Abstract
Over the past 15 years, there has been a significant shift in biomedical research toward a major focus on stem cell research. Although stem cells and their derivatives exhibit potential in modeling and mitigating human diseases, the ongoing objective is to enhance their utilization and translational potential. Stem cells are increasingly employed in both academic and commercial settings for a variety of in vitro and in vivo applications in regenerative medicine. Notably, accessibility to stem cell research in low-Earth orbit (LEO) has expanded, driven by the unique properties of space, such as microgravity, which cannot exactly be replicated on Earth. As private enterprises continue to grow and launch low-orbit payloads alongside government-funded spaceflight, space has evolved into a more viable destination for scientific exploration. This review underscores the potential benefits of microgravity on fundamental stem cell properties, highlighting the adaptability of cells to their environment and emphasizing physical stimuli as a key factor influencing cultured cells. Previous studies suggest that stimuli such as magnetic fields, shear stress, or gravity impact not only cell kinetics, including differentiation and proliferation, but also therapeutic effects such as cells with improved immunosuppressive capabilities or the ability to identify novel targets to refine disease treatments. With the rapid progress and sustained advocacy for space research, we propose that the advantageous properties of LEO create novel opportunities in biomanufacturing for regenerative medicine, spanning disease modeling, the development of stem cell-derived products, and biofabrication.
Historical Context and Spaceflight
The International Space Station (ISS) National Laboratory stands as a significant achievement in human space exploration and cooperation, originating in the 2000s through the efforts of the United States Congress. This extraordinary facility is dedicated to conducting scientific research with the goal of benefitting humanity on Earth, employing a public–private partnership model that involves private companies, academic institutions, and various organizations. 1,2 Collaborating with the United States and selected international partners, the ISS provides a stable microgravity environment in low-Earth orbit (LEO), enabling researchers to investigate the impacts of this unique environment on biology and physiology.
The initial research conducted has yielded valuable insights into utilizing microgravity to advance biomanufacturing in space, with direct implications for enhancing human life and promoting commercial enterprise on Earth. 2 Given the growing emphasis on stem cell research in biomedicine, recent attention has been directed toward examining how microgravity influences properties such as cell proliferation, differentiation, culture, and potency, offering insights into potential changes compared with Earth conditions. 3
Earlier studies on stem cells conducted on Earth utilized artificial microgravity simulators, and more recently, investigations have extended to LEO aboard the ISS, often in tandem with ground-based studies for comparison. 4,5 The ongoing development of private space stations further positions them as prospective research laboratories in LEO. Despite these advancements, there is still considerable work to be done in comprehensively understanding the impact of microgravity on fundamental stem cell properties. This includes exploring how microgravity influences the modeling of human physiology during spaceflight and the potential enhancements it might bring to tissue biomanufacturing processes. 6
Cell Culture in Space
In recent times, there has been a growing interest in leveraging microgravity as a potential means to augment various stem cell properties, including pluripotency, multipotency, division, differentiation, and the maturation of stem cell derivatives. 7 The potency of stem cells, characterized by their ability to generate derivative cells within a tissue-specific lineage of interest (multipotency) or across multiple germ lineages (pluripotency), is a defining aspect. Despite the evolution of differentiation protocols over several decades, the persistent variability in stem cell potency and differentiation among different cell lines remain significant challenges in the field.
This variability poses a substantial obstacle to experimental reproducibility. 8 Consequently, there is a pressing need to identify methodologies capable of preserving or enhancing the potency and differentiation capacities of both multipotent and pluripotent stem cell lines.
The challenge of safely and efficiently expanding stem cells and their derivatives persists, particularly in the context of regenerative medicine where cells such as pluripotent stem cells (PSCs) and multipotent mesenchymal stem cells (MSCs) are frequently employed. Recent studies have revealed that human MSCs can be successfully cultured aboard the ISS, demonstrating enhanced immunosuppressive capabilities compared with their counterparts grown on Earth. 9 Such comparative investigations are crucial for understanding the impact of microgravity on pluripotency and multipotency, as they have the potential to unveil new molecular or cellular mechanisms capable of improving biomanufacturing processes or clinical therapy on Earth.
