Abstract
Abstract
Background:
Clostridium difficile colitis is associated with increased age, antibiotic usage, and hospitalization. Severe C. difficile colitis refractory to medical therapy may require surgical intervention including subtotal colectomy. We initiated an adjuvant intracolonic vancomycin (ICV) enema protocol for inpatients with severe C. difficile colitis and compared the response to this therapy in patients from the community and nursing homes.
Methods:
A single-hospital, retrospective chart review was done on 47 consecutive patients with C. difficile colitis treated with ICV (1 g/500 mL normal saline q6h) from January 2007 through October 2009. The proportions of patients with the outcomes of response to the ICV protocol, need for subtotal colectomy, and death were described. Associations of patient characteristics with these outcomes were examined with bivariate tests and multivariable logistic models with adjustment for age, hypoalbuminemia, acidosis, and nursing-home status.
Results:
Thirty-three of 47 patients (70%) with severe C. difficile colitis responded to adjunct ICV with complete resolution without surgery. Incomplete responders who had surgery were more likely to survive than those patients who did not undergo subtotal colectomy (p<0.01). Seven of nine patients who underwent surgery survived >90 d, and overall, 37 of 47 patients (79%) survived after ICV therapy. Nursing-home residence, acidosis, and hypoalbuminemia were significantly associated with the non-resolution of colitis in bivariate analyses (all p<0.01), whereas nursing-home residence and hypoalbuminemia showed non-significant trends toward association with death (p=0.07 and p=0.06, respectively). Multivariate logistic-regression models showed significant associations of acidosis with an incomplete response to ICV (p=0.02), of older age with death (p=0.04), and of hypoalbuminemia with both an incomplete response to ICV and death (both p=0.04). No complications were attributable to ICV.
Conclusion:
Complete resolution without surgery was achieved in 70% in this series of patients with severe C. difficile colitis who received adjunct ICV therapy. A clinical trial will be needed to determine whether ICV as compared with standard therapy alone can reduce the need for surgery with non-inferior or superior outcomes.
S
Intracolonic delivery of vancomycin (ICV) has been proposed by several groups as a modality for treating patients with C. difficile colitis in whom oral vancomycin cannot reach the colon because of paralytic ileus [14–19]. We hypothesized that adjunct therapy with ICV enemas would decrease mortality and the need for surgical resection of the colon. On the basis of our clinical observations that many of these patients were admitted from nursing homes, we also hypothesized that these patients would have worse outcomes. We performed multivariable statistical analyses to assess this observation after adjusting for potential confounders.
Patients and Methods
Study population and design
We initiated a protocol in our inner-city hospital by which patients with presumed or documented severe C. difficile colitis were considered candidates for ICV under the supervision of infectious diseases and surgical consultants. All patients who received adjunct ICV at Jacobi Medical Center (Bronx, NY) were identified by a hospital pharmacy data base. A standard data collection form was developed and completed for each patient. The following data were evaluated: age, gender, comorbidities (hypertension, diabetes mellitus, renal failure, acquired immune deficiency syndrome [AIDS], chronic respiratory disease, inflammatory bowel disease, and malignant disease), previous antibiotic use, history of recent hospitalization or admission from a nursing home, and previous episodes of C. difficile colitis. Clinical symptoms of C. difficile colitis (fever, diarrhea, abdominal pain, distention, and tenderness) were recorded. Evidence was obtained for colitis by computed tomography (CT) or for pseudomembranous colitis by sigmoidoscopy. Laboratory data including white blood cell (WBC) count, serum albumin, and serum bicarbonate concentration, and arterial base deficits were recorded.
