Abstract
Globally, more than 300 million individuals experience chronic pain. Chronic inflammation with increased infiltration of activated inflammatory cells is a major cause of chronic pain. Mesenchymal stem cells (MSCs) are known to suppress excessive inflammation, and their mechanism of action has been shown to be a gap junction-mediated interaction with the endothelium and circulating white blood cells. In vitro-expanded autologous adipose tissue-derived MSC were transplanted intravenously into patients with chronic pain. The degree of pain was evaluated before and after treatment using the Faces Pain Scale and Pain Disability Assessment Scale. This study included 28 patients. The potential of MSCs for gap junction-mediated transfer of small water-soluble molecules was evaluated in vitro. Autologous adipose tissue-derived MSC significantly attenuated chronic pain compared with pain before cell transplantation. In vitro analysis confirmed that about 80% of transplanted MSC could transfer small molecules via gap junctions. Our results indicate that transplantation of in vitro-expanded adipose tissue-derived MSC, which can transfer small molecules via gap junctions, is safe and may suppress chronic pain. Further double-blinded clinical studies are required to confirm the effect.
Introduction
Chronic pain, such as osteoarthritis and cervicobrachial syndrome, is a common orthopedic disorder affecting >300 million patients. 1 Frayed cartilage with persistent inflammation causes chronic joint pain, although the detailed pathogenetic mechanisms remain controversial. 1 Inflammation has been suggested to be correlated with cervicobrachial syndrome. 2 Various experimental therapies, such as transplantation of platelet-rich plasma, microfragmented adipose tissue containing stromal vascular fraction, plasma concentrate enriched for alpha 2 macroglobulin, and mesenchymal stem cells (MSCs), have been initiated with promising results, although the therapeutic mechanisms remain controversial. 2,3
Gap junctions connect the cytoplasm of two adjacent cells and allow the rapid intercellular movement of small water-soluble molecules according to their concentration gradients. 4 Recently, we showed that the intravenous injection of MSCs after stroke reduced the permeability of the blood–brain barrier and suppressed inflammation in the poststroke brain by removing metabolites from endothelial cells via gap junctions in a murine model. 5 Furthermore, MSCs have been shown to interact with circulating white blood cells (WBCs) via gap junctions 5 and may remove various metabolites from WBCs. 6
Here, we evaluated the clinical effects of adipose tissue-derived MSCs on chronic pain and their potential for small-molecule transfer via gap junctions using in vitro analysis.
Methods
The clinical treatment was designed according to the Law for Securing the Safety of Regenerative Medicine in Japan. The protocol was approved by the Certified Special Committee for Regenerative Medicine of the Japan Regenerative Medicine Association (Protocol Nos. 01C2311033 and 01C2312024) and was registered with the Japan Registry of Clinical Trials (jRCT1030230333). Written informed consent was obtained from all participants.
Harvest and in vitro expansion of MSCs
One milliliter of adipose tissue was obtained from abdominal fat tissue under local anesthesia using 0.5% lidocaine (Sandoz, Tokyo, Japan), as described previously. 7 Adipose tissue-derived MSCs were expanded in vitro according to a previous report, 7 with modifications. Briefly, adipose tissue was washed twice with phosphate buffered saline (PBS) (−), and MSCs were isolated using an MSC purification kit according to the manufacturer’s protocol (Fukoku, Gunma, Japan). The MSCs were cultured in KBM ADSC-1 medium (Kohjin Bio, Saitama, Japan) up to the eighth passage. The cells were frozen with freeze-damage protection fluid (Cellbanker; Zenogen Pharma, Fukushima, Japan) using a programmable freezer (Kryo360; Asahi Life Science, Saitama, Japan).
Intravenous injection of MSCs
A total of 1 × 108 cells were intravenously injected slowly over 30 min. Changes in symptoms, such as breathing trouble and palpitations, were carefully observed. The degree of pain was evaluated before and approximately 4 months (123 ± 48 days) after the first treatment using the Faces Pain Scale (FPS) 8 and Pain Disability Assessment Scale (PDAS). 9 Patients were given the opportunity to receive a second injection upon their request after evaluating the safety of the first injection without asking or documenting the reason for their choice. Patients who received a second injection underwent an additional evaluation approximately 3 months (102 ± 29 days) after the second injection.
