Abstract
Oligodendrocyte precursor cells (OPCs) can differentiate into myelinating oligodendrocytes during embryonic development, thereby representing an important potential source for myelin repair or regeneration. To the best of our knowledge, there are very few OPCs from human sources (human-derived OPCs [hOPCs]). In this study, we aimed to evaluate the safety and remyelination capacity of hOPCs developed in our laboratory, transplanted into the lateral ventricles of young animals. Several acute and chronic toxicity experiments were conducted in which different doses of hOPCs were transplanted into the lateral ventricles of Sprague–Dawley rats of different ages. The toxicity, biodistribution, and tumor formation ability of the injected hOPCs were examined by evaluating the rats' vital signs, developmental indicators, neural reflexes, as well as by hematology, immunology, and pathology. In addition, the hOPCs were transplanted into the corpus callosum of the shiverer mouse to verify cell myelination efficacy. Overall, our results show that transplanted hOPCs into young mice are nontoxic to their organ function or immune system. The transplanted cells engrafted in the brain and did not appear in other organs, nor did they cause tissue proliferation or tumor formation. In terms of efficacy, the transplanted hOPCs were able to form myelin in the corpus callosum, alleviate the trembling phenotype of shiverer mice, and promote normal development. The transplantation of hOPCs is safe; they can effectively form myelin in the brain, thereby providing a theoretical basis for the future clinical transplantation of hOPCs.
Introduction
Newborns are one of the most at-risk groups for developing demyelinating diseases [1], characterized by loss of or damage to the central nervous system's oligodendrocytes (OLs), the primary myelin sheath-forming cells in the brain and spinal cord. Recovery from this disease requires myelin replacement through normal proliferation and differentiation of OLs [2]. Over the past few decades, a series of studies have been conducted on treatment of demyelinating diseases with cell transplantation. Stem cells have nerve regeneration, neuroprotection, and neurotrophic properties [3,4]; therefore, stem cell transplantation is considered one of the most promising treatment methods. Studies have shown that OLs can be differentiated from oligodendrocyte precursor cells (OPCs), and OPCs have greater proliferation and migration ability than OLs [5]; therefore, OPCs are more suitable for post-transplant survival, which also increases the extent of myelination.
Studies have shown that stem cell transplantation can cause adverse reactions, including vomiting, low-grade fever, headache, occasional chest tightness, intercostal neuralgia, abdominal distension, meningitis, and rash. It does not cause side effects such as tumors, seizures, arrhythmia, allergic reactions, and local infections [6,7]. Currently, there are few reports about adverse reactions related to cell transplantation, and it is relatively safe. Safety studies on stem cell transplantation for other neurological diseases have been reported: bone marrow mesenchymal stem cell transplantation for spinal cord injury [8]; human allogeneic central nervous system stem cells (hNSCs) transplantation for Pelizaeus-Merzbacher disease [9]; human neural stem cell (hNSC) transplantation for Alzheimer's disease [10]; and olfactory ensheathing cells for cerebral infarction sequela [11]. These have confirmed the safety of stem cells for the treatment of neurological diseases. However, neonatal nervous, immune, and digestive systems are not mature, and newborns are more prone to headaches, low-grade fever, and vomiting [12,13]. Establishing the preclinical safety of cell transplantation for treatment of neonatal demyelinating diseases is, therefore, extremely important.
Although several studies have confirmed the effectiveness of OPCs transplanted into neonatal animals to form myelin, most transplanted cells were of rat or mouse origin [14 –16], and only a small number used human-derived OPCs (hOPCs) [17 –19]. The transplanted cells in our study are OPCs of human origin. Previously, our group isolated hNSCs from 10- to 13-week-old aborted infants, which were then differentiated into hOPCs. In a separate study, we verified myelination ability of cells by transplanting hOPCs into an injured animal model (white matter damage) [20]. However, the safety of cell transplantation needs further study. In this study, we use shiverer mice (an uninjured model) to briefly verify the remyelination ability of hOPCs.
Materials and Methods
Cultivation and identification of hOPCs
The Pediatric Laboratory of the Sixth Medical Center of the Chinese People's Liberation Army General Hospital provided human fetal central nervous system tissue obtained from 10- to 13-week-old embryos. Written informed consent was obtained from each woman who donated their aborted fetuses. Tissue was prepared as primary cells (Supplementary Text S1). The preparation of the hOPCs was approved by the Institutional Review Board of The Sixth Medical Center of PLA General Hospital (Application No. 2015013). Before cell transplantation, hOPCs were identified by flow cytometry and cell morphology. HOPCs were surface stained with PDGFR-α BV421 mouse anti-human (Cat. #562799; BD Biosciences, Franklin Lake, NJ), A2B5 PE mouse anti-human (Cat. #130-093-581; Miltenyi Biotec, Bergisch-Gladbach, Germany), NG2 APC mouse anti-human (Cat. #FAB2585A; R&D Systems, Minnesota), and Ki67 PE mouse anti-human (Cat. # ab270650; Abcam, Cambridge, UK) antibodies for flow cytometry (FACSanto II; BD Biosciences). To further identify the transplanted cells, we performed immunofluorescence staining on brain tissue from 21-day transplanted rats (from the acute toxicity test described below). First, we stained brain tissue with STEM121 to confirm that the transplanted cells were human derived. PDGFR-α, A2B5, NG2, and Ki67 were performed on the brain tissue to determine that the transplanted cells were hOPCs (Supplementary Text S2).
Animals
Sprague-Dawley (SD) rats and homozygous shiverer mice (Jackson Laboratory, Maine) were maintained in a specific pathogen-free environment at the hospital. All animal experiments were performed according to protocols approved by the hospital's Animal Care and Use Committee (Application No. SCXK-2012-0001). Room temperature was set to 23°C ± 2°C, humidity was maintained at 60% ± 10%, and there were light and dark cycles of 12 h each. The animals had ad libitum access to sterile food and water.
Safety of hOPC transplantation
In the acute toxicity experiments, transplantation was performed on postpartum day 6. We measured weight, tibia length, righting reflex, and cliff avoidance reaction every 2 days for 14 days following transplantation (21 days after birth). On day 14, the young rats were sacrificed by cervical dislocation. In the chronic toxicity experiment, a total of three transplants were performed; transplantation time points corresponded to postpartum days 6, 20, and 40. After the first transplantation, body weight and tibial length of the mice were measured every 3 days until the end of the experiment (postpartum day 90). After the third transplantation, blood was taken from the tail vein every 2 weeks for routine and biochemical blood tests. After the third blood collection, blood immune factor detection and blood immune cell detection were carried out simultaneously. Immunofluorescence was used to detect residual hOPCs in various tissues. Before being sacrificed, all transplanted rats were photographed. After sacrifice, coefficients of the heart, liver, spleen, lung, kidney, brain, thymus, spinal cord, testes, and ovary were calculated using the formula OC = m/M%, where m represented the organ's weight and M represented the rat's weight.
