Abstract
Abstract
Background:
The aim of this study was to characterize the elution of four antibiotics from pharmaceutical-grade calcium sulfate beads and show that the eluted antibiotics retained efficacy.
Methods:
Calcium sulfate was combined with gentamicin, tobramycin, vancomycin, or rifampicin (ratio: 20 g of calcium sulfate, to 240 mg, 500 mg, 900 mg, and 600 mg of antibiotic, respectively). Three grams of beads were immersed in 4 mL of sterile phosphate-buffered saline (PBS) at 37°C. At each time point (4, 8, 24 h; 2, 7, 14, 28, 42 d), eluates were removed for analysis by liquid chromatography–mass spectrometry. The antimicrobial efficacy of antibiotics combined with calcium sulfate beads after 42 d was tested by a modified Kirby-Bauer disc diffusion assay.
Results:
All samples showed a generally exponential decay in the eluted antibiotic concentration. At the first time point, both gentamicin and tobramycin had eluted to a peak concentration of approximately 10,000 mcg/mL. For rifampicin, the peak concentration occurred at 24 h, whereas for vancomycin, it occurred at 48 h. The eluted concentrations exceeded the minimum inhibitory concentration for common periprosthetic joint infection pathogens for the entire span of the 42 study days. Mass spectrometry confirmed all antibiotics were unchanged when eluted from the calcium sulfate carrier. Antimicrobial efficacy was unaltered after 42 d in combination with calcium sulfate at 37°C.
Conclusions:
Pharmaceutical-grade calcium sulfate has the potential for targeted local release of tobramycin, gentamicin, vancomycin, and rifampicin over a clinically meaningful time period.
A
One of the most adopted materials for local release of antibiotics is polymethylmethacrylate (PMMA) [14,15]. However, this material has a number of significant disadvantages. It is a dense, acrylic, and non-resorbing material, which generally must be removed in a second surgical procedure when its function has been fulfilled to avoid becoming a nidus for future infection [16]. Also, it can release subinhibitory concentrations of drugs over an extended implantation period, and bacteria adherent to PMMA cement thus can acquire resistance to the loaded antibiotic because of long exposure to the sub-inhibitory concentrations [17–19]. In addition, PMMA is not suitable for delivery of thermosensitive antibiotics because of the high temperatures generated during PMMA curing [20,21].
Fully bioabsorbable carriers are now the subject of extensive investigation [22–24]. One such material is calcium sulfate, with recent literature discussing its application in combination with antibiotics [16,25,26], including its safe and efficacious use in infected soft tissue sites [27].
There is a variety of data in the literature on the in vitro elution rates of various antibiotics from a number of carrier materials [23,28–38], including calcium sulfate [20,39–43]. Literature reports a large variation in the in vitro experimental methods used to determine antibiotic elution. Methods differ in the quantity of material tested, the nature by which the sample is presented to the solution (single small beads [20,42,43] and larger cast cylinders [40,41]), the volume and nature of the solution into which the sample is immersed, the volume removed for analysis, and the eluent sampling intervals. It is difficult to correlate in vitro with in vivo conditions.
When investigating the elution of tobramycin from calcium sulfate beads, McLaren et al. [44] considered the assumptions made when approximating surgical site conditions. Those authors estimated that many orthopedic surgical sites have a volume of 100 mL or less and are commonly filled with as much as 40 antibiotic-loaded calcium sulfate/antibiotic beads of unspecified size. Despite this observation, this in vitro elution study immersed only eight beads in 20 mL of buffered saline. In their discussion, the need for additional investigation to characterize elution from calcium sulfate during the first 24 h of implantation was noted.
The current study was designed to establish an experimental protocol that mimics more closely the clinical situation for the material–antibiotic combinations in order to provide elution data of greater clinical relevance. This protocol was then used to determine the elution profiles of four commonly used antibiotics, namely tobramycin, gentamicin, vancomycin, and rifampicin over a period of 42 d, the typical time period over which calcium sulfate beads would be resorbed fully in vivo [45,46].
Materials and Methods
Experimental design
The in vitro conditions were intended to mimic the implantation of calcium sulfate beads in a small bone defect. Two bead sizes were investigated. Beads were hemispherical with diameters of either 3 mm or 6 mm.