This system of treatment evolved at our institution from a multi-disciplinary response to an unexpected presentation of 15 patients with fulminant C. difficile colitis in 2007. As subsequently reported by researchers in Quebec and internationally, such severely ill patients were suspected of harboring a highly toxigenic strain of C. difficile [11]. The clinical course of these patients was characterized by diarrhea, a robust leukemoid reaction (mean WBC count=29±14x103 cells/nL), acidosis (bicarbonate=16±7 mmol/L), abdominal tenderness and distention, followed by a precipitous clinical demise. Polymerase chain reaction testing for deletion of the TcdC gene of C. difficile was done in all 15 of our patients, and nine tested positive for the strain that produces high levels of C. difficile toxin. Four of the most severe cases with the TcdC gene deletion had a rapidly fulminant course of disease, underwent colectomy, and died. Because the window for aggressive intervention in these patients was narrow, a multidisciplinary team with representatives from our departments of surgery, infectious diseases, pharmacy, and infection control formulated an aggressive treatment protocol based on the available literature in 2007 [13–21]. Any patient experiencing diarrhea, with or without a fever, a WBC count >15,000/nL, or a bicarbonate <21 mmol/L would require infectious diseases and surgical consultations to help guide the patient's therapy. In the critically ill patients, primarily those who did not have a rapid response to oral vancomycin therapy, a surgical evaluation was required and a protocol for administration of an adjunct ICV retention enema was instituted in 47 consecutive patients as an intermediary step before surgery. The inpatient pharmacy of the hospital was required to screen inpatient orders and help to identify these patients, enforce the protocol, and notify the infection control department.
From January 2007 through October 2009, 250 inpatients had positive findings of C. difficile toxin and were reported to the New York State Department of Health by the Infection Prevention Department at Jacobi Medical Center. The criteria for inclusion in this patient group were stool samples positive for C. difficile toxin by cytotoxicity assay with or without evidence of pseudomembranous colitis by sigmoidoscopy, and failure to improve with standard treatment with oral vancomycin and intravenous metronidazole, as demonstrated by severe leukocytosis, hypotension, need for pressors, evidence of end-organ damage, diarrhea, or abdominal distention with tenderness on physical examination. Intravenous vancomycin solution was prepared as an oral formulation and given at a dose of at least 125 mg PO q6h. Metronidazole was given in intravenous doses of 500 mg IV q8h. Criteria for exclusion from the analysis included presumed C. difficile colitis in the differential diagnosis with a negative finding of C. difficile toxin by stool analysis.
A patient with presumed or documented severe C. difficile colitis was considered a candidate for ICV, which was given under the supervision of infectious diseases and surgical consultants. Forty-seven patients met the entry criterion of “severe” for toxin-positive C. difficile colitis, which included the presence of any one of the following: a WBC count >15,000/nL, abdominal tenderness, or an abnormal base deficit consistent with acidosis (Table 1). Acute Physiology and Chronic Health Evaluation II (APACHE II) severity scores were determined by standardized calculation [22]. During this period from January 2007 through October 2009, every patient who met these inclusion criteria for severe C. difficile colitis received ICV. The study was reviewed by the Albert Einstein College of Medicine Institutional Review Board (IRB) and was determined to be exempt from IRB approval.
Treatment
Adjunctive ICV was initiated for patients with severe C. difficile colitis in whom standard C. difficile therapy of oral vancomycin and intravenous metronidazole failed to prove effective. Intracolonic vancomycin was prepared by the hospital pharmacy in accord with the hospital protocol. The standard dosage was 1 g of vancomycin dissolved in 500 mL 0.9% normal saline given rectally q6h via retention enema [19,20], in addition to oral vancomycin and intravenous metronidazole. Patients were then monitored by the surgical service for the need for potential surgical resection by subtotal colectomy. No patients went immediately to surgery before instillation of ICV.
Treatment outcomes
Outcomes of treatment with ICV included response to the ICV protocol, need for subtotal colectomy, and death <90 d after admission. Response to the ICV protocol was categorized as complete resolution or incomplete resolution of C. difficile colitis. Incomplete resolution was defined as some or no clinical response despite continued adjunct ICV therapy and standard C. difficile therapy, and consisted of persistent diarrhea, leukocytosis, abdominal distension, tenderness or pain, persistent pancolitis on CT scan, or abnormal base deficits. Complete resolution was defined as the eradication of signs and symptoms of C. difficile colitis after adjunct ICV. All patients to whom surgery was recommended had clearly documented hypotension with an increasing need for pressors, respiratory failure, a rising WBC count, increasing acidosis, progressive renal failure, worsening findings on abdominal examination, or changes in mental status. Although there is no consensus in the literature about specific criteria for surgery for C. difficile colitis [1–3,10,11,23,24], these are the clinical guidelines we have used to recommend surgery. Complications were defined as any adverse reactions documented after the intracolonic administration of vancomycin.