Transplantation of hOPCs
HOPCs were transplanted into the right lateral ventricle of the experimental group (n = 12), and saline was injected into the right lateral ventricle of the control group (n = 12). The subgroups were (1) male cell (n = 6), (2) female cell (n = 6), (3) male saline (n = 6), and (4) female saline (n = 6). Rats anesthetized with isoflurane were fixed on a rat brain stereotaxic apparatus (Stoelting, Wood Dale, IL), and their heads were disinfected before the skin was incised to expose the skull. The anterior fontanelle was the zero point. The injection site (AP, anterior and posterior; ML, midline-lateral; and DV, depth) was approached based on the zero point [21]. In the acute toxicity experiments, the lateral ventricle of each rat was injected with either 5 μL of hOPCs (1 × 106 cells) or 5 μL of saline; injection rate: 0.5 μL/min; and the lateral ventricle coordinates were AP: 0.5 mm, ML: 1 mm, and DV: 2.0 mm. In the chronic toxicity experiments, each rat was transplanted thrice, once every 2 weeks. During the first, fourth, and seventh week, each rat was injected with a volume of 5, 10, and 10 μL, respectively, of either hOPCs (1 × 106 cells, 2 × 106 cells, and 2 × 106 cells) or saline. The coordinates of the lateral ventricle corresponding to the three transplants were AP: 0.5 mm, ML: 1 mm, and DV: 2.0 mm; AP: 0.5 mm, ML: 1 mm, and DV: 1.0 mm; and AP: 0.5 mm, ML: 1 mm, and DV: 1.0 mm. After transplantation, the incision was sutured, skin was disinfected, and the young rats were returned to their mothers for feeding and observed closely for 48 h. After weaning, female and male rats were reared in separate cages.
Neural reflex detection
As part of the acute toxicity experiments, a neural reflex test was performed. Righting reflex: after transplantation, whether the supine young rats could turn over and touch the ground within 2 s was observed and assessed; if all young rats were positive for three consecutive tests on the same day, the day was considered a standard day. Cliff avoidance reflex: starting from the first day after transplantation, young rats were placed on the edge of a workbench with a height of 30 cm; if the rats retreated or turned around from the edge within 90 s, the reaction was considered positive; if all rats in the same cage were positive, the day was considered a standard day.
Blood routine and serum biochemistry
In the chronic toxicity experiment, we collected blood thrice after the final transplantation, at 2-week intervals (the 9th, 11th, and 13th week after birth). Detection indicators included red blood cells, white blood cells (WBCs), platelets (PLTs), hematocrit, hemoglobin, and mean PLT volume. Using a biochemical analyzer (HITACHI 7020, JAPAN), we measured alanine aminotransferase, aspartate aminotransferase, albumin, creatinine, urea, glucose (GLU), total protein, total cholesterol, and triglycerides.
Detection of cytokines and immune cells in peripheral blood
At the last blood collection in the chronic toxicity experiment (13th week after birth), an enzyme-linked immunosorbent assay (ELISA; R&D Systems) was used to detect interferon-gamma, tumor necrosis factor alpha (TNF-α), interleukin (IL)-2, IL-4, IL-5, and IL-6. The assay was performed according to the manufacturer's instructions. Another fresh blood sample was made as a cell suspension. Finally, the rats were sacrificed by cervical dislocation. The spleen was removed under aseptic conditions and was made a cell suspension. These two cell suspensions were surface stained with CD3 APC mouse anti-human (Cat. #562799; BD Biosciences), CD4 APC-A750 (Cat. #130-093-581; Miltenyi Biotec), CD8 PC5.5 (Cat. #FAB2585A; R&D Systems), and CD45RA PC7 mouse anti-human antibodies for flow cytometry (FACSanto II; BD Biosciences). The assay was performed according to the manufacturer's instructions.
Cell distribution, histopathology, and tumor formation analysis
After the chronic toxicity experiments, the heart, liver, spleen, kidney, thymus, spinal cord, testes, ovary, brain, and lung were fixed in 10% formalin. Immunofluorescence staining with STEM121 was performed to label residual hOPCs in each tissue. Tissue sections were stained with hematoxylin and eosin (H&E), and an upright optical microscope was used for observation, inspection, and evaluation of histopathological lesions. Ki67 immunofluorescence staining was used to assess the proliferation of brain tissue, so as to assess whether the transplanted cells form tumors in the brain tissue (Supplementary Text S3).
Study of effectiveness
hOPC transplantation in shiverer mice and tremor assessment
Newborn shiverer mice (n = 6) were transplanted within 1 day after birth. A mouse brain stereotaxic apparatus (Stoelting) was used to inject hOPCs (1.5 μL; 2 × 105 cells; the cells used in this part were the same batches used in the safety study) into the right corpus callosum. Transplant coordinates were AP, 0.5 mm; ML, 1 mm; and DV, 0.5 mm. The control group consisted of six randomly selected nontransplanted homozygotes. After surgery, body weights were recorded at the same time every 7 days until the end of the experiment. At 30, 60, and 90 days after transplantation, tremor was assessed. Indicators included amplitude of tremor and duration of continuous tremor.
Electron microscopy and tissue immunofluorescence staining
Three months after transplantation, mouse brain samples were imaged using a transmission electron microscope (TEM, H7650-B; HITACHI, Tokyo, Japan). A field of view was randomly selected for each mouse, and the G-ratio was calculated (myelin inner diameter/myelin outer diameter) according to the amount of myelin in the field of view. The remaining mouse brain tissues were immunostained with myelin basic protein (MBP) (rat anti-MBP antibody, Cat. #Ab7349; Abcam). A fluorescence microscope (IX-70; Olympus Corporation) was used to image the samples; average optical density of MBP was calculated using Image Pro Plus 6.0.
Statistical analysis
Statistical analyses were computed using SPSS version 22.0 (Armonk, NY). Data were expressed as mean ± standard error. Mean of continuous data was analyzed using analysis of variance. The Dunnett's t-test was used to compare groups. Values of P < 0.05 were considered statistically significant.
Results
hOPCs identified by main markers
Before transplantation, hOPCs were identified by morphology, and main markers were quantified using flow cytometry. Brightfield microscopy verified that hOPCs had a typical bipolar, bead-like morphology (Fig. 1A), and were able to be stably passaged up to the fifth generation. Flow cytometry analyses showed that the proportion of PDGFR-α+ cells was 72.27% ± 3.01%. A2B5+ cells accounted for 25.87% ± 3.02%, NG2+ cells accounted for 18.38% ± 1.51%, and Ki67+ cells accounted for 47.28% ± 0.96% (Fig. 1B) of the total cell population. After transplantation, the four markers in the animal brain tissues were examined using tissue immunofluorescence staining (Fig. 1C).
