Abstract
The development of acute graft-versus-host disease (GvHD) after allogeneic hematopoietic stem cell transplantation (allo-HSCT) is facilitated by damage-associated molecular patterns (DAMPs) released upon tissue damage due to the conditioning regimen. Heme oxygenase-1 (HO-1) is a stress-inducible enzyme responsible for the breakdown of the DAMP cell-free heme. HO-1 plays a protective role in diseases characterized by systemic inflammation such as sepsis, but its role in the development of acute GvHD remains unclear. Here, we characterized the expression of HO-1 in a small cohort of allo-HSCT recipients with and without acute GvHD. We found HO-1 protein levels in plasma to be elevated in patients just before their acute GvHD diagnosis compared with baseline. Furthermore, HO-1 mRNA expression was increased in patients with acute GvHD at 1 and 3 months after allogeneic HSCT compared with patients without acute GvHD. Finally, induction of HO-1 in a humanized mouse model for acute GvHD led to lower disease scores and a reduction in weight loss. Overall, our data indicate that HO-1 expression is increased in patients with acute GvHD and that HO-1 induction might be able to provide protection against the disease, warranting further research into HO-1 as a target for clinical application.
Introduction
For many patients with malignant or nonmalignant blood disorders, an allogeneic hematopoietic stem cell transplantation (HSCT) is the only curative option. One of the main limitations of this treatment is acute graft-versus-host disease (GvHD), which develops in up to 50% of all allogeneic HSCT recipients. 1 Acute GvHD is a potentially severe complication that involves the destruction of host tissues by alloreactive donor lymphocytes. Morbidity and mortality in patients with acute GvHD is high, with mortality rates reaching 50% in severe cases. 2
The development of acute GvHD is facilitated by damage-associated molecular patterns (DAMPs) that are released upon tissue injury and cell death caused by the conditioning regimen. 3 The release of DAMPs induces the activation of antigen-presenting cells of the host, which can in turn facilitate the activation and alloreactivity of donor lymphocytes. 4 DAMPs consist of a large variety of molecules, many of which have been indicated to play a role in acute GvHD. 1,3,5 As such, molecules that are able to prevent the release of DAMPs or inhibit their negative effects in the context of disease are of great clinical interest. We have shown previously that levels of cell-free (cf)DNA, which acts as a DAMP, are increased in patients at the time of acute GvHD diagnosis compared with pretransplantation and in mice with acute GvHD. 6 In recent years, there has been a growing interest in another DAMP in the context of inflammatory diseases: cell-free (cf)heme. Cfheme is released from mitochondrial membranes and hemoproteins, such as hemoglobin and myoglobin, upon cell death. Cfheme can bind to Toll-like receptor 4 (TLR4) and potently induces the production of reactive oxygen species. Moreover, cfheme itself is a lipophilic molecule, allowing it to intercalate into cell membranes inducing lysis and promoting further release of heme into the circulation. 7 In plasma, cfheme and cfhemoglobin (a source of cfheme) are rapidly cleared by the two scavenger proteins hemopexin (HPX) and haptoglobin (HPT), respectively. The resulting HPX-heme and HPT-hemoglobin complexes are subsequently taken up by macrophages where heme is broken down into CO, biliverdin, and ferrous iron by heme oxygenase-1 (HO-1). 8,9 HO-1 is an intracellular, stress-inducible enzyme encoded by the HMOX1 gene that plays a pivotal role in preventing the deleterious effects of cfheme. HO-1 has been the subject of extensive research and both HO-1 and the breakdown products of heme have been shown to have anti-inflammatory and cytoprotective effects in a wide range of diseases as well as transplantation settings. 8,10 –12 In the context of acute GvHD, the role of HO-1 as a protector against heme-driven tissue damage and inflammation is still largely unknown. Studies that made use of mouse-mouse models for acute GvHD have shown that increased expression of HO-1 in target organs alleviated severity of experimental acute GvHD 13 and that donor-derived HO-1 is able to provide protection against the development of acute GvHD. 14 In humans, the expression of HO-1 in bone marrow mononuclear cells was found to be significantly higher in patients who did not develop acute GvHD. 15
Here, we aimed to elucidate whether HO-1 might be of importance in the development of acute GvHD using a human cohort of allogeneic HSCT recipients and a humanized mouse model for acute GvHD. We measured protein levels of HO-1, HPX, and HPT in plasma by enzyme-linked immunosorbent assay (ELISA), protein levels of HO-1 intracellularly by flow cytometry, and HO-1 expression levels by quantitative PCR (qPCR) in patients. Furthermore, we induced the expression of HO-1 in a humanized mouse model for acute GvHD to study its effects on the development and perpetuation of the disease.
