Abstract
The proliferation of neural stem cells (NSCs) is precisely regulated by extracellular environmental factors. In situ hypoxia, one of the key factors involved in the regulation of NSC characteristics, has attracted increasing amounts of attention. Numerous studies have demonstrated that hypoxia can significantly promote the formation of neurospheres and the proliferation of NSCs in vitro and that intermittent hypoxia can promote the proliferation of endogenous NSCs in vivo. In this article, the effects of different concentrations of oxygen on NSC proliferation and differentiation both in vivo and in vitro are reviewed, and the potential applications of hypoxia-preconditioned NSCs, as well as research progress and challenges in the treatment of central nervous system diseases, are further summarized. Here, the critical role of oxygen in the neurogenesis of NSCs is emphasized, and insights into the use of hypoxia to regulate NSC characteristics are provided.
Introduction
Physiological normoxia is defined as the oxygen content or pressure in situ for most primate cells representing a hypoxic state that is lower than normal oxygen concentrations (atmospheric O2 concentrations 20%–21%). 1,2 These comparatively low-oxygen tensions (hypoxia) maintain the undifferentiated states of pluripotent stem cells in vitro, such as hematopoietic, mesenchymal, and neural stem cell (NSC) phenotypes, and influence proliferation and cell fate commitment. 3 –5 Increasing evidence has revealed the breadth of the effects of hypoxia on the properties of NSCs and the benefits of these effects for NSC amplification and transplantation. 6,7 Given the emerging understanding of these effects, this review focuses on the regulatory effects of hypoxia on NSC proliferation and on the potential application of hypoxia in the treatment of central nervous system diseases. In addition to recent findings on NSCs, trends in the research and development of hypoxia-treated NSCs are discussed.
The Niche of NSCs Is Hypoxic
Hypoxia is characteristic of the niches of NSCs. As inhaled air enters the lungs and spreads throughout the body, the oxygen concentration gradually decreases. 8 Therefore, physiological normoxia is regarded as hypoxia. In the central nervous system of mammals, the physiological concentrations of oxygen range from as low as 0.55% in the midbrain to 8% in the leptomeninges. In particular, in the subventricular zone niche, the oxygen concentration ranges from 2.5% to 3%. 9 Large changes in oxygen concentration have been measured within a small distance from the dura mater: 3% at a depth of 22–27 mm and 4% at a depth of 7–12 mm. 1 Zhang et al. used an optical fiber light-emitting oxygen sensor to measure the oxygen content in the brains of adult rats and found that the partial pressure of oxygen in the ventricles fluctuates dynamically in the range of 42–48 mmHg. In the hippocampus, the partial pressure of oxygen in the CA1 region and hilus is ∼2 mmHg, whereas the partial pressure of oxygen in the dentate gyrus fluctuates in the range of 6–8 mmHg. 10 As a result, the oxygen concentration in the brain is much lower than that in air. These data convincingly show that hypoxia is a normal physiological state.
In previous studies, oxygen partial pressure was measured in different species during embryonic neurogenesis; for example, the partial pressure of oxygen does not exceed 50 mmHg in the uterus of hamsters and rats, does not exceed 60 mmHg in the fallopian tube, and is <10 mmHg in the uterus of rhesus monkeys. 11 –13 After embryo implantation, the concentration of oxygen in amniotic fluid remains low in all three trimesters of pregnancy. 14 –16 Lord et al. used strong immunological staining of the hypoxia marker EF5 in the neural tube of the hindbrain and midbrain region of embryos to prove that these areas were hypoxic. 17 Lee et al. used the hypoxia marker Hypoxyprobe™-1 to label the neural tube of the telencephalon, diencephalon, and hindbrain of mouse embryos as an anoxic region. 18 Taken together, these results indicated that both the embryo and the neural tube develop in a hypoxic environment.
To date, most stem cells cultured in vitro are still cultured under normoxic rather than hypoxic conditions, which may influence normal metabolic responses. However, some scientists have begun to control the partial pressure of oxygen to provide an in vitro environment that resembles the hypoxic niche of stem cells, which is very important for stem cell survival. 19 –23 Hence, the interaction between stem cells and their niche is ultimately necessary for the design of neural stem cell transplantation. Characteristic hypoxia in the niche is an important factor in the regulation of neural stem cells.