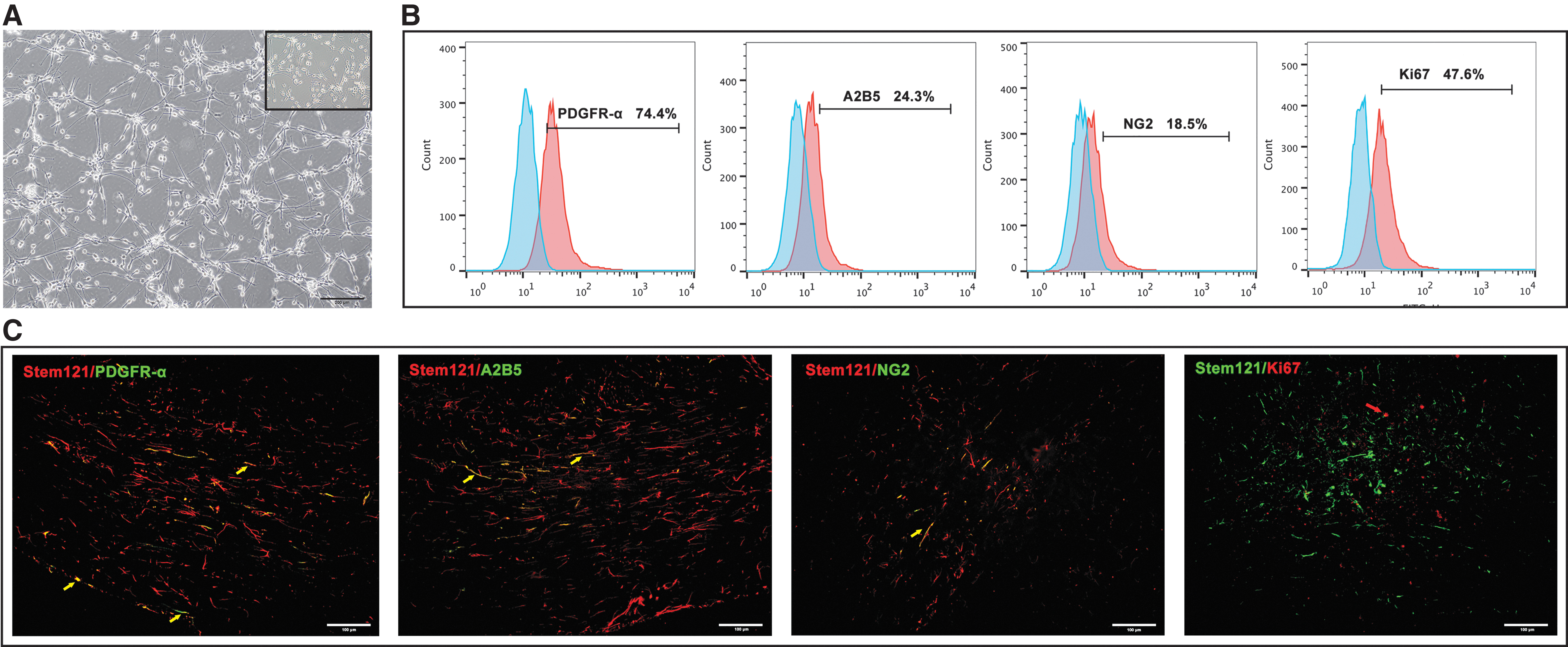
Culture and identification of hOPCs. The transplanted cells were identified based on their morphology and main markers.
Safety of hOPC transplantation
Evaluation of growth and neural reflex
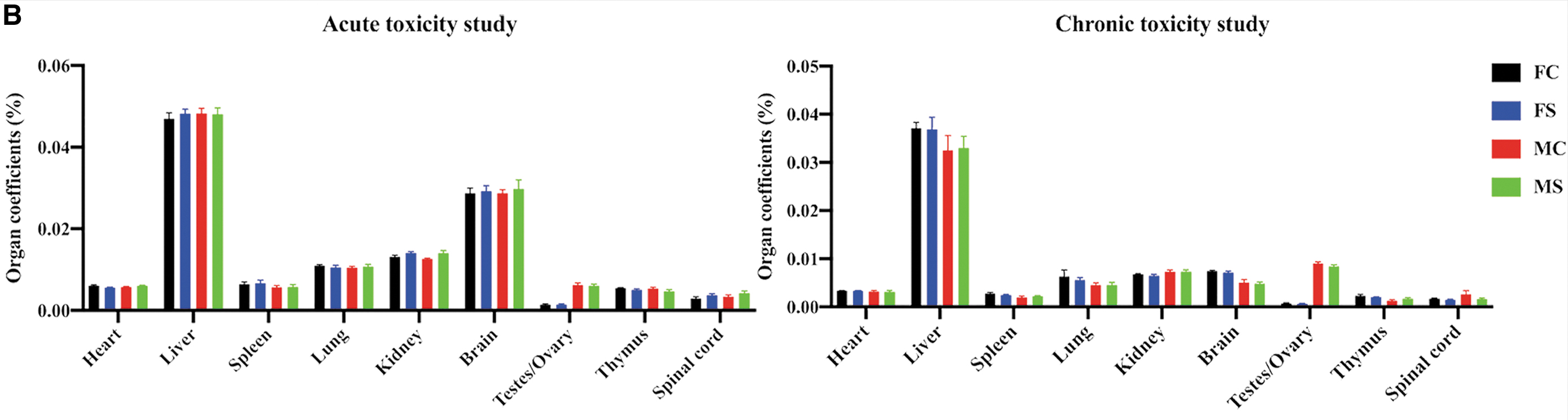
Body surface characteristics of both hOPCs transplanted and controls were normal. Major organs were normal in appearance and displayed no sign of inflammation, exudation, bleeding, tissue proliferation, or tumor formation (Fig. 2A). Organ coefficient results showed no statistically significant difference between hOPCs transplanted and controls (Fig. 2B). Results of the tibia length test showed that there was no significant difference between hOPCs transplanted and controls (acute: P = 0.99 and chronic: P = 0.91; Fig. 3A). Weight measurement results showed that the difference between hOPCs transplanted and controls was not statistically significant (acute: P = 0.64 and chronic: P = 0.78); however, weight difference between females and males in the chronic toxicity experiment was statistically significant (P < 0.05, Fig. 3B). In the acute toxicity experiment, righting reflex evaluation results showed that the time the rats needed to reach a standard day was 14.66 ± 0.88 days (hOPCs transplanted) and 14.25 ± 1.06 days (controls), but was not statistically significant (P = 0.31). Cliff avoidance reflex evaluation results showed that the time the rats needed to achieve a standard response was 14.92 ± 0.67 days (hOPCs transplanted) and 14.83 ± 0.94 days (controls), but was not statistically significant (P = 0.80) (Fig. 3C).