Quantification of the gap junction-mediated cellular potential of MSCs
An outline of the procedures is shown in Supplementary Figure. Randomly selected MSCs (n = 4) were defrosted and divided into two tubes. MSCs in the first tube (5 × 106 cells) were incubated with DiD (Thermo Fisher, Massachusetts, USA) to label the cell membrane for 30 min at 37°C according to the manufacturer’s protocol. MSCs in the second tube (5 × 106 cells) were incubated with 5 μM calcein acetoxymethyl ester (Calcein-AM; Dojindo, Kumamoto, Japan) to load water-soluble green-fluorescent dye, calcein, into the cytoplasm for 30 min at 37°C according to the manufacturer’s protocol. Calcein-AM is a nonfluorescent, cell-permeable compound that is converted by intracellular esterases into calcein, a water-soluble fluorescent form with no cell permeability. 10 DiD-labeled and calcein-loaded MSCs were washed twice with PBS before coculture. DiD-labeled MSCs and calcein-loaded MSCs were mixed and cocultured to evaluate the transfer of calcein from calcein-loaded MSCs to DiD-labeled MSCs via gap junctions for 3 h at 37°C in Dulbecco's modified Eagle medium (Thermo Fisher) with 10% fetal bovine serum (Thermo Fisher) in a CO2 incubator. After incubation, cells were resuspended to single cell with 0.25% trypsin-EDTA (Thermo Fisher) and washed twice with PBS before analysis by FACS (FACS Lyric; BD Biosciences, New Jersey, USA). DiD-labeled MSCs that received calcein from calcein-loaded MSCs were identified as calcein-positive cells in DiD-labeled cells. In contrast, DiD-labeled MSCs that did not receive calcein were identified as calcein-negative cells in DiD-labeled cells. The percentage of MSCs that have the potential to receive small water-soluble molecules from other cells via gap junctions was calculated as (number of calcein- and DiD-positive cells)/(number of total DiD-positive cells) × 100.
Data analysis
Individual comparisons were performed using Student’s t-test. Normal distribution of the data was confirmed using JMP 7 (JMP Statistical Discovery, NC, USA) with the Shapiro–Wilk test. In all experiments, the mean ± standard deviation is reported. Chi-square test was used for nonparametric statistics.
Results
Enrolled patients
This study included 28 patients who experienced chronic pain and did not show satisfactory relief from standard treatments for chronic pain. Patients with rheumatoid arthritis were excluded. Eighteen of the 28 patients received second treatment approximately 4 months after the first treatment.
Safety assessment
None of the enrolled patients showed symptoms of allergic reactions, pulmonary embolism, or other adverse events after cell injection.
Changes in FPS scores after cell transplantation
The changes in FPS scores of the patients are shown in Figure 1A. Compared with before treatment, significant relief of pain was observed after the first injection. However, 10 of the 28 patients did not show an improvement in FPS scores after cell injection. The backgrounds of the patients who showed or did not show improvement in FPS scores are shown in Table 1. No statistically significant differences were observed between groups. Sequential changes in FPS scores of the patients who received a second treatment are shown in Figure 1B. No statistically significant pain relief was observed after the second injection. The backgrounds of the patients who received only the first or second treatment are shown in Table 2. All the patients who suffered knee pain chose a second treatment.
