Abstract
Autologous hematopoietic stem cell transplantation is used to restore bone marrow function after high-dose chemotherapy. For apheresis, granulocyte colony-stimulating factor (G-CSF) is standard of care, but obtaining sufficient stem cells can be challenging. Other mobilization agents include plerixafor and PEGylated G-CSF (PEG-G-CSF). While efficacy of these is established in adults, limited data for their use in pediatric patients are available. Here, we compare Good versus Poor Mobilizers and study success of different mobilization regimens in regard to CD34+cell-collection, -quality, -phenotype and hematologic reconstitution in pediatric patients. In this multi-center retrospective study, we analyzed data of 278 patients with solid tumors and lymphoma, mobilized with either G-CSF (n = 224), PEG-G-CSF (n = 34), or G-CSF/PEG-G-CSF with additional plerixafor (n = 20). In Poor Mobilizers (13.7% of all patients), addition of plerixafor to G-CSF augmented CD34+cell collection, without adverse effects on hematologic reconstitution and CD34+cell quality. PEG-G-CSF-aided mobilization was successful as first-line treatment in two-thirds of patients and did not impair hematological reconstitution, compared to G-CSF-only. Within the Poor Mobilizer group, G-CSF+plerixafor increased primitive (CD45RA-CD38-CD90+CD49f+) and CXCR4-expressing CD34+cells in apheresis products compared to G-CSF-only, without exceeding levels of Good Mobilizers. No plerixafor-related increase in tumor cells was observed in apheresis products. In conclusion, our comprehensive study supports the use of plerixafor and furthermore demonstrates the potential of patient-friendly PEG-G-CSF for mobilization of pediatric patients.
Introduction
Autologous hematopoietic stem cell transplantation (aSCT) aims to restore bone marrow (BM) function after high-dose chemotherapy and is used in high-risk or recurrent pediatric tumors. This involves mobilization and collection of sufficient hematopoietic stem and progenitor cells (HSPCs; CD34+cells) that are capable of rapid and lasting hematologic reconstitution. Granulocyte colony-stimulating factor (G-CSF; Filgrastim) is commonly used to mobilize CD34+cells from the BM into the circulation. However, 10%–30% of pediatric cancer patients are Poor Mobilizers, with insufficient numbers of harvested CD34+cells. 1,2 Predicting poor mobilization remains a challenge 3 and little is known about the intrinsic factors resulting in poor mobilization.
Other mobilization agents include (off-label) use of PEGylated G-CSF (PEG-G-CSF; pegfilgrastim) and plerixafor. The addition of polyethylene glycol (PEG) to G-CSF prolongs half-life from 3.5 h to 15–80 h, offering benefits in terms of fewer injections and consequently reduced injection-related stress. 4,5 Plerixafor is a CXCR4-antagonist that disrupts binding of CD34+cells to stromal cells. 6 Based on successful use in adults, plerixafor has been used for pediatric Poor Mobilizers off-label since 2009 and is approved by the European Medical Agency for use in children since 2019. 7 The efficacy and safety of both agents are well established in adults, 8 –13 but limited data for its use in pediatric patients with solid tumors are available. In children, successful mobilization and hematological reconstitution after use of PEG-G-CSF 14 –17 and plerixafor 1,18 –24 are reported, but the cohort size is small in most studies, and detailed analyses on graft composition and quality are often lacking.
In this retrospective multi-center study, we compared the success of CD34+cell collection with different mobilization regimens (ie, G-CSF vs. PEG-G-CSF with or without addition of plerixafor in the subgroup of Poor Mobilizers) and determined the effect on hematological reconstitution after aSCT in pediatric cancer patients. This was combined with a comprehensive analysis of mobilized CD34+cells by studying viability, clonogenic capacity, and phenotype.
Materials and Methods
Study population
In this retrospective multi-center study, data were collected from 278 Dutch pediatric patients with solid tumors, brain tumors and lymphomas, scheduled for mobilization in the Academic Medical Center Amsterdam (Institute 1), Princess Máxima Center Utrecht (Institute 2), and University Medical Center Groningen (Institute 3) between 2000 and 2019 (Table 1, Supplementary Table S1, and Fig. 1). The institutional review board of Institute 2 approved the study. The study was conducted in accordance with the ethical guidelines of the Declaration of Helsinki. Informed consent for use of clinical data collected in standard of care approaches and for additional experimental procedures was obtained from patients, parents, or legal representatives.
Patient Characteristics Good and Poor Mobilizers
Chi square test.
median test.
prior to apheresis.
at the start of stem cell mobilization.
Patients were classified as Poor Mobilizers when they did not meet the criteria to proceed to apheresis (see Methods), when plerixafor was needed to reach these criteria, or when the harvest yield was <2*106 CD34+cells/kg.
BM, bone marrow; MIBG, Meta-Iodo-Benzyl-Guanidine.
