic literature review (SLR) and meta-analysis of surgical site infections (SSIs) after surgical incision closure with triclosan-coated sutures (TS) compared with non-antibacterial coated sutures (NTS) published previously by the authors suggested that fewer SSIs occurred in the TS study arm. However, the results were vulnerable to the removal of one key randomized controlled trial (RCT) because of insufficient data. Furthermore, recently published RCTs highlighted the need for an update of the SLR to challenge the robustness of results.
Methods:
The protocol for the new SLR included more stringent tests of robustness than used initially and the meta-analysis was updated with the results of two new RCTs as well as the count of patients and SSIs by U.S. Centers for Disease Control and Prevention (CDC) incision class.
Results:
The updated SLR included 15 RCTs with 4,800 patients. No publication bias was suggested in the analysis. The predominant effect estimated a relative risk of 0.67 (95% CI: 0.54–0.84, p=0.00053) with an overall lower frequency of SSI in the TS arm than in the NTS arm. Results were robust to sensitivity analysis.
Conclusions:
The two additional peer-reviewed double-blind RCTs of this update confirmed the predominant effect found in the authors' previous meta-analysis and established the robustness of conclusions that were lacking previously. This SLR and meta-analysis showed that the use of triclosan antimicrobial sutures reduced the incidence of SSI after clean, clean-contaminated, and contaminated surgery. The two additional peer-reviewed double blind RCTs reinforced the evidence level of this SLR (CEBM level 1a).
T
Tests and clinical trials have also shown that surgeons cannot differentiate the presence or absence of triclosan in braided or monofilament sutures, making it possible to design randomized double-blind trials for clinical comparison.[9,10] Several randomized, controlled clinical trials (RCTs) have compared the frequency of SSIs after closure of surgical incisions with triclosan-coated sutures (TS) compared with non-triclosan sutures (NTS) in different clinical settings, and surgical procedures, to ensure the comparability of study arms, diagnostic criteria, suture materials, patient demographics, background diseases, and surgical operations. These clinical trials are the subject of this updated systematic literature review (SLR) and meta-analysis.
An earlier SLR and meta-analysis, published by the authors of this current article, was conducted according to a predefined written protocol, and identified 13 peer-reviewed eligible RCTs that compared TS vs. NTS in different clinical settings (different surgical operations, suture materials, underlying diseases, concentration of incision contamination, and methods to assess SSI occurrence) [9,11–23]. The result was a lower risk of SSI in the TS arm with a point estimate relative risk (RR) of 0.69, and a 95% confidence interval (CI) of 0.52–0.92 (p<0.011). Sensitivity analysis showed that this result was vulnerable to the removal of one trial, resulting in a borderline p-value, suggesting that the conclusions of the meta-analysis depended on that trial alone [16]. This article reports an update of the prior SLR and meta-analysis with inclusion of new clinical trials as well as additional information about the study methods and patient characteristics communicated by the authors of the previously reported eligible trials. The primary objective was to determine if incision closure presented the same risk of SSI when triclosan was present or absent on the surface of the sutures. The secondary objectives were to assess potential bias or confounding factors that could invalidate the primary conclusion and to determine the extent to which the primary conclusion could be generalized to the various types of patients included in the pooled RCTs.
Materials and Methods
The SLR protocol of the first meta-analysis reported previously was developed according to PRISMA recommendations and is repeated in the current communication with a number of differences described below.[24]
Study selection
Embase/Medline, the Cochrane database (Central Register of Controlled Trials [CENTRAL], Cochrane Database of Systematic Reviews [CDRS], Health Economic Evaluations Database [HEED], and Database of Health Technology Assessments [HTA]) as well as www.clinicaltrials.gov were searched on July 30, 2013 using their own search engines with a syntax that combined the following keywords: Triclosan AND sutur* AND (random* OR RCT). Clinicaltrials.gov was searched to identify potentially completed relevant RCTs. The “*” character at the end of keyword radicals indicated to search engines to include any character sequence that ended the keyword. This syntax differed from the previous meta-analysis by not searching broader keyword combinations that had proved to retrieve only non-randomized studies or irrelevant publications.
Study eligibility
Study eligibility criteria were the same as reported previously [11]. The results of the clinicaltrials.gov search and the reference lists of all relevant publications were meticulously searched to “catch back” any potentially eligible RCTs that might have been missed by the electronic study extraction and filtering process.
Data extraction
Data extraction was performed according to the same rules as previously described [11]. The four data items extracted from each study were the number of patients in the TS group (N_TS) with the number of patients presenting an SSI in the TS arm (n_TS), and their counterparts in the NTS arm (N_NTS, n_NTS).
Whenever a study reported outcomes on both an intention-to-treat (ITT) basis and a per-protocol (PP) basis, ITT results were used. Whenever a study reported outcomes PP only, but indicated the number of randomized patients together with demographics on an ITT basis, the PP sample size was replaced by ITT sample size.
In the case of RCTs reporting outcomes of the closure of several incisions per patient, only outcomes of the incision defined with the primary endpoint were included. For example, when the primary endpoint was the occurrence of SSI in the closure of a leg incision, after venous graft harvesting for coronary artery bypass surgery (CABG), outcomes of chest closure in the same patient were reported as a secondary endpoint. Trials where the same patient was systematically used in the active arm and the control arm were excluded as these trials were not “head-to-head” comparisons and did not provide independence of groups.
Compared with the previous protocol, the extraction also recorded the blinding method (double-blind, single-blind, open-label), the SSI diagnostic method used (U.S. Center for Disease Control and Prevention [CDC] criteria or other) as well as the number of patients with and without SSI per treatment arm broken down by class of incision contamination (class I/clean, class II/clean-contaminated, class III/contaminated, class IV/dirty-infected) [25,26]. Whenever these data were not reported in the publications and could not be deducted from the study context, the reviewers contacted the corresponding authors of the publications to obtain the missing information.
Quality of evidence
As performed with the RCTs included in the previous SLR, newly published studies were tested against the eligibility criteria defined in this SLR, against the concentration of evidence criteria proposed by the Centre for Evidence-Based Medicine (CEBM) at the University of Oxford, and the Cochrane criteria for quality and low risk of bias [27,28]. Full publications of all new RCTs were acquired and reviewed.
Statistical analysis
The data extracted from eligible RCTs was analyzed using meta-analytic techniques as previously, and the same CMA software (Comprehensive Meta-Analysis v2.2.027, Englewood, NJ) and reproducibility of calculations was checked by running an auditable script in STATA 12 (StataCorp LP, College Station, TX, USA). A rigorous 13-step analytical strategy was formulated (Fig. 1) and implemented in order to include the additional questions to be compared to the meta-analysis. Subgroup meta-analysis was considered to be powered sufficiently if it pooled a minimum of five estimates. Similarly, the comparison of subgroups was considered powered sufficiently if all compared subgroups pooled a minimum of five estimates. Subgroups that pooled four estimates were considered only to describe the available evidence and to assess its internal validity, but not for generalization of conclusions.