Follow-up data for recurrence
Telephone interviews were conducted with the patients in the study population or their next of kin in August 2010, through the use of contact information in hospital records. Subjects were questioned about their general health since discharge from the hospital, recurrence of C. difficile colitis, and, for patients who had died, cause of death, through relatives or other sources.
Statistical analyses
Categorical variables were reported as percentages and compared in bivariate analyses through the Fisher exact test. Continuous variables were reported as mean±SD or median with interquartile range (IQR) and were compared for nursing home versus non-nursing-home residents using the t-test for independent samples or the Mann–Whitney U test as appropriate. Multivariate binary logistic regression models were constructed for the outcomes of death and resolution of symptoms of C. difficile colitis. Models were limited to three covariates at a time because of the small number of outcome events. In addition to nursing-home status and age, a third covariate was added separately if it showed evidence of bivariate association with both nursing-home status and one or both outcome variables. For the logistic-regression models we reported the Naglekerke R2, the Hosmer-Lemeshow goodness-of-fit statistic, and discrimination data using the C statistic, along with odds ratios (ORs) and 95% confidence intervals (CIs) for the covariates. A two-tailed α=0.05 was used to denote statistical significance. All analyses were performed using SPSS version 19 (SPSS, Chicago, IL).
Results
There were 47 patients with severe C. difficile colitis treated with ICV (Table 1). These patients all had documentation of at least one of the criteria for entry into the study (abdominal tenderness, WBC count >15,000/nL, or abnormal base deficit) listed in Table 1. Of these 47 patients, 36 patients had two of the three entry criteria and 24 patients had documentation of all three entry criteria.
Demographic and baseline clinical characteristics and pre-existent comorbidities for these 47 consecutive patients with C. difficile toxin-positive colitis are presented in Table 2 according to nursing-home status. The demographics of the patient group are similar to the characteristics of large populations with C. difficile colitis reported in the United States [7]. The mean (±SD) age for patients in the study population was 65±19 years, and in our series more than one-half of the patients who received ICV were nursing-home residents. All 47 of the patients had high APACHE II scores, with a mean of 23±8 consistent with severe illness. Sixteen of the 47 patients (34%) were hypotensive and were started on intravenous vasopressor therapy. The blood pH range of these patients was 7.1–7.5. In resemblance to what had been reported in other studies, 70% of the patients had received antibiotics within the previous 12 mos, and 75% had been hospitalized within the past year [24]. Notably, patients from nursing homes were more likely to present with prior episodes of C. difficile colitis, older age, hypertension, a low serum bicarbonate concentration, and hypoalbuminemia.
AIDS=Acquired immune deficiency syndrome; APACHE=Acute Physiology and Chronic Health Evaluation; WBC=White blood cell.
Values are % or mean±standard deviation (SD). Values of p are derived from comparisons of nursing home patients with non-nursing-home patients on the basis of the χ2 test and t-test for categorical or continuous variables, respectively.
Treatment with ICV was instituted after the progression of C. difficile colitis during treatment with oral vancomycin and intravenous metronidazole. In the 33 patients who responded to ICV therapy with complete resolution of their condition, the median duration of standard treatment before the initiation of ICV therapy was 1 d. For the patients who displayed incomplete resolution with ICV therapy, the median (IQR) duration of standard medical therapy for patients who experienced incomplete resolution of their condition with ICV therapy was also 1 d (1,1). There was no statistically significant difference in the duration of standard therapy in these two groups (p=0.51).