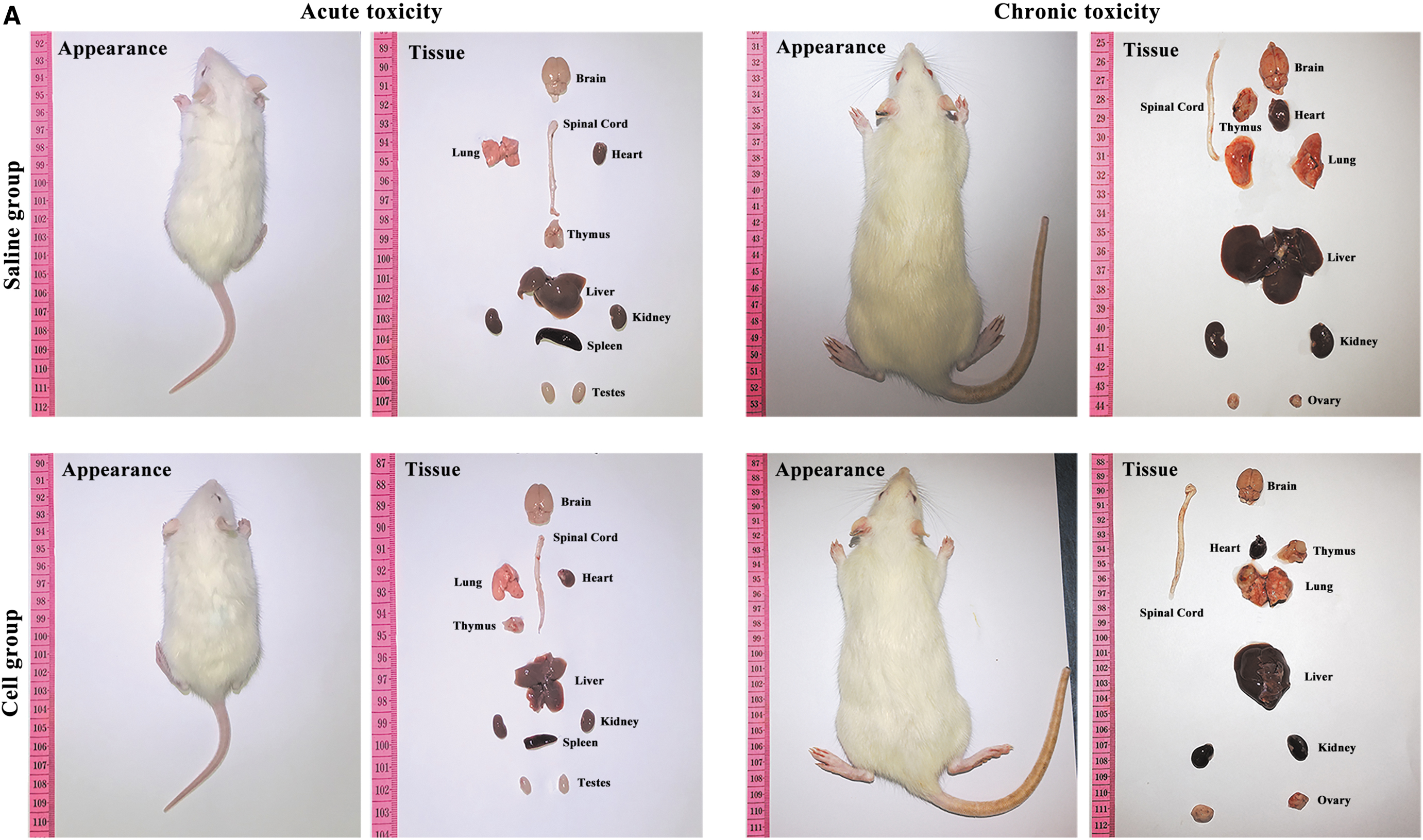
Rat body surface and main internal organs. The rat's body surface and main organs were evaluated for symptoms, including inflammation, exudation, bleeding, tissue proliferation, or tumor formation.
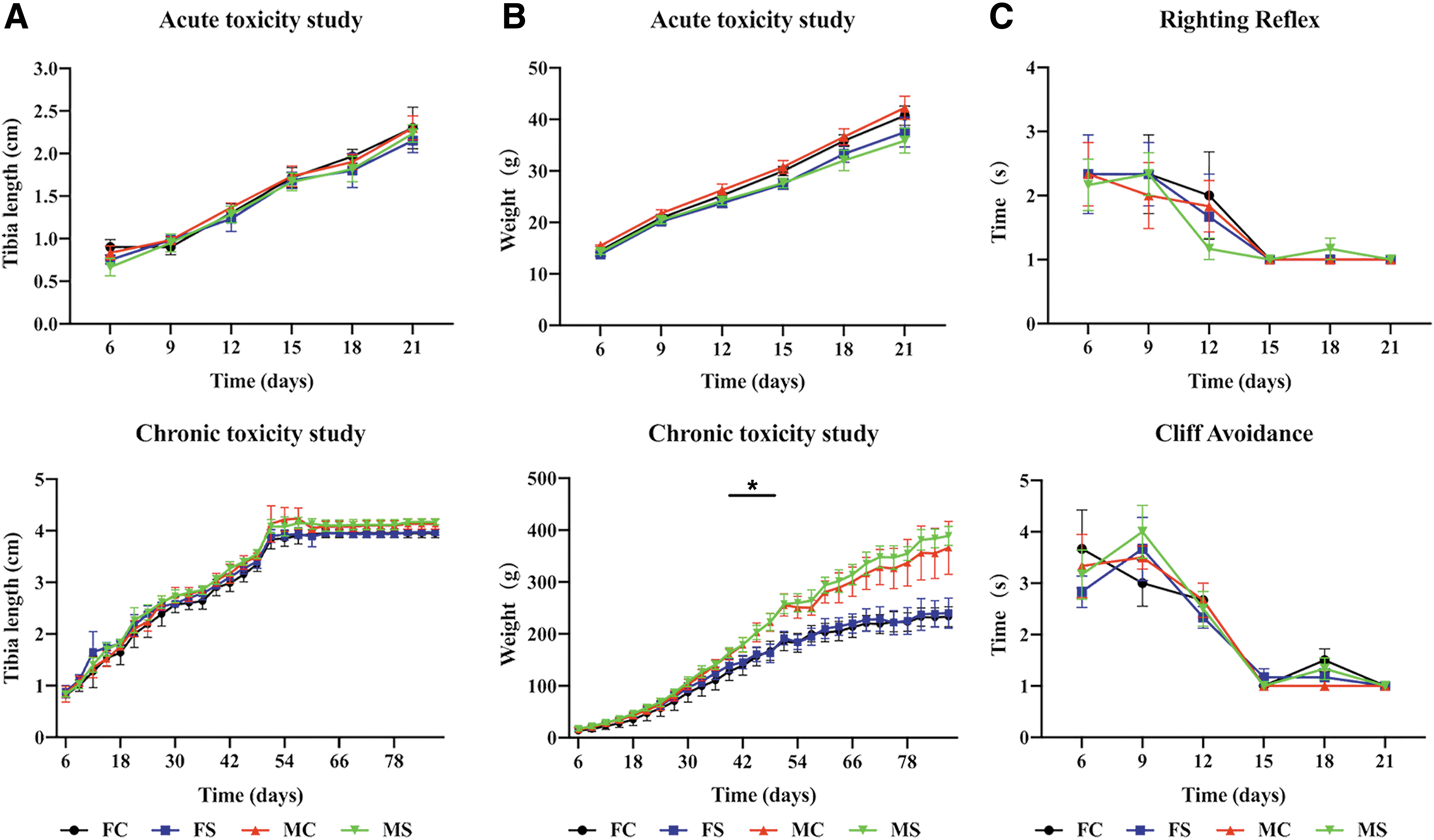
Rat growth and neural reflex. The growth of the rats and the development of the nervous system were assessed.