General Introduction to Studies on NSCs Under Hypoxia
The oxygen concentration is one of the most important environmental conditions for cell proliferation and differentiation both in vitro and in vivo. Increasing attention has been given to the effects of mild hypoxia on the neurogenesis and development of NSCs. A comprehensive search of PubMed using the keyword “hypoxia and neurogenesis” revealed only 30 articles published prior to 2003. However, 237 relevant studies were published from 2004 to 2013, reflecting exponential growth. Remarkably, in the decade beginning in 2014, the cumulative number of related articles reached nearly 400, surpassing the total number published before 2013 (Fig. 1).
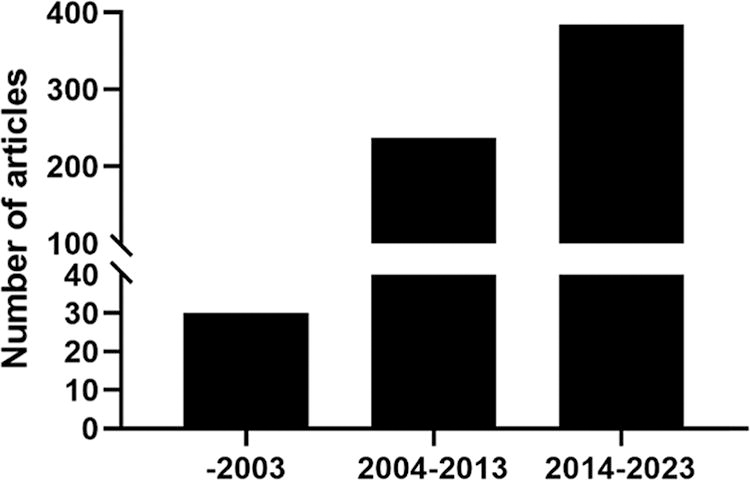
A comprehensive search of PubMed using the keyword “hypoxia and neurogenesis.”
In 1992, Reynolds et al. 24 isolated NSCs from the striatum of the adult mouse brain for the first time; these cells can continuously divide and proliferate in vitro and can differentiate into neurons and astrocytes, contradicting the traditional theory that nerve cells are incapable of regeneration. Researchers then began to culture and study NSCs in vitro. Hypoxia can induce multiple types of stem cells to proliferate, differentiate, and develop into specific cell types. In 2000, Morrison and Studer first used progenitor cells from the E14.5 rat sciatic nerve and the E12 rat midbrain central nervous system to show that hypoxic culture can promote the survival, proliferation, and differentiation of NSCs. 25,26 Storch et al. also reported that hypoxia promoted the proliferation and differentiation of human NSCs. They cultured 9- to 12-week-old human fetal brain neural precursor cells (NPCs) under hypoxic conditions (3%) and found that these cells grew and proliferated in vitro for a long time, surviving up to 11 months. 27 By 2010, oxygen content was identified as a key characteristic of the stem cell niche, 3 and the roles of hypoxia not only in promoting neurogenesis in embryos, newborns, and adults but also in promoting neuronal differentiation from nerve stem/progenitor cells in vitro were elucidated. 28 –31 Moreover, oxygen plays important roles not only in the development of the nervous system but also in the pathology of many brain diseases. Researchers have explored oxygen levels in physiological states and brain diseases and in high- and low-oxygen environments and have begun to investigate the oxygen-sensitive molecular mechanisms that regulate the proliferation and differentiation of NSCs, including the Notch, bone morphogenetic protein (BMP), and Wnt pathways.
In addition, researchers have shown that intermittent hypoxia therapy exhibits neuroprotective effects in multiple models of central nervous system disease and have further explored the possible underlying mechanism; for example, intermittent hypoxia promoted the in situ proliferation of NSCs in the rat brain, which suggested that targeted hypoxia therapy has great potential in the treatment of central nervous system diseases. 32 –35 Hypoxia-induced NSCs are more beneficial than untreated NSCs in transplantation therapy for nervous system diseases. NSCs and neural progenitor cells preconditioned with hypoxia exhibit significantly increased survival and regeneration after transplantation into the host, thus increasing therapeutic efficacy in various disease models. 36,37 Therefore, hypoxia not only promotes the survival, proliferation, and differentiation of NSCs in vitro but also acts as an effective way to stimulate neurogenesis in neurological diseases.