Materials and Methods
Patients
All patients included in the study were admitted to the Amsterdam University Medical Center, location Academic Medical Center (AMC), Amsterdam, for an allogeneic HSCT. All patients and healthy donors signed written consent forms and approval of the Institutional Medical Ethics Committee of the AMC was obtained for all patients before initiation of the study. All characteristics of patients and healthy donors are summarized in Table 1. A total of 69 patients were included and analyzed by ELISA and a total of 20 patients were included and analyzed by flow cytometry and qPCR (Table 1). Nine patients were part of both analyses.
Figure-Specific Characteristics of Patients and Healthy Donors
Data are expressed as median (range) or n (%).
ALL, acute lymphoid leukemia; AML, acute myeloid leukemia; AUL, acute undifferentiated leukemia; CLL, chronic lymphocytic leukemia; CML, chronic myeloid leukemia; HO-1, heme oxygenase-1; MDS, myelodysplastic syndrome; MF, myelofibrosis; MH, morbus Hodgkin; MM, multiple myeloma; NHL, non-Hodgkin lymphoma; SAA, severe aplastic anemia; MA, myeloablative conditioning; RIST, reduced-intensity stem-cell transplantation conditioning; MUD, matched-unrelated donor; Sib, sibling donor; aGvHD, acute graft-versus-host disease.
Sample processing
Whole blood samples were drawn in ethylenediaminetetraacetic acid (EDTA) tubes before allogeneic HSCT (baseline) and at different time points (1 month and 3 months) after allogeneic HSCT. Samples were processed before analysis as described previously. 16
ELISA
HO-1, HPT, and HPX were measured in plasma samples by ELISA as described previously. 17 Nucleosomes were measured in mouse plasma as described previously. 6
Flow Cytometry
The following antibodies were used for staining: BUV496 anti-CD3 (BD Biosciences), Pe-Cy7 anti-CD4 (BD Biosciences), BUV805 anti-CD8 (BD Biosciences), BV421 anti-CD14 (Biolegend), PerCP-Cy5.5 anti-CD16 (Thermofisher, clone 3G8), PE-Texas Red anti-CD19 (Thermofisher, clone SJ25-C1), PE anti-CD45 (Invitrogen, clone HI30), FITC anti-CD56 (BD Biosciences), BV605 anti-CD69 (Biolegend), APC-Cy7 Near IR-Live/Dead (Invitrogen), BV510 anti-Ki-67 (BD Biosciences), and AF647 anti-HO-1 (Santa Cruz Biotechnology, clone F-4). Cells were first stained with near-IR live/dead dye (Invitrogen) at room temperature in the dark for 30 min. After washing, cells were (extracellularly) stained with antibodies diluted in phosphate-buffered saline (PBS)/0.5% Bovine Serum Albumin (BSA) at room temperature in the dark for 30 min. Cells were then permeabilized and fixed using a BD Cytofix/Cytoperm Fixation/Permeabilization kit and subsequently stained with intracellular antibodies for 45 min in the dark at 4°C. Cells were measured on a BD FACSymphony A5 cell analyzer and data were analyzed using FlowJo v10.8.
qPCR
For each sample, total RNA was isolated from 0.5 × 106 cells using the RNeasy kit (Qiagen) and mRNA was reversely transcribed into cDNA using random primers (Invitrogen) and SuperScript II Reverse Transcriptase (invitrogen) following manufacturer’s instructions. cDNA was stored at −20°C until further analysis.
For all patient cDNA samples, the fold-change expression of HO-1 and HO-2 was determined compared with a cDNA sample derived from a pool of 10 healthy donors. 18S was used as a household gene. Gene expression levels were determined using the SYBR green method with Power SYBR Green (Applied Biosystems). The following primers were used: HO-1; 5′-GGGTGATAGAAGAGGCCAAGACT-3′ (forward) and 5′-AGCTCCTGCAAC TCCTCAAGA-3′ (reverse), HO-2; 5′-CCCTGGACCTGAACATGAA-3′ (forward) and 5′-ACCCATCCTCCAAGGTCTC-3′ (reverse), 18S; 5′-CGGCTACCACATCCAAGGAA-3′ (forward) and 5′-GCTGGAATTACCGCGGCT-3′ (reverse). Samples were measured in triplo on a StepOnePlusTM instrument. Fold-change expression was calculated using the ΔΔCt method.