In addition, these studies lay the groundwork for future advancements in in-orbit biomanufacturing products. 10 The microgravity-induced expansion of stem cells could address the limitations associated with scalability, availability, and compatibility of cell types such as hematopoietic stem cells on Earth, which are commonly utilized in clinical stem cell therapies. 11,12
Ongoing discussions revolve around the potential of a microgravity environment to enhance the differentiation efficiency and yield of stem cells into somatic cell types currently achievable on Earth. The level of maturity for cells is also considered as a variable. On Earth, diverse factors such as culturing conditions, cell source, and environmental cues contribute to significant variations in stem cell differentiation. 13,14 Factors such as cell passage number, freeze/thaw cycles, human error, and clone survival during reprogramming can impact the efficiency of differentiation batches. 15
Developing highly reproducible differentiation protocols, both in two-dimensional (2D) and three-dimensional (3D) contexts, becomes challenging due to these variables. Considering the existing variability in Earth-based protocols, it is essential to conduct parallel differentiation studies in LEO and on Earth simultaneously to provide groundside controls (Fig. 1). 1 To identify any microgravity-induced phenotypes, high-throughput and real-time analyses are necessary to assess the integrity and function of cell and tissue batches produced both on Earth and in LEO.
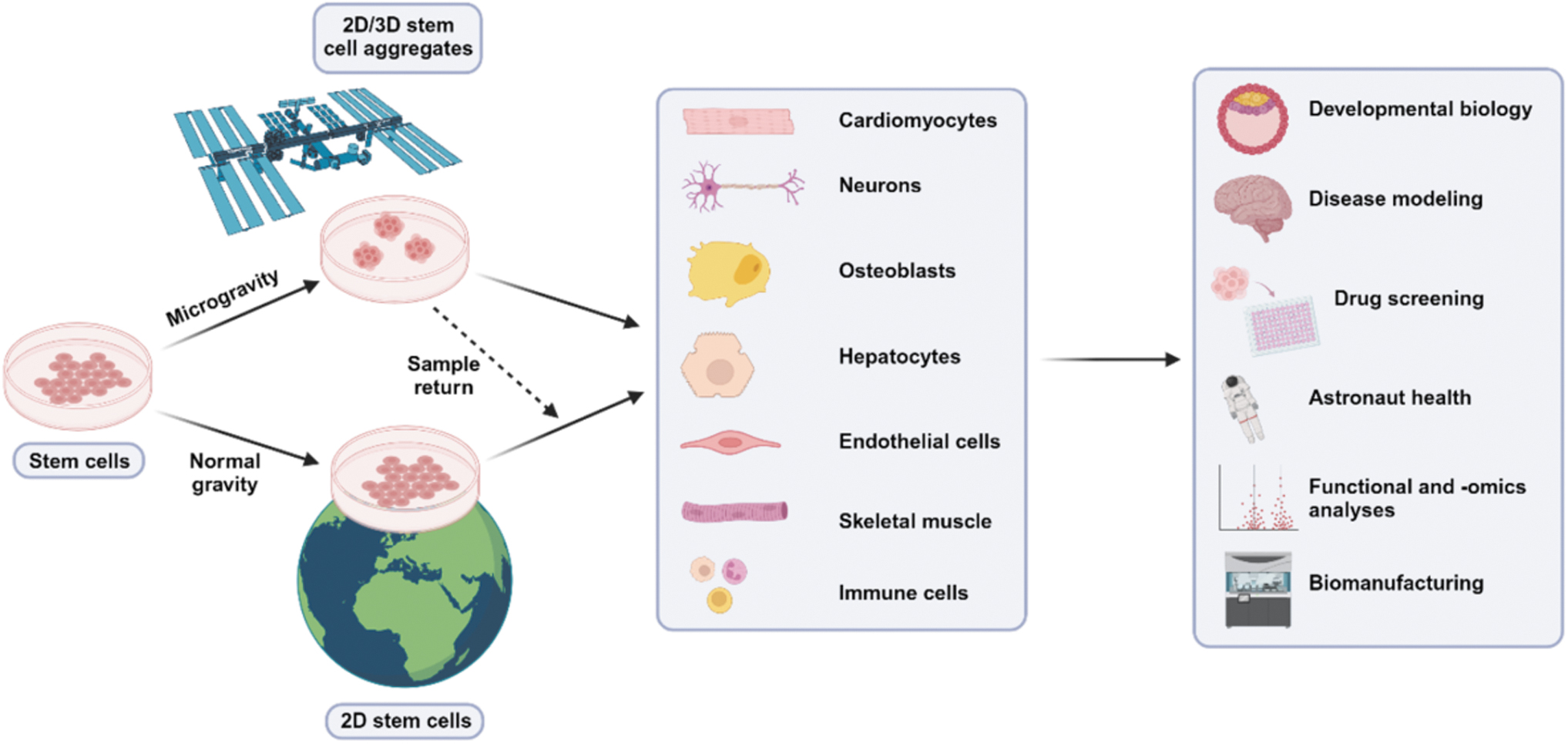
Testing the effects of microgravity on stem cell culture and downstream applications. Parallel stem culture studies conducted both in low-Earth orbit and on Earth may highlight potential mechanisms of microgravity that can improve the differentiation and production of cell types for use in biomanufacturing, clinical therapies, and better understanding human diseases. Figure made with BioRender.
Sample return from the ISS facilitates continued Earth-based studies on how stem cells respond to the environmental changes associated with microgravity. For instance, microgravity might act as a naturally accelerated environment for 3D stem cell growth due to expedited aggregation compared with Earth, where a gravitational force acts downward on culture plates. 16
3D Tissue Culture in Space
Tissue-engineered multilineage 3D models represent valuable tools that can recapitulate the intricate architecture, microenvironment, and functionality of living human tissues and organs. 7 Recognizing that 2D cell monocultures often fall short in replicating a physiologically relevant tissue environment, various advancements, such as tissue chips, organoids, assembloids, and 3D bioprinting, are accelerating drug discovery, advanced tissue modeling, and disease treatment on Earth. 7 The microgravity environment on the ISS is a crucial element in mechanistic exploration, particularly in understanding how weightlessness and gravitational unloading during spaceflight impact human health.