Effects of Hypoxia on NSC Proliferation and Differentiation In Vitro
Mild hypoxia is known to increase the proliferation of various stem cells in vitro and play a vital role in their differentiation. Morrison et al. made a seminal contribution to this field in 2000, as reported in the Journal of Neuroscience, demonstrating that hypoxia facilitates both the proliferation and the differentiation of NSCs. They extracted neural crest stem cells from the sciatic nerves of E14.5 rats and explored their status under hypoxic conditions. The findings revealed that when cultured in standard medium with 3%–6% hypoxia for 6 days, these cells exhibited a significant increase in survival rate and colony formation capacity. Furthermore, the addition of BMP2 and forskolin under hypoxic conditions promoted the differentiation of these cells into dopaminergic neurons and increased their synthesis and release of dopamine and norepinephrine. 25 Furthermore, Studer et al. isolated NPCs from the midbrains of rat embryos (E12) and cultivated them under reduced oxygen levels (3 ± 2%). They observed that hypoxia led to increased proliferation and decreased apoptosis of these NPCs, indicating that hypoxia can make it possible to obtain definite numbers of precursor cells and differentiated cells, available for therapeutic transplantation, providing an effective method for the large-scale production of specific neurons for brain repair. 26 Zhao et al. demonstrated that hypoxia significantly increased the proliferation of primary NSCs extracted from the midbrain of rat embryos (E13.5): both the number of neurospheres and the proportion of BrdU-positive cells at 3% and 10% O2 were markedly elevated compared with the corresponding numbers under normoxic conditions. 38
Moreover, to assess the effects of oxygen concentration on the differentiation of NSCs, Horie et al. extracted NSCs from the ganglionic eminence of Institute of Cancer Research (ICR) mouse embryos (E15.5) for cultivation and observed a significant increase in the proportions of BrdU-labeled cells and Tuj1-positive cells under mild hypoxia (2% O2). Furthermore, NSCs differentiated into gamma-aminobutyric acid (GABA)-positive neurons under normoxic conditions and into glutamate-positive neurons under mild hypoxia. Considering the above observations and the established ability of mild hypoxia (2% O2) in the ischemic penumbra to facilitate neuronal differentiation, the authors suggested that the reduction in oxygen levels attributable to cerebral ischemia might promote NSC proliferation and neural differentiation. 39 Similarly, Zhao et al. isolated NSCs from the midbrain of embryonic Wistar rats (E13.5) and subjected them to hypoxia (3% O2) for 3 days, followed by differentiation under normoxic conditions for 5–7 days. The results revealed that the percentages of TH-positive cells obtained by the differentiation of NSCs under normoxic and hypoxic conditions were 10.25 ± 1.03% and 19.88 ± 1.44%, respectively. The dopamine content in the culture medium of the low-oxygen group was approximately twice that of the normoxic group. Under hypoxic conditions, NSCs exhibit an increased capacity to differentiate into TH-positive neurons. 40
Pistollato et al. reported that, compared with those cultivated in 17% O2, human neural precursor cells cultivated in 5% O2 exhibited a 20-fold increase in oligodendrocyte production; moreover, during subsequent differentiation in 20% O2, oligodendrocytes produced under hypoxic conditions underwent 2.5-fold greater maturation, indicating that mild hypoxia can regulate cell fate and maturation. 41 Santilli et al. established the v-myc immortalized human NSC (IhNSC) line and investigated the impact of 1%, 2.5%, 5%, and 20% O2concentrations on the proliferation and differentiation of IhNSCs in vitro; the results showed that mild hypoxia (2.5%–5% O2) increased the proliferation of human NSCs in vitro. In subsequent differentiation experiments, hypoxia promoted the differentiation of lhNSCs into neurons and oligodendrocytes and increased the number of GABAergic neurons and glutamatergic neurons. 9 Therefore, it can be inferred that the decrease in oxygen levels that occurs in neuronal diseases, such as cerebral ischemia, might transiently cause NSCs to remain stationary and that mild hypoxia promotes NSC proliferation and differentiation into neurons and oligodendrocytes, providing a potential pathway for the treatment of cerebral ischemia and other neurological diseases in which NSCs participate. Stacpoole et al. produced NPCs from human embryonic stem cells (hESCs) under hypoxic (3% O2) conditions. In the first stage, they performed suspension culture of hESC colonies under 3% O2 conditions, and within 2 weeks, the cells exhibited neuroepithelial characteristics. From day 14 to day 28, either the continuous application of retinoic acid and purine amine (PM) was used to induce hESC differentiation into spinal motor neurons or the addition of fibroblast growth factor-8 and PM was used to promote hESC differentiation into midbrain dopaminergic neurons. This study revealed that specific cell types are produced at 3% O2, which represents a significant advance in in vitro disease modeling. 42 In summary, a low-oxygen concentration of 3%–10% can promote the proliferation and differentiation of NSCs from different sources, such as mice, rats, and humans, in vitro (Table 1).