Animals
Male NOD.Cg-PrkdcscidIL2tmlWjl/Szj (NOD-SCID IL-2rγnull) (NSG) mice were obtained from Jackson Laboratory and bred in-house at the Netherlands Cancer Institute, Amsterdam. All animal experiments were performed in accordance with the Experiments on Animals Act and with approval of the Central Animal Testing Committee (CCD) and the Animal Welfare Body (IvD, protocol ID 25.3.10658) of the Netherlands Cancer Institute. NSG mice were kept, irradiated, treated, and scored for acute GvHD as described previously. 6 In short, mice were injected with human peripheral blood mononuclear cells (PBMC) (1.0 × 105 cells/g bodyweight) or PBS and scored for symptoms of acute GvHD (Supplementary Table S1) daily until a humane endpoint (HEP) was reached or until the end of the experiment. On day 0 and every 7 days thereafter, 50 μL of blood was collected in EDTA tubes. Upon reaching a HEP or at the end of the experiment, mice were euthanized, blood was collected by heart puncture and liver, spleen, bone marrow (sternums and hind limbs), and intestines were collected for pathological analysis. Cobalt(III) protoporphyrin IX chloride (CoPP) (Frontier Specialty Chemicals) was dissolved in 100% Dimethyl Sulfoxide (DMSO) and subsequently diluted and adjusted to a pH of 7.4 in NaCl 0.9% (Fresenius). Mice in the CoPP-treated group received intraperitoneal (i.p.) injections of 5 mg/kg bodyweight CoPP on days −2, −1, and 7. After collection, blood was processed and analyzed for chimerism levels by flow cytometry as described previously. 6
Pathology
Livers, intestines, spleens, and bone marrow (derived from sternums and hind limbs) were fixed in 4% formaldehyde. Organs were subsequently embedded in paraffin and tissue sections were stained with hematoxylin and eosin (HE). Tissues were then examined and evaluated blindly by an expert animal pathologist at the Netherlands Cancer Institute for lesions of acute GvHD.
Statistical analyses
Data are shown as mean ± standard deviation (SD) or mean ± standard error of the mean (SEM). All statistics were performed using Graphpad Prism 9.1.1 software. For comparisons of multiple groups a Kruskal–Wallis test with Dunn’s multiple comparisons test was performed. For comparisons of two groups a two-tailed Mann–Whitney U test was performed. Differences between survival curves were calculated using a Gehan–Breslow–Wilcoxon test and correlations were calculated by simple linear regression. P values ≤ 0.05 were considered statistically significant.
Results
Allogeneic HSCT is associated with increased plasma levels of HO-1
We measured plasma levels of HO-1, HPX, and HPT in samples collected from a cohort of 69 allogeneic HSCT recipients before (baseline) and after allogeneic HSCT (1 and 3 months) and in 10 age-matched healthy controls (Table 1). Plasma levels of HO-1 were significantly elevated in all allogeneic HSCT recipients (regardless of acute GvHD status) at baseline, 1, and 3 months after allogeneic HSCT compared with healthy controls (Fig. 1A–C). The difference between patients with acute GvHD and healthy controls seemed larger at the 1- and 3-month time points than at baseline. For HPX and HPT, no significant differences were seen between patients with and without acute GvHD and healthy controls at any of the time points (Fig. 1A–C). We then compared levels of HO-1, HPX, and HPT at baseline to levels measured in the last sample taken before diagnosis of acute GvHD. Plasma levels of HO-1 were significantly higher close to the time of acute GvHD diagnosis compared with baseline, while no significant differences were seen for HPX or HPT (Fig. 1D).