The volume fill of 3 g of 3-mm calcium sulfate beads is 3.4 mL, whereas the volume fill of 3 g of 6-mm calcium sulfate beads is 4.2 mL. The use of this quantity of beads for the elution experiment was comparable to the quantity of beads used clinically in small bone defects/voids of approximately 4 mL volume, a size comparable to that observed in osteomyelitis [25,47]. The addition of 4 mL of phosphate-buffered saline (PBS), pH 7.4, to the elution vessel enabled a sufficient volume of liquid for eluent collection whilst ensuring fluid coverage of the beads, mimicking a small bone void, with the limited volume available for both bead placement and subsequent filling of the interstitial space between the beads (e.g., blood, serous fluid, extracellular matrix).
Considering a dynamic surgical site environment, balancing bleeding and capillary leaking at the site with resorption with venous and lymphatic return, a partial exchange of eluent was used (1.5-mL daily exchange of a total volume of 4 mL) in an attempt to represent the antibiotic concentrations present in a relatively stable surgical site more accurately [44].
A concern when incorporating antibiotics into a delivery vehicle is the effect of the carrier material on the antibiotic and the potential for it to cause chemical changes to the antibiotic that may affect both its antimicrobial efficacy and the biological response. The elution concentrations in this study were determined using liquid chromatography–mass spectrometry (LC-MS), a powerful technique with high sensitivity and selectivity. An advantage of using this technique was that the identity of the eluted antibiotics could be confirmed in order to determine if there had been any change in the antibiotic chemistry. To identify any change in the antimicrobial properties of the antibiotics following combination with calcium sulfate over 42 d, the aged and non-aged beads were tested using a modified Kirby-Bauer disc diffusion assay [48–50].
Preparation of beads
Ten-milliliter kits containing 20 g of pharmaceutical-grade calcium sulfate alpha-hemihydrate powder (CSH; Stimulan®, Biocomposites Ltd, Keele, Staffordshire, United Kingdom) were used to prepare the antibiotic-impregnated beads; 20 g of CSH was mixed with 6 mL of gentamicin sulfate solution (40 mg/mL, Hospira UK Ltd., London, UK) for gentamicin-loaded beads; For the tobramycin-containing beads, 20 g of CSH was mixed with 500 mg of tobramycin sulfate powder (Gallipot Inc., now Fagron US, St. Paul, MN) and blended thoroughly prior to the addition of 6 mL of sterile water. For the vancomycin-containing beads, 20 g of CSH was mixed with 1 g of vancomycin hydrochloride powder (Hospira UK) and blended thoroughly prior to the addition of 6 mL of sterile water. For the beads containing rifampicin, 20 g of CSH was mixed with 600 mg of rifampicin powder (Sigma–Aldrich UK, Dorset, United Kingdom) and blended thoroughly prior to the addition of 6 mL of sterile water.
In each case, all components were mixed thoroughly for 30 to 60 sec to form a smooth paste, which was pressed into 3.0- or 6.0-mm diameter cavities in the flexible mold (Fig. 1A). The beads were left undisturbed for 30 to 60 min to hydrate and set according to the reaction:

Preparation of calcium sulfate beads using flexible mold.
When set hard, the beads were removed from the mold by flexing it (Fig. 1B). By using thorough mixing in the preparation of the beads, a homogenous mixture of antibiotic and CSH was achieved. Therefore, the concentration of antibiotic per weight of beads could be calculated (Table 1).
Calculated as the maximum achievable concentration of antibiotic when 3 g of prepared beads are placed in 4mL of phosphate-buffered saline.
Antibiotic elution from 3 g of beads
Three grams (±0.05 g) of each antibiotic and bead size combination (Table 1) was placed in sterile 7-mL tubes, to which 4 mL of sterile PBS was added. Tubes were incubated in the dark at 37°C. The time points chosen to determine elution were 4, 8, and 24 h and 2, 7, 14, 28, and 42 d. At each time point, three 0.5-mL samples of the PBS were removed for analysis by LC-MS. The same amount of fresh PBS was replaced and the tubes returned to the incubator.
The three individual eluate samples from each vial were frozen immediately at −20°C. All eluate samples were stored under these conditions until analyzed together at the end of the final elution time point of 42 d. Prior to LC-MS analysis, the eluates were defrosted on ice. The 0.5-mL aliquots were transferred to fresh tubes. These were centrifuged at 4°C, and then 0.2 mcm from each sample was syringe-filtered directly into glass autosampler vials to remove any particulate matter. The autosampler holding the vials was held at 4°C.