Differentiation Characteristics of NSCs Under Hypoxia In Vitro
hNPC, human neural precursor cell; NPC, neural precursor cell; NSC, neural stem cell; SVZ, subventricular zone.
Effects of Hypoxia on Neurogenesis In Vivo
Mild hypoxia not only promotes the proliferation and differentiation of NSCs in vitro but also stimulates the proliferation of endogenous NSCs. Studies on the effects of hypoxic preconditioning on neurogenesis in vivo have been reported in various animal models of neurological disease including Alzheimer’s disease (AD), depression, hypoxic–ischemic (HI) injury, and middle cerebral artery occlusion (MCAO). For example, as a newly identified external stimulus, intermittent hypoxia fosters postnatal hippocampal neurogenesis through a series of steps that include the proliferation of NSCs, the survival and migration of new neurons, and the synaptic morphogenesis of mature neurons. 34 Zhu et al. found that hypoxia promoted the proliferation of NSCs in an in vivo experiment. The rats were subjected to low pressure and low oxygen at simulated altitudes of 3,000 m or 5,000 m for 4 h/day for 2 weeks. The experimental results showed that, compared with those in the control group, the numbers of BrdU-positive cells in the inferior gyrus of the lateral ventricle and in the dentate gyrus of the hippocampus in rats increased by 62% and 35%, respectively, indicating that intermittent hypoxia promoted the in situ proliferation of NSCs. 35 Similarly, Varela-Nallar et al. assessed whether chronic hypoxia induced neurogenesis in the subgranular zone of the hippocampal dentate gyrus. They subjected both adult wild-type mice and AD dual transgenic APPswe-PS1ΔE9 model mice to a chronic hypoxic environment at 10% O2 for periods ranging from 6 to 72 h. The results indicated that hypoxia promoted cellular proliferation and neurogenesis in the hippocampal regions of both mouse models, suggesting that mild hypoxia may have therapeutic value for neurodegenerative diseases and conditions with impaired neurogenesis. 43
Depression is often accompanied by neurological damage and a decrease in neurogenesis, 44,45 whereas hypoxia can promote neurogenesis; thus, hypoxic treatment has potential for ameliorating the pathological symptoms of depression. Zhu et al. developed a depression model in rats by subjecting them to chronic stress. Preconditioning rats with low-pressure hypoxia at altitudes of 3,000 m and 5,000 m for 4 h/day for 14 days showed that intermittent low-pressure hypoxia had a marked antidepressant effect. 46 Similarly, in 2016, Kushwah et al. investigated the neuroprotective effect of intermittent hypobaric hypoxia (IHH) on depression-like behavior induced by unpredictable chronic mild stress (UCMS) in rats. They established a model of depression in male Sprague–Dawley rats and administered intermittent low-pressure hypoxia treatment at a simulated altitude of 5,000 m for 4 h/day for 2 weeks. They observed alleviation of the depressive behavior of the rats in the open-field test, the forced swim test, and the elevated-plus maze test. Further verification via histological and molecular biology methods confirmed that IHH mitigated the decrease in neurogenesis induced by UCMS. 47 These findings suggest that intermittent hypoxia has therapeutic potential similar to that of antidepressants in animal models of depression and can act as a preventive treatment for this pathological and physiological state.
