Abstract
Intravascularly transplanted bone marrow cells, including bone marrow mononuclear cells (BM-MNC) and mesenchymal stem cells, transfer water-soluble molecules to cerebral endothelial cells via gap junctions. After transplantation of BM-MNC, this fosters hippocampal neurogenesis and enhancement of neuronal function. Herein, we report the impact of transplanted BM-MNC on neural stem cells (NSC) in the brain. Surprisingly, direct transfer of water-soluble molecules from transplanted BM-MNC and peripheral mononuclear cells to NSC in the hippocampus was observed already 10 min after cell transplantation, and transfer from BM-MNC to GFAP-positive cortical astrocytes was also observed. In vitro investigations revealed that BM-MNC abolish the expression of hypoxia-inducible factor-1α in astrocytes. We suggest that the transient and direct transfer of water-soluble molecules between cells in circulation and NSC in the brain may be one of the biological mechanisms underlying the repair of brain function.
Introduction
Gap junctions connect the cytoplasm of two adjacent cells. They allow the rapid intercellular movement of small water-soluble molecules according to their concentration gradient. 1 The importance of permanent cytosol linkage between heterogeneous cells is well established for various cell types, such as the myocardium, smooth muscle cells, and astrocytes. However, although transient cytosol linkage between heterogeneous cells exists, its significance is still unclear. 2
We have previously demonstrated that direct transfer of water-soluble molecules between intravascularly transplanted bone marrow mononuclear cells (BM-MNC) and cerebral endothelium via gap junctions induces angiogenesis by activation of hypoxia-inducible factor-1α (HIF1α) in a subacute stroke model. 3 Moreover, we have shown that direct transfer of water-soluble molecules between transplanted bone marrow mesenchymal stem cells (BM-MSC) and cerebral endothelium suppresses inflammation in an acute stroke model. 4 A surprising observation was that the transient cytosol linkage between transplanted cells and endothelium occurred already 10 min after cell transplantation. Although this phenomenon does not last long, it may have an enduring impact on tissue repair. For example, we recently reported that transplantation of BM-MNC into aged mice improves neurogenesis at the hippocampus followed by improvement of neuronal function. 5,6 Collectively, these findings signify the role of transient and direct transfer of water-soluble molecules between heterogeneous cells in steady state and disease.
We have now investigated the link between transplanted BM-MNC and neural cells, that is, hippocampal neural stem cells (NSC) and cortical astrocytes, and found that direct transfer of water-soluble molecules occurs also between these populations.
Materials and Methods
The following study was approved by the Animal Care and Use Committee of Institute of Biomedical Research and Innovation at Kobe and complies with the Guide for the Care and Use of Animals published by the Ministry of Education, Culture, Sports, Science, and Technology in Japan. Experiments and results are reported according to the Animal Research: Reporting of In Vivo Experiments guidelines.
Preparation of murine BM-MNC and circulating MNC
After euthanasia by intraperitoneal administration of Secobarbital (Nichiikou, Toyama, Japan), bone marrow was obtained from 6-week-old male CB-17 mice (CLEA Japan, Tokyo, Japan). The femoral and tibial bones were dissected, and bone marrow was extracted from the bones with phosphate-buffered saline (PBS). Bone marrow was mechanically dissociated into single cells, and BM-MNC were isolated by Ficoll–Paque (GE-Healthcare, Little Chalfont, UK) density-gradient centrifugation, as described previously. 3 Blood was obtained by heart puncture of CB-17 mice, and peripheral MNC in circulation were isolated by Ficoll–Paque density-gradient centrifugation.
Loading of low molecular weight fluorescence molecules into the cytoplasm of BM-MNC and peripheral MNC
BM-MNC or peripheral MNC were incubated with 5 µM Calcein acetoxymethyl ester (Calcein-AM; Dojindo, Kumamoto, Japan) or Calcein Deep Red acetoxymethyl ester (Calcein DR-AM; Dojindo) for 30 min at 37°C according to the manufacturer’s protocol. Calcein- or Calcein DR-loaded cells were washed twice with PBS before transplantation.
Cell transplantation
Calcein-loaded BM-MNC or peripheral MNC were injected through the left common carotid artery (CCA), as described previously. 7 Briefly, the left CCA was isolated under 3% isoflurane inhalation anesthesia. After clipping the proximal portion of CCA, 1 × 106 BM-MNC or circulating mononuclear cells in 100 µL PBS were infused through a 35G needle. Subsequently, the needle insertion damage was infilled with medical bond (Aron alpha A; Sankyo, Tokyo, Japan), and the clip was released.