Flowchart of the meta-analysis.
Step 1
Calculated the risk ratio (RR) as the measure of effect between treatment arms in each RCT. RR=(n_TS/N_TS)/(n_NTS/N_NTS), where in each treatment arm, n is the number of patients with SSI and N is the number patients treated.
Step 2
Tested the null hypothesis (H0) of absence of publication bias. H0 was rejected if the Egger intercept test (significant if p<0.05) detected an asymmetric inverted funnel shape in the funnel plot [29–31].
Step 3
Tested the null hypothesis (H0) of no heterogeneity between trials, meaning that the RRs of all RCTs were drawn from a common population with the same true mean. H0 was rejected if the Cochran Q-test was significant (if p<0.05) [32]. The I2 estimated the percentage of variability of study RRs that could not be explained by random error only [33,34]. Given differences between trials in terms of methods and clinical settings (blinding, diagnostic criteria, compared suture materials, types of operations, CDC incision classes, and site/organ location), the algorithm temporarily maintained the assumption of heterogeneity between study populations until all these factors would be ruled out further in the analysis.
Step 4
Tested the effect between treatment arms across studies by calculating the random effects pooled RR (weighted average) [35,36]. This estimated the predominant effect among the populations from which the RCTs were drawn but not the true mean RR of any study population in particular. Under the null hypothesis (H0), the pooled RR is one, meaning the same frequency of SSIs between treatment arms. H0 would be rejected in favor of the alternative that the predominant effect was significant if the pooled RR's 95% CI included one.
Step 5
Sensitivity analysis of the robustness of meta-analysis was performed on all eligible RCTs. This consisted of repeating steps two, three, and four iteratively after removing one RCT at a time, then two RCTs at a time (any pair of RCTs), then three RCTs at a time, and so on, until 95% CI of the pooled RR reached one, thus non-significance. This process enabled identification of the RCTs upon which depended significance.
Step 6
Assessed the potential bias in the predominant effect potentially caused by differences in blinding method between RCTs (double-blind, single-blind, open-label). The pooled random effects RR and 95% CIs of each blinding subgroup with more than four individual RRs were calculated. The null hypothesis (H0), that subgroups shared the same population mean RR, was rejected if p<0.05, in favor of the alternative where subgroups had heterogeneous population means. Sensitivity analysis of robustness was planned similarly to step five in the double-blinded subgroup if the subgroup RR was significant and pooled more than four individual RRs.
Step 7
Assessed the potential bias in the predominant effect potentially caused by differences in SSI diagnostic method between RCTs (CDC criteria, other methods). The process was similar to step six but no sensitivity analysis was planned.
Step 8
Assessed potential bias in the predominant effect caused by differences in compared suture materials between RCTs (polyglactin 910, polydioxanone, poliglecaprone, combinations of various materials, silk sutures) as performed [25]. The process was similar to step six with sensitivity analysis.
Step 9
Assessed potential bias in the predominant effect caused by differences in CDC incision class reported either between RCTs or class of contamination. The process was similar to step six with sensitivity analysis.
Step 10
Assessment of the potential bias in the predominant effect caused by differences in incision depth or site, between RCTs or within RCTs, was planned if reviewers were able to extract that information from publications. Incisional and organ/space surgical site definitions were those proposed by the CDC [37]. The process was similar to step six with sensitivity analysis.
Step 11
Assessed potential bias in the predominant effect caused by differences in operation type as reported by RCT authors. The process was similar to step six with sensitivity analysis.
Step 12
Assessed the potentially confounding relationship between the RR and the SSI incidence rate (IR) in each RCT. Incidence rate is an outcome computed with the same inputs as RR but examines the frequency of SSI as an average, ignoring differences between treatment arms: IR=(n_TS+n_NTS)/(N_TS+N_NTS). In order to enable a linear comparison of RR and IR, the Log(RR) of each RCT was plotted against the corresponding RCT's IR in a meta-regression. The null hypothesis (H0) that RR was independent of IR was tested by testing the slope of Log(RR) as a function of IR. The alternative hypothesis that RR did vary with IR was accepted if that slope was (i.e. p<0.05) different from 0.
Step 13
Conclusion: If previous steps showed that the overall random-effects pooled RR was robust, and that RR did not dependent on the IR, four conclusion options were possible:
i. If the pooled RR of the double-blind RCTs subgroup was robust AND if no heterogeneity was found among all RRs, AND all tested potential bias/heterogeneity factors were ruled out: The conclusion was a similar treatment effect across study populations probably caused by triclosan. ii. If the pooled RR of the double-blind RCTs subgroup was robust BUT heterogeneity was found among all RRs, OR any tested potential bias/heterogeneity factors was not ruled out: The conclusion was a significant but parameter treatment effect across study populations probably caused by triclosan. iii. If the pooled RR of the double-blind RCTs subgroup was not robust BUT no heterogeneity was found among all RRs, AND all tested potential bias/heterogeneity factors were ruled out: The conclusion was a similar treatment effect across study populations but an uncertain role of triclosan in the effect. iv. If the pooled RR of the double-blind RCTs subgroup was robust AND if heterogeneity was found among all RRs, OR if any tested potential bias/heterogeneity factors was not ruled out: The conclusion was a parameter treatment effect across study populations and an uncertain role of triclosan in the effect.
Results
Seventy-six references were identified and one additional reference was caught back [40]. Forty-three duplicate references were removed, 34 references were screened and 15 were confirmed eligible RCTs (Fig. 2). The eligible trials included the 13 RCTs from the previous meta-analysis and two double-blind prospective RCTs published later [11,38,39]. The new RCTs met all eligibility criteria of this SLR protocol as well as CEBM and Cochrane criteria for quality and low risk of bias [27,28]. The 19 ineligible references included four abstracts of non-peer reviewed RCTs [40–43]. The eligibility of those four abstracts could not be determined because of insufficient methods descriptions or incomplete reporting of results reporting.