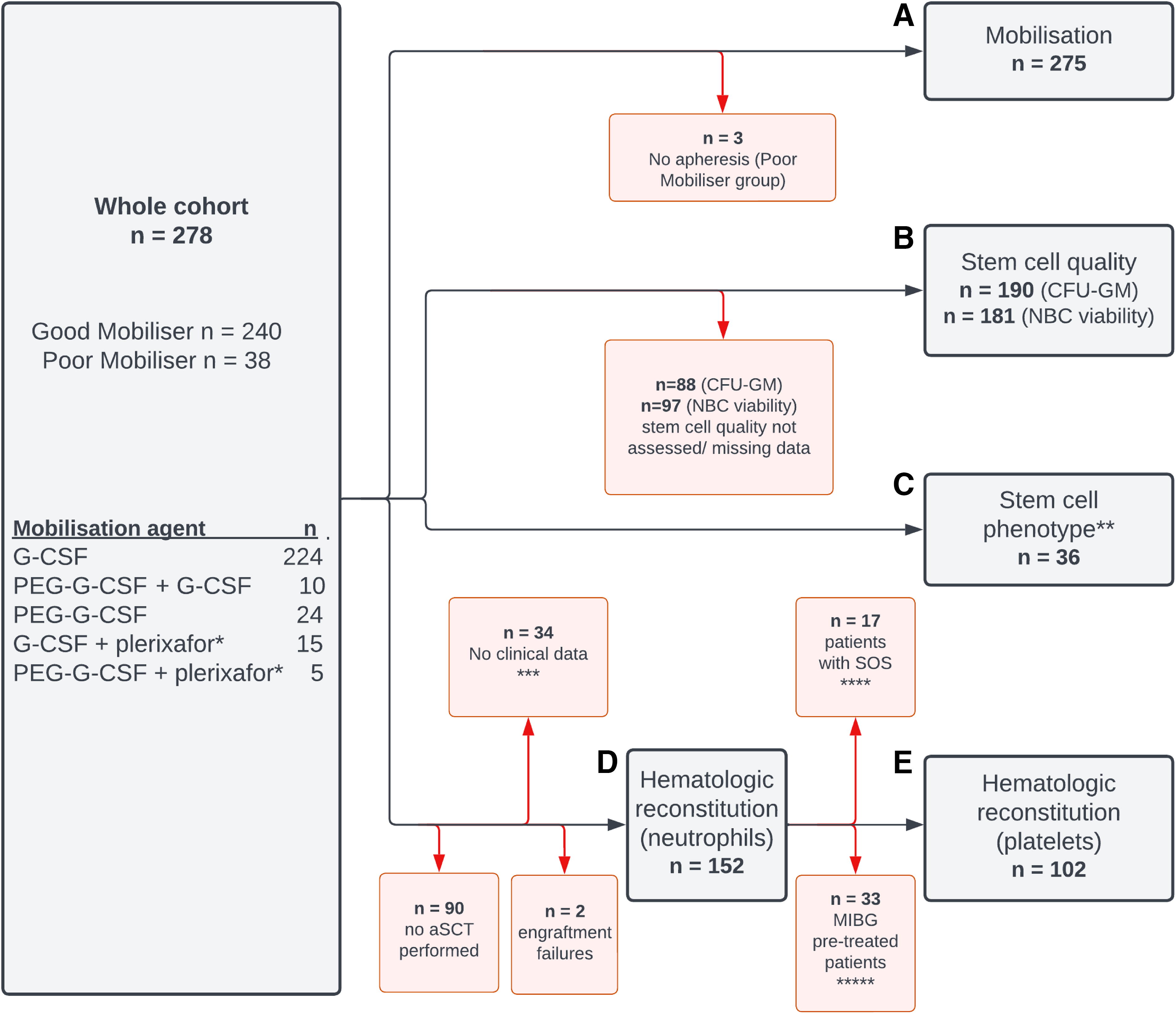
Overview of patient cohort. The number of patients included (and reasons for exclusions) are indicated for analysis of
Apheresis and hematologic reconstitution
Mobilization was performed according to local standard of care protocols (Supplementary Fig. S1), using G-CSF or PEG-G-CSF, aiming to collect >2*106, >6*106 or >9*106 CD34+cells/kg, depending on the treatment protocol. PEG-G-CSF was the standard of care at Institute 2 from 2019 onwards, with the aim of reducing treatment burden. Eligibility criteria to proceed to apheresis were, for Institute 1 and 3: peripheral blood (PB)-CD34+cell counts >0.1% of mononuclear cells and leukocytes ≥2*109/L, and for Institute 2: PB-CD34+cell counts ≥20 cells/μL. If these criteria were not met or if harvest failure was expected, mobilization regimens were adjusted (Supplementary Fig. S1): G-CSF was doubled to twice-daily 10 mcg/kg. In case of previous PEG-G-CSF administration, G-CSF was added. Plerixafor 0.24 mg/kg/day was administered for 2 days (median, range 1–4) to patients who still failed to reach PB-CD34+cell thresholds or after insufficient harvest yield in a prior apheresis session. For this study, patients were classified as Poor Mobilizers when they did not meet the criteria to proceed to apheresis, when plerixafor was needed to reach these criteria, or when the harvest yield was <2*106 CD34+cells/kg. An apheresis session could consist of multiple consecutive apheresis-days.