Immunohistochemistry
For histochemical analysis, mice brains were fixed with 2% paraformaldehyde after euthanasia by intraperitoneal administration of secobarbital (Nichiikou) or a mixture containing medetomidine (0.3 mg/kg), midazolam (4 mg/kg), and butorphanol (5 mg/kg). The brain was carefully removed and cut into coronal sections (20 µm) using a vibratome (Leica, Wetzlar, Germany). Sections were immunostained with the antibodies, as listed in Table 1. Vessels were stained by intravenous injection of 100 µl fluorescence-labeled isolectin [Lycopersicon Esculentum Tomato Lectin (1 mg/mL); Vector, CA)] before sacrifice. Microscopic investigations were performed using a LSM990 confocal microscope (Carl Zeiss, Oberkochen, Germany).The number of glial fibrillary acidic protein (GFAP), nestin-, or Doublecortin (DCX)-positive cells with or without Calcein signal at dentate gyrus was counted by blinded investigators (N = 20 views in each sample).
Primary and Secondary Antibodies for Immunostaining
Loading of low molecular weight fluorescent molecules into the cytoplasm of murine cerebral endothelial cells
After sacrifice, male CB-17 mice were perfused with 10 mL medium for endothelial cell culture (HuMedia; Kurabo, Osaka, Japan) at 37°C for 3 min to remove esterase in the serum, followed by perfusion of with 50 mL medium containing 10 µM Calcein-AM (Dojindo) at 37°C for 15 min.
Quantitative polymerase chain reaction analysis
Brain tissue was harvested at 3 h after transplantation of BM-MNC (1 × 105 cells in 100 µL PBS) or PBS (100 µL). After harvesting the brain, coronal sections (4 mm thick) of the forebrain between 2 mm and 6 mm from frontal pole were cut followed by immersion in RNAlater (Thermo Fisher, Waltham, MA) to prevent RNA degradation. Under stereoscopic microscope (Olympus, Tokyo, Japan), the hippocampus was dissected by medical tweezers, as described previously. 5 Total RNA was isolated using RNeasy Plus Universal Mini Kit (Qiagen, CA) according to the manufacturer’s instructions. cDNA was synthesized from 1 µg total RNA using PrimeScriptTM II 1st strand cDNA Synthesis Kit (TAKARA, Kyoto, Japan) according to the manufacturer’s protocol. Transcription of mRNA at hippocampus was analyzed using PowerUpTM SYBRTM Green Master Mix (Applied Biosystems, Foster City, CA) and the Agilent AriaMx real-time quantitative polymerase chain reaction (PCR) System. For the reference gene, RNA18S5 (18s ribosomal RNA) was used. The list of target genes, primer sequences, and amplification protocols is shown in Table 2. For this experiment, aged CB-17 mice (more than 80 weeks) and young mice (5 weeks) were used.
Target Genes, Primer List and Amplification Protocol
NCBI, National Center for Biotechnology Information.
In vitro assay
Human astrocytes were purchased from ScienCell Research Laboratories (#1800, Carlsbad, CA) and cultured according to the manufacturer’s protocol. Astrocytes at passage 3 were coincubated with BM-MNC. For the dye transfer assay, BM-MNC were incubated with 5 µM Calcein DR-AM for 20 min at 37°C. Cells were washed twice with PBS and cocultured with astrocytes for 2 h. The expression of GFAP, HIF1α, and connexin43 was evaluated using the respective antibodies, as listed in Table 1.
Fluorescence activated cell sorting
Small molecule transfer from BM-MNC to GFAP-positive astrocyte in vitro was evaluated using Calcein DR. Calcein DR-loaded BM-MNC were cocultured with GFAP-positive astrocytes for 2 h, and the level of Calcein DR in GFAP-positive cells was evaluated by fluorescence activated cell sorting (FACS). To block the gap junction, Calcein DR-loaded BM-MNC were incubated with 1 mM 1-octanol (Wako, Osaka, Japan) for 10 min before coculture, as described previously. 3
Data analysis
The chi-squared test was used for the analysis of the ratio of Calcein-positive cells in each NSC. Individual comparisons were performed using Student’s t-test. All data are shown as mean ± standard deviation. Correlation between RNA transcription of gap junction and Na+/K+-ATPases was evaluated with linear regression analysis.
Results
Transfer of small molecules from transplanted BM-MNC to cerebral endothelium and hippocampal NSC
The water-soluble small fluorescent molecules (Calcein) were loaded into BM-MNC, which were subsequently transplanted along with fluorescence-labeled isolectin for vessel staining. After 10 min, the Calcein signal was observed at isolectin-positive endothelial cells and nestin-positive NSC in dentate gyrus (Fig. 1A). Many dot-like green fluorescence signals were observed in the dentate gyrus. To investigate the origin of the dot-like green fluorescence signal, non-Calcein-loaded BM-MNC were transplanted. After this, the dot-like green fluorescence signals were still observed in the dentate gyrus, although collocated neither with nestin- nor with isolectin-positive cells (Fig. 1B). This finding indicates that the dot-like green fluorescence signals are just autofluorescence, for instance, originating from lipofuscin that is present even in young mice. 8 Immune staining with anti-GFAP and anti-DCX antibody revealed that GFAP- and DCX-positive cells were also Calcein-positive (Fig. 1C, D), although no NeuN-positive cells were Calcein-positive (data not shown). These findings indicate that direct and/or indirect small molecules transfer from systemically transplanted BM-MNC to hippocampal NSC, including nestin-, GFAP-, and DCX- positive cells.