Hypoxic preconditioning refers to the administration of brief, mildly hypoxic stimuli, a process that can significantly enhance the body’s tolerance to subsequent more severe ischemic or hypoxic events. 49 Ara et al. first demonstrated in a neonatal piglet model that hypoxic preconditioning protects against HI injury by enhancing neurogenesis within the brain. One-day-old piglets were placed in a large organic glass low-oxygen chamber and pretreated with 8% O2 for 3 h. After 24 h, the piglets were exposed to a low-oxygen (5% FiO2) environment for 30 min, followed by a 10-min low-blood pressure treatment to simulate an HI state. Their results showed that hypoxic preconditioning increased the number of BrdU-positive cells in the brains of newborn piglets, promoted the proliferation of progenitor cells in the brains of newborn piglets, and might promote postnatal neurogenesis by regulating the proliferation and destiny of neural stem/progenitor cells in the subventricular zone (SVZ) region, thereby contributing to the repair of damaged brain tissue. Furthermore, following hypoxic preconditioning, the neural stem or progenitor cells in the SVZ of neonatal piglets subjected to hypoxia–ischemia exhibited multipotency, and the proliferation of these cells increased. These results showed that hypoxic preconditioning can induce the proliferation and neurogenesis of endogenous NSCs. 50,51 Recently, to investigate the endogenous neuroprotective mechanisms against neonatal HI encephalopathy, researchers exposed a mouse model of neonatal HI encephalopathy to a hypoxic environment for 20 min on the 7th day of life (P7) for hypoxic preconditioning. In these mice, the number of Na+/Ca2+ exchange 1 (NCX1)- and NCX3-positive cells in the brain increased, and the motor abilities of the mice were enhanced. Furthermore, a substantial decrease in ischemic damage was observed in these mice. The results of this study indicated that in a mouse model of neonatal HI encephalopathy, hypoxic preconditioning promoted the proliferation of neuroblasts in the hippocampal dentate gyrus and triggered neurogenic processes related to the maintenance of ion homeostasis, which provided ideas for endogenous neuroprotection against neonatal HI encephalopathy. 52 Huang et al. tied silk sutures at the proximal and distal ends of the common carotid artery in Sprague–Dawley rats. Then, the proximal common carotid artery was ligated, and the distal common carotid artery was clamped with an artery clamp for 10 min to simulate double common carotid artery occlusion. Using this MCAO model, they found that hypoxic preconditioning equivalent to an altitude of 5,000 m for 3 h/day for 14 days significantly ameliorated MCAO-induced axonal and neuronal damage, and the protective effect of hypoxic preconditioning against ischemic brain injury was confirmed by a rat behavioral test, a cognitive score, and the 2, 3, 5-triphenyltetrazolium chloride test. 53 These studies provide an important theoretical basis for the treatment of MCAO.
In addition to the beneficial effects of hypoxic preconditioning, Becerra-González et al. reported that hypoxic preconditioning did not lead to motor deficits in animal models. They subjected male mice (CD1, GFAP, EGFP, and Pax2-GFP mice) to hypoxic preconditioning by placing the mice in 50-mL conical tubes and securely sealing the caps. When the mice began panting, they were removed, and the process was repeated three times. The results revealed that under hypoxic conditions, the expression of cellular markers such as nestin, NeuN, and Iba1 was upregulated in cells at the apex of the fourth ventricle, suggesting a trend toward increased proliferation and differentiation of brain cells. Moreover, behavioral tests such as the rotarod test and the horizontal bar test confirmed that hypoxic preconditioning did not induce significant motor deficits in mice. 48 In summary, mild hypoxia stimulation has the potential neurorestorative and therapeutic effects on diseases such as decreased nerve regeneration ability or nerve damage in the nervous system.
The Mechanism by Which Mild Hypoxia Regulates Neurogenesis
In studies of development of the nervous system and various diseases associated with decreased neurogenesis, the mechanisms by which hypoxia regulates neurogenesis primarily include the following aspects.
Notably, hypoxia-inducible factor 1 (HIF-1) is the principal regulator of the cellular response to hypoxia. 54 Under hypoxic conditions, the expression of C-terminal binding protein (CTBP) and HIF-2α in hESCs decreases, accompanied by a reduced rate of glycolytic metabolism. When CTBP is silenced, the expression of pluripotency markers in the cells also decreases. These findings suggest that HIF-2α and the metabolic sensor CTBP are involved in the survival and self-renewal of hESCs under hypoxic conditions. 55 Using adenoviral vectors, Zhao et al. demonstrated that the overexpression of HIF-1α at 20% O2 and 10% O2 promoted the proliferation of brain-derived neural stem/progenitor cells (NPCs) in rat embryos (E13.5). In contrast, when the pSilencer 1.0-U6 plasmid was used to target HIF-1α mRNA, the induction of NPC proliferation by 10% O2 was inhibited by HIF-1α knockdown, which demonstrated the crucial role of HIF-1α in the hypoxia-driven proliferation of NPCs. 38 Francis et al. also demonstrated that mild hypoxia can promote both the survival and the neural differentiation of hESCs by upregulating the expression of components in HIF-1α and HIF-2α signaling pathways. 56