Hematology and immunological assessment
On the chronic toxicity test, routine blood examination and peripheral blood biochemical results showed no significant difference between the two groups (Fig. 4, Supplementary Tables S1 and S2). Analyses for serum cytokines and immune cell detection in the peripheral blood and spleen showed no significant differences between the two groups (Fig. 5A–E and Supplementary Table S3).
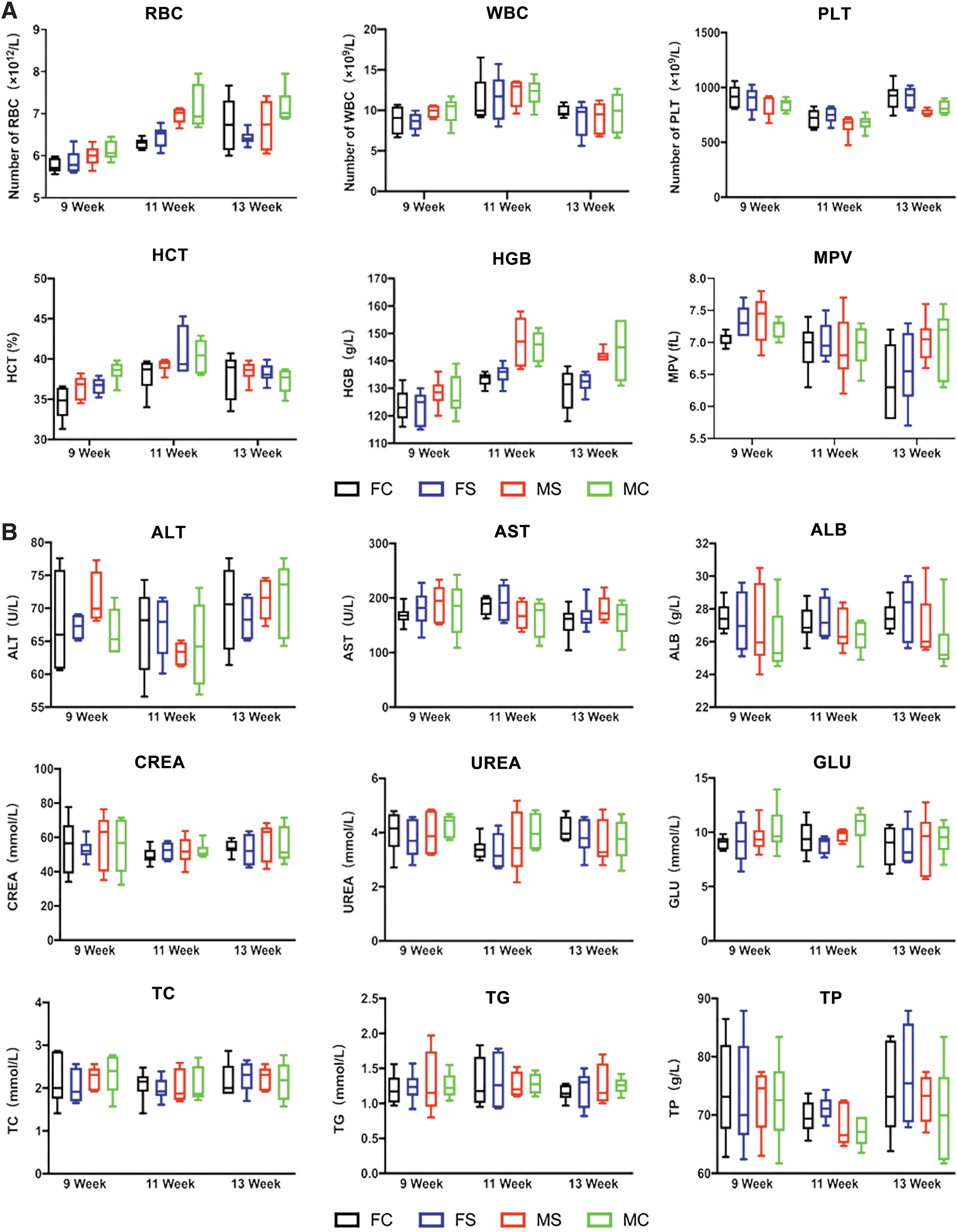
Chronic toxicity laboratory hematology examination. Symptoms of anemia and infection, as well as normality of liver and kidney functions were assessed in the rats.
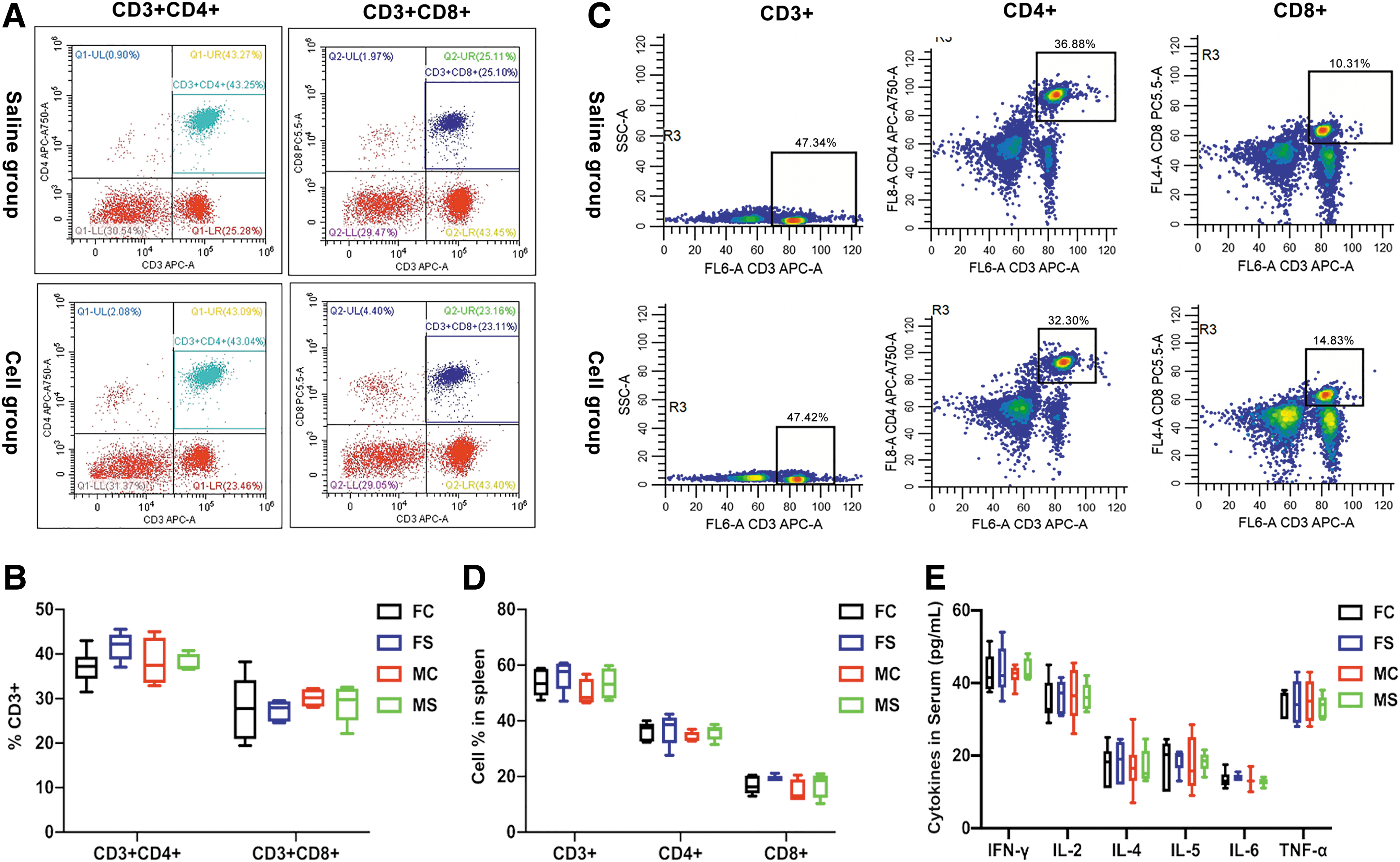
Immunological evaluation of chronic toxicity experiment. Effects of the transplanted cells on the immune system of the rats were evaluated.