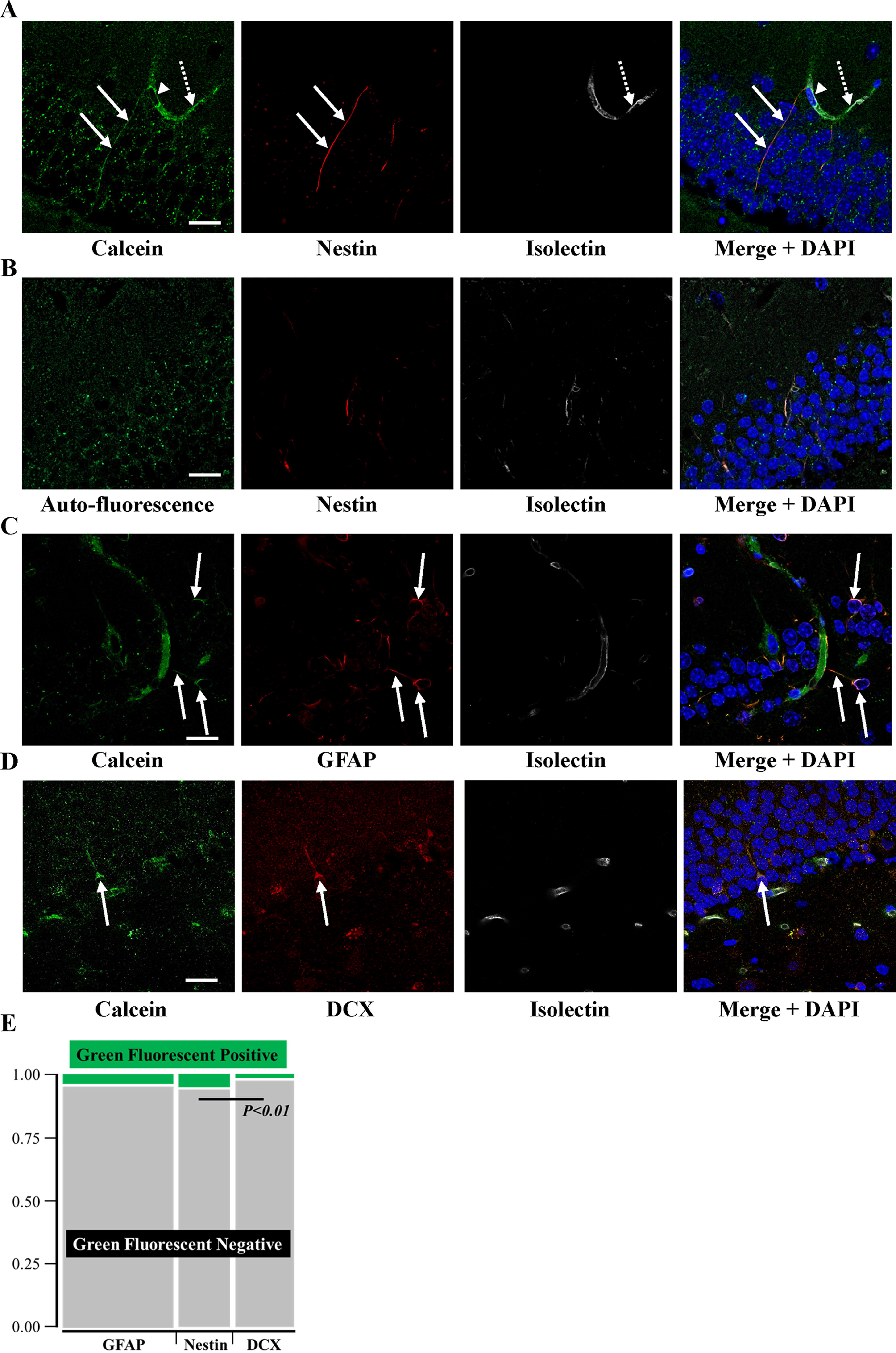
Transfer of the small molecules from systemically transplanted BM-MNC to hippocampal NSC 10 min after cell transplantation.