Thirty-seven of the 47 patients (79%) with severe C. difficile colitis who were treated with adjunct ICV survived. There were no complications deemed attributable to ICV. After our treatment protocol with ICV in combination with oral vancomycin and intravenous metronidazole, 33 of the 47 patients (70%) demonstrated complete resolution of their condition (Fig. 1). The median (IQR) duration of ICV therapy in the 33 patients with complete resolution was 6 d (range 4–9.5 d), as compared with a median of 3.5 d (range 2–7.5 d) for the 14 patients with incomplete resolution (p=0.15). Fourteen of the 47 patients (30%) with severe C. difficile colitis showed incomplete resolution of their colitis. Nine of the 14 incomplete responders to ICV therapy underwent surgery. Of these nine patients, seven survived. The median (IQR) duration of ICV therapy for the seven patients who survived after surgery was three days (range, 2–6 d). Five patients treated who had incomplete resolution of their condition with ICV did not undergo surgery. For three of these patients the family refused to give consent for subtotal colectomy, and the attending surgeon deemed two other patients to be “poor surgical candidates” because of multiple organ dysfunction syndrome and hemodynamic instability. None of the five non-responders to ICV who did not undergo surgery survived, whereas seven of the nine patients with incomplete responses to ICV who underwent surgery survived (p=0.02). The three deaths of patients who had responded completely to ICV were attributed to Candida fungemia, E. coli sepsis, and Candida pneumonia with urosepsis, respectively.
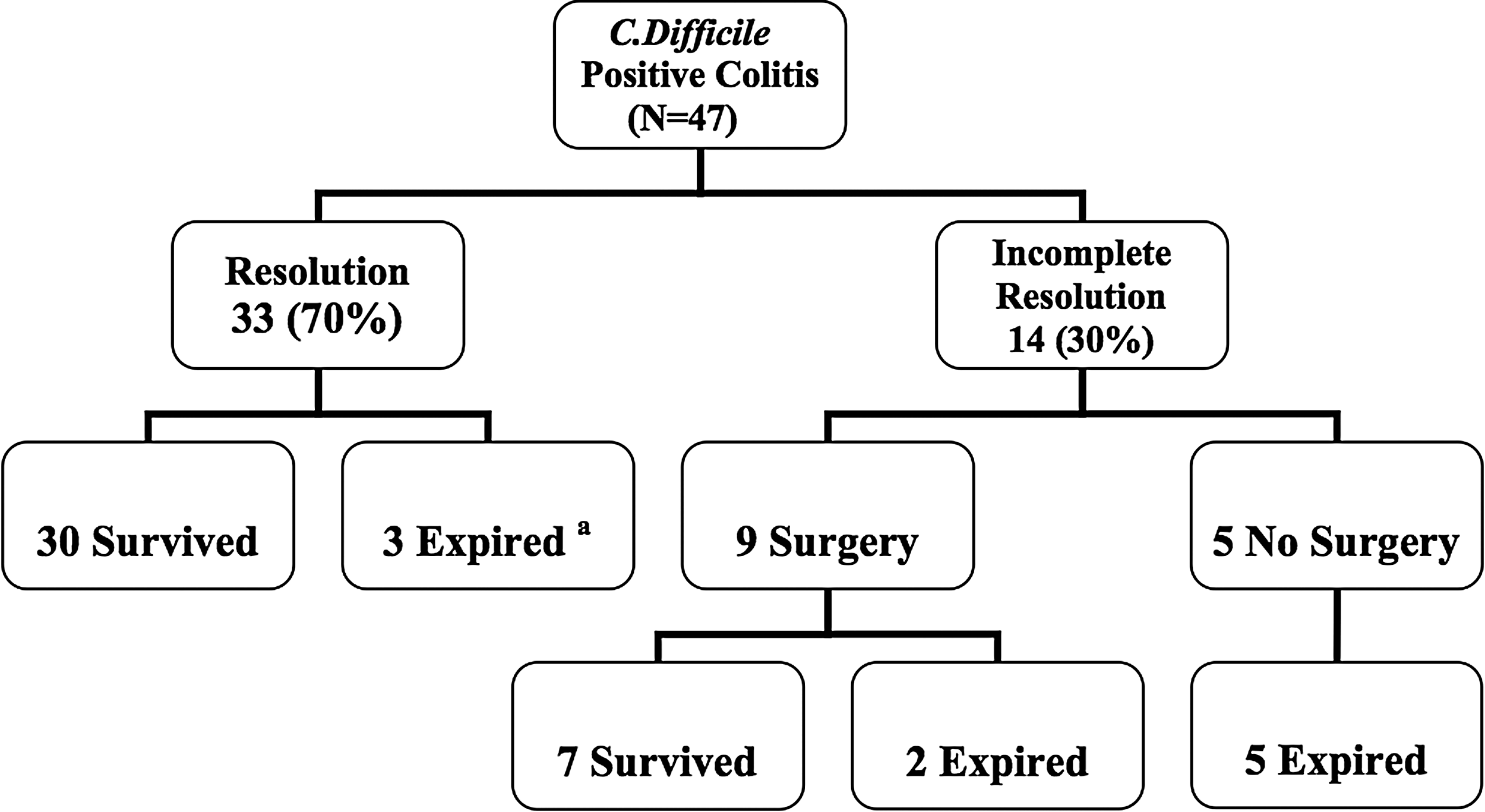
Overall outcomes of 47 patients with fulminant Clostridium difficile colitis treated with intracolonic vancomycin. Asterisk (*) represents all-cause mortality.