Hematologic reconstitution post-aSCT was defined as a neutrophil count >0.5*109/L and platelet count >20*109/L (unsupported by transfusions), according to European Society for Blood and Marrow Transplantation criteria 26 (see Supplementary Data).
Graft quality
Nucleated blood cell (NBC) viability after cryopreservation was determined by flow cytometry (Institute 2 and Institute 3) or by trypan blue exclusion (Institute 1). In Institute 1 and 2, progenitor capacity was assessed by colony forming unit-granulocyte macrophage (CFU-GM) assays, which were performed before (Institute 1) or after (Institute 2) cryopreservation at local stem cell facilities, according to protocol (Supplementary Data).
Phenotypic testing
Samples were gradually thawed to room temperature in IMDM/20% FCS medium with DNAse (100 µg/mL) and magnesium chloride (5 mM). After a 30-minute recovery period, cells were washed and stained —according to the flow cytometry protocol (Supplementary Data)—for 13 markers in order to analyze: (1) viability, (2) expression of homing- and adhesion molecules (CD49d, CD49e, CXCR4), (3) stemness (CD45RA, CD38, CD90, CD49f 27 ), (4) proliferation (Ki-67), and (5) lineage commitment (CD33, CD41, CD62L 28 ) (Supplementary Figs. S2 and S3).
Statistical analysis
Characteristics between poor- and good- mobilizers were investigated using Chi-Square tests for nominal variables and median tests for continuous variables. To assess predictors for mobilization, univariate logistic regression models were estimated and odds ratiosalong with 95% confidence intervalswere reported. Differences in surface marker expression were compared by Mann–Whitney U tests (2 groups) or Kruskal–Wallis tests (3 groups). For reverse transcription-quantitative polymerase chain reaction (RT-qPCR) results, Fisher’s exact test was used to compare categorical variables between different groups. The level of significance was set at 0.05. Statistical analyses were performed using SPSS version 25 and GraphPad Prism version 9.1.1.
Results
We included 278 patients (median age 5 years, range 0–19) with various solid tumors (majority neuroblastoma), brain tumors and lymphomas diagnosed between 2000 and 2019 at three Dutch institutes (Table 1, Supplementary Table S1, and Fig. 1
Mobilization
First, apheresis data were compared (Table 2, Supplementary Table S2, and Supplementary Fig. S4). In total, 38 patients (13.7%) were classified as Poor Mobilizers (Table 1). To investigate which factors were associated with poor mobilization, we estimated univariate logistic regression models. Low leukocyte count at start of mobilization, neuroblastoma diagnosis, prior chemotherapy and BM metastasis at both diagnosis and start of mobilization were significant predictors of poor mobilization (Supplementary Table S3).
Apheresis
Combined analysis of aphereses of all institutes. Harvest yield indicates the median per apheresis day. For Good versus Poor Mobilizer analyses, data from successful first apheresis sessions without plerixafor were included. For PEG-G-CSF analyses, all sessions upon PEG-G-CSF-aided mobilization without plerixafor were included. For plerixafor versus G-CSF analyses, all sessions upon plerixafor-aided mobilization were included, excluding PEG-G-CSF-treated patients (n = 5). Right panel: paired data (before and after plerixafor).
PEG-G-CSF only: n = 24, PEG-G-CSF+G-CSF: n = 11 (includes apheresis data of one patient of the PEG-G-CSF+plerixafor-group who did not yet receive plerixafor).
based on 5 patients for which paired harvest yield data were available. For analysis of patients that received PEG-G-CSF and additional plerixafor, see Supplementary Fig. S4 and Supplementary Table S5.
G-CSF, granulocyte colony-stimulating factor; PEG-G-CSF, pegylated G-CSF.
To evaluate PEG-G-CSF-based mobilization, we conducted a sub-analysis of data from 2014 to 2019 of Institute 2 (Supplementary Table S4), as PEG-G-CSF was only implemented in Institute 2 in more recent years, and apheresis protocols were most comparable from 2014 onwards. This sub-analysis showed a median harvest yield of 5.89*106 CD34+cells/kg for G-CSF (n = 81), 9.99*106 CD34+cells/kg for PEG-G-CSF-only (n = 25) and 7.38*106 CD34+cells/kg for PEG-G-CSF+G-CSF (n = 11; Supplementary Table S4). Although the CD34+cell collection goal could have differed for individual patients, so comparisons between groups can therefore not be made, the data demonstrate that PEG-G-CSF-aided mobilization as first-line treatment was at least comparable to G-CSF, and in two-thirds of patients no additional G-CSF was needed.
For 20 Poor Mobilizers (52.6%), plerixafor was added to G-CSF (n = 15; Table 2) or PEG-G-CSF (n = 5; Supplementary Table S5). Plerixafor was administered during the first apheresis session (n = 9) or a follow-up session (n = 11), resulting in a successful mobilization in 75% of patients (Table 3). In 9 patients with matched samples (before and after plerixafor), a median 3.8-fold increase in PB-CD34+cell count was observed after plerixafor administration (Table 2). For 5 out of these 9 patients with paired samples, harvest yield data was available, showing a median 3.7-fold higher CD34+cell harvest yield (Table 2). No significant adverse events were observed during the mobilization procedure. Thus, our findings demonstrate that addition of plerixafor to G-CSF augmented CD34+cell collection.