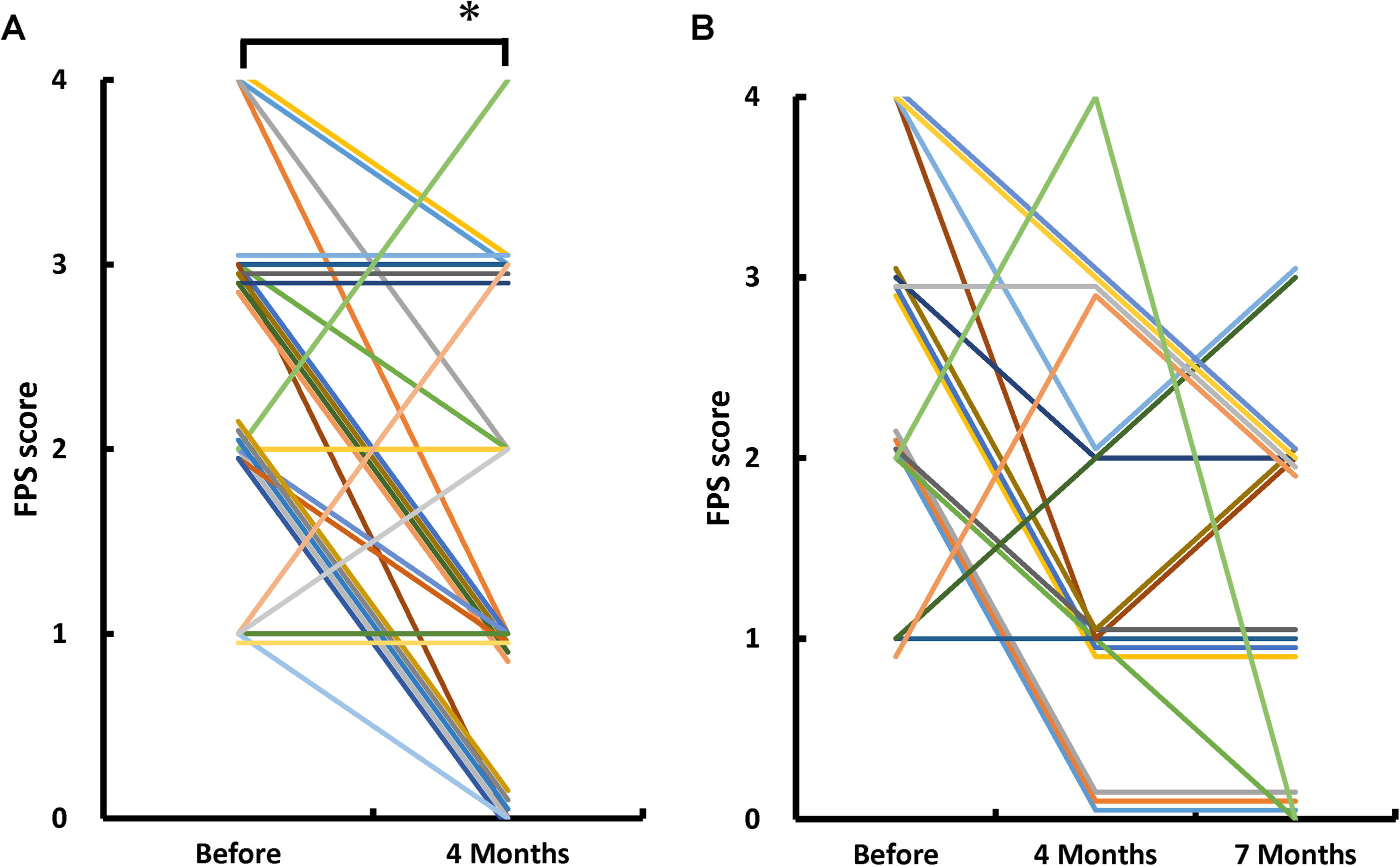
Changes in Faces Pain Scale (FPS) scores.
Patients That Showed or Did Not Show Improvement of FPS Scores by First Treatment
FPS, Faces Pain Scale; SD, standard deviation.
Patients That Received Only First or Second Treatment
FPS, Faces Pain Scale; SD, standard deviation.
Changes in PDAS scores after cell transplantation
The changes in PDAS scores of the patients are shown in Figure 2A. Similar to the FPS evaluation, significant relief of pain was observed after the first injection, compared with before treatment. Sequential changes in PDAS scores of the patients who received a second treatment are shown in Figure 2B. No statistically significant pain relief was observed after the second injection.