In addition, the Wnt/β-catenin and BMP signaling pathways, which are closely related to HIF-1, also play key roles in neurogenesis under hypoxia stimulation. For example, the Wnt/β-catenin signaling pathway is instrumental in determining the fate, proliferation, and differentiation of NPCs during neurodevelopment in mice and humans, as well as in directing neurogenesis of the central nervous system in adult mammals. The Wnt protein serves as a pivotal regulatory element in NSC behavior during both embryonic development and adult hippocampal neurogenesis. 57,58 Mazumdar et al. discovered that HIF-1α facilitates the activation and expression of the β-catenin protein by upregulating the downstream effectors LEF-1 and TCF-1 to modulate Wnt/β-catenin signal transduction in embryonic stem cells and primary cultured NSCs under hypoxic conditions. 59 Cui et al. cultivated hippocampal NSCs from transgenic newborn mice (0 day) with green fluorescent protein under 5% O2. The results showed increased expression of the β-catenin protein in these cells, and the overexpression of β-catenin promoted the proliferation of NSCs. Consequently, they posited that Wnt/β-catenin signaling is involved in the induction of hippocampal NSC proliferation by hypoxia. 60 Pistollato et al. demonstrated that hypoxia suppresses BMP signaling, hindering the differentiation of central nervous system precursor cells into glial cells, whereas an increase in oxygen concentration was observed to initiate SMAD activation within 30 min. Researchers proposed that hypoxia might influence the fate of NPCs by modulating SMAD proteins 1, 5, and 8, which are key molecules in BMP signal transduction. Moreover, HIF-1α silencing also resulted in SMAD activation, suggesting that HIF-1α is essential for BMP inhibition. 41,61
Notch signaling also plays a crucial role in determining the fate of NSCs. Felling et al. discovered that following HI damage, the levels of leukemia inhibitory factor increase significantly, leading to the activation of Notch-1 and Delta-like 1 and consequently promoting the expansion of neural stem/progenitors (NSPs) in the brain. These data confirm the importance of the Notch–Delta–Serrate–Lag2 signaling pathway in the expansion of NSPs after HI damage. 62 Gustafsson et al. further determined that, 63 compared with that under normoxic conditions, the expression of the Hes-1 gene, which is directly downstream of Notch, increased by 1.8 times after 4 h of hypoxic treatment of NSCs. By further investigating the process by which hypoxia regulates Notch signal transduction, they discovered that hypoxia substantially elevated Hes-1 mRNA and protein expression in the embryonic cancer cell line P19. In addition, hypoxia induced a 3.9-fold increase in the expression of the highly Notch-specific promoter 12XCSL-luc. Moreover, γ-secretase inhibitors were shown to block the cleavage of Notch receptors, releasing the Notch intracellular domain and reducing the hypoxia response of downstream Notch genes and promoters. Therefore, hypoxia can increase endogenous Notch signal transduction, leading to increased activation of downstream Notch gene transcription and thereby regulating the fate of NSCs.
In addition, the sphingosine kinase (SphK) and BDNF TrkB signaling pathways play more pronounced roles in hypoxia protection. SphK, a key rate-limiting enzyme, facilitates the conversion of sphingosine (Sp) to sphingosine 1-phosphate (S1P). S1P exerts neuroprotective effects through mechanisms such as diminishing leukocyte adhesion, attenuating endothelial cell apoptosis, and stimulating signaling molecules such as AKt and eNOS. Hypoxic preconditioning was found to increase the expression of the SphK2 protein in the cerebral vasculature of mice, effectively decreasing both the infarct area and the edema ratio postischemia. 64 To elucidate the molecular mechanism of neuroprotection induced by hypoxic preconditioning, Guan et al. performed hypoxic preconditioning on propofol-treated Sprague–Dawley pups (postnatal day 7) and reported that cAMP upregulation and CREB phosphorylation in the hippocampus led to a significant decrease in caspase-3 levels. The results of this study suggested that hypoxic preconditioning ameliorated propofol-induced nerve injury through the cAMP/PKA/CREB signaling pathway. 65 Zhu et al. 46 reported that in a rat model of depression induced by chronic stress, the antidepressant effect of IHH disappeared when the brain-derived neurotrophic factor-tyrosine kinase B (BDNF-TrkB) signaling pathway was blocked. This finding suggested that hypoxia can promote hippocampal neurogenesis through the BDNF-TrkB signaling pathway. Interestingly, Platero-Luengo et al. reported that the growth of NSCs in adult carotid bodies is also regulated by hypoxia. Hypoxia was shown to regulate NSCs through mature spherical cells that express endothelin-1 (ET-1, an O2-sensitive element responsive to hypoxia), thus indirectly affecting the proliferation of NSCs. The results of this study suggested that hypoxia might regulate the proliferation of NSCs in the brain through a more subtle mechanism. 66
Advantages of Hypoxia-Induced NSCs for Transplantation
The transplantation of NSCs into the brain can ameliorate neurological functional deficits in neurological disorders. In this process, the survival and functional recovery of transplanted cells within the host brain are paramount. According to numerous studies in disease/injury models, both hypoxic preconditioning and the pharmacological induction of a cellular hypoxia response can facilitate cellular adaptation and increase the survival rate of transplanted cells. To date, hypoxia-induced NSC transplantation has been applied in the treatment of central nervous system diseases, including traumatic brain injury (TBI), MCAO, cerebral hemorrhage, spinal cord injury (SCI), Parkinson’s disease, and neurogenic pain (Table 2). Low-oxygen preconditioning can also increase cellular communication between transplanted cells.