Success of Mobilization with Plerixafor in Poor Mobilizers
Apheresis session can consist of multiple consecutive apheresis days. Successful harvest: > 2*106 CD34+cells/kg (cumulative of a maximum of three consecutive sessions).
Graft quality
For 181 patients, viability of the harvested NBCs was assessed. As protocols and median NBC viability values differed among institutes (Supplementary Table S6), these analyses were performed per institute (Table 4 and Supplementary Fig. S5). When comparing either Poor- versus Good Mobilizers or G-CSF+plerixafor- versus G-CSF-treated Poor Mobilizers no differences were observed, but NBC viability was relatively lower in the PEG-G-CSF-group compared to the G-CSF-group: 68% versus 80%. Nevertheless, the clonogenic capacity (CFU-GM) was comparable for all groups (Table 4 and Supplementary Fig. S5).
Graft Quality and Hematological Reconstitution
For PEG-G-CSF analyses, patients that received PEG-G-CSF in combination with plerixafor were excluded (n = 5). Reconstitution data from all reinfusion sessions combined. CFU-GM data and reconstitution data were not available for Institute-3. In Institute-2, 5 patients (graft quality data) and 2 patients (hematologic reconstitution data) received plerixafor in combination with PEG-G-CSF instead of G-CSF. Exclusion criteria: see flowchart in Figure 1.
Institute-2 only.
Institute-1 only.
NA, no patients of Institute-1 were treated with PEG-G-CSF; CFU-GM, Colony Forming Unit Granulocyte/ Macrophage; NBC, nucleated blood cell.
Hematologic reconstitution
The median time to neutrophil- and platelet-reconstitution after aSCT was respectively 11 and 14 days in Good Mobilizers versus 12 and 20 days in Poor Mobilizers (Table 4 and Supplementary Fig. S6). For the PEG-G-CSF group, the median time to reconstitution was comparable to the G-CSF group. In the plerixafor-treated Poor Mobilizers, the median time to neutrophil- and platelet-reconstitution was 12 and 15 days, respectively. Although the number of patients in the plerixafor group was insufficient to draw definitive conclusions about comparisons with the Poor Mobilizers-control-group (Table 4 and Supplementary Fig. S6), the data suggest that plerixafor has no adverse effect on hematological reconstitution.
CD34+cell phenotype
We additionally performed a detailed phenotypic analysis of mobilized CD34+cells to get insights into potential alterations that might underly poor mobilization and into the effect of adding plerixafor to mobilization regimens. For a selection of 36 patients (Supplementary Table S7), an extensive flow cytometry analysis was performed on cryopreserved apheresis aliquots (Supplementary Fig. S2, Supplementary Fig. S3).