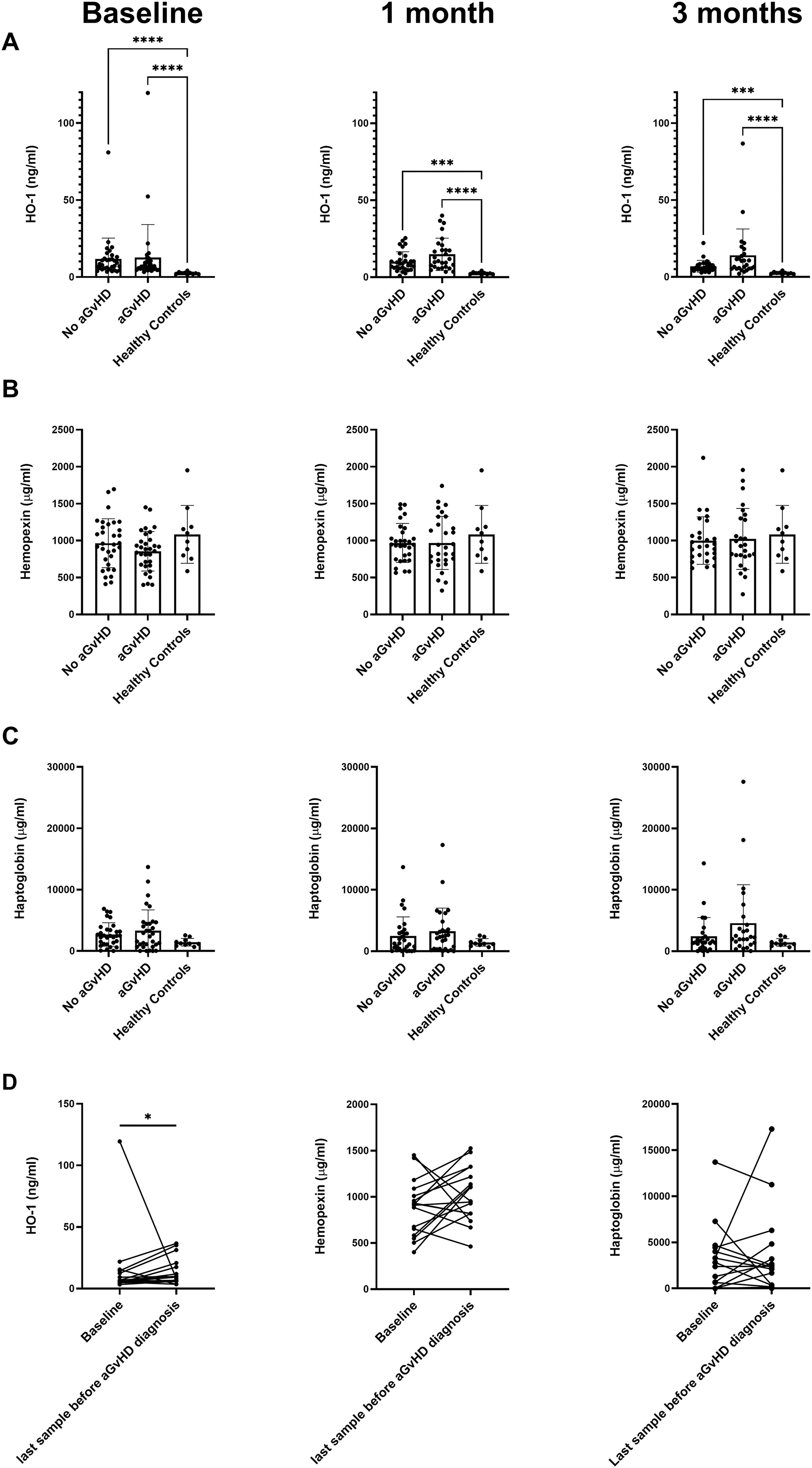
Plasma levels of HO-1 are increased in allogeneic HSCT recipients and at time of acute GvHD diagnosis. Plasma levels of HO-1
Patients with acute GvHD have increased HO-1 expression levels but not protein levels
As HO-1 is an intracellular enzyme, we then performed flow cytometry experiments (gating strategy can be seen in Supplementary Fig. S1) to study intracellular protein levels of HO-1 in PBMC samples taken from 20 allogeneic HSCT recipients. Expression of HO-1 was exclusively found in the different monocyte populations and was virtually absent in all lymphocyte populations (Supplementary Fig. S2). The percentage of HO-1-expressing cells was highest in the intermediate monocyte population (CD14+CD16+), and lower in the classical (CD14+CD16−) and nonclassical monocyte (CD14−CD16+) populations. We found no significant differences in the percentage of HO-1+ cells in the three different monocyte populations when comparing patients with acute GvHD to patients without acute GvHD at baseline or at 1 month or 3 months after allogeneic HSCT (Supplementary Fig. S3). The overall cell composition in the PBMC samples was comparable in all patients (Supplementary Fig. S4). We also measured overall expression of HO-1 in the PBMC samples by qPCR. No significant difference in HO-1 expression levels at baseline between patients with and without acute GvHD was found. In contrast to the flow cytometry data, however, we did find a significantly higher expression level of HO-1 in patients with acute GvHD at 1 and 3 months after allogeneic HSCT (Fig. 2).