Studies on the Application of Transplanted NSCs Under Hypoxia
EpoSV, erythropoietin enhancer-SV; ES-NPC, embryonic stem cell-derived neural progenitor cell; GM-CSF, granulocyte macrophage colony-stimulating factor; HP, Hypoxic preconditioning; iPSC, induced pluripotent stem cells; PD, Parkinson's disease; SCI, spinal cord injury; SFI, sciatic nerve function index; TBI, traumatic brain injury.
Wei et al. preconditioned iPSC-derived neural progenitor cells (iPSC-NPCs) in mice under hypoxic conditions (0.1%–0.3% O2) and subsequently transplanted them into the brains of a Wistar rat model of TBI at P14. The results revealed that the transplanted rats exhibited improvements in food preference, social interaction, and social novelty tests, suggesting that the transplantation of cells subjected to hypoxic preconditioning into the brain can alleviate neuropsychiatric deficits in young mice. 67 This approach has demonstrated considerable potential for improving the prognosis of patients with neuropsychiatric defects. Similarly, Theus et al. transplanted the embryonic stem cell-derived neural progenitor cells (ES-NPCs) subjected to hypoxia preconditioning at 1% O2 into the brains of MCAO model rats, and cell death and caspase-3 activation were reduced by 30%–40% after 3 days. Moreover, hypoxia-preconditioned transplanted ES-NPCs demonstrated extensive neuronal differentiation within the ischemic brain, facilitating the recovery of sensory-motor functions. 37 This research suggested that this approach, which enhances the survival of transplanted cells and their ability to repair tissue, has the potential to optimize the process of cell transplantation for disease treatment. Wakai et al. exposed NSCs to hypoxic conditions (5% O2) for 24 h, after which they transplanted these preconditioned NSCs into the brains of mice 3 days after cerebral hemorrhage. They found that hypoxic preconditioning notably strengthened the functionality of NSCs in a mouse model of cerebral hemorrhage. This treatment not only increased the survival rate of the transplanted cells but also enabled the preconditioned NSCs to demonstrate tissue-protective capabilities and to expedite the restoration of mouse behavioral functions. 36 The results of this research suggested that hypoxic preconditioning can markedly improve the therapeutic efficacy of NSC transplantation for treating cerebral hemorrhage.
SCI can cause cell apoptosis and lead to motor dysfunction. 68,69 Lian Jin et al. demonstrated that the transplantation of mouse NSCs (American Type Culture Collection NE-4C) preconditioned with 1% O2 for 24 h into a rat model of SCI significantly increased neuronal survival in the injured spinal cord. This research confirmed that the in vivo transplantation of mouse NSCs modified with a hypoxia-inducible VEGF gene expression system increased cell viability in SCI rat models. 70 Similarly, Kim et al. preconditioned NSCs that overexpressed granulocyte macrophage colony-stimulating factor (GM-CSF) with 1% O2 or the hypoxia simulator CoCl2, followed by transplantation into a rat model of SCI. They observed that hypoxic preconditioning resulted in a significant increase in the number of surviving NSCs and in the neuronal differentiation of NSCs in rats after transplantation, as well as a significant improvement in motor function. 71 Fan et al. isolated and cultured primary NSCs from the cortex of E14.5 offspring of rat embryos. After 4 h of hypoxic preconditioning at 1% O2, these NSCs were transplanted into a rat model of SCI. The findings indicated that hypoxic preconditioning of the transplanted NSCs promoted neuronal survival in the damaged spinal cord, particularly in 5-HT and ChAT neurons, and upregulated the expression of neurotrophin-3, glial-derived neurotrophic factor, and growth factors, contributing to functional recovery in SCI rats, as evidenced by improvements in behavior, locomotion, and hypersensitivity responses. 72 In a subsequent study in 2022, Fan et al. transplanted NSCs preconditioned with 5% O2 into SCI rats. After 1 week, the levels of neurotrophic factors such as vascular endothelial growth factor (VEGF) and brain-derived neurotrophic factor (BDNF) in the SCI rats treated with preconditioned NPCs increased. In addition, the levels of transmembrane protein 1 (TMIGD1) and TMIGD3 in the immunoglobulin domain were correlated with a significant decrease in the levels of proinflammatory cytokines such as TNF-α. 73 These studies demonstrate that hypoxic preconditioning of NSCs prior to transplantation can enhance neuronal survival at the injury site and improve motor function.