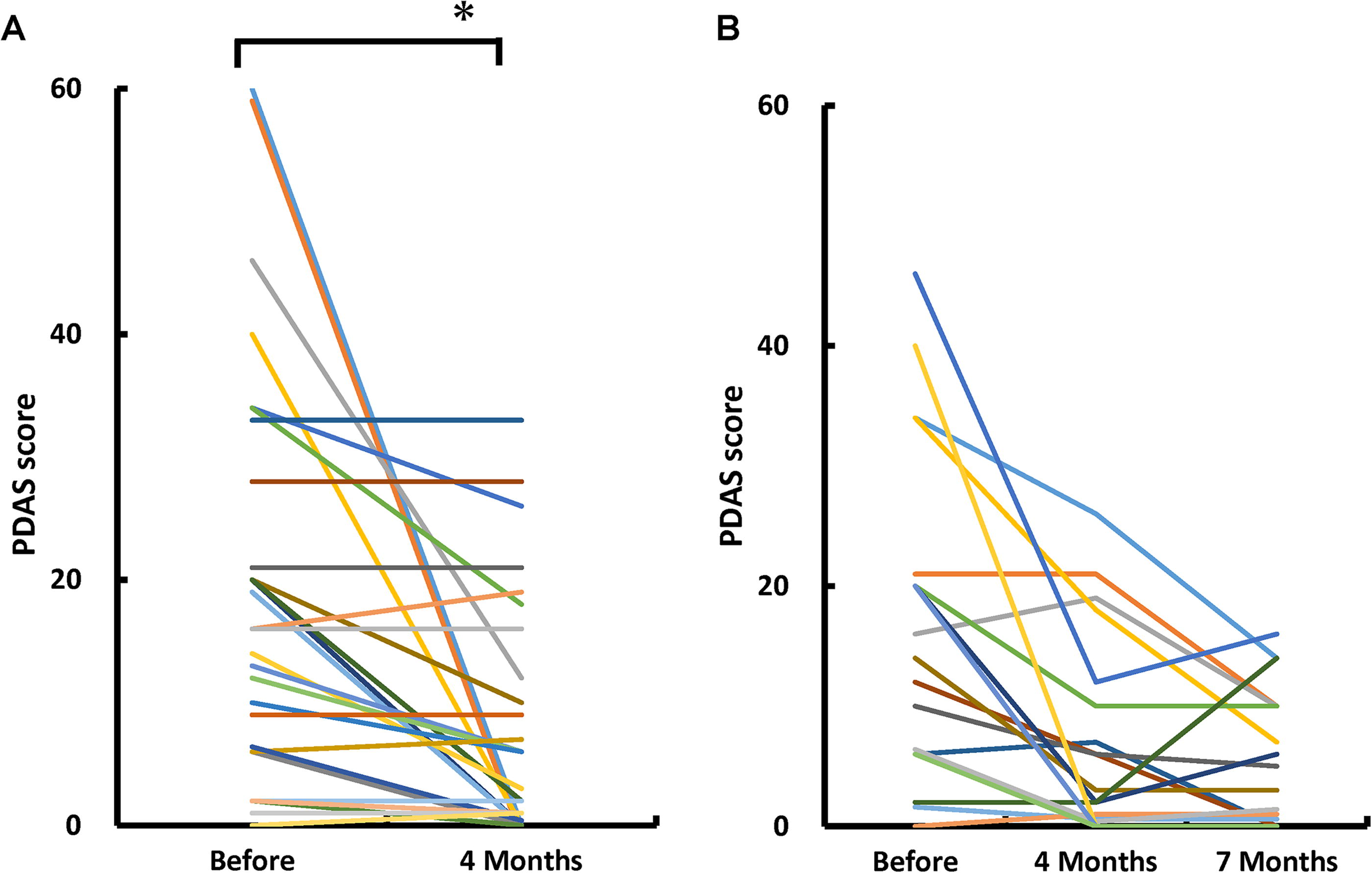
Changes in Pain Disability Assessment Scale (PDAS) scores.
Potential of gap junction-mediated small-molecule transfer of MSCs
Previously, we showed that a major mechanism of action of bone marrow-derived MSC therapy is the removal of metabolites from activated cells, such as endothelial cells and WBCs, via gap junctions. 5 To evaluate the potential gap junction-mediated cellular interactions of in vitro-expanded adipose tissue-derived MSCs, MSCs from four randomly selected patients were evaluated using FACS. As shown in Figure 3, about 80% (83% ± 8%) of adipose tissue-derived MSC removed small molecules from other cells. Similar to bone marrow-derived MSC, this finding indicates that adipose tissue-derived MSCs have the potential to remove small water-soluble molecules from endothelial cells and circulating WBCs.