Antibiotic solutions prepared from the same stocks as those used to prepare the beads were used as standards for assay development and quantification by LC-MS. Quantification was carried out using a triple quadrupole (QQQ) mass spectrometer (MS) coupled to a 1200 series rapid-resolution high-performance liquid chromatography (HPLC) stack (Agilent Technologies, Microlease, Morrisville, NC). Depending on the concentration, samples were analyzed either neat or diluted to ensure values fell within the linear range of the standard curves. Concentrations were calculated from peak areas using standard curves developed from dilutions of antibiotic prepared to known concentrations.
The HPLC conditions were as follows: Buffer A was 5% acetonitrile (ACN), 0.1% formic acid and B was 95% ACN, 0.1% formic acid. The gradient was held with 0% B for 2 min before increasing linearly to 100% over 4 min. The 100% B was maintained for a further 2 min before rapid requilibration in 100% A. The flow rate was constant at 0.3 mL/min.
The gas temperature in the source was 325°C with a flow rate of 9 L/min. The nebulizer pressure was 35 pounds per square inch and the capillary voltage 3.5 kV. Fragmentor and collision energy voltages were optimized for each antibiotic and are listed in Table 2 along with the relevant multiple reaction-monitoring (MRM) values.
Quantification by LC-MS
Standard curves of each antibiotic were used to determine the limits of detection (LoD) and linear ranges of detection using peak areas. Test solutions were prepared for which the concentrations were unknown to the user. These were run twice, once directly and once spiked with a known concentration of the relevant antibiotic, ensuring peak areas fell within the linear range. This ascertained whether ion suppression would prevent quantification directly from a standard curve without further dilution. If there was no ion suppression, eluates were analyzed directly from a standard curve comprising a minimum of five concentrations. Otherwise, samples were diluted to obtain a concentration low enough to prevent this problem.
Antibiotic stability
In elution experiments for antibiotics combined with calcium sulfate published previously [20], samples were frozen for a maximum of 10 d before quantifying the antibiotic content so as to minimize the risk of the antibiotics degrading over time. As samples in this experiment were stored frozen for a maximum of 42 d, an antibiotic control was used, whereby a sample of the antibiotic solution used for LC-MS calibration and testing was kept aside and processed alongside the eluate samples (both incubation and freezing). The results were checked against the original standard curve to confirm the stability of the antibiotic. Therefore, tubes containing 4 mL of prepared antibiotics at the maximum concentration shown in Table 1 were incubated alongside the elution study samples.
Antibiotic efficacy
In order to determine the preservation of antimicrobial efficacy of the antibiotics in the beads over the 42-d period, samples of antibiotic-loaded beads were prepared as described above. These beads were tested using a modified Kirby-Bauer disc diffusion assay [48–50]. The control was beads either aged or non-aged and prepared without antibiotic. The beads were prepared either immediately prior to testing or stored for 42 d at 37°C in sealed sterile moisture-impermeable foil pouches prior to testing. Zone of inhibition (ZOI) testing of aged and non-aged beads containing the antibiotics and antimicrobial susceptibility testing discs (AST discs) (tobramycin 30 mcg, vancomycin 30 mcg, rifampicin 5 mcg, and gentamicin 200 mcg (Oxoid Ltd, a division of ThermoFisher Scientific, Basinstoke, Hamphire, United Kingdom) was performed in triplicate against Staphylococcus aureus American Type Culture Collection strain (ATCC) 6538 and S. epidermidis ATCC 12228. Tryptone soy agar (TSA) plates were seeded (0.2 mL) with a suspension of the relevant organism containing approximately 108 colony-forming units [CFU]/mL. The plates were transferred to an incubator operating at 35±2°C for 30 min. The plates were removed from the incubator and the beads or discs placed on the surface. The plates were then incubated at 35±2°C for 24 h, after which time, they were removed from the incubator and examined for any clear zones around the test sample. Zones were measured from the edge of the bead or control disc to the zone perimeter, calculating the total zone diameter including the sample. Images were taken of the plates.