In post-thaw apheresis samples of Poor Mobilizers, a lower percentage of viable cells within the CD34+cell population was found compared to Good Mobilizers (Fig. 2A). When testing the viable CD34+cells for adhesion/homing markers (Fig. 2B–D), a minor decrease by 0.6% in CD49e-expressing CD34+cells was observed (Fig. 2C) and a more substantial, albeit not statistically significant, decrease by 12.5% in CXCR4-expressing CD34+cells in Poor-compared to Good-Mobilizers (Fig. 2D). To study the percentage of primitive stem cells in the apheresis product, we gated for CD34+CD45RA-CD38-cells and further distinguished between CD90+CD49f+ long-term-HSCs and CD90-CD49f
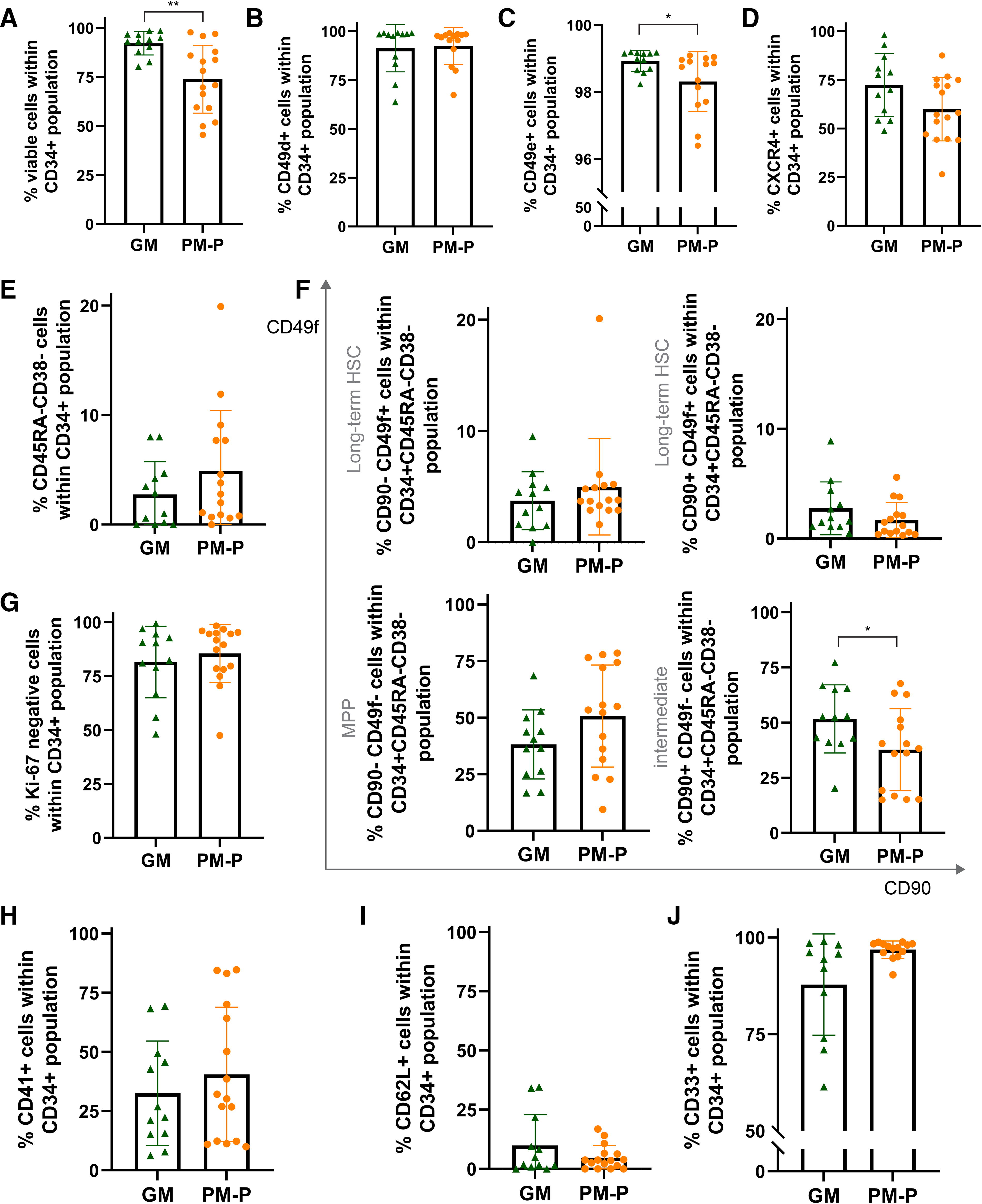
Phenotype of CD34pos cells in apheresis products of Good versus Poor Mobilizers. Flow cytometric analysis on cryopreserved reference aliquots of a subgroup of Good Mobilizers (GM) and Poor Mobilizers not treated with plerixafor (PM-P).
To assess whether plerixafor influences mobilization of (specific) CD34+cell subpopulations, we compared Poor Mobilizers treated with plerixafor to those who did not receive plerixafor, analyzing apheresis samples from different patients. The viability of CD34+cells did not differ between the groups (Fig. 3A). Interestingly, the homing molecule CXCR4 was expressed in greater intensity per cell (Supplementary Fig. S7A) and on a higher percentage of CD34+cells in the plerixafor-group (Fig. 3D), reaching expression levels similar to Good Mobilizers (Supplementary Fig. S7B). Analysis of primitive HSC-markers indicated a modest preferential mobilization of long-term-HSCs in the plerixafor-group: the most primitive CD90+CD49f+ population was increased, while the more committed CD90−CD49f− MPP population was decreased, albeit not statistically significant (Fig. 3F). Importantly, the primitive HSC subsets of Poor Mobilizers treated with plerixafor did not significantly differ from the Good Mobilizer group (Supplementary Fig. S7C and D). For proliferation- and lineage commitment-markers, no differences were observed (Fig. 3G–J). Taken together, our findings demonstrate that within the Poor Mobilizer group, G-CSF+plerixafor increased the levels of primitive and CXCR4-expressing CD34+cells in apheresis products compared to G-CSF-only, without exceeding the levels observed in the Good Mobilizer group.