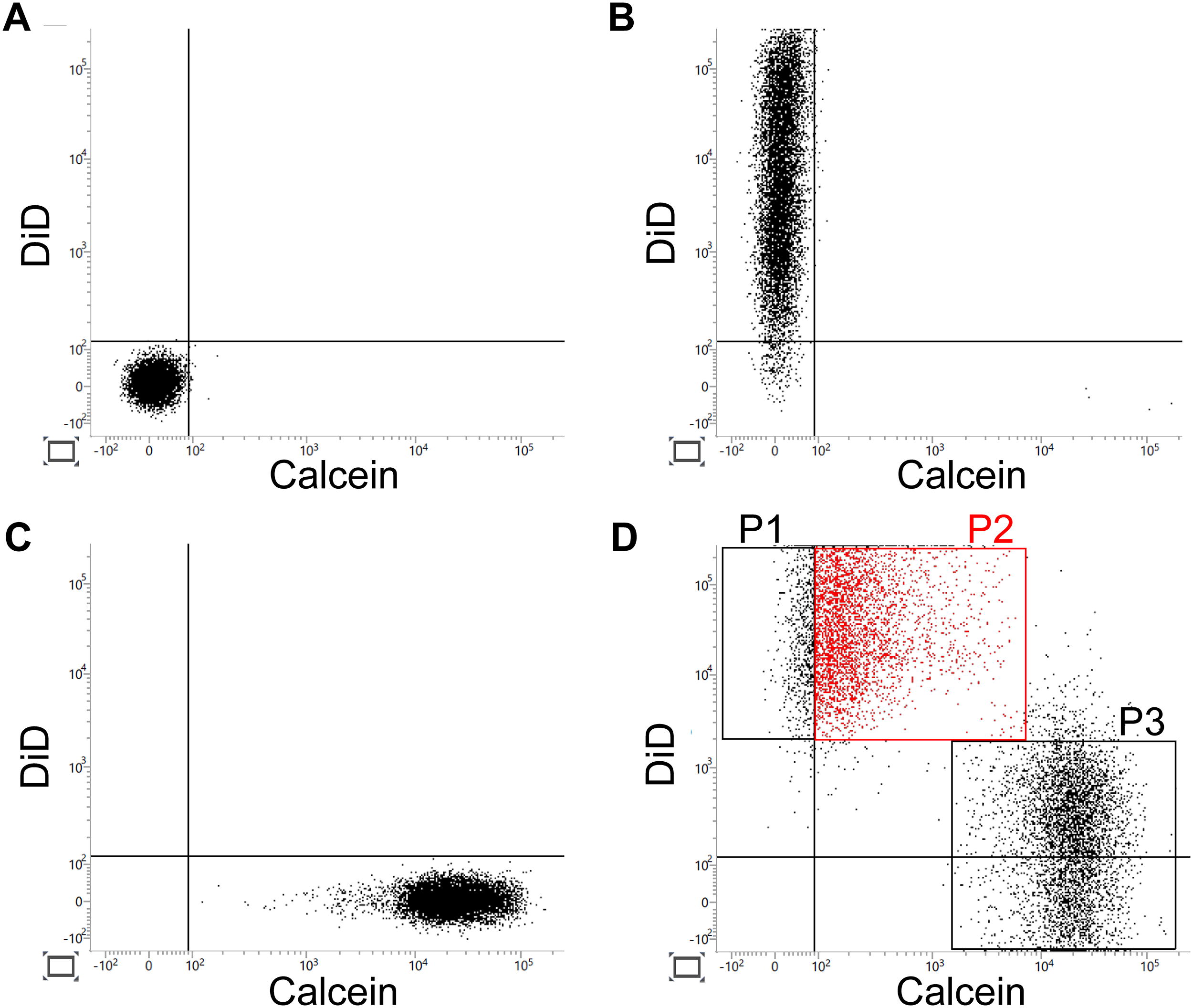
FACS analysis of mesenchymal stem cells. Results of the FACS analysis of nonstained
Discussion
Here, we demonstrated that the injection of in vitro-expanded adipose tissue-derived MSCs is safe and may suppress chronic pain after the first injection. Furthermore, our results showed that the potential gap junction-mediated small-molecule transfer of MSCs can be quantified by in vitro analysis.