The ratio of Calcein-positive cells in GFAP, Nestin, or DCX-positive was quantified, and it was found that the ratio of Calcein-positive cells is significantly higher in nestin-positive cells, compared with DCX-positive cells (Fig. 1E). This finding indicated that transplanted BM-MNC have the potential to provide small molecules to all of NSC but the affinity to BM-MNC varies between NSC.
Transfer of small molecules from endothelium to NSC was not observed in the hippocampus
To investigate whether small molecules transfer from systemically transplanted BM-MNC to NSC was direct and/or indirect via cerebral endothelial cells, Calcein was loaded directly into endothelial cells, along with the injection of fluorescence-labeled isolectin. All isolectin-positive endothelial cells were clearly loaded with Calcein (Fig. 2A). However, no nestin-positive NSC was Calcein-positive, including nestin-positive NSC adjacent to Calcein-positive endothelial cells (Fig. 2B). This indicates that direct, not indirect, endothelial cell-mediated transfer of small molecules occurs from systemically transplanted BM-MNC to hippocampal NSC. This result is consistent with previous reports that circulating leukocytes have the potential to penetrate the blood–brain barrier (BBB) in physiological and pathological conditions. 9
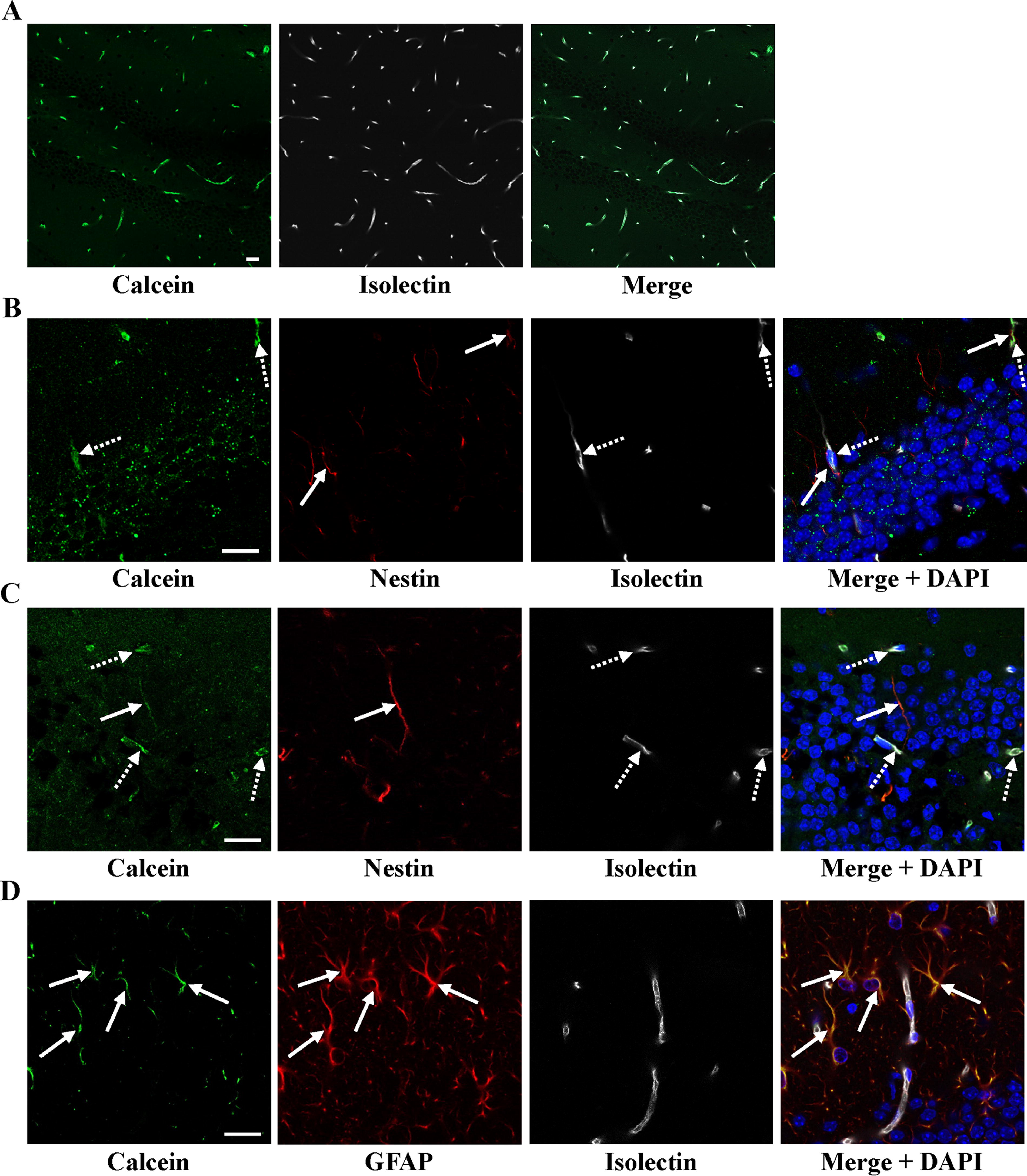
Transfer of the small molecules from transplanted peripheral MNC to hippocampal NSC.