Table 3 presents the results of bivariate analysis of variables from Table 2 that showed some evidence of association with nursing home status. Of those variables associated with nursing home status, hypoalbuminemia and acidosis showed statistically significant bivariate associations with incomplete resolution of C. difficile colitis (both p=0.001), and age showed a significant association with death (p=0.01). Hypoalbuminemia, a marker of frailty and malnutrition, did show a trend toward association with death that was not statistically significant (p=0.06). A history of prior nursing home residence had a significant association with incomplete resolution of C. difficile colitis (p=0.002) and a borderline association with death (p=0.07). With adjunct ICV therapy, only one-half of the nursing home patients had a complete resolution of C. difficile colitis, and 67% of the nursing home residents with persistent symptoms required surgical intervention (p=0.02). However, in multivariable logistic-regression models (Table 4) in which age, albumin <2.5 g/dL, nursing home status, and serum bicarbonate were used as independent variables, we found that resolution of C. difficile colitis with ICV therapy was independently associated with albumin concentration <2.5 g/dL (p=0.04) and serum bicarbonate (p=0.02), but not significantly with nursing home status or age. In separate multivariable logistic models (Table 5), age (p=0.04) and albumin <2.5 g/dL (p=0.04), but neither nursing home status nor serum bicarbonate, were independently associated with death.
In addition to nursing-home residence itself, the variables selected were those that were significantly associated with nursing-home status in Table 2. Values shown are percentages for categorical variables and mean±SD for continuous variables. Values of p are derived from the χ2 (or Fisher exact test) and t-test, respectively.
Because of the number of outcomes, variables in models were limited to nursing-home residence, age, and one additional covariate. The additional covariates were selected from those that were significantly associated both with nursing-home status and one or both outcomes.
Model statistics for Model 1 with hypoalbuminemia: Nagelkerke R2=0.42; Hosmer-Lemeshow test p=0.26; C statistic=0.82.
Model statistics for Model 2 with serum bicarbonate: Nagelkerke R2=0.46; Hosmer-Lemeshow test p=0.76; C statistic=0.86.
Due to number of outcomes, models were limited to nursing home residence, age, and one additional covariate. The additional covariates were selected from those that were significantly associated both with nursing home status and one or both outcomes.Model Statistics:Model 1 with Hypoalbuminemia: Nagelkerke R2=0.43; Hosmer Lemeshow Test P=0.57; C statistic=0.87.Model 2 with Serum Bicarbonate: Nagelkerke R2=0.27; Hosmer Lemeshow Test P=0.26; C statistic=0.78.
Phone interviews were conducted with the survivors of C. difficile colitis 10 mos after completion of the study to assess overall health since discharge from the hospital, recurrence of C. difficile colitis, and death since the study. Eleven of the 33 survivors (33%) of C. difficile colitis analyzed in this study were not able to be contacted by telephone and were therefore lost to follow-up. Nine patients (27%) who were contacted died from other causes. The causes of mortality at ten months post-analysis included malignancy, sepsis, and liver failure. There were two of 22 patients contacted who reported recurrence of C. difficile colitis that required re-hospitalization; both of these patients contacted had expired from recurrent C. difficile colitis.