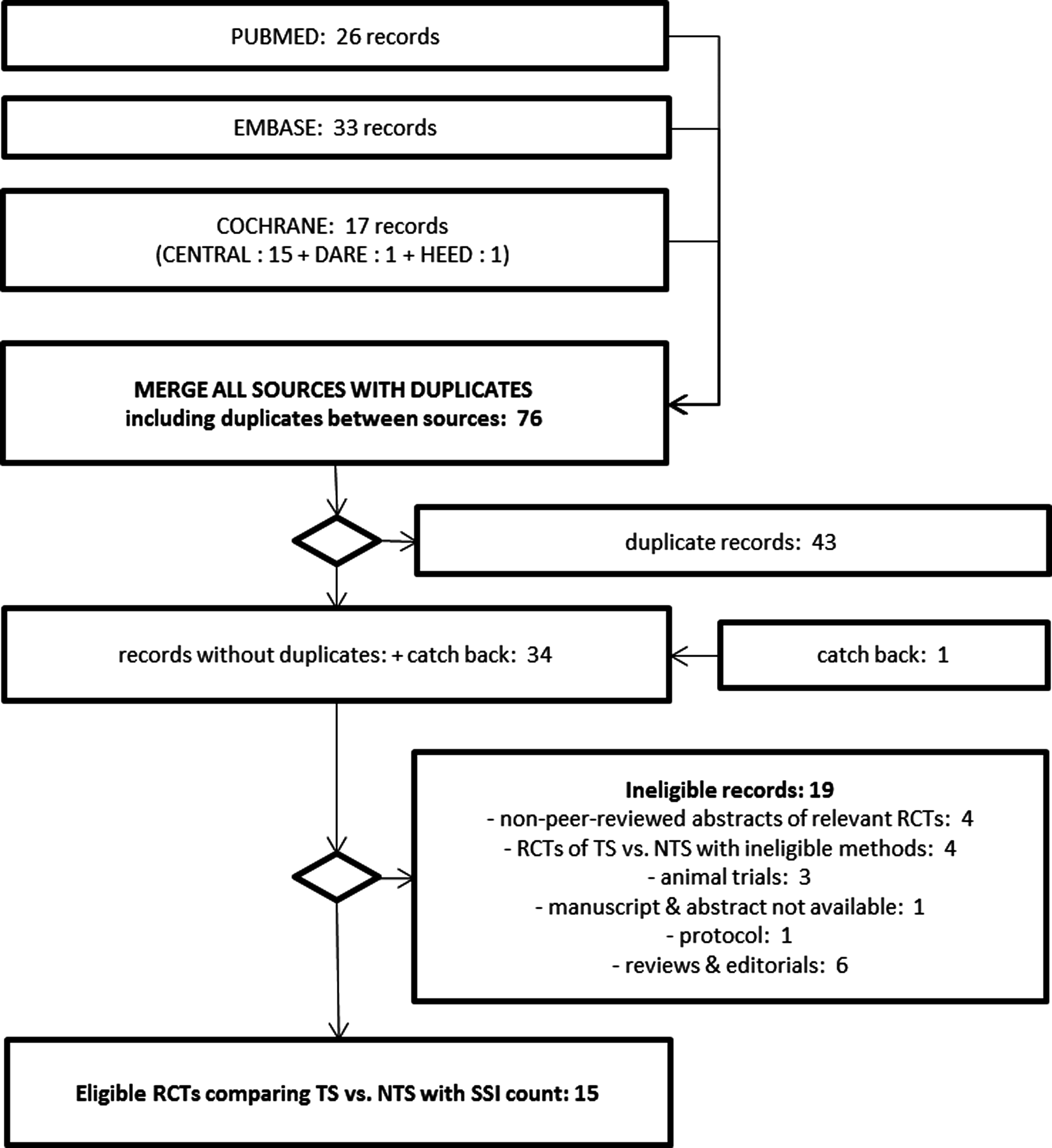
PRISMA diagram of study extraction and eligibility selection.
The SSI diagnostic method used and CDC incision class were reported in four RCTs and CDC incision class could be deducted as clean from the type of surgery in two others. Ten authors were contacted to obtain or confirm the count of patients with and without SSI by CDC class and the confirmation of the SSI diagnostic method when these were not specified in the publication. Seven authors responded, two of whom also added that SSI had been diagnosed using CDC criteria. The lead author of one trial on colorectal surgery, who did not use the CDC criteria, responded that all cases were clean or clean-contaminated so the reviewers assumed that all cases were clean-contaminated, as in other colorectal trials [17]. Three authors did not respond. Altogether outcomes were available by class of incision contamination in 12 RCTs. Table 1 summarizes the characteristics of the eligible RCTs obtained from publications and authors directly. Overall, the 15 RCTs enabled the extraction of outcomes in 4,800 patients; (TS: n=2,323 and NTS: n=2,477), with head-to-head comparisons of a single incision per patient. There were 453 patients who had an SSI (TS: n=180 and NTS: n=273). The 13-step analysis revealed the following:
RCT=randomized controlled trial; TS=triclosan-coated sutures; NTS=non-triclosan-coated sutures; n=number of patients, SSI=surgical site infections; ITT=intention-to-treat; PP=per-protocol; CDC=U.S. Centers for Disease Control and Prevention; CSF=cerebrospinal fluid; CABG=coronary artery bypass grafting.