Biodistribution and tumor formation
STEM121 immunofluorescence staining of main organ tissues showed that human-derived cells were found only in the brain tissue (Fig. 6A). In the chronic toxicity test, histopathological evaluations of the rat's main organs did not reveal any sign of inflammation (Fig. 6B). Tumor formation of transplanted cells was evaluated using Ki67 immunofluorescence staining of brain tissues at different time points of transplantation (Fig. 6C and Supplementary Fig. S1A). Results showed that differences between the transplanted group and controls were not statistically significant (Fig. 6D and Supplementary Fig. S1B).
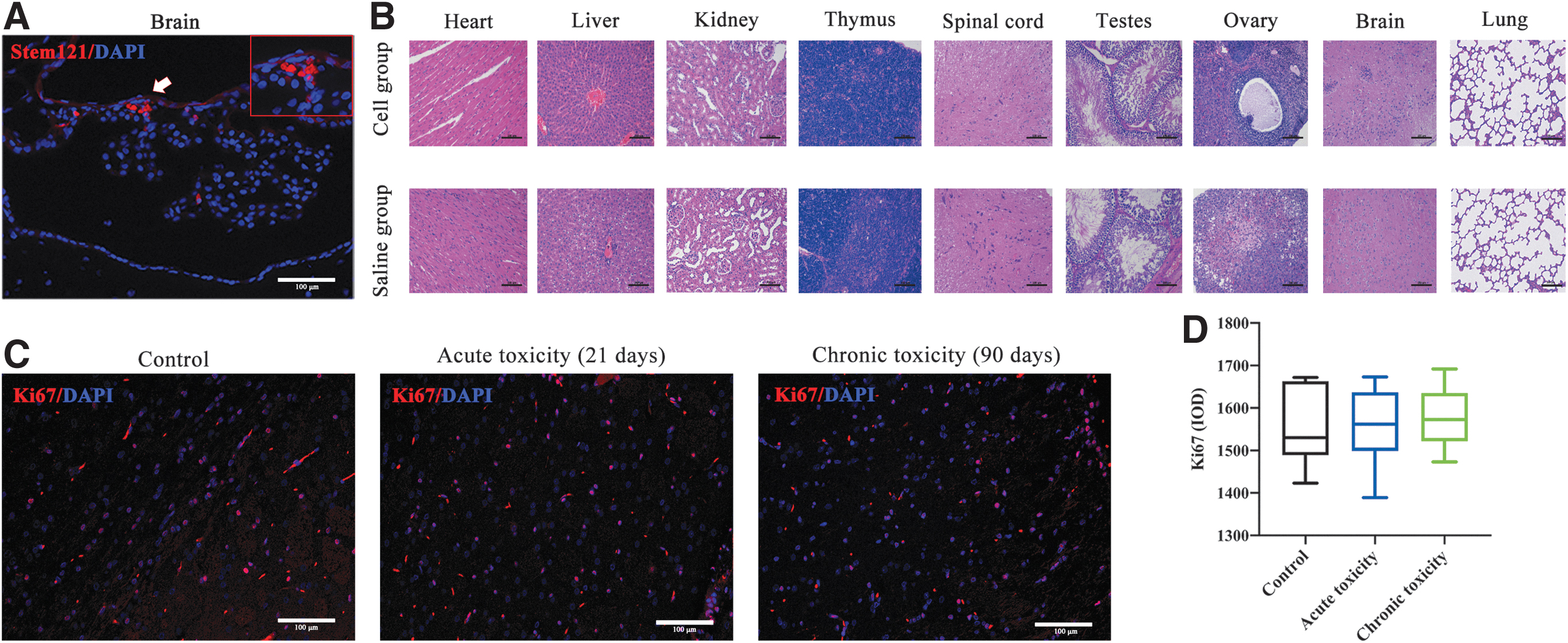
Cell distribution, histopathology, and tumor formation analysis in chronic toxicity experiment. Biodistribution of transplanted cells and whether they caused pathological damage or tumor formation to major organs were assessed.
Confirmation of myelination
Three months post-hOPC transplantation, brain imaging with transmission electron microscope revealed a small amount of mature myelin in the homozygous nontransplanted shiverer mice control group. Most of the myelin sheaths did not form dense lines, and the overall structure was loose. In the hOPC transplanted group, the number of mature myelin sheaths was large, and dense lines were formed (Fig. 7A). The results of the myelin G-ratio analysis showed that hOPCs transplanted and control values were 63.59% ± 3.36% and 94.42% ± 1.42%, respectively, which was statistically significant (P < 0.0001, Fig. 7B). MBP immunostaining confirmed myelination in the corpus callosum (Fig. 7C). Mean optical density values, derived from MBP labeling, were 0.071 ± 0.005 (hOPCs transplanted) and 0.017 ± 0.002 (controls) and were statistically significant (P < 0.0001, Fig. 7D). Body mass index test results showed that within 90 days post-transplantation, there was a statistically significant weight gain difference between the two groups, (P < 0.005, Fig. 7E). Results of tremor amplitude and duration of a single continuous tremor observation within 91 days post-transplantation were statistically significant between the groups (Fig. 7F, G and Supplementary Table S4).