Transfer of small molecules from peripheral MNC to hippocampal NSC
To investigate the possible interaction between peripheral MNC and hippocampal NSC, Calcein-loaded peripheral MNC were injected systemically. Transfer of Calcein from peripheral MNC to hippocampal nestin- and GFAP-positive cells was observed (Fig. 2C, D). These results indicate that small water-soluble molecules transfer also occurs from peripheral MNC.
BM-MNC transplantation to aged mice increased transcription of Connexin 37 (Cx37) at hippocampus
BM-MNC transplantation to aged mice had been shown to increase the number of NSC at hippocampus with improvement of learning memory. 6 To investigate the effect of BM-MNC transplantation on gap junction, the change of RNA transcription of connexins was evaluated by qPCR. The results show that the expression of Cx37 was significantly increased by BM-MNC transplantation in aged mice (Fig. 3A), although the increase in Connexin43 (Cx43) was not statistically significant (Fig. 3B). The activity of Na+/K+-ATPases, which use more than 50% adenosine triphosphate (ATP) in the brain, 10 has a prominent role in learning memory. 11 As gap junction is known to be one of the key molecules that maintain brain metabolism, 12 the correlation between RNA level of connexin and Na+/K+-ATPase at hippocampus was investigated in aged mice. The results showed a significant correlation between the level of connexins and Na+/K+-ATPases (Fig. 3C, D). To further investigate this link, young mice underwent PBS or BM-MNC transplantation, and the RNA transcription of connexin and Na+/K+-ATPases was evaluated. The results, including aged and young mice with or without BM-MNC transplantation, showed a strong correlation between RNA transcription of connexin and Na+/K+-ATPases (Fig. 3E, F).
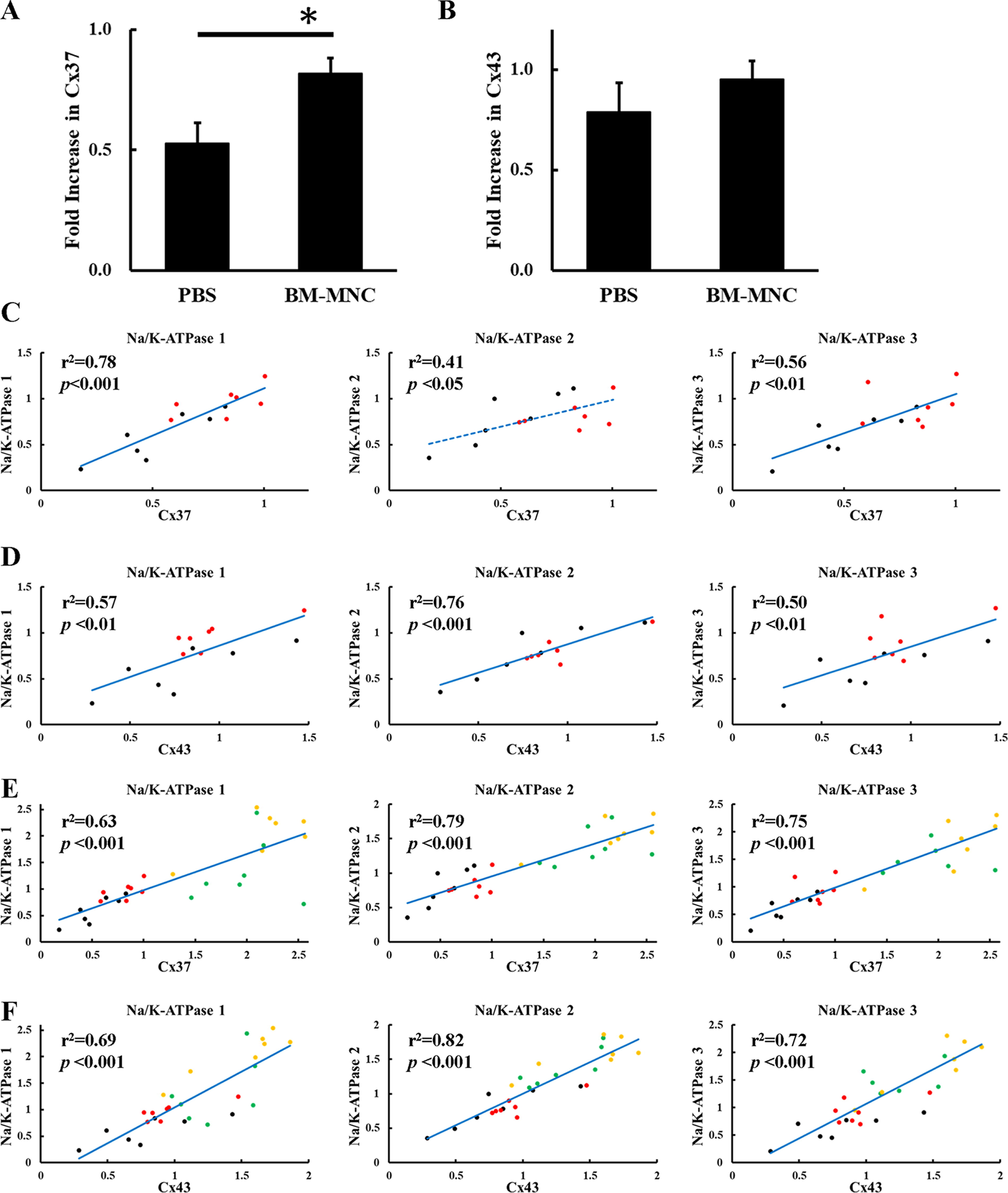
BM-MNC transplantation increased transcription of Cx37 at hippocampus.