The 15 trial RRs and their 95% CIs were calculated with data reported (Table 1) on an intention to treat (ITT) basis in nine RCTs, with data reported per protocol (PP) in four RCTs, and with the number of SSIs per protocol and the number of randomized patients according to ITT methods in two RCTs (
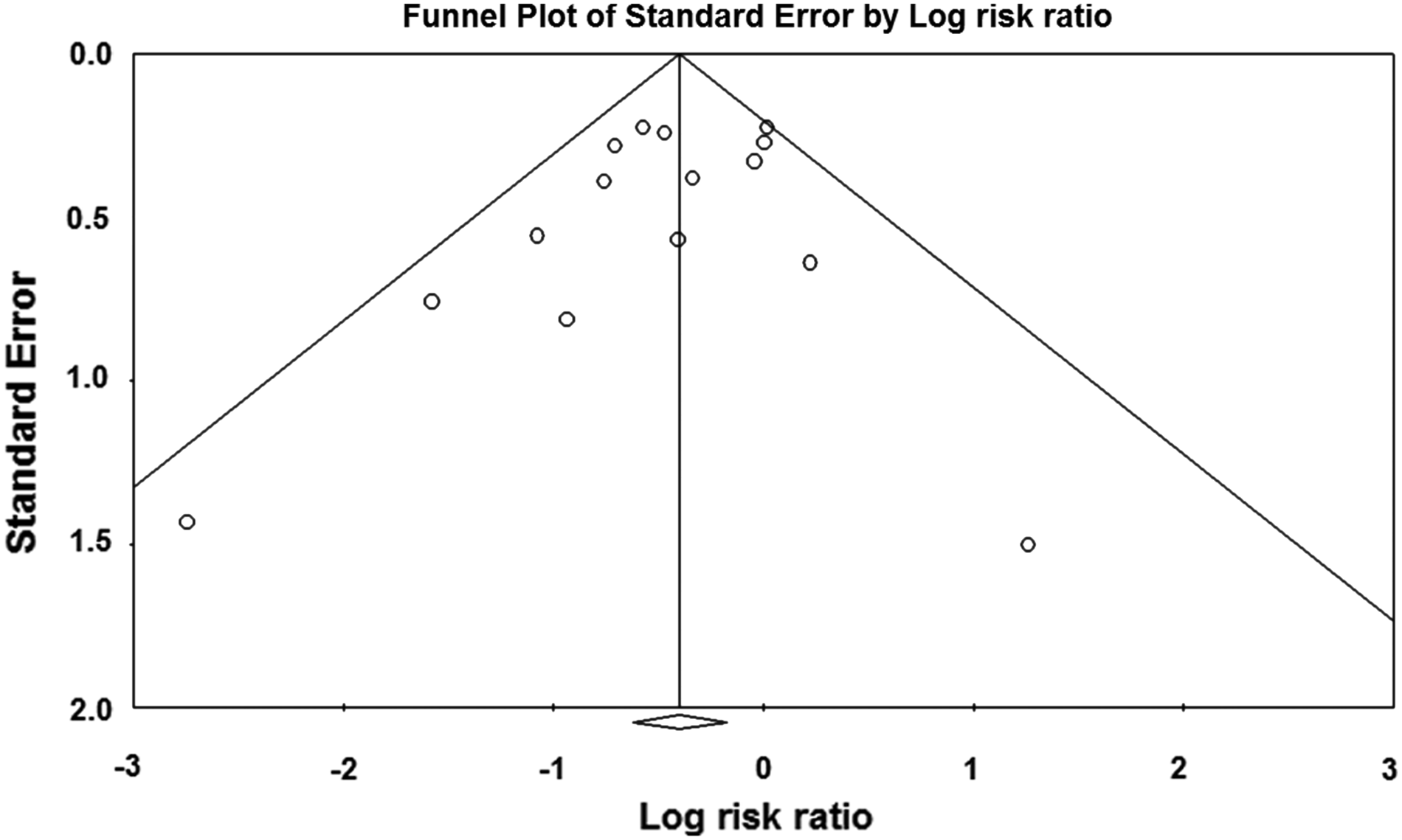
Funnel plot of the risk ratios of eligible RCTs.
The overall pooled RR was 0.67, 95% CI 0.54–0.84, p=0.00053 (
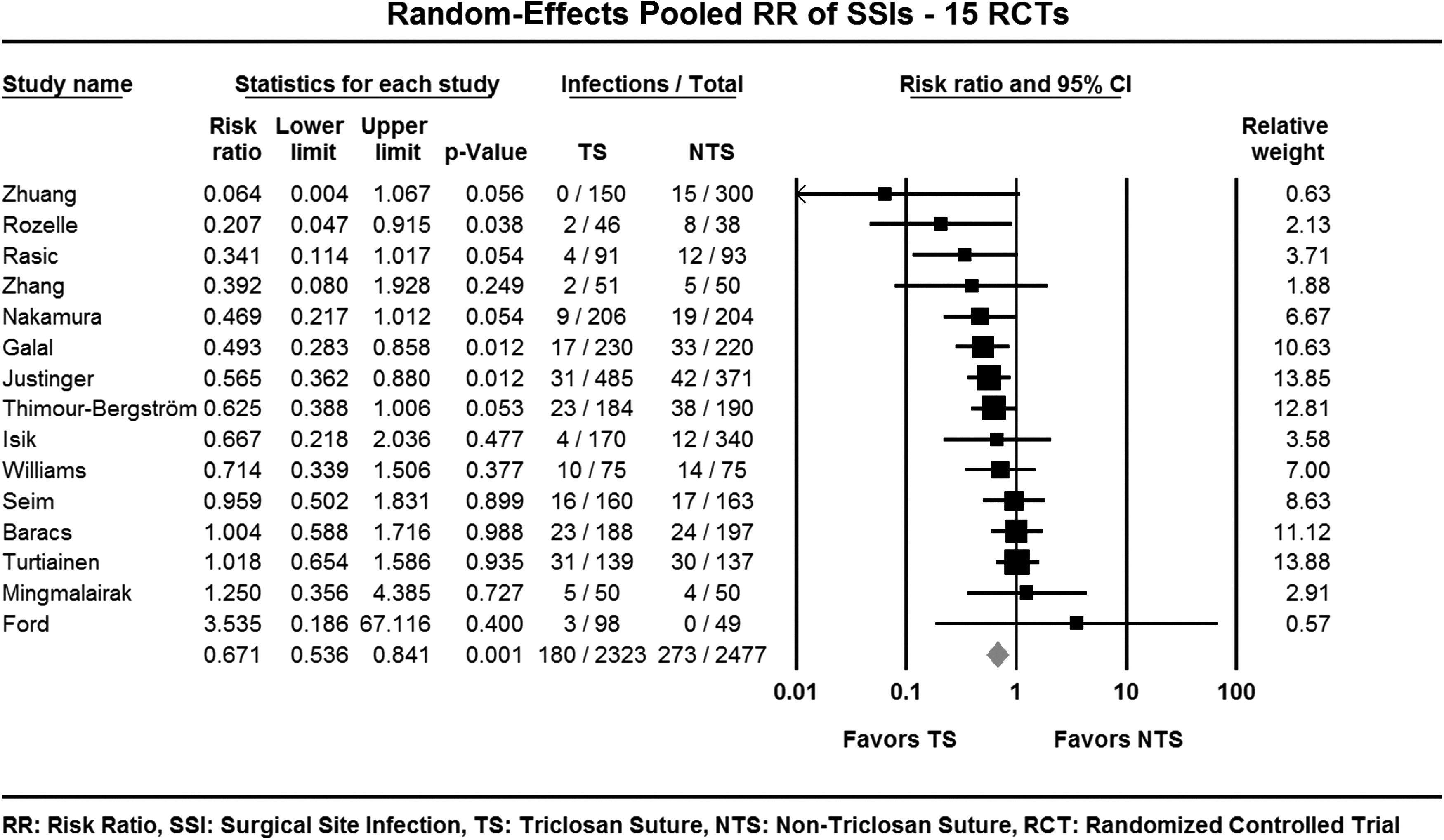
Forest plot of the risk ratios–Overall.
RR=risk ratio; CI=confidence interval; LL=lower limit; UL=upper limit; dF=degrees of freedom; TS=triclosan sutures; NTS=non-triclosan sutures; N=number of patients; SSI=surgical site infections; IR=incidence rate.