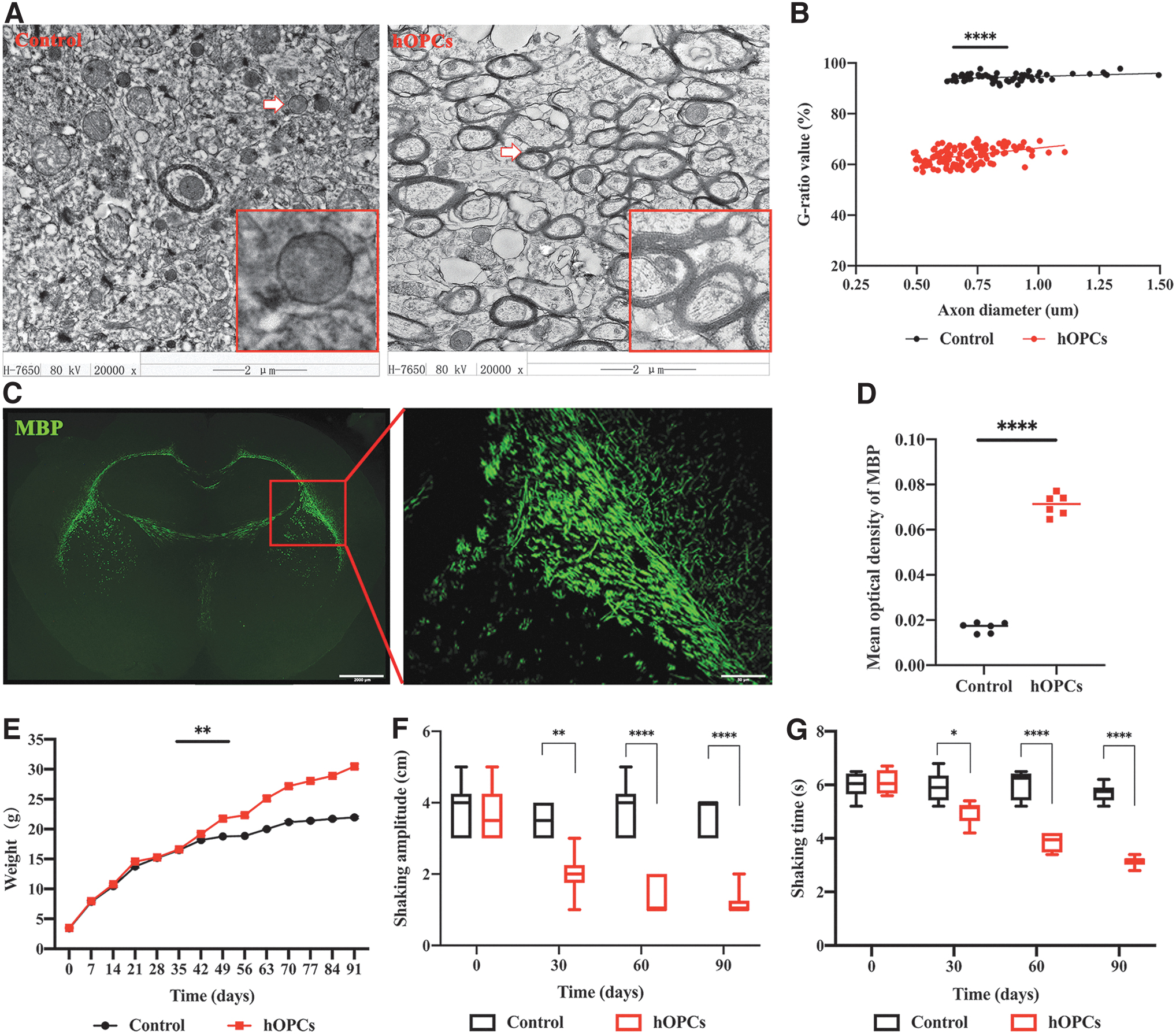
Evaluation of the effectiveness of hOPC transplantation into shiverer mice. Myelination ability of transplanted cells and their effects on the body weight and tremor symptoms of mice were evaluated.
Discussion
Building on current research [22], safety studies of stem cell transplantation usually include purity and stability of stem cell line, cell dose, species and age of animals, experimental period, and selection of test indicators [23]. In this study, we transplanted different doses of hOPCs into the lateral ventricles of SD rats of different ages, and performed both acute short-term (21 days) and chronic long-term (90 days) toxicity tests. During the experiment, we assessed growth and development of the rats, and assessed their nervous system, hematology, immunology, and histopathology; findings demonstrated that hOPC transplantation had no toxic side effect. Next, we showed that biodistribution of the transplanted hOPCs was limited to the brain and that transplanted cells did not result in tissue proliferation or tumor formation. In addition, this work confirms that hOPCs transplanted into the corpus callosum of shiverer mice can form myelin, alleviating the shivering behavior caused by demyelination, and thereby promoting the normal growth of these mice.
Current research shows that there are many ways to obtain hOPCs from the nervous system. For example, using fluorescence-activated cell sorting (FACS) or magnetic-activated cell sorting technology to sort the target cells from the brain tissue of the fetus or adult [24,25]. However, in addition to limited sources of human tissue, this method has ethical issues associated with the use of human tissue. Another way to obtain hOPCs is to induce human pluripotent stem cells (hPSCs) or human embryonic stem cells to differentiate into hOPCs [26]. However, directional differentiation of hPSCs into hOPCs requires a stage of directional differentiation of hPSCs into neural progenitor (NP) cells, and then a stage of differentiation of NPs into hOPCs. This cell induction method not only consumes a large number of different types of cytokines but also takes 21–150 days to obtain high-purity hOPCs [27,28]. This increases cost and limits the potential productivity of hOPCs.
hOPCs in this study are induced from hNSCs. There are many advantages to using hNSCs as the source of hOPCs: (1) with informed consent from the pregnant woman, obtaining hNSCs from the fetal brain of aborted infants avoids serious ethical problems; (2) current methods for obtaining hNSCs from fetal brain tissue are very mature, including selection of gestational age (10–15 weeks), isolation and culture methods, and cryopreservation and resuscitation [29,30]; (3) because a large number of hNSCs can be obtained from the fetal brain tissue, a large number of hOPCs can be obtained as long as the targeted differentiation can be achieved; (4) it only takes 6–7 days to induce hNSCs into hOPCs, saving precious time for clinical transplantation; (5) this type of hOPC has good proliferation ability and can be stably expanded to the fifth generation; and (6) in our hOPC culture system, only a small number of cytokines need to be consumed. In this study, we first assessed the purity and stability of the hOPCs through cell morphology and biomarker analysis. According to previous reports, hOPCs are typical bipolar bead-like cells; they express PDGFR-α, A2B5, NG2, SOX10, and Olig2 [31], which is consistent with our results. Findings showed that the cells before and after transplantation expressed PDGFR-α, A2B5, NG2, and Ki67.
The current selection of toxicology animal models usually includes time of physiological development and organ development similar to humans, costs, and ease of obtaining experimental animals [32]. In this study, we selected SD rats for the safety investigation. In the investigation that evaluated myelin forming ability of transplanted cells, we used shiverer mice, an animal model with congenital myelination loss, very suitable for testing the myelinating ability of transplanted cells [33]. Considering developmental characteristics of animal species, choice of age is very important. In this study, we selected newborn SD rats and newborn shiverer mice for the experiments.