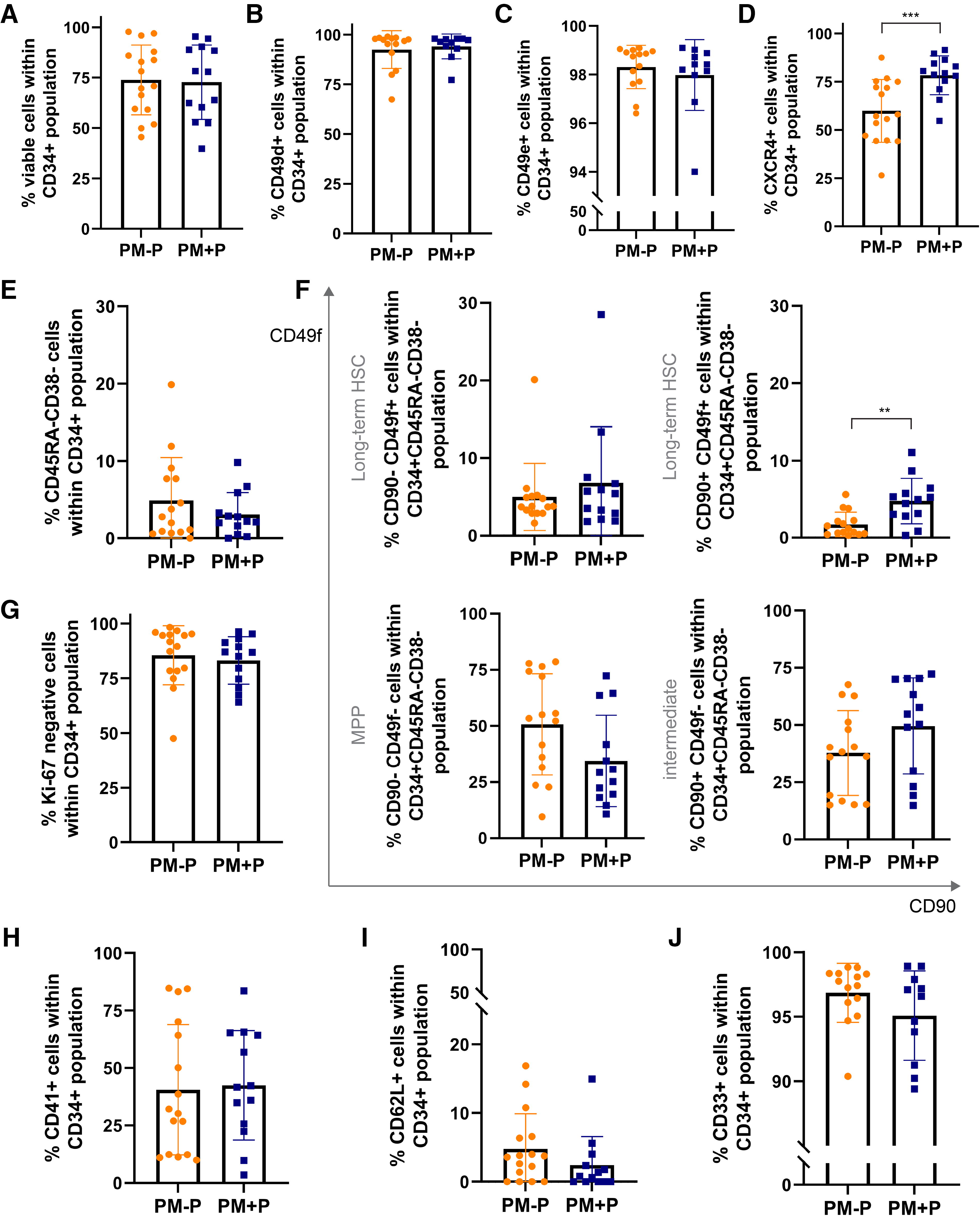
Phenotype of CD34pos cells in apheresis products of Poor Mobilizers that received plerixafor. Flow cytometric analysis on cryopreserved reference aliquots of a subgroup of Poor Mobilizers (PM) treated with (PM+P) and without plerixafor (PM-P, same group as in Fig. 2). Comparison of different patients; no paired data.
Low risk of unintentional tumor cell mobilization by plerixafor
Since plerixafor blocks the CXCR4/CXCL12 interaction and some tumors including neuroblastoma also express CXCR4, the concern was raised that tumor cells might unintentionally be mobilised from BM concomitantly with HSPCs. Therefore, we analyzed tumor cell presence in cryopreserved apheresis aliquots of a subgroup of 19 patients with neuroblastoma by RT-qPCR (Supplementary Data). This revealed that 22% (2/9) of the samples were positive for neuroblastoma-markers in the plerixafor-group, while 40% (4/10) were positive in the control-group (G-CSF-only) (Fig. 4). Of note, RT-qPCR-positive apheresis samples contained on average only 0.02% tumor cells (range 0.005%–0.2%, unpublished results). Overall, we did not observe a plerixafor-related increase in tumor cell presence in apheresis products.