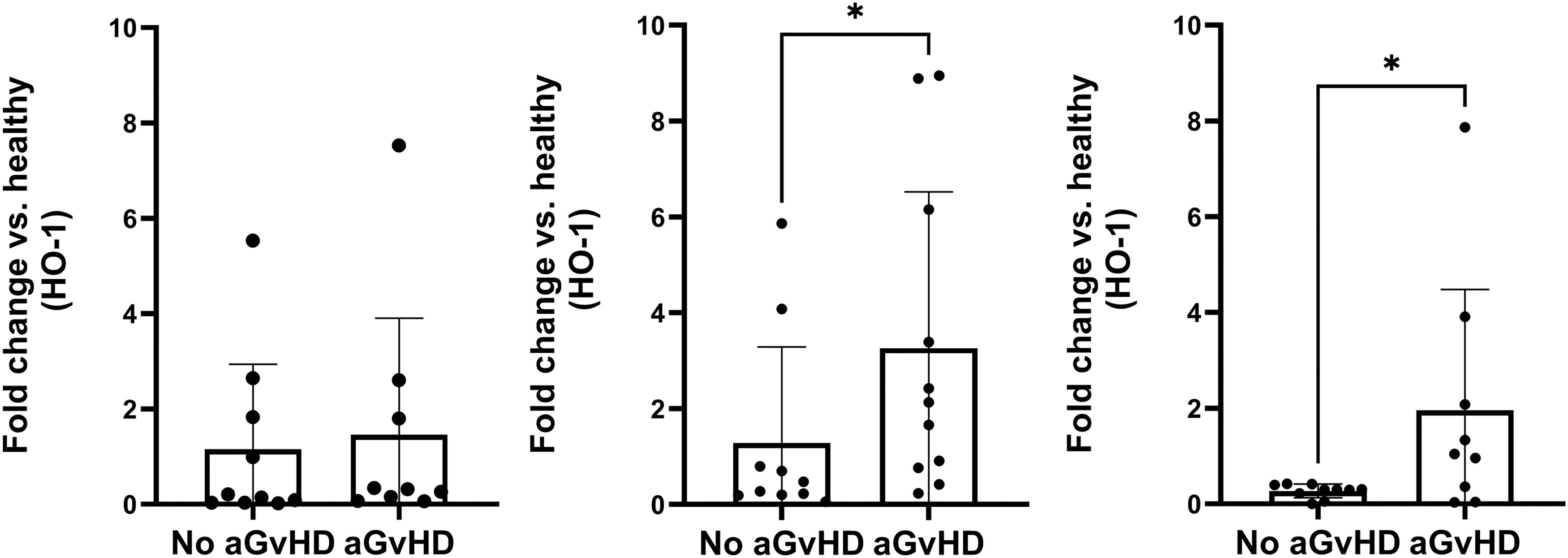
Fold-change expression of HO-1 is higher in PBMC from patients with acute GvHD. The fold-change expression of HO-1 is shown for PBMC samples from patients with (n = 10) and without (n = 10) acute GvHD taken at baseline (left), 1 month after allogeneic HSCT (middle), and 3 months after allogeneic HSCT (right) compared with a pooled PBMC sample of 10 healthy donors. Data are derived from qPCR experiments. Data are shown as mean ± SD. *P < 0.05. GvHD, graft-versus-host disease; HO-1, heme oxygenase-1.