Results
Quantification by LC-MS
Correlation of standard curves (R2) for all antibiotics was ≥0.975. Figures 2 and 3 show the mean antibiotic concentrations of eluate samples taken from tubes containing 3g of beads. All samples showed a generally exponential decay in eluted antibiotic concentration with respect to time. At the first, 4-h, time point, gentamicin had eluted to peak concentrations (3-mm beads 6,400 mcg/mL, 6-mm beads 10,400 mcg/mL). The elution of gentamicin was maintained at a higher level for a longer duration with the 6-mm beads. By comparison, the 3-mm beads showed a sharp drop in detectable concentrations at 14 d.
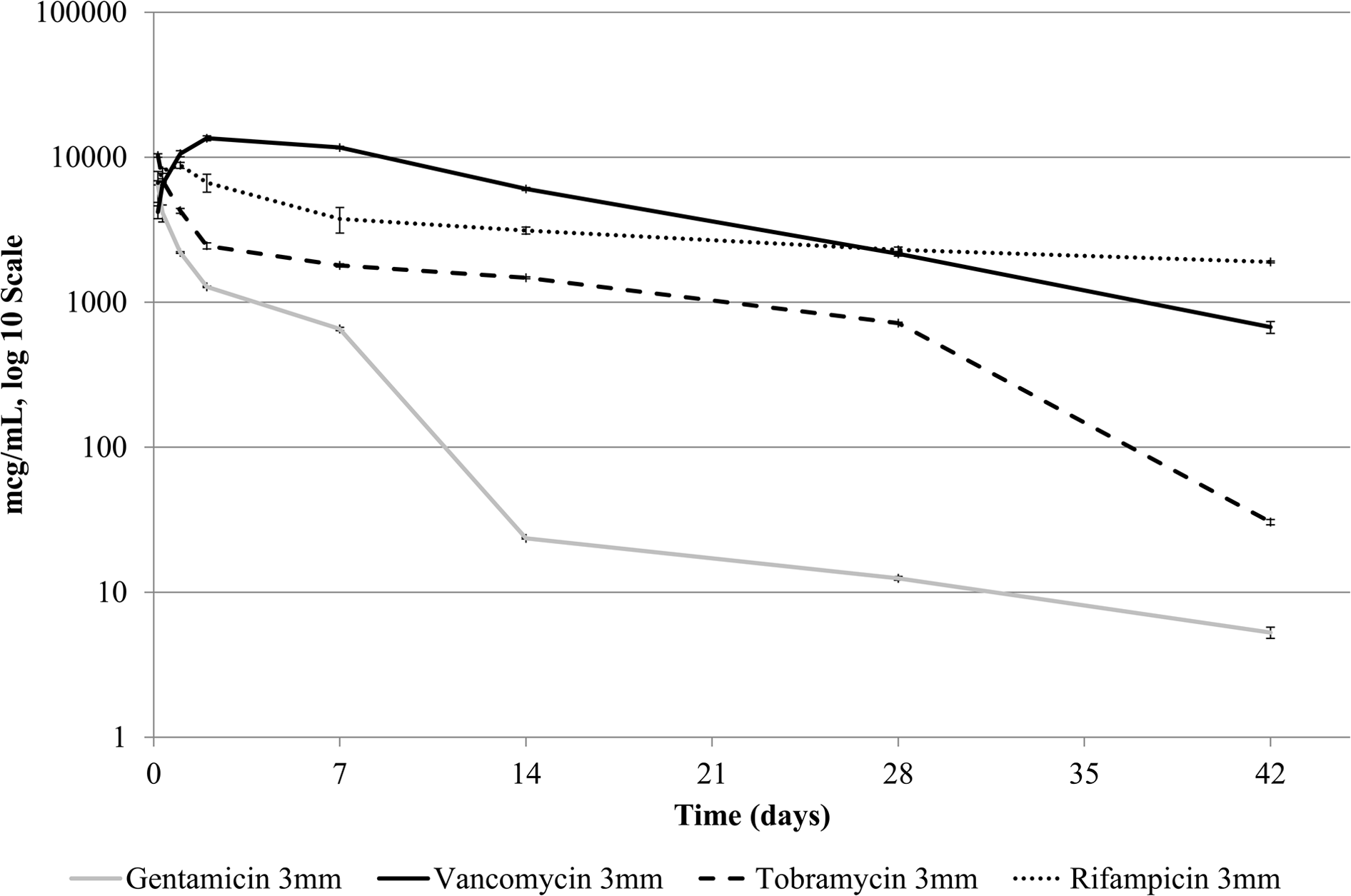
Concentration (mcg/mL) of gentamicin, vancomycin, tobramycin, or rifampicin determined by liquid chromatography–mass spectrometry over time in phosphate-buffered saline removed from tubes containing 3 g of 3-mm beads. Bars represent standard error of the mean.