In the case of Parkinson’s disease, which has a high incidence rate, the primary pathological characteristic is the reduction of dopaminergic neurons in the substantia nigra of the brain, 74 and using the differentiation potential of NSCs to produce dopaminergic neurons is one of the most promising therapeutic strategies. Guo et al. used 0.5 mM valproic acid, 3 μM CHIR99021, and 1 μM Repsox to induce the reprogramming of rat embryonic (E16-18) fibroblasts into neural progenitor cells under 5% O2, endowing the cells with characteristics similar to those of rat brain-derived NSCs. These cells were subsequently transplanted into the substantia nigra of a 6-hydroxydopamine-damaged rat model of Parkinson’s disease. Through open field, Morris water maze, and rotational experiments, they ascertained that behavioral function in a Parkinson’s disease rat model improved after transplantation. Transplanted neural progenitor cells differentiated into functionally active neurons that integrate into the brain in the Parkinson’s disease rat model. 75 The results of this study suggested that the transplantation of hypoxia-preconditioned neural progenitor cells can ameliorate neurological functional deficits in a 6-OHDA-induced PD rat model. Hence, hypoxic preconditioning may emerge as a significant approach for treating neurodegenerative diseases.
Neurogenic pain can be caused by disease or injury to the nervous system. 76,77 Lee et al. transplanted mouse erythropoietin enhancer-SV40 promoter-VEGF NSCs (EpoSV-VEGF-NSCs) preconditioned with 1% O2 into a male Sprague–Dawley rat model of neuropathic pain with sciatic nerve injury. They found that the transplantation of hypoxia-preconditioned EpoSV-VEGF-NSCs effectively improved the sciatic nerve function index (SFI), ameliorated mechanical dysfunction, and increased myelin regeneration. 78 These results suggest that NSC therapy involving hypoxic preconditioning and VEGF expression can be used safely and effectively for the treatment of neuropathic pain and is suitable for the treatment of sciatic nerve injury.
Cellular communication between transplanted NSCs and host cells is crucial for many functions of implanted NSCs. Researchers exposed the mouse NSC line C17.2 to hypoxia for 3 h and found that the number of connexin 43 aggregates in NSCs, a key protein responsible for cell communication, increased by 31%. 79 These findings suggested that hypoxic preconditioning before transplantation surgery may be a reliable method for improving communication between the transplant and host. In summary, transplanted NSCs exert beneficial effects not only by replacing damaged cells, regulating inflammation, and promoting neurotrophic regulation but also by promoting cell communication between the graft and the host. Therefore, the rational use of the transplantation of hypoxia-regulated NSCs has the broad potential in the treatment of neurological diseases.
Conclusions
Hypoxia serves as a crucial niche for neurogenesis. In vitro, hypoxia not only maintains the pluripotency of NSCs but also regulates their differentiation properties. Hypoxia-preconditioned NSCs can be used for transplant therapies for various neurological diseases. Moreover, intermittent hypoxia or hypoxic preconditioning is a noninvasive physical stimulation method that is a significant strategy for regulating endogenous NSC proliferation and has been proven to be very effective in the treatment of AD, depression, UCMS, hypoxia ischemic encephalopathy, MCAO, TBI, SCI, and other disease models in mice, rats, pigs, and other animals. Therefore, hypoxia has the potential for application in the treatment of human neurological disorders and may be a highly effective adjunctive therapeutic approach.
The important regulatory role of the hypoxic microenvironment on NSCs is an important factor that must be considered in the future application of NSCs in the process of neural injury repair. This microenvironment may affect the survival and directed differentiation of NSCs and even affect the formation of neural circuits during brain injury repair. Because mild hypoxia can induce neurogenesis in vivo, the use of mild hypoxia in vitro to induce NSC proliferation and differentiation can address the problem of insufficient NSC sources for the transplantation treatment of neurological diseases. Moreover, mild hypoxia plays a pivotal role in the transplantation process of neurological diseases, increasing the success rate of transplantation and improving patient prognosis.
Footnotes
Acknowledgment
We thank members of the Zhu laboratory for their contribution.
Author Disclosure Statement
No competing financial interests exist.
Funding Information
This study was supported by the