In the safety study, we examined toxicity, biodistribution, and tumorigenicity of transplanted cells. Herein, the acute toxicity experiment showed that within 21 days of cell transplantation, there were no transplant-associated deaths among the young rats. Furthermore, growth indicators such as skin color, abdominal hair, body weight, and bone development were normal. Breathing, exercise, digestion, and nervous system-related indicators were also normal. In the chronic toxicity test, to avoid complications such as infection, hematoma, and brain herniation that can be caused by multiple transplants, we performed transplantation every 2 weeks, using a very slow injection speed (0.5 μL/min). Hematological indicators and immunological tests taken from multiple time points were normal. Relevant indicators of routine blood tests were at normal values. Although WBC and PLT fluctuated among the three tests, no symptom of infection or anemia was found. Liver and kidney function and blood lipids were normal [34], as were GLU levels. Levels of immune-related factors and immune cells in the blood and spleen were also within the normal range. H&E staining of major organs showed normal structure without inflammation. We observed that transplanted cells were found in the brain tissues of both the 21- and 90-day-old rats (Fig. 1C), and although they were not cleared quickly, they did not cause a recognizable immune response or other adverse symptoms.
Regarding hOPC biodistribution after lateral ventricle transplantation, STM121 immunofluorescence staining of major peripheral organs showed that transplanted hOPCs only engrafted in brain tissue and did not localize to other regions (at 90 Ridays). In a study of treatment for spinal cord injury, migration distance of OPCs in the spinal cord parenchyma was only about 3 cm [35]. Therefore, it was not expected that hOPCs transplanted into the lateral ventricle would migrate.
As hOPCs have several stem cell characteristics, risk of tumor formation after transplantation cannot be ignored [36]. In this study, even on the 13th week of the chronic toxicity experiment, no neoplasm was observed on macroscopic and microscopic aspects of SD rats in the transplantation group; moreover, the weight change and organ coefficient of the transplantation group were not different from those of the control group. In addition, pathological analysis and brain tissue Ki67 immunofluorescence staining showed no tissue proliferation or tumor formation in any major organ.
Shiverer mice were used to evaluate the myelinating ability of hOPCs. Consistent with other studies, transmission electron microscope and MBP immunofluorescence staining conducted 90 days after hOPC transplantation confirmed myelination of the corpus callosum. Furthermore, the tremor phenotype was relieved in the transplanted group. The appetite of the transplanted mice also increased, resulting in greater body weight than in the controls. Studies have shown that the internal environment of the brain can affect function of transplanted cells, and some cytokines secreted by transplanted cells can affect brain development. Compared with the adult brain, the internal environment of the neonatal brain is more beneficial to transplanted cells, and the transplantation effect is better [37,38]. First, there are more silent synapses in the brain tissue of newborns (including newborn animals) than in adults. In early stages of neonatal neurodevelopment, because either the presynaptic structure cannot release neurotransmitters or the postsynaptic membrane lacks related receptors, many synapses are in a silent state. However as the nervous system develops, the number of silent synapses gradually decreases [39,40]. Second, there are many neurotrophic factors in neonatal brain tissue that may help activate these silent synapses [41,42], including glial cell-derived neurotrophic factor, neurotrophin-3, brain-derived neurotrophic factor (BDNF), and neural growth factor (NGF) [43,44]. In addition, transplanted OPCs can secrete neurotrophic factors including BDNF, NGF, and ciliary neurotrophic factor, activating more silent synapses [45,46]. Importantly, OPCs can express neurotransmitter receptors and a variety of ion channels, and form synaptic connections with myelinated neurons, thereby promoting myelination of neuronal axons [47,48]. Compared with adults, transplantation of OPCs to newborns or young children has better effects; the earlier, the better.
The animal models used in this study are uninjured models. Studies have shown that transplanted OPCs can survive longer in uninjured models, having a better effect on remyelination than injured models [49]. This may be because aggressive neuroinflammation in injured models can inhibit survival and differentiation of transplanted OPCs [50]. Other studies have shown that inflammatory factors in brain tissue, such as TNF-α and transforming growth factor-beta, may promote the proliferation and migration of OPCs, which is beneficial to myelin regeneration [51,52]. Therefore, to exclude the impact of the injured environment on the transplanted cells, we ultimately chose an uninjured model for our research.
Findings from this work provide theoretical guidance for future clinical trials of lateral ventricle transplantation of hOPCs for treatment of demyelinating diseases. First, we determined that the safe dose of hOPCs for single transplantation of cells in 1-week-old newborn SD rats is 1 × 106, with an injection volume of 5 μL. At postpartum week 7, the safe dose can be increased to 2 × 106, with an injection volume of 10 μL. The efficacious number of myelinating cells needed to alleviate tremor phenotype in newborn Shiverer mice is 2 × 105, with an injection volume of 3 μL. These experimental doses do not cause brain herniation, and the cell suspension does not overflow. We recommend experimental doses to not greatly exceed this.
Second, detection indicators after transplantation showed potential to provide references for clinical trials. The choice of observation indicators plays an important role in experimental study of juvenile animal toxicity. According to published guidelines, general evaluation indicators usually include gross observation, vital signs, growth and development, hematology, and pathology. If a specific target organ or system is considered, it can be combined with function indicators, including reproductive, sexual development, immune, and central nervous system [53,54].
In addition, length of observation period, transplantation time points, and index detection time points from this study can be used as reference values for future clinical trials. Studies have shown that within 22 days of birth, rats are equivalent to human infants/toddlers (<2 years), in which basic physiological development characteristics have been established. Moreover, within 46 days of birth, rats are equivalent to human children at 2–12 years of age. Important organs are fully developed when mice are 13 weeks old, equivalent to the adolescent period (12–16 years) in humans. The development of neural reflexes, learning and memory, kidneys, and other systems are relatively complete when rats are about 20 weeks old, equivalent to human adulthood [55,56]. In summary, cell transplantation dose, test indicators, and time design have guiding significance for future investigations.
This study has some limitations. First, it involved rodent models and did not use large animals, such as canines or primates. To date, rats have been the most commonly used and preferred rodents for experimental research in young animals, accounting for about 80% of research in this field, with dogs as the most commonly used nonrodent species, accounting for ∼8% [57,58]. Second, we demonstrated remyelination ability using the same batch of hOPCs from the safety study, and did not use magnetic resonance [59] or diffusion tensor imaging technology [60] to quantify remyelination or changes in brain development. Moreover, there was no further study on the degree of proliferation of transplanted cells in the AP axis of the mouse brain and the dynamic process of remyelination. Although these do not affect our conclusions, high-quality research is warranted in these to further confirm findings.
Conclusion
In conclusion, this study provides evidence of the safety of hOPC transplantation in young animals. Our study also confirms the efficacy of its myelination potential. Together, these results provide a theoretical basis for future clinical studies of hOPC transplantation for the treatment of neonatal myelin diseases.
Footnotes
Acknowledgments
The authors appreciate the technical support and other help from Leping Zhang and Tiantian Qiu. We would like to thank Editage (
Author Disclosure Statement
No competing financial interests exist.
Funding Information
This research was supported by the National Key R&D Program of China (NO.2017YFA0104200).
Supplementary Material
Supplementary Text S1
Supplementary Text S2
Supplementary Text S3
Supplementary Figure S1
Supplementary Table S1
Supplementary Table S2
Supplementary Table S3
Supplementary Table S4
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