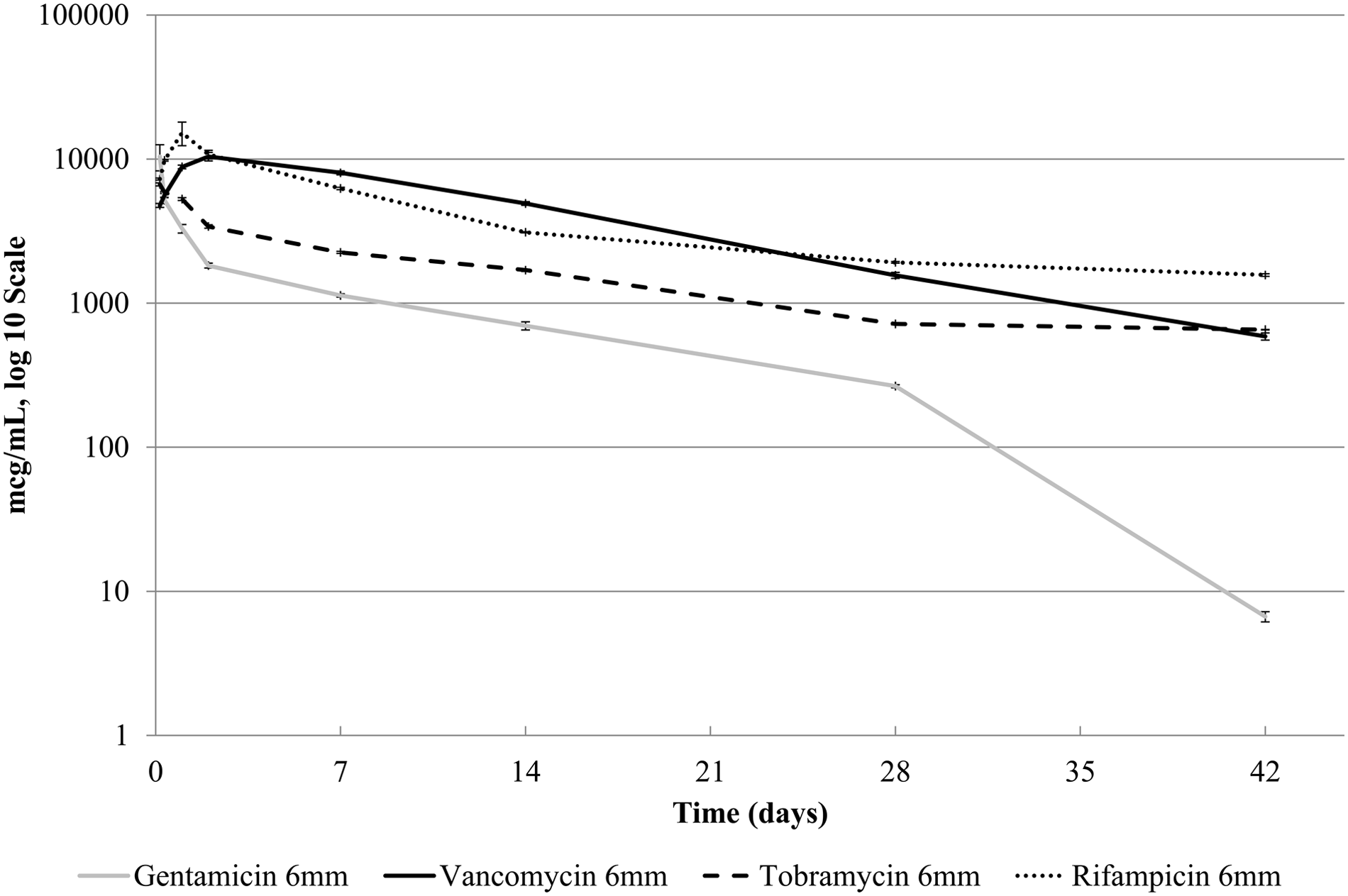
Concentration (mcg/mL) of gentamicin, vancomycin, tobramycin, or rifampicin determined by liquid chromatography–mass spectrometry over time in phosphate-buffered saline removed from tubes containing 3 g of 6-mm beads. Bars represent standard error of the mean.