HO-1 induction seems to improve survival, disease score, and weight loss in a humanized mouse model for acute GvHD
To study the effects of HO-1-induction on the development of acute GvHD in vivo, we made use of a humanized mouse model. Male NSG mice were first irradiated and then injected with human PBMC to induce acute GvHD. Mice injected with human PBMC developed symptoms of acute GvHD a median of 16 days (range 12–20) after injection. HO-1 expression was induced on days −2, −1, and 7 by i.p. injections with 5 mg/kg bodyweight CoPP (Fig. 3A). CoPP injections were generally well-tolerated and did not lead to visible discomfort. Survival of mice treated with CoPP (n = 16) seemed to improve compared with mice that were left untreated (n = 16), but the difference was not significant. We found a significant improvement in disease severity score and weight loss in the CoPP-treated group compared with the untreated control group (Fig. 3C, D). Histopathological analysis showed lesions of acute GvHD irrespective of whether mice received CoPP or not. Lesions of acute GvHD presented as hepatitis characterized by local to extensive leukocyte infiltrations accompanied by necrosis of hepatocytes and mild to moderate fibrosis of the liver parenchyma. Partial to severe depletion of hematopoiesis was observed in bone marrow as well as in spleen (Fig. 3E). Mild colitis was also encountered in all groups. The radiation prior to the experiment had a mild effect on weight as can be seen by the increased initial weight loss in the PBS control group (n = 12) compared with the PBS radiation control group (n = 6) (Fig. 3D).
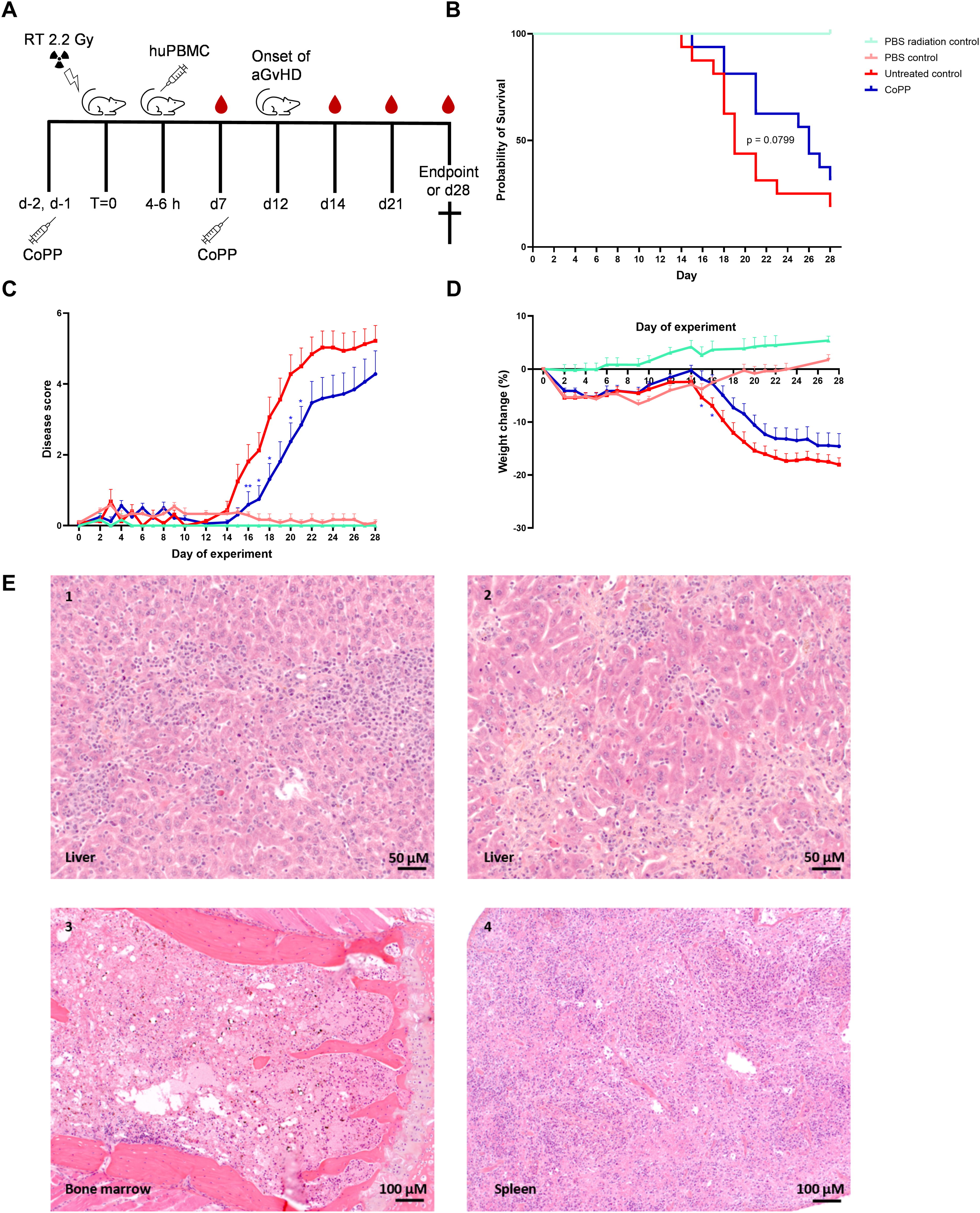
HO-1 induction improves survival, disease score, and weight loss in NSG mice with acute GvHD.