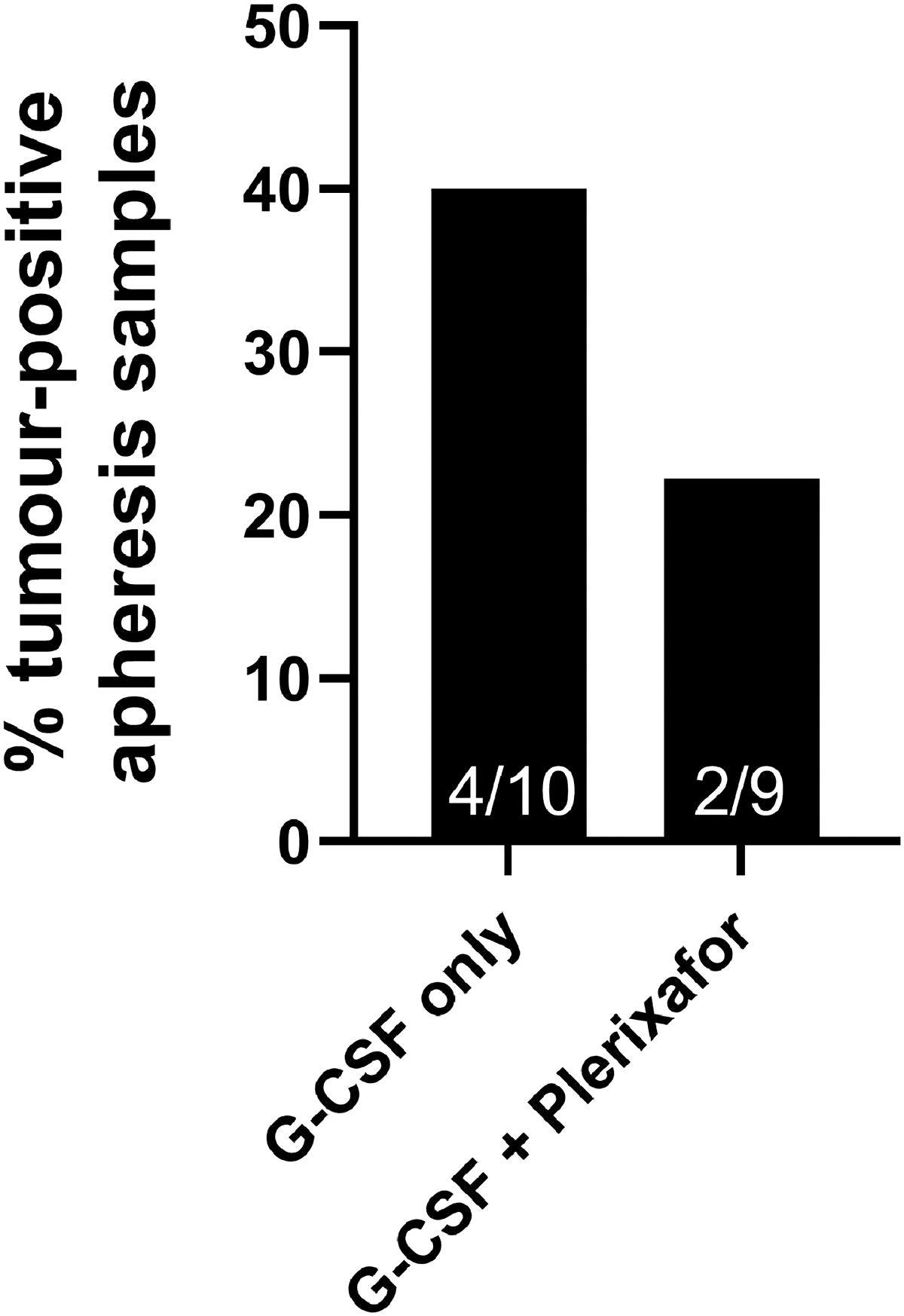
Assessment of tumor cell presence in apheresis material of patients treated with and without plerixafor. Comparison of cryopreserved reference apheresis aliquots of a subgroup of 19 neuroblastoma patients: 10 mobilized with G-CSF-only and 9 with G-CSF + plerixafor. Tumor-positivity was determined by RT-qPCR, using a NB-specific marker panel (Supplementary Data). Bar graph shows percentage of RT-qPCR-positive apheresis samples within each group. Number of positive samples (/of total) is indicated at the bottom of each bar. Statistical analysis was performed using the Mann–Whitney U test. Results were not significant. RT-qPCR, reverse transcription-quantitative polymerase chain reaction.
Discussion
In this retrospective multi-center study, we had the unique opportunity to compare Good versus Poor Mobilizers and evaluate the success of PEG-G-CSF and plerixafor administration in pediatric cancer patients; not only regarding mobilization efficiency, but also hematologic reconstitution and CD34+cell characteristics such as clonogenic capacity, viability, stemness, quiescence, lineage commitment, and expression of homing- and adhesion molecules.
In line with 10%–30% reported in other studies, 1,2 13.7% of the patients were classified as Poor Mobilizers. Our search for predictors of poor mobilization identified both known (tumor infiltration in BM 25,29 and prior chemotherapy 25,30 ) and novel (neuroblastoma diagnosis) risk factors. Notably, the incidence of BM tumor infiltration at the time of neuroblastoma diagnosis is high, particularly in high-risk neuroblastoma. This suggests that tumor cells may disrupt processes in the BM niche related to HSC retention, potentially contributing to poor mobilization. Further studies with larger cohorts are needed to determine whether neuroblastoma diagnosis is an independent risk factor for poor mobilization. Upon reinfusion of the harvested CD34+cells, our data indicate a minor delay in hematologic reconstitution in Poor Mobilizers compared to Good Mobilizers, albeit not significant like for adults. 31,32 This may be attributed to the reduced viability of CD34+cells in post-cryopreservation apheresis samples from Poor Mobilizers, as detected by flow cytometry in the CD34+ population but not in the total nucleated blood cell pool. Apart from that, the flow cytometric analysis did not uncover substantial intrinsic differences in collected CD34+cells of Poor versus Good Mobilizers, for example, with respect to homing- and adhesion molecules.