Despite efforts on Earth to simulate microgravity using methods such as rotating bioreactors and other microgravity-stimulating devices, the variability in cellular responses may not entirely mirror true microgravity experienced in LEO. 16 Although these systems can partially eliminate hydrostatic fluid stress on cells and tissues, they fall short of fully replicating the promotion of cell–cell aggregation, alterations in differentiation, or effects on protein turnover induced by microgravity. 17,18 Both on Earth and in LEO, certain essential components for better replicating 3D architecture, such as vascularization and nutrient perfusion, remain absent from tissue-engineered constructs.
Consequently, maintaining the functionality of stem cell-derived tissue over extended periods remains complex, especially when considering the additional intricacies associated with microgravity. Future studies in the 3D bioengineering of tissues and organs in microgravity should account for the fine tuning of numerous elusive variables on Earth before spaceflight.
Commercial Opportunities in Space
Despite the high costs associated with space travel, the growing involvement of private and commercial enterprises in crafting vehicles for research, development, and interplanetary exploration is poised to enhance the feasibility and frequency of studying the effects of microgravity on human health. This emphasis on understanding the impact of microgravity extends beyond astronauts, encompassing future space residents and innovative techniques to enhance human health on Earth. 2
At present, the primary consumers of stem cell-derived products on Earth are individuals utilizing them for research tools and therapeutic applications. However, the potential for large-scale production of specific stem cell-derived products suggests that developing such facilities in LEO could offer advantages not achievable on Earth. 2 Microgravity presents an opportunity for innovative insights that could significantly impact the stem cell research and development community, potentially influencing therapeutic options. Immediate efforts should be initiated to establish collaborations among biomanufacturing facilities, commercial space station developers, the stem cell research community, and government agencies.
A major focus should be on scaling up infrastructure for good manufacturing processes-grade manufacturing facilities in LEO. The realization of these opportunities hinges on the success of examples demonstrating how microgravity enhances stem cell properties. These examples will serve as a framework for further building and validating the commercial success of biomanufacturing and biomedical research in the unique environment of LEO.