Pooled RR by blinding subgroup (
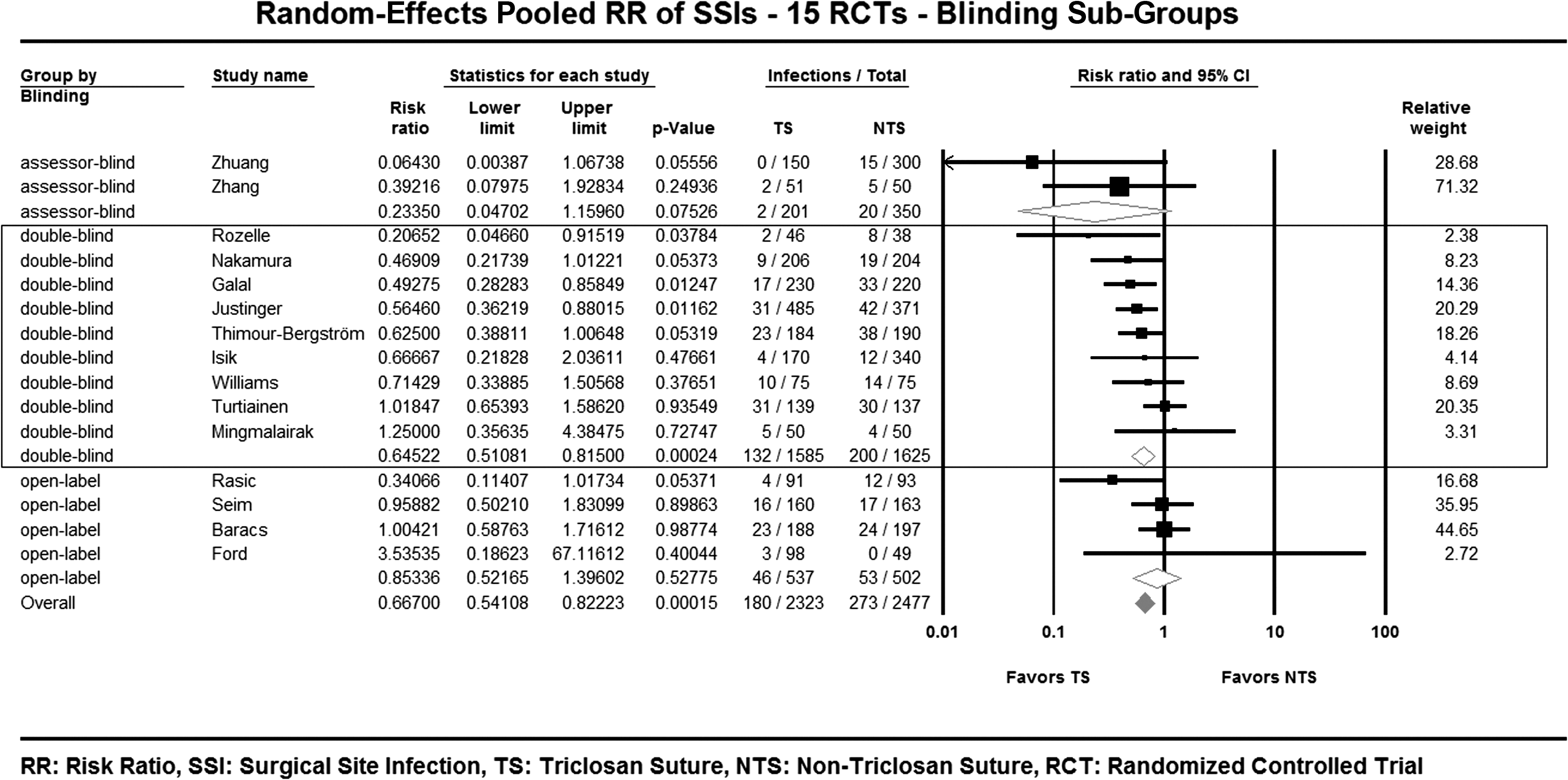
Forest plot of risk ratios–Blinding subgroup.
RR=risk ratio; CI=confidence interval; LL=lower limit; UL=upper limit; dF=degrees of freedom; TS=triclosan sutures; NTS=non-triclosan sutures; N=number of patients; SSI=surgical site infections; IR=incidence rate.