Transfer of small molecules from transplanted BM-MNC to GFAP-positive cortical astrocytes
BM-MNC transplantation into aged mice is known to improve various neurological functions, including cognitive and motor functions. 5 As hippocampal NSC are known to be essential for memory formation, 13 we also investigated the effect of transplanted BM-MNC on cortical cells. Ten minutes after Calcein-loaded BM-MNC transplantation, GFAP-positive astrocytes were found to be Calcein-positive (Fig. 4). Although the functional difference between GFAP-positive and GFAP-negative astrocytes is unclear, 14 our observation indicates that GFAP-positive astrocytes can receive water-soluble molecules from transplanted BM-MNC.
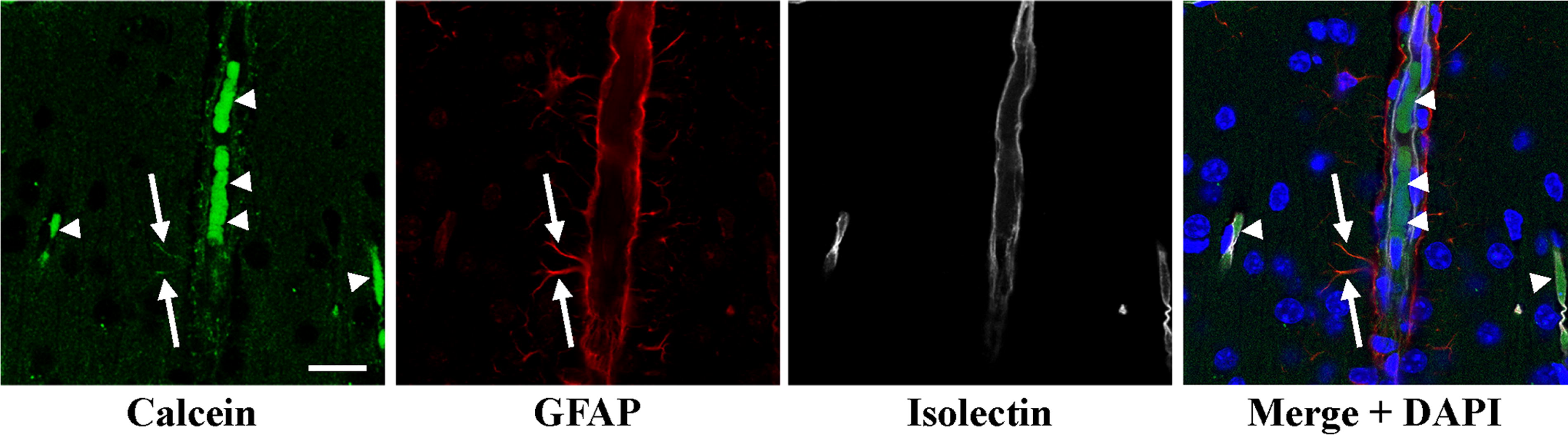
Transfer of the small molecules from transplanted BM-MNC to GFAP-positive cortical astrocytes. Calcein signals were observed in GFAP-positive cortical astrocytes (arrows) already 10 min after BM-MNC transplantation. Arrow heads indicate the transplanted BM-MNC located in the intravascular lumen. Scale bar = 20 µm.