Elution of tobramycin followed the same pattern, with peak concentrations at 4 h (3-mm beads 10,300mcg/mL, 6-mm beads 6,700 mcg/mL). However, higher elution was achieved with the 3-mm beads.
For rifampicin, the peak concentration occurred at the 24-h time point (3-mm beads 88,000 mcg/mL, 6-mm beads 15,200 mcg/mL), whereas for vancomycin, it occurred at 48 h (3-mm beads 13,500 mcg/mL, 6-mm beads 10,400 mcg/mL).
For 3-mm beads, rifampicin and vancomycin maintained a higher concentration in the PBS than tobramycin and gentamicin from 8 h onwards. Rifampicin 6-mm beads initially maintained a higher concentration (8 h to 48 h), whereas vancomycin was at a higher concentration at 7 d and 14 d.
Significant concentrations of eluted antibiotic were noted at 42 d (gentamicin: 3-mm beads 5 mcg/mL, 6-mm beads 6mcg/mL; tobramycin: 3-mm beads 30 mcg/mL, 6-mm beads 650 mcg/mL; vancomycin: 3-mm beads 670 mcg/mL, 6-mm beads 590 mcg/mL; rifampicin: 3-mm beads 1900 mcg/mL, 6-mm beads 1,600 mcg/mL).
Antibiotic stability
Analysis of stability standards confirmed stability of the antibiotics at the chosen concentration over the 42-d sampling period.
Antibiotic efficacy
Results from the ZOI testing indicated that antimicrobial efficacy of all the antibiotics was unaffected after 42 d at 37°C in the loaded beads (Tables 3 and 4). The zones of inhibition observed around the beads tested were larger than those observed with the AST discs for each antibiotic.
Available zone interpretation of Susceptible (S) in accordance with Clinical Laboratory Standards Insitute Performance Standards for Antimicrobial Susceptibility Testing [70].
NT=not tested.
Available zone interpretation of Susceptible (S) in accordance with Clinical Laboratory Standards Insitute Performance Standards for Antimicrobial Susceptibility Testing [70].
Zones starting to merge; therefore, impossible to measure accurately.
NT=not tested.
Discussion
This study was intended to address experimental design and the clinical use of the material–antibiotic combinations in order to provide elution results of greater clinical relevance. The experimental parameters described above were intended to mimic a small bone defect/void of approximately 4 mL in a relatively stable environment. The model does have some limitations. Whilst the chosen parameters are intended to mimic an in-vivo scenario in terms of quantity of material used, volume of eluent, and eluent exchange, the model does not take into account factors such as clot formation, which will have an effect on the diffusion of antibiotic into the surgical site, and protein binding [51], which will affect the antimicrobial efficacy of the antibiotics released into the extracellular matrix. Silverman et al. [52] attempted to mimic the in-vivo environment by determining the effect of clot formation around gentamicin-impregnated β-tricalcium phosphate granules on antibiotic elution, with results showing that clotted blood slowed drug release.
Common pathogens reported in orthopaedic infections are S. aureus [53–55], S. epidermidis [55,56], coagulase-negative staphylococci [54], and Pseudomonas aeruginosa [54,55]. The eluted concentrations of all four antibiotics tested, for both the 3-mm and 6-mm beads, exceeded reported MICs for pathogens commonly observed in periprosthetic joint infections [57–68] for the 42-d study period. This suggests that the study material has potential for effective local antimicrobial therapy when placed in a stable environment for as long as 42 d. For surgical sites with higher fluid transfer, the elution period may be reduced.
Care should be taken when drawing comparisons of antibiotic elution between different studies, as the design of elution experiments can have an effect on the concentrations and duration of antibiotic release. Two studies that have investigated elution from a single bead of calcium sulfate showed a shorter duration of elution than was demonstrated by the results of this experiment. Miclau et al. [43] investigated the elution of tobramycin from individual beads of calcium sulfate containing 25,000 mcg/bead and found that 17% of the antibiotic load was released in 24 h, with only trace amounts remaining at three weeks.