Mice treated with CoPP show reduced levels of chimerism but no changes in nucleosome levels
Blood was drawn weekly starting on day 0 of the experiment and on the day of offering to monitor levels of donor chimerism (measured by %huCD45+ cells) and inflammation (measured by plasma nucleosome levels). On the day of offering, mice treated with CoPP (n = 16) showed significantly lower donor chimerism compared with the untreated control mice (n = 16) (Fig. 4A). Interestingly, no significant differences were found in the level of nucleosomes on the day of offering between both groups (Fig. 4B). A significant correlation was found between the level of chimerism and the level of nucleosomes in the plasma (Fig. 4C), which is in line with our previous findings. 6
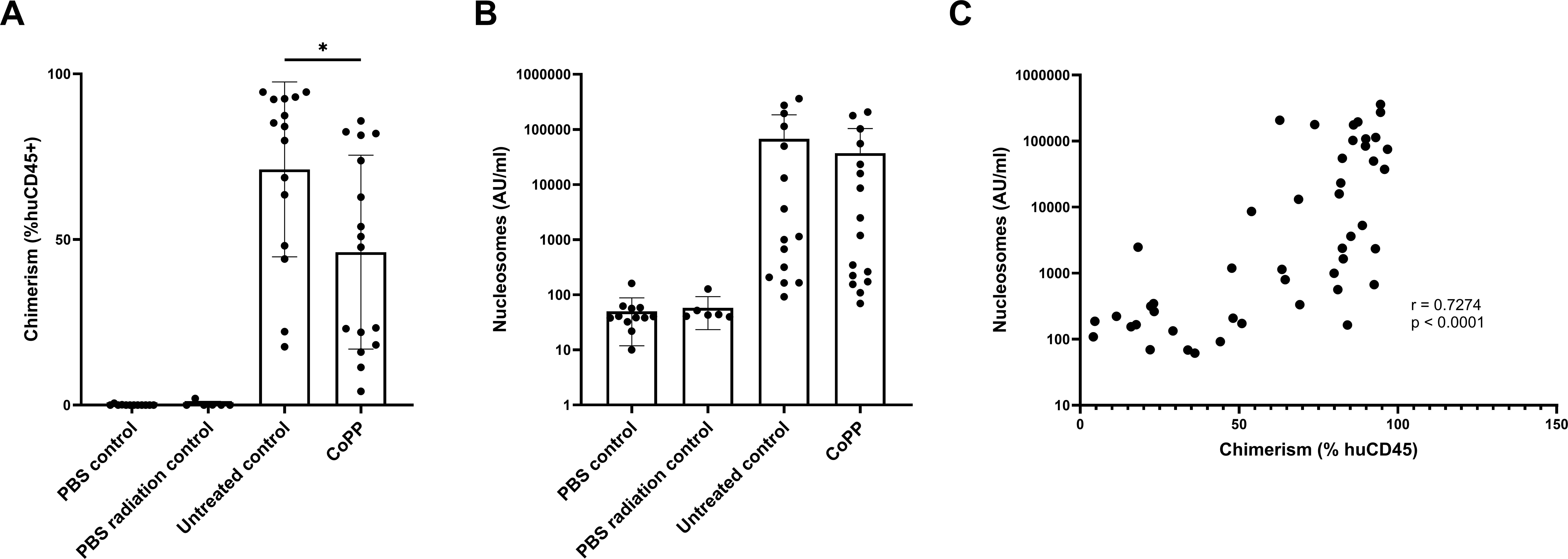
Treatment with CoPP leads to a reduction in the percentage of chimerism but not in nucleosome levels. Levels of chimerism measured by the percentage of human CD45+ cells in blood
Discussion
The development of acute GvHD is, at least in part, driven by pro-inflammatory signals from DAMPs that are released from damaged cells and tissues of the patient due to the conditioning regimen that is given prior to transplantation. 3 Here, we aimed to determine the importance of HO-1, an anti-inflammatory enzyme responsible for the rapid breakdown of cfheme (a potent DAMP) in acute GvHD pathogenesis. In the present study, we measured extracellular and intracellular protein levels of HO-1 as well as HO-1 expression in a cohort of allogeneic HSCT recipients and analyzed the effects of HO-1 induction in a humanized mouse model for acute GvHD.