The scarcity of primary research data or biological replicates from stem cell experiments in LEO has created numerous gaps that necessitate further exploration to render commercial opportunities realistic. Although research conducted on Earth provides valuable controls and comparisons for experiments in LEO, the inherent differences between the environments and the challenges of reproducing conditions in space often result in disparate or irreproducible variables. Researchers involved in LEO-based stem cell studies should adhere to current best practices established in Earth-based studies to enhance the translatability of their findings. 19
Leveraging advancements in high-throughput assays, miniaturization, microfluidics, robotics, machine learning, and artificial intelligence can significantly increase both the quantity and quality of data obtained, while concurrently reducing the crew time required to complete experiments in LEO. Furthermore, special attention should be given to the selection of cell sources for experiments in LEO, incorporating a diverse population that may either experience spaceflight or benefit from potential therapeutic opportunities.
Validation for Space Research
As humans spend more time in space, it becomes imperative to prioritize a deeper comprehension of the physiological effects induced by spaceflight. Furthermore, the potential for biomanufacturing in space to drive scientific and technological breakthroughs not attainable on Earth offers the promise of products with economic value and terrestrial benefits. The costs and time associated with LEO-based manufacturing are expected to decrease as more commercial entities engage in spacecraft and in-orbit platform development. 2 Ongoing experimentation in LEO may facilitate the integration of off-the-shelf products from Earth into current experimental hardware for stem cell culture in microgravity.
Although existing devices designed by space implementation partners adhere to NASA safety protocols and provide fully contained environments for research aboard the ISS, their production is expensive and limited in quantity. 20 Streamlining equipment and devices used for stem cell research in microgravity would further align to studies conducted on Earth for comparison purposes. This parallels with the simultaneous terrestrial growth in the utilization of stem cells and stem cell-derived products, given the ability of pluripotent stem cells to transform into various human somatic cell types for applications such as disease modeling, drug screening, and cell therapy.
Overcoming challenges in large-scale stem cell production on Earth, attributed to slow cell growth or the accumulation of genetic changes, necessitates a thorough understanding of how microgravity influences crucial stem cell properties such as production, maintenance, and differentiation. Such understanding is critical for expediting the validation of biomanufacturing applications in LEO.
Outlook
Despite the regular application and availability of 2D and 3D model systems on Earth, the importance of exploring and adapting these systems for long-term culture in space persists. Although experiments involving specialized cells and tissues are routinely conducted in LEO, offering valuable insights into how microgravity can impact various systems, it is imperative to replicate studies to substantiate the validity of experimental findings. This necessitates maximizing the data generated from each experiment per missions and investing in research to facilitate such missions. Although documentation and regulations are in place that each experiment aimed at being conducted aboard the ISS must follow and align with, there remains a lack of standardization across protocols due to the distinctive nature of each project and design.
Each project requires independent specifications and adaptations to make it possible to be conducted in microgravity. With the push for financial investment in space-based research, an additional goal should be made toward aligning all objectives for projects not yet completed with the outcomes of previously completed projects. Standardizing stem cell-based projects toward agreed-upon parameters would better aid in data production and replication. This would also help to more regularly implement research and design that has been tested in artificial gravity simulators on Earth but not in space.
As technology progresses in 3D modeling and high-throughput screening platforms, it becomes crucial to prioritize opportunities that demonstrate the clear economic value of LEO-based research. This clarity is essential to encourage future investments and sustain the growth of research activities in this unique environment.
Footnotes
Author Disclosure Statement
No competing financial interests exist.
Funding Information
The authors are grateful for support from the American Heart Association Career Development Award 856987 (to A.S.). A.S. is also supported by the Board of Governors Regenerative Medicine Institute at Cedars-Sinai, an In-Space Production Award NNJ13ZBG001N (InSPA) from NASA, and the Donna and Jesse Garber Award for Cancer Research. A.S. and J.M. are supported by a California Institute for Regenerative Medicine (CIRM) Bridges Award.