Discussion
The treatment of C. difficile-associated diarrhea (CDAD) has evolved over the past several years from the routine use of oral metronidazole to routine oral vancomycin and intravenous metronidazole for severe disease [20,23]. Numerous studies describe oral therapy with vancomycin for severe CDAD, but only a few case reports describe the use of ICV for this condition [14–19]. We here report our experience with ICV in an underserved patient population that has been treated for severe C. difficile colitis. Our data are consistent with the hypothesis that when used in addition to standard therapy with oral vancomycin and intravenous metronidazole, ICV can be an effective adjunct means of treating severe C. difficile colitis for decreasing both mortality and the need for subtotal colectomy. Most importantly, the results of our study are consistent with the recommendations of the Society for Healthcare Epidemiology (SHEA) and the Infectious Diseases Society of America (IDSA) [23] for the initiation of ICV for severe C. difficile colitis.
Interestingly, we found that in bivariate analysis, nursing home residents were at a statistically greater risk than were non-nursing home patients for severe C. difficile colitis that may not be amenable to medical management and have poorer outcomes without surgery. Nursing-home patients were more likely to be elderly, hypoalbuminemic, and to present with recurrent C. difficile colitis, all of which are factors that probably contributed to their poor outcomes. Whether nursing-home patients are predisposed to high-toxin-producing C. difficile or represent a more immunocompromised segment of the population is unknown [25,26] and could not be discerned with the data available to us.
The management of severe C. difficile colitis is difficult because of the high morbidity and mortality of a subtotal colectomy with ileostomy in an aged, frail population with multiple pre-existent comorbidities [2,10,27]. Mortality for this fulminant disease has been reported to exceed 50% [9,28]. The appropriate selection criteria for patients who require surgery are elusive [27]. Although we demonstrated ∼21% mortality with ICV therapy, careful monitoring by a surgical service of patients with severe C. difficile infections is mandatory. Statistical analysis of the patients in our study who did not respond to ICV showed that those who underwent surgery were more likely to survive than those who did not undergo surgery. However, refusal of surgery by either the patient's family or the surgical team might be an indication of a poor prognosis.
It will never be known whether any of the patients in our study for whom surgery was refused would have survived surgery. More research is needed to determine the best management of these sickest patients, but our data do support the finding in other studies that early surgery for elderly, hypoalbuminemic patients who live in nursing homes and develop C. difficile colitis may improve survival [24].
Nonetheless, subtotal colectomy with ileostomy is necessary in as many as 3.5% of patients with CDAD [30], and surgery to remove the colon remains a highly morbid therapy for patients with fulminant colitis. Five of our patients who failed to improve with intracolonic vancomycin therapy died without surgical intervention. During the period of our study in which there was aggressive surveillance for C. difficile colitis, all patients who fit our criteria for severe illness received ICV because of heightened awareness for the disease through aggressive initiation of a hospital-wide protocol. Moreover, the strong presence and vigilance of early and ongoing surgical consultation, in addition to adjunct ICV, may have contributed to improved outcomes for the severely ill patients who usually came from non-surgical services [31].
Reasons for an incomplete response to ICV therapy include the possibility that the enema solution may not have been able to reach all of the C. difficile spores, as well as active and vegetative bacteria in the colon, because bowel wall edema can restrict the passage of enema contents to the right colon by narrowing the gut lumen [3]. Surgical therapy as source control for C. difficile infection and toxin production then becomes paramount, because organ failure ensues otherwise. Indeed, lavage of the colon with the saline enemas used in ICV therapy may have contributed to its salutary effect by flushing out the with C. difficile toxin. Lavage of the colon with vancomycin enemas may also have contributed to the effects seen by Neal et al. in their recent report of intra-operative antegrade washout of the colon with 8 L of polyethylene glycol solution via loop ileostomy for fulminant C. difficile colitis [4,24].
Systemic absorption of ICV has been reported [21], and metabolic acidosis can be a rare side effect of the instillation of normal saline into the colon. Perforation of the colon or rectum is a theoretical risk of the use of rectal tubes and enemas. None of these effects were observed in our study. It is noteworthy that in the study by Neal et al., all patients were treated for 10 d post-operatively with antegrade vancomycin enemas, a critical element of their treatment protocol that probably contributed to the overall effects seen with a less invasive surgical approach and similar outcomes to our study [24].