Wichelhaus et al. [20] investigated the elution of vancomycin, teicoplanin, gentamicin, and clindamycin from individual calcium sulfate beads. Initial release was especially high for gentamicin and clindamycin, at greater than 25,000 mcg/bead within the first 24 h. Elution kinetics demonstrated an initial elution phase, with beads releasing approximately 45% of the glycopeptide antibiotic and about 80% of gentamicin and clindamycin within the first 24 h. A more gradual second phase was observed over a further 10 d.
Recently, Roberts et al. [69] reported elution of vancomycin from individual calcium sulfate beads for as long as 20 d from vancomycin-loaded beads and as long as 40 d from beads loaded with both vancomycin and tobramycin. The ability of the presence of tobramycin to prolong vancomycin elution was noted.
In comparison, results from this study demonstrate elution of gentamicin, tobramycin, vancomycin, and rifampicin in significant amounts for as long as 42 d. However, it must be noted that our elution results were from a larger number of beads per vial (a total combined weight of 3 g) and a different sampling regimen than in many published elution studies, and therefore caution should be exercised when directly comparing results.
The molecular fingerprint of the antibiotics was measured using their mass and products of fragmentation. If they had degraded in any way, such as by oxidation or hydrolysis, the molecular fingerprint would change. Any structural change in the antibiotics would be unlikely to lead to identical ion breakup. No change was observed in the molecular fingerprints for the antibiotics for the duration of the elution experiment in both the elution samples and the antibiotic stability standards over the 42-d period of testing.
The Kirby-Bauer disc diffusion assay is used routinely to test bacterial sensitivity to antibiotics. The modification of this technique has been successful previously in determining the shelf-life of antibiotic-impregnated calcium sulfate beads [49]. The results of this previous study suggested that calcium sulfate beads containing either vancomycin or tobramycin, when stored at room temperature and ambient humidity, appeared to maintain their antimicrobial characteristics for at least 120 d. The disc diffusion data presented herein indicate that the antimicrobial efficacy of gentamicin, tobramycin, vancomycin, and rifampicin remained unchanged after combination with calcium sulfate and storage for 42 d at 37°C. The combined observations from the LC-MS analysis and the modified Kirby-Bauer assay demonstrated that the antibiotics eluted from the calcium sulfate beads were unchanged throughout the 42-d period and confirm that their antimicrobial efficacy was maintained.
An antimicrobial, or other therapeutic agent, can be present in a water-based delivery matrix in essentially three forms, all dependent on the relative hydrophilicity/hydrophobicity and solubility: (1) Simply dissolved in the aqueous phase; (2) present as a solid dispersion throughout the matrix; or (3) adsorbed onto the surface of the solid component of the matrix. The differences in the elution profiles for each antibiotic tested here may be attributable to differences in their solubility in the calcium sulfate matrix. Differences were observed in the elution between the 3-mm and 6-mm beads. Relative elution of both vancomycin and rifampicin were different for the two beads sizes over the first 48 h of testing. Also, elution of gentamicin was maintained at a higher level for 6-mm beads for the first 28 d of testing. The differences in elution profiles are unclear and may be attributable to a number of factors. The 6-mm and 3-mm beads have different surface-to-volume ratios, with the smaller beads having a higher ratio by a factor of eight. Therefore, the 3-mm beads have a larger surface area and a reduced diffusion path for each antibiotic from the center to the surface of each bead. However, this must be balanced against the solubility of the antibiotic, its molecular weight, and its potential for binding to the calcium sulfate. Further studies are required to determine the mechanism of release kinetics for antibiotics from calcium sulfate.
Conclusions
The study data from the LC-MS analysis were confirmed by the ZOI results from the Kirby-Bauer analysis. The data presented here indicate that synthetic pharmaceutical-grade calcium sulfate may have useful application as a delivery matrix for the targeted local release of tobramycin, gentamicin, vancomycin, and rifampicin and may be an effective tool in the management of bone and soft tissue infections. Calcium sulfate has the potential to deliver effective local antibiotic concentrations for a period of at least 42 d without affecting the chemical nature and antimicrobial efficacy of these antibiotics.