Pooled RR by diagnostic method subgroup (
Pooled RR by suture material (
Pooled RR by incision contamination (
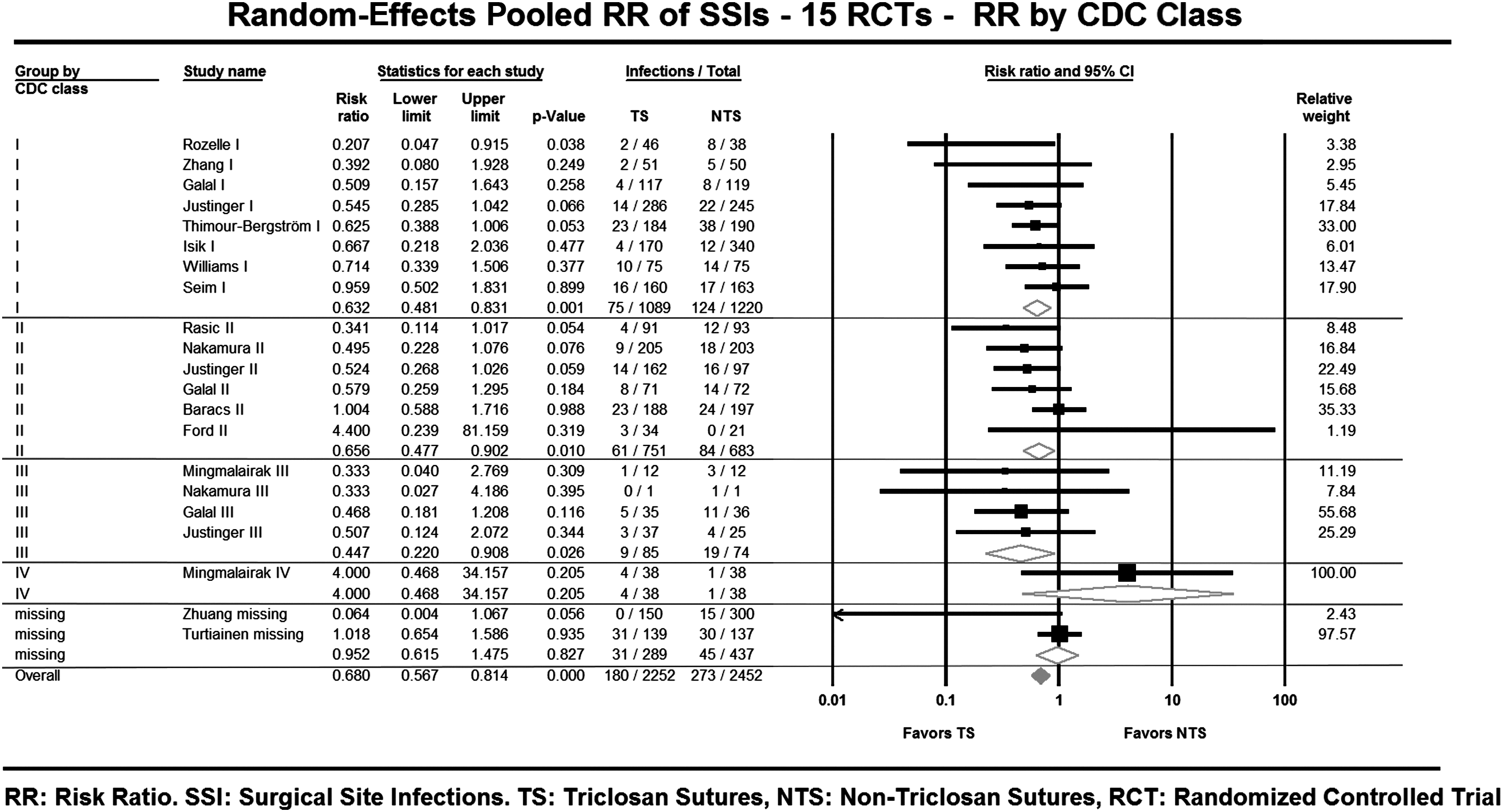
Forest plot of risk ratios–CDC incision class subgroup
Pooled RR by incision depth and organ/space infection (
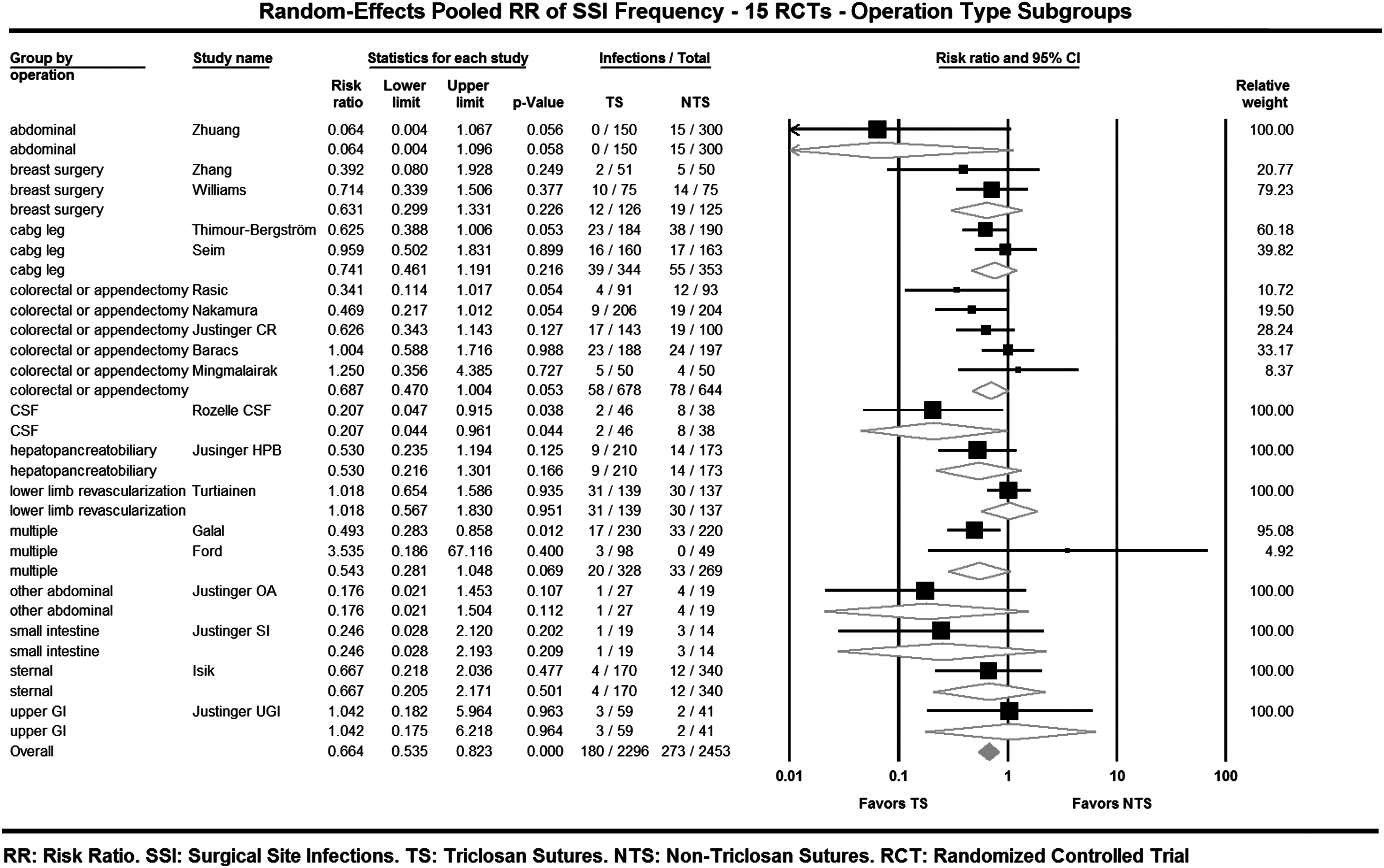
Forest plot of risk ratios–Operation type subgroup.