Transfer of small molecules from BM-MNC to GFAP-positive astrocytes in vitro
Calcein DR-loaded BM-MNC were cocultured with GFAP-positive astrocyte in vitro. The transfer of Calcein DR from BM-MNC to astrocyte was observed (Fig. 5A). To confirm the significance of gap junction mediated cellular interaction between GFAP-positive and BM-MNC, gap junction was blocked by 1-octanol and the transfer of small fluorescence molecules was quantified by FACS. The transfer was significantly impaired by the gap junction blocker (Fig. 5B).
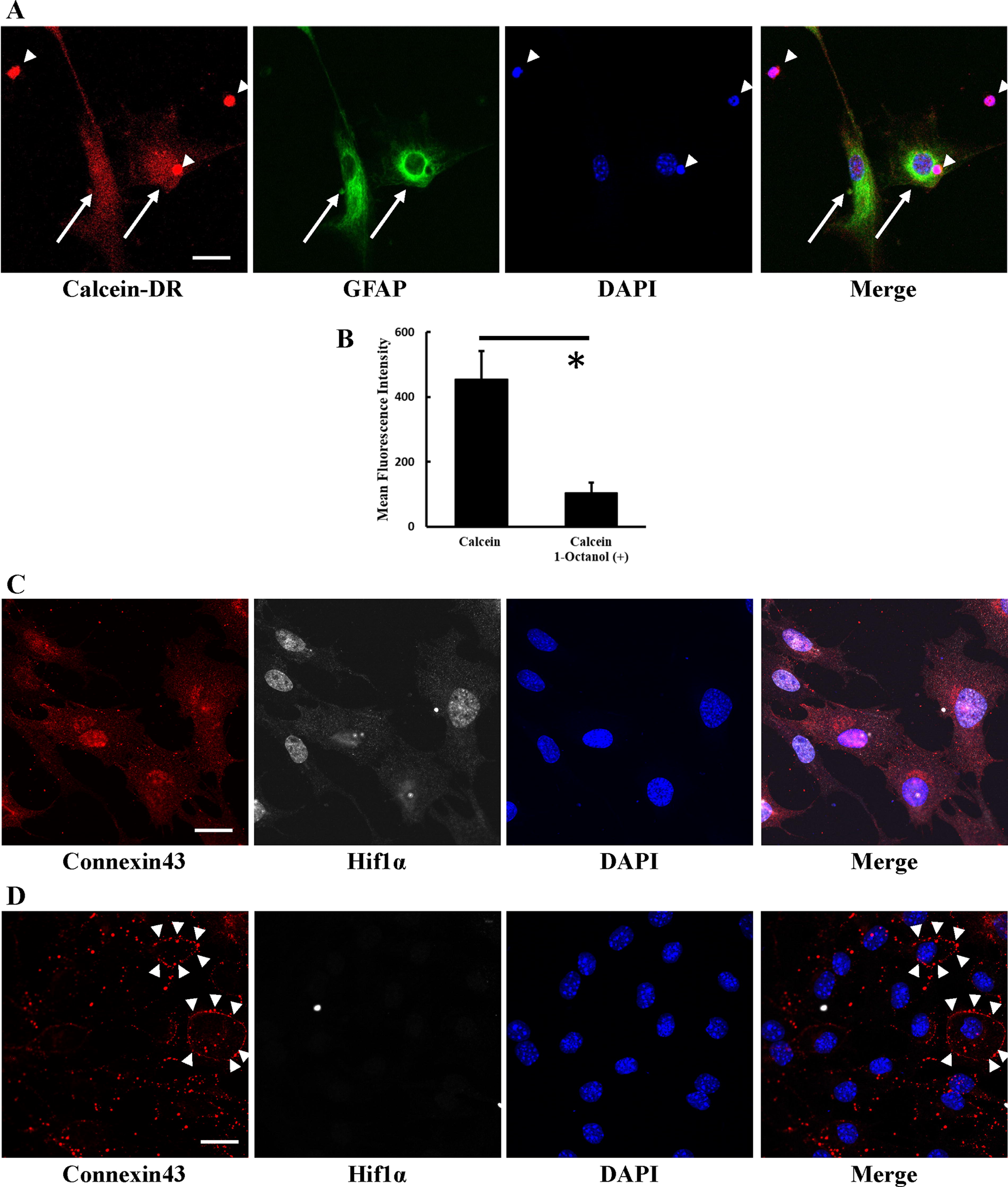
Transfer of small molecules from BM-MNC to GFAP-positive astrocytes in vitro.
To investigate the effect of gap junction-mediated interaction between BM-MNC and GFAP-positive astrocytes, changes in HIF1α expression were evaluated. We have previously demonstrated that gap junction mediated interaction between BM-MNC and endothelial cells induces HIF1α expression in endothelial cells. 3 However, BM-MNC and astrocyte coculture resulted in abolishment of the HIF1α expression (Fig. 5C, D). In addition, clustering of connexin43 3,4 was observed in astrocytes during the coculture with BM-MNC.