There are several limitations to our study. First, it was a non-randomized, retrospective, single-center case review study of a small number of patients with C. difficile colitis. This limitation alone makes our conclusions tentative and in need of confirmation. The definitions of resolution as well as indications for ICV and surgery were not standardized and may have varied on a case-by-case basis. A defined cohort-control group of patients with severe C. difficile colitis but without ICV was not available for comparison. We also do not have available complete data on the outcomes of patients who did not have severe C. difficile colitis and who did not receive ICV. A randomized, prospective trial of the use of ICV before and/or after surgery is needed to determine its role and effectiveness in tackling the various virulent strains of C. difficile that cause colitis.
The recurrence rate reported in the literature for C. difficile colitis is as high as 25% [32]. We were able to contact 22 patients, two of whom developed fatal, recurrent disease after ICV. Because one-third of our patients could not be contacted in follow-up by telephone, an accurate rate of recurrence of C. difficile colitis could not be determined for ICV therapy. Therefore, we do not believe that this retrospective review can resolve the question of whether ICV can decrease the recurrence of C. difficile colitis. Comparisons of ICV with other modalities in decreasing the recurrence of C. difficile colitis require future investigation [32,33].
Most of the patients in our study were receiving standard medical therapy for C. difficile colitis for 24 h before the initiation of ICV, and complete responders received a median of 6 days of therapy. On the basis of our retrospective study with a small sample size and with an untreated control group, we cannot definitively determine that ICV had a role in the resolution of the disease over and above that provided by the increased duration of oral vancomycin and intravenous metronidazole. Furthermore, it was not our intention to study the duration of standard medical therapy that should be given before the initiation of ICV therapy or the duration of ICV therapy itself. The sickest patients who required surgery, however, did not respond in less than 24 h, and it is not known whether a longer course of therapy may have prevented the need for surgery. At this point we recommend continuous evaluation and communication by critical care, infectious diseases, and surgical services to determine the times for initiation and termination of ICV therapy, and particularly the need for early surgical intervention.
Another limitation of our study is that our criteria for a diagnosis of severe C. difficile colitis differed from the SHEA/IDSA guidelines that were published in May 2010 [23], which was released substantially after our protocol was initiated in January 2007. Because of the high rate of mortality in the epidemic of C. difficile colitis in the prior year, we included in our criteria, beginning in 2007, a broad definition of a WBC count >15,000/nL, abdominal tenderness, or an abnormal base deficit. Our chosen dose of ICV was extrapolated from case reports available at the time [14–19]. The SHEA/IDSA guidelines for 2010 [23] define “severe” as a WBC count 15,000/nL or higher, or a serum creatinine concentration that exceeds the pre-morbid level by 1.5-fold or more. In our retrospective study, 38 patients had a WBC count>15,000/nL and met these criteria. Creatinine concentration was not used initially as criteria for severe illness in our study. Further studies are needed to determine whether a dose-dependent failure of ICV therapy to improve severe C. difficile colitis should be followed by a consideration of surgery in the form of subtotal colectomy or by less invasive strategies such as the laparoscopic creation of a loop ileostomy or percutaneous colostomy with total colonic washout [3,24,34]. Future multi-center research that compares the duration of ICV therapy before surgery with that of alternative medical therapies, such as fecal transplantation or intravenous monoclonal antibodies as anti-toxins to C. difficile toxin, will be necessary but ethically difficult [35–38]. Nevertheless, the estimated cost of ICV therapy during our study was $29/d. On the basis of our experience, ICV therapy remains a simple, inexpensive treatment with a low side-effect profile that continues to be a part of the standard treatment guidelines for severe C. difficile colitis in our hospital.
Conclusion
Our data are consistent with the hypothesis that ICV can have an important role in the management of severe C. difficile colitis as a simple, inexpensive adjunct to oral vancomycin and intravenous metronidazole, without apparent side effects. A clinical trial is needed, however, to determine whether ICV compared to standard therapy does in fact improve patient outcomes as compared with standard therapy, including a reduction in the need for surgery.
Acknowledgment
This work was supported in part by CTSA Grants UL1 RR025750, KL2 RR025749, and TL RR025748 from the National Center for Research Resources (NCRR), a component of the National Institutes of Health (NIH), and the NIH Roadmap for Medical Research. Its contents are solely the responsibility of the authors and do not necessarily represent the official view of the NCRR or NIH.