Clinical trials of MSC transplantation have been initiated for various disorders. The source of MSCs varies among trials, and both positive and negative results have been reported. 11 However, the mechanisms of action of MSC therapy in tissue repair 12,13 and immune suppression 14 remain controversial. Therefore, a consensus on the mechanism and standardization of cell quality remain lacking. Recently, we showed that the mechanism of action of stem cell therapy with hematopoietic stem cells involves direct gap junction-mediated cellular interactions between transplanted cells and the endothelium soon after cell transplantation. 15 Similarly, we showed that a major mechanism of action of stem cell therapy with bone marrow-derived MSCs is the gap junction-mediated direct cellular interaction between transplanted cells and endothelium/WBCs. 5 Gap junctions connect the cytoplasm of two adjacent cells and allow the rapid intercellular movement of small water-soluble molecules according to their concentration gradients. 4 The study results showed that adipose tissue-derived MSCs have the potential to remove small molecules from other cells. These results would encourage further clinical and basic studies that link transplanted MSCs and the endothelium/WBCs via gap junctions. A possible mechanism is shown in Fig. 4.
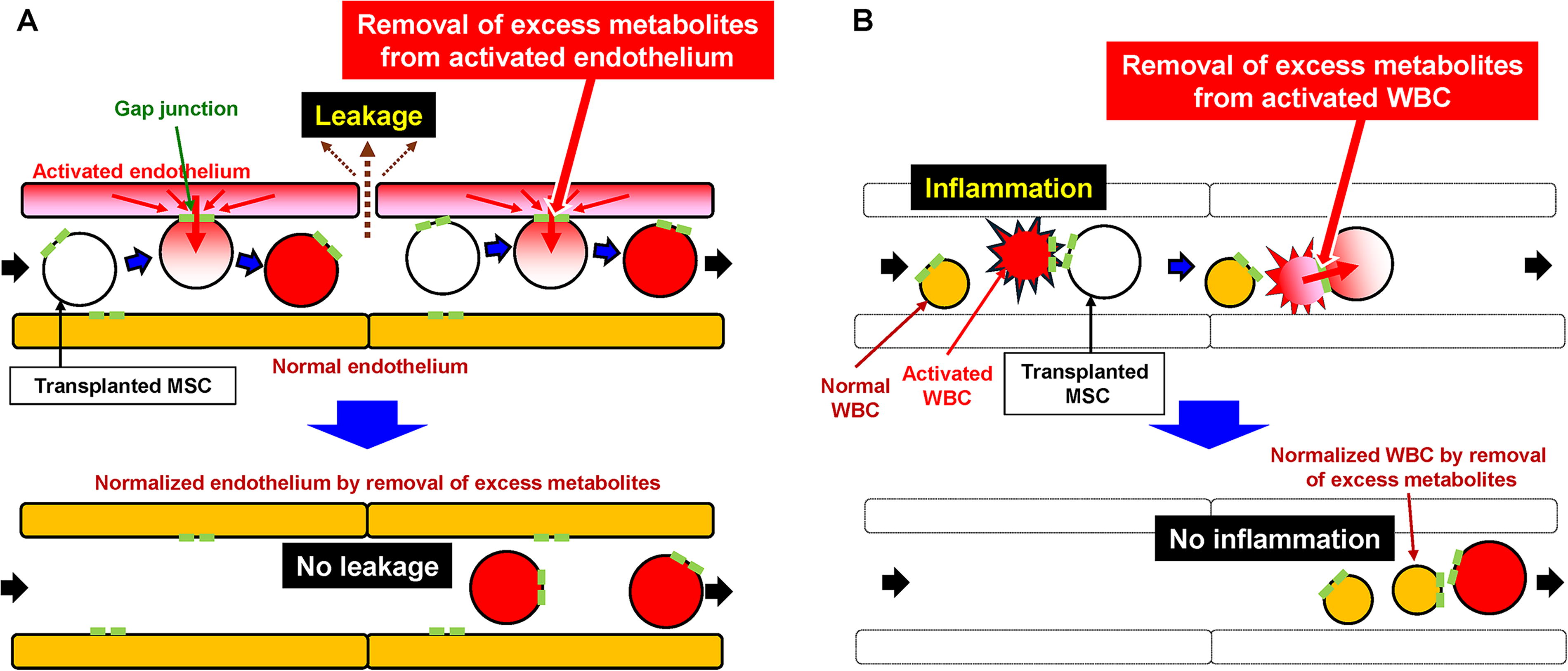
Schematic illustration of our hypothesis that links transplanted mesenchymal stem cells (MSCs) and over activated endothelium/WBCs via gap junction. Direct cell–cell interaction between intravenously transplanted MSCs and over activated endothelium
Although a statistically significant improvement in FPS scores was observed after the first injection in this clinical trial, it should be noted that 10 of the 28 patients did not show an improvement in FPS scores after the cell injection. Nonresponse can be caused by the insufficient ability of transplanted MSCs, differences in pathogenic conditions causing pain, or both. Recently, expression of adhesion molecule, integrin-β2, was shown to have a major impact on the outcome of hematopoietic stem cell transplantation for stroke. 16 This finding indicates that the expression of adhesion molecules on MSCs may play a key role in their therapeutic effects. Furthermore, mRNA expression in circulating WBCs has been proposed as a marker of chronic inflammation associated with aging. 17 Further studies elucidating the detailed mechanisms of MSC transplantation on chronic pain with standardization of transplanted MSCs and identification of markers for responders are required to improve MSC therapy.