The heterogeneity of average incidence rate (IR) of SSI in treatment arms described in step three was visible on the Forrest plot (Fig. 8). The meta-regression was conducted using the mixed effects method of moments on the 15 RCTs with the IR as independent parameter and Log(RR) as dependent parameter. The regression slope was 3.35, 95% CI −0.60–7.31 (p=0.096), thus non-significant (Fig. 9). The null hypothesis that the RR is independent of IR was therefore not rejected (
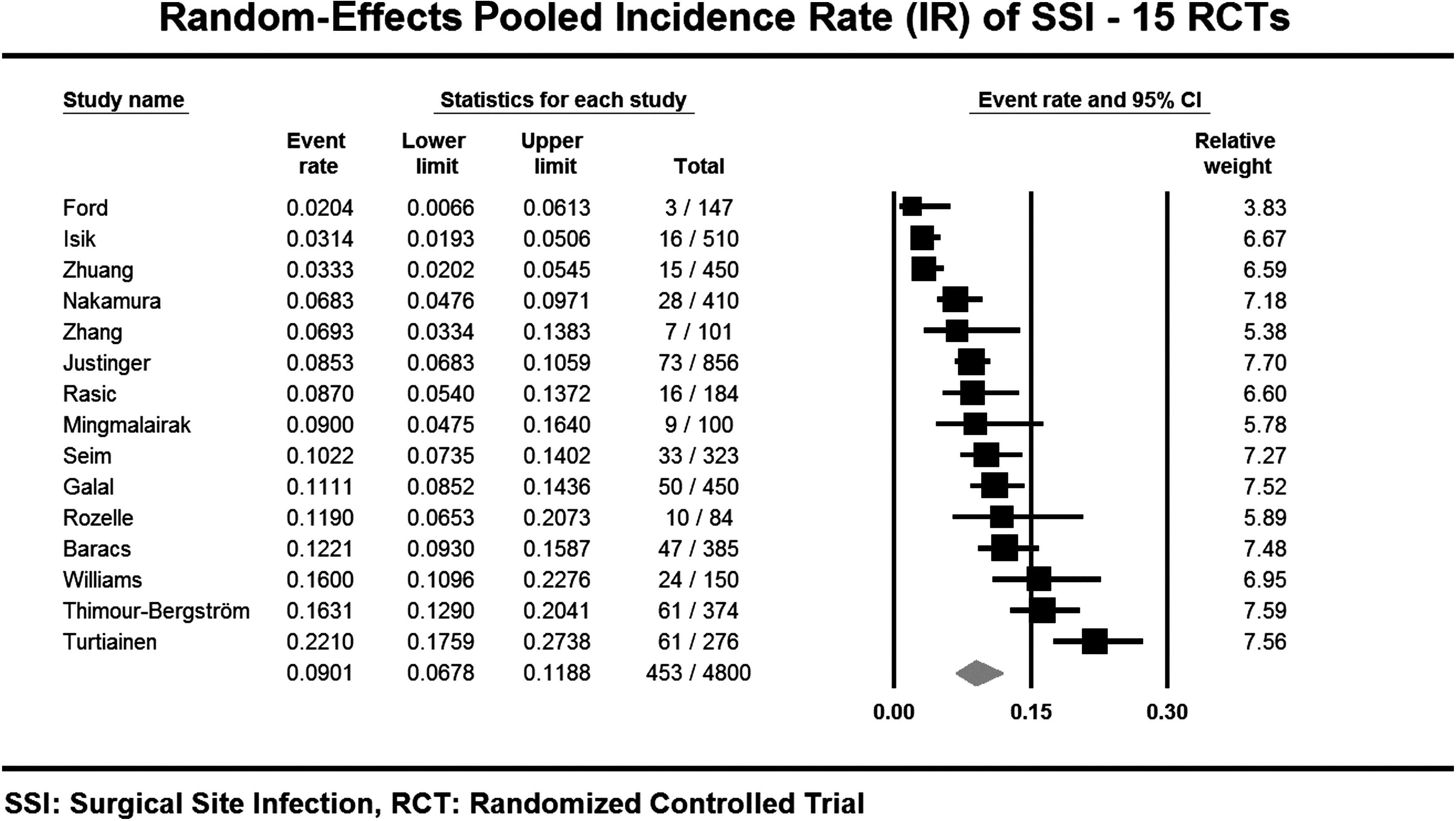
Forest plot of the incidence rates of SSI–Overall.
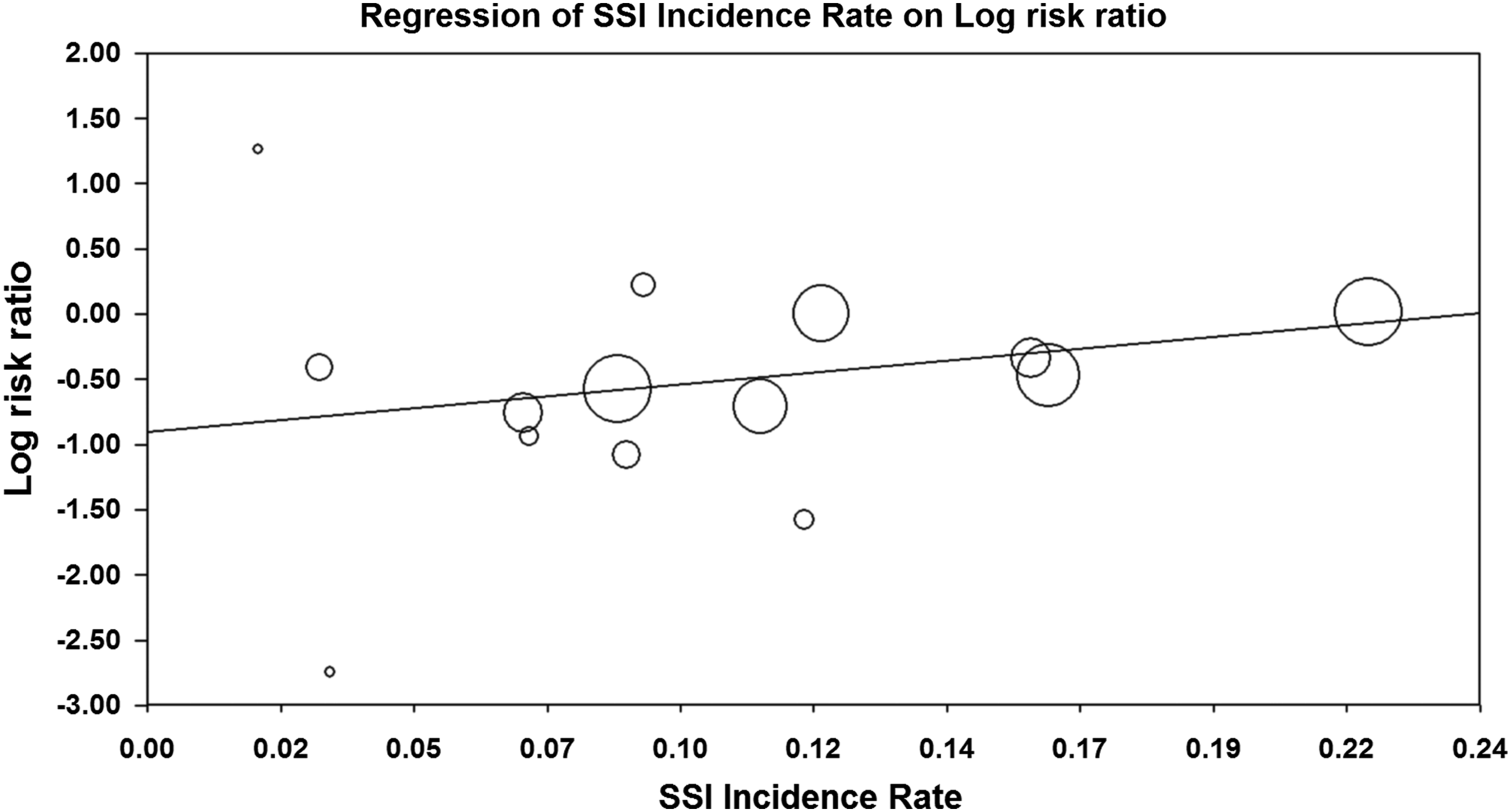
Meta-regression of (Log) risk ratio depending on incidence rate.