Discussion
We report that a transient direct cytosol link between systemically transplanted MNC and hippocampal NSC as well as cortical astrocytes is established across the BBB. These findings indicate that biological systems undergo repair and maintenance of brain function facilitated by circulating MNC.
The significance of a continuous cytosol linkage between heterogeneous cells is well established, although the role of transient heterogeneous cells interaction via gap junctions is mainly unclear. 2 We have previously shown that gap junctions play a critical role in hematopoietic 3 and mesenchymal stem cell therapies 4 by establishing transient direct cytosolic interactions. These observations are consistent with previous findings that a lower number of circulating hematopoietic stem cells correlate with worse stroke outcome 15 and progress of cognitive impairment in patients after stroke. 16 Our new findings provide mechanistic insights into the transient but significant role of heterogeneous cell interaction via the gap junction, in addition to our previous findings that RNA transcription in circulating white blood cell (WBC) is correlated to short-term memory. 6
Our findings show that, similar to hippocampal NSC, GFAP-positive cortical astrocytes can receive water-soluble molecules from systemically transplanted BM-MNC. BM-MNC transplantation in aged mice improves various neurological functions, including cognitive and motor function. 5 Overall, our findings of small molecules transfer to hippocampal NSC, GFAP-positive astrocytes, and cerebral endothelium can help explaining the beneficial effect of BM-MNC transplantation on various neuronal functions in aged mice, 5 stroke mice, 17 and, at least to some extent, stroke patients 18 without the necessity of permanent central nervous system homing and residence.
Circulating leukocytes are known to pass through the BBB under physiological conditions with very low frequency. 9 In this article, we found that although the main body of leukocytes was observed within blood vessels, small molecules transfer to NSC and cortical astrocytes can take place. These findings indicate that small molecules transfer can be achieved by transient partial BBB penetration of circulating leukocytes. As chemokines induce WBC penetration 19 and the local expression of chemokines has a critical role in NSC migration, 20 the difference in localization and stemness of NSC would explain the difference in their affinity to BM-MNC. Although further studies are required to elucidate the mechanism of leukocyte BBB penetration induced by various NSC, our findings suggest that direct interaction between circulating leukocytes and cerebral cell populations is not a rare phenomenon.
Most water-soluble molecules smaller than 1,000 Dalton can traverse gap junctions in the direction of concentration gradients. 2 We observed that there are significant differences in metabolites between cells 21 and that their transfer between heterogenous cells impacts cell activation/inactivation. 3,4 The in vitro findings in this study demonstrate that BM-MNC suppress the expression of HIF1α in GFAP-positive astrocyte. Astrocytes are known to undergo anaerobic metabolism, 22 and the suppressed HIF1α expression in astrocytes during coculture with BM-MNC may potentially be explained by the removal of glycolytic substrates by BM-MNC through gap junctions. Moreover, HIF1α plays a significant role in hippocampal neurogenesis, 23 and the blockade of the HIF1α cascade is known to induce differentiation of neuronal stem cells. 24,25 Further studies will be required to reveal detailed mechanisms and effects of the interaction between BM-MNC and peripheral MNC on HIF1α-mediated processes. One possible working hypothesis may be that peripheral MNC can have an impact on hippocampal neurogenesis through the regulation of HIF1α.
Recent studies have demonstrated that short-term memory is mediated by hippocampal neurogenesis. 26,27 Pathological examination of brain tissue from patients suffering from Alzheimer’s disease showed a drastic reduction of hippocampal neurogenesis. 28,29 In this article, we have shown that transplanted BM-MNC can interact with NSC via gap junction with activation of RNA transcription of connexin. Although the general mechanism and impact of gap junction mediated heterogeneous cellular interaction is unclear, even in highly researched areas such as vascular inflammation 2 requiring further studies, our findings suggest that the neuronal activity would be affected by gap junction mediated cellular interaction with transplanted BM-MNC and circulating leukocytes. In the light of our findings, it will be worthwhile exploring whether systemic transplantation of BM-MNC or stem cells may mitigate Alzheimer’s disease by activating hippocampal neurogenesis and metabolism. 30
Conclusions
We have demonstrated transient and direct transfer of water-soluble molecules between cells in circulation and neuronal stem cells in the brain. Our findings suggest that the transient and direct transfer of water-soluble molecules between cells in circulation and NSC in the brain may be one of the biological mechanisms underlying repair of brain function.
Footnotes
Data Availability
The data supporting the findings of this study are available from the corresponding author upon reasonable request.
Author Disclosure Statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Funding Information
A.T. is supported by the Suzuken Memorial Foundation (R4-International Exchange Grants). J.B. is supported by funding from The Academy of Medical Sciences (Newton Advanced Fellowship NAF/R11/1010).