Various studies have reported the safety of MSC transplantation. 18 Consistent with these previous reports, no serious adverse events were observed in the present study. However, it should be noted that there are case reports of pulmonary embolism after repeated intravenous adipose tissue-derived stem cell therapy. 19 These findings indicate that there remain safety concerns regarding MSC transplantation, especially with repeated treatments.
This study has several limitations. Our results suggest that the injection of in vitro-expanded adipose tissue-derived MSCs may suppress chronic pain. However, double-blind, placebo-controlled studies using appropriate surrogate markers are required to properly evaluate the effects of cell transplantation. Hematopoietic stem cell transplantation has been shown to reduce the RNA expression of metabolism-related genes in circulating WBCs in a murine model. 20 These findings may provide a hint for the exploration of new surrogate markers that reflect the effects of stem cell therapies. In this study, all patients with knee pain chose to receive a second injection. Osteoarthritis is the most common joint pain associated with aging; however, few effective treatments have been established. 21 The reason for patients with knee pain choosing to receive a second injection may be related to the insufficient effect of the first injection on knee pain or the experience of its therapeutic effect. This concern should be addressed in future studies that focus on knee pain. Furthermore, the correlation between the potential of gap junction-mediated small-molecule transfer of MSCs and their therapeutic effects was not evaluated in this study, as not all MSC samples after transplantation were stored for analysis. Further studies are required to link the changes in chronic pain by MSC transplantation, the potential of transplanted MSC, and changes in surrogate markers.
Overall, our results indicated that a single transplantation of adipose tissue-derived MSCs is safe and may suppress chronic pain. Furthermore, the potential of MSCs was quantified using FACS analysis. These findings have encouraged blinded clinical trials with exploration of novel surrogate biomarkers.
Conclusion
Although further double-blinded clinical studies are required to confirm the effect, our results indicate that the injection of in vitro-expanded adipose tissue-derived MSCs, which can transfer small molecules via gap junctions, is safe and may suppress chronic pain.
Data Availability Statement
All the data are available upon request from the corresponding author. A proposal is required to address these requests.
Footnotes
Author Disclosure Statement
A.T. has received research funding from BestCell. S.S., T.T., and A.I. are employees of BestCell.
Funding Information
A.T. has received research funding from BestCell.
Supplementary Material
Supplementary Figure S1
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