Given the above results, it was concluded that results were significant with robust conclusions, and that triclosan was the only known cause of difference in SSI frequency between treatment arms (
Discussion
Internal validity
The two newly included trials increased the number of double-blind RCTs to nine studies compared with seven in the previous meta-analysis [11]. The double-blind RCTs, being designed to ensure equipoise of the patient sample and comparability of study arms from baseline through follow-up completion (given TS and NTS made of the same material), could not be distinguished. All double-blind RCTs used CDC criteria of SSI except the Rozelle trial, which used microbiologic culture proof of shunt infection [12]. The core of double-blind studies yielded a highly robust predominant effect, with a small proportion of heterogeneity (I2 close to 0), no indicator of publication bias and robustness of results to the removal of up to three RCTs.
Certain potential heterogeneity factors that could have affected the pooled RR were not ruled out owing to an insufficient number of estimates per subgroup. Such was the case with suture material and operation type. However, the proportion of heterogeneity among the nine double-blind RCTs was close to zero (percentage calculated using the I2 statistic). The incidence rate of SSI, which is the expression of the differences in risk through the trials' clinical settings, was ruled out as factor of bias in the double-blind RCTs below outlier IR of 22% and in the 15 RCTs including the outlier [20]. Finally, the pooled RR was significant throughout three incision classes, clean, clean-contaminated, and contaminated classes, both among the double-blind RCTs and all 15 RCTs, which included approximately 80% of enrolled patients. This distribution of a predominant effect throughout the incision contamination class case-mix indicated that the overall predominant effect was not biased by that case-mix. Therefore, the core of double-blind RCTs enabled this updated SLR to meet the many rigorous criteria of internal validity that were not met by previous SLRs on this topic. In addition, no indicator suggested that the conclusions of double-blind RCTs should not apply to the entire set of 15 RCTs [11,45,46].
The results of this analysis suggest strongly that the presence of triclosan-coated sutures within the surgical incision bed appears to be the predominant factor associated with a reduction in the incidence of SSIs reported in the 15 RCTs included in this meta-analysis. Given the insufficient number of trials that included the size of effect according to operation type, definition of SSI incisional and organ/space sites, it would be prudent to continue to report the predominant effect size obtained with a random-effects model, to account for potentially underestimated heterogeneity across study populations.
The two trials where RR was calculated with the number of SSIs reported per protocol and the number of randomized patients for whom demographics were also reported, according to ITT method avoided decreasing the sample size by 11.9% (12 in 101 patients) yielding an RR of 0.39 ITT instead of 0.37 PP in the Zhang trial, and avoided decreasing the sample size by 15.3% (23 in 150 patients) yielding an RR of 0.71 ITT instead of 0.66 PP in the Williams trial.[15,18] This approach thus maintained sample size without overestimating treatment effect.
External validity
The applicability of these pooled results to future patient care depends on the similarity of the case-mix of future surgical patient populations based upon the case-mix reported in the 15 RCTs, where case-mix is defined in terms of incision contamination class. External validity should be quantifiable when examining patients with incisions classified as clean, clean-contaminated, and contaminated, with high confidence that the risk reduction of SSIs found in clean incisions should apply, given the robustness of that subgroup but with a lower confidence for clean-contaminated and contaminated incisions. External validity could not be established for the effect of triclosan coated sutures to close dirty incisions or for operations where incision contamination was not described.
Conclusion
This systematic literature review (SLR) and meta-analysis identified 15 randomized controlled trials and produced a risk ratio of 0.67, 95% CI 0.54–0.84 (p=<0.00053), demonstrating a highly statistically significant, lower risk of SSI following operative procedures in incisions which were closed with triclosan-coated sutures compared to non-antimicrobial closure technology. This result was robust to the removal of up to three trials. Similar results, yielded by the core of nine double-blind randomized controlled trials, supported the hypothesis that triclosan coated sutures were responsible for the reported reduction in SSI.
Trials comparing triclosan polyglactin to polyglactin demonstrated a robust risk ratio in favor of triclosan. Subgroup analysis was underpowered to determine heterogeneity between studies comparing different suture materials.
Diagnostic method was ruled out as a cause of bias and the incidence of SSIs, which ranged between 2% and 22%, had no significant effect on risk reduction effect of the triclosan coated sutures and was therefore ruled out as a cause of bias. Based upon this analysis there was a high concentration of confidence that a 20% to 50% reduction in SSIs should be expected in surgical procedures involving clean surgical incisions. A smaller and statistically significant reduction could be expected in clean-contaminated and contaminated incisions but these results were not robust when considered separately from the clean incisions. No conclusions could be drawn based upon this analysis on the impact of triclosan sutures as a risk reduction strategy for SSIs involving dirty incisions or surgical procedures where the composite incidence rate of infections exceeded 17%. The two additional peer-reviewed double-blind RCTs reinforced the evidence level of this SLR (CEBM level Ia).
Footnotes
Acknowledgments
FCD, CEE, and DJL thank the authors of the compiled studies who responded to our request for additional information: Henri R. Ford, MD (University of Southern California Keck School of Medicine, USA); Chatchai Mingmalairak, MD (Thammasat University Hospital, Pathumthani, Thailand); Jozsef Baracs, MD (University of Pecs, Pecs, Hungary); Toru Nakamura, MD (Graduate School of Medicine, Sapporo, Hokkaido, Japan); Anders Jeppsson, MD, on behalf of Thimour-Bergström L. et al. (Sahlgrenska University Hospital, Gothenburg, Sweden); Sahin Sahey, MD on behalf of Isik I. et al., (Marmara University, Istanbul, Turkey); Marko Sever, MD, on behalf of Rasic Z. et al. (University Hospital Sveti Duh, Zagreb, Croatia); and David Leaper, MD, FACS,(Imperial College, London, UK) on behalf of Williams et al.; who were co-authors of the studies included in the meta-analysis.
Author Disclosure Statement
This systematic analysis was conducted independent of any external funding support from non-profit or for-profit entities. The design, execution, analysis, and conclusions of this analysis are the sole responsibility of authors Daoud, Edmiston, and Leaper.