We found a general increase in plasma HO-1 levels in patient samples compared with samples of healthy controls. As active transport of HO-1 out of cells has, to our knowledge, not been described, this is most likely due to the enhanced tissue damage and cell death seen in patients, leading to release of intracellular HO-1 into the extracellular space. Levels of HPX did not differ between patients with or without acute GvHD and healthy controls. While previous studies have shown that low-grade hemolysis and/or disintegration of mitochondria during cell death can lead to the release of cell-free heme and a subsequent consumption of HPX, this does not seem to be the case in our cohort of allogeneic HSCT recipients. Plasma levels of HPT were generally higher (although not significant) in the patient groups compared with the healthy controls. These data are in line with what we could observe in patients with sepsis, albeit that the difference between sepsis patients and healthy controls was significantly larger. 17 HO-1 levels in plasma were increased in patients shortly before their acute GvHD diagnosis compared with baseline, while no such differences were seen for HPX or HPT. As acute GvHD is characterized by widespread tissue damage, this is most likely due to release of HO-1 from damaged cells.
Our flow cytometric analysis showed that HO-1 is predominantly expressed in the monocyte lineage. We found no differences between patient groups in the percentage of HO-1-expressing classical monocytes, intermediate monocytes, or nonclassical monocytes at any of the time points. When investigating the expression of HO-1 in total PBMC by qPCR, we observed a significant increase of expression in patients with acute GvHD at 1 and 3 months after transplantation. As most patients developed acute GvHD within this 1–3 month window after allogeneic HSCT, this indicates that the development of acute GvHD coincides with an overall elevation in HO-1 expression. These data also seem to indicate that cellular stress induces an increase in the expression of HO-1 in cells that already express the enzyme, but that it does not lead to novel expression of HO-1 in cells. However, it is also possible that the sensitivity of our flow cytometry assay is limited, preventing us from detecting possible subtle differences between groups. As HO-1 is generally seen as a protective enzyme, these data may seem difficult to interpret. However, HO-1 is a strongly inducible enzyme that responds to a wide range of cellular stressors. 9 As such, it is to be expected that expression of HO-1 increases when patients suffer from acute GvHD, a major causative agent of cellular stress.
In a humanized model for acute GvHD, we were able to show that induction of HO-1 by CoPP (shown to be a potent inducer of HO-1 18 ) leads to a subtle but significant decrease in disease severity and weight loss. Interestingly, we did not see this effect reflected in plasma levels of nucleosomes, a known DAMP and marker for inflammation. We observed a decrease in the percentage of chimerism in the group treated with CoPP. The question whether these differences are due to decreased expansion of human cells or a decrease in death of mouse cells remains open. The decrease in chimerism in the CoPP-treated group may also suggest that HO-1 induction could prevent proliferation of human PBMC in this model, thereby reducing the severity of acute GvHD. However, further experiments are required to determine this and to explore options for clinical application.
In conclusion, we found increased levels of HO-1 in the plasma of allogeneic HSCT recipients compared with plasma of healthy donors and in patients around the time of acute GvHD diagnosis compared with baseline. We found no differences in the percentage of HO-1+ monocytes, but did see an increase in overall HO-1 expression in patients with acute GvHD at 1 month and 3 months after transplantation compared with patients without acute GvHD. In a humanized mouse model for acute GvHD, HO-1 induction significantly reduced the severity of acute GvHD and disease-related weight loss. Overall, our data indicate that expression of HO-1 might be able to provide protection against- or reduce the development of acute GvHD and that the induction of HO-1 in patients undergoing allogeneic HSCT could be a valuable treatment modality for acute GvHD to explore further.
Footnotes
Acknowledgment
The authors would like to thank N.V.D. Brule for providing us with patient information.
Author Disclosure Statement
The authors declare no competing financial interests.
Funding Information
This work was financially supported by Landsteiner Foundation for Blood Transfusion, Research Grant F1719 (M.V., S.Z., I.B., and C.V.) and AMC foundation Grant 20868 (S.Z. and C.V.).
Supplementary Material
Supplementary Figure S1
Supplementary Figure S2
Supplementary Figure S3
Supplementary Figure S4
Supplementary Table S1