Adding plerixafor to G-CSF, resulted in a 75% success rate which is comparable to the published success rates ranging from 66.7 to 93.9%. 1,3,18 –20,22 –24 Our study further confirmed successful hematologic reconstitution after reinfusion of plerixafor-mobilised CD34+cells in pediatric patients. 19,20 Phenotypically, plerixafor-mobilised CD34+cells differed from the Poor Mobilizers control group only in a few surface markers, among which the increased expression of the homing molecule CXCR4—both on a per-cell basis and in a higher percentage of cells—was the most prominent. This might be explained by the fact that plerixafor is a CXCR4-antagonist and may therefore lead to decreased retention of CXCR4-expressing CD34+cells in BM and thus a concomitant rise in the apheresis product. A second difference was an increased proportion of primitive CD90+CD49f+ long-term HSCs in the plerixafor-group. Although our findings on primitive HSCs should be treated with caution due to the low number of cells in this subpopulation, they are in line with adult studies reporting increased mobilization of primitive subsets 33 –36 and increased CXCR4 expression 34 upon plerixafor. Contradictory findings are also reported, 34,37,38 possibly reflecting differences in sample condition (post-thaw vs. pre-cryopreservation), control-groups (Poor vs. Good Mobilizers) and/or disease context (cancer type and pediatric vs. adult). However, based on literature, 34 our findings of increased CXCR4 expression as well as CD90+CD49f+ long-term HSCs are expected to have, if any, a favorable effect on engraftment. Moreover, levels of these CD34+cell subsets in the plerixafor-treated Poor Mobilizers did not significantly exceed the levels of Good Mobilizers, indicating that they are qualitatively equivalent to cells collected after traditional G-CSF mobilization in Good Mobilizers. Lastly, like others, 3 we did not find indications for plerixafor-related mobilization of neuroblastoma tumor cells into peripheral blood.
Analysis of the PEG-G-CSF-treated patient group revealed that CD34+cell collection and hematologic reconstitution were comparable to G-CSF, corroborating other small pediatric studies showing safe and effective stem cell mobilization with PEG-G-CSF. 15,16,39 Given that PEG-G-CSF reduces the number of injections and is therefore considered more patient-friendly, our data suggest that PEG-G-CSF could be a favorable option for mobilization compared to G-CSF.
Comprehensive analyses of PEG-G-CSF and plerixafor are scarce in pediatric cohorts. This while optimization of mobilization for pediatric patients is of importance as studies examining two successive aSCTs (double/tandem transplants) are being performed with promising results [clinicaltrials.gov: NCT03165292, NCT04221035 40 ], underscoring the need to maximize stem cell yields and determining which patient is at risk for poor mobilization. The data presented here are thus relevant for clinical practice. Our study has, however, some limitations, which are as follows: (1) Our study population was very heterogeneous in terms of underlying disease and treatment prior to mobilization, and apheresis/treatment protocols changed in time; (2) a future larger cohort study is desirable, in particular to study platelet reconstitution in plerixafor-treated patients, due to the relatively small size of this subgroup; and (3) phenotypic analyses could not be performed for the PEG-G-CSF-group due to unavailability of sufficient reference apheresis aliquots.
Altogether, our study showed that administration of PEG-G-CSF as well as plerixafor (with addition of G-CSF) resulted in successful collection of CD34+cells and no adverse consequence on subsequent hematologic reconstitution after reinfusion, when compared to G-CSF. For plerixafor we additionally showed that there were no adverse effects on CD34+cell phenotype/characteristics and tumor cell mobilization. Therefore, our study, albeit using a limited patient number, supports the use of patient-friendly PEG-G-CSF and the addition of plerixafor for mobilization of stem cells in pediatric patients with harvest problems.
Patient Consent Statement
Informed consent for use of clinical data collected in standard of care approaches and for additional experimental procedures was obtained from patients, parents, or legal representatives.
Footnotes
Acknowledgments
The authors thank A. Javadi, S. Tol and N. Weterings for technical support.
Ethics Approval Statement
The Institutional Review Board of Princess Máxima Center Utrecht approved the study. The study was conducted in accordance with the ethical guidelines of the Declaration of Helsinki.
Author Disclosure Statement
The authors have no competing interests.
Funding Information
This work was supported by KiKa (grant number 374); Sanquin Research grant for Product and Process development for Cellular products (grant number 19–27). The sponsors had no involvement in study design or collection, analysis and interpretation of the data.
Supplementary Material
Supplementary Data
Supplementary Figure S1
Supplementary Figure S2
Supplementary Figure S3
Supplementary Figure S4
Supplementary Figure S5
Supplementary Figure S6
Supplementary Figure S7
Supplementary Table S1
Supplementary Table S2
Supplementary Table S3
Supplementary Table S4
Supplementary Table S5
Supplementary Table S6
Supplementary Table S7
