Abstract
Spaceflight impacts cardiovascular function in astronauts; however, its impact on cardiac development and the stem cells that form the basis for cardiac repair is unknown. Accordingly, further research is needed to uncover the potential relevance of such changes to human health. Using simulated microgravity (SMG) generated by two-dimensional clinorotation and culture aboard the International Space Station (ISS), we assessed the effects of mechanical unloading on human neonatal cardiovascular progenitor cell (CPC) developmental properties and signaling. Following 6–7 days of SMG and 12 days of ISS culture, we analyzed changes in gene expression. Both environments induced the expression of genes that are typically associated with an earlier state of cardiovascular development. To understand the mechanism by which such changes occurred, we assessed the expression of mechanosensitive small RhoGTPases in SMG-cultured CPCs and observed decreased levels of RHOA and CDC42. Given the effect of these molecules on intracellular calcium levels, we evaluated changes in noncanonical Wnt/calcium signaling. After 6–7 days under SMG, CPCs exhibited elevated levels of WNT5A and PRKCA. Similarly, ISS-cultured CPCs exhibited elevated levels of calcium handling and signaling genes, which corresponded to protein kinase C alpha (PKCα), a calcium-dependent protein kinase, activation after 30 days. Akt was activated, whereas phosphorylated extracellular signal-regulated kinase levels were unchanged. To explore the effect of calcium induction in neonatal CPCs, we activated PKCα using hWnt5a treatment on Earth. Subsequently, early cardiovascular developmental marker levels were elevated. Transcripts induced by SMG and hWnt5a-treatment are expressed within the sinoatrial node, which may represent embryonic myocardium maintained in its primitive state. Calcium signaling is sensitive to mechanical unloading and directs CPC developmental properties. Further research both in space and on Earth may help refine the use of CPCs in stem cell-based therapies and highlight the molecular events of development.
Introduction
A
Research in our own laboratory has shown that simulated microgravity (SMG) impacts the developmental profile of human cardiovascular progenitor cells (CPCs) in an age-dependent manner [4]. Interestingly, microarray analysis in those experiments identified small RhoGTPases and Wnt signaling as being some of the systems affected by the SMG environment in neonatal CPCs.
The effect of mechanical unloading on mouse embryonic stem cells (mESCs) has been shown to impact differentiation and stemness, with experiments by Blaber et al. [5] demonstrating that embryoid bodies retain markers of self-renewal and exhibit reduced definitive germ layer marker expression when flown in space. However, in the same studies, when mechanically unloaded embryoid bodies returned to Earth, they were able to differentiate more readily into contractile cardiomyocyte colonies. Similarly, Jha et al. [6] found that human induced pluripotent stem cells more readily differentiate into cardiomyocytes using three-dimensional culture coupled with transient, early exposure to SMG.
These separate experiments may represent a similar phenomenon in which a low gravity culture promotes an enhanced state of stemness under SMG or MG that results in increased differentiation ability when the cells are returned to normal gravity conditions. Thus, stem cell therapies relevant to cardiac repair may be improved by manipulating the mechanisms relevant to mechanical signaling in cardiovascular progenitors. In particular, inducing enhanced stemness in cardiovascular progenitors may facilitate a correspondingly enhanced clinical effect upon transplantation.
Alterations in mechanical sensing molecules, such as the small RhoGTPases, are believed to be involved in the molecular adaptation to MG [7,8]. Importantly, these molecules are also able to impact intracellular signaling pathways, such as calcium oscillations, which subsequently can activate AKT [9] and extracellular signal-regulated kinase (ERK) [10]. In the context of cardiogenesis, these processes are critical to maintaining a balance between inductive and proliferative cues [11,12]. Therefore, manipulating the normal gravity environment of early CPCs may highlight important mechanisms by which early cardiac progenitors develop or expand. Such insights may be applied to further understand cardiovascular development and enhance the outcomes of stem cell-based regenerative therapies.
In the context of cardiac repair, early clinical trials of these types of therapies are promising [13 –15], but are stymied by a failure of cell engraftment and controversy over the appropriate cell type [16]. Therefore, the application of findings from MG experiments to Earth-based experiments may help overcome the shortcomings of current clinical trials involving the use of CPCs for cardiac repair.
In an effort to characterize both the effects of MG on a population of early CPCs as well as the potential use of these changes on Earth, we cultured neonatal human CPCs using a two-dimensional clinostat and in the National Laboratory aboard the International Space Station (ISS). We sought to identify changes in the transcription of genes involved in signaling in response to SMG and MG as well as the effects of such signaling changes on stemness. We then modeled features of these molecular changes in vitro using a small molecule under normal gravity conditions. In doing so, we present components of the adaptive cellular response to MG and their implications for enhancing the regenerative potential of neonatal CPCs.
Materials and Methods
Isolation and culture of early CPCs
The Institutional Review Board of Loma Linda University approved the protocol for use of tissue that was discarded during cardiovascular surgery, without identifiable private information, for this study with a waiver of informed consent. CPCs were isolated from cardiac tissue of neonates (1 day–1 month), as previously described [17]. Briefly, atrial tissue was cut into small clumps (∼1.0 mm3) and then enzymatically digested using collagenase (Roche, Indianapolis, IN) at a working concentration of 1.0 mg/mL. The resulting solution was then passed through a 40-μm cell strainer. Cells were cloned in a 96-well plate by limiting dilution to a final concentration of 0.8 cells per well to create populations for expansion.
Then, clones were screened for the coexpression of Isl1 and c-Kit and supplemented with growth media comprising 10% fetal bovine serum (Thermo Scientific, Waltham, MA), 100 μg/mL penicillin–streptomycin (Life Technologies, Carlsbad, CA), 1.0% minimum essential medium nonessential amino acids solution (Life Technologies), and 22% endothelial cell growth media (Lonza, Basel, Switzerland) in Medium 199 (Life Technologies). The MycoAlert PLUS Mycoplasma Detection Kit (Lonza, Basel, Switzerland) was used to test for mycoplasma contamination.
Flow cytometry
Progenitor cell populations were fluorescently labeled with antibodies, as recommended by their respective manufacturers, and then analyzed using a MACSQuant® analyzer (Miltenyi Biotec, Auburn, CA). Quantification of data was performed using FlowJo software version 10 (Ashland, OR). Isotype controls and unstained cell populations were used to define positive and negative gates. Antibodies used for cytometric analysis are described in Table 1.
n/a, not applicable.
Simulated microgravity
A two-dimensional clinostat (BioServe Space Technologies, Boulder, CO) was used to simulate the effects of MG on Isl1+c-Kit+ CPCs, as previously described [4]. Clinorotation simulates MG by generating a state of relative motionlessness through the combined effects of gravity, centrifugation, and Brownian motion [18]. Using a rotation rate of 3.94 ± 0.01 rotations per minute, CPCs experienced a relative centrifugal force of <0.5 mG. Cells were seeded at a density of 200,000 cells per Opticell or Biocell, gassed with a mixture of 5% CO2 and 95% air, and then subjected to clinorotation. After 6–7 days of clinorotation, cells were trypsinized, counted, and used for experiments. Controls were similarly seeded and grown under static conditions within a 5% CO2 cell culture incubator for a matched period of time.
CPC culture aboard the ISS
For experiments described herein, neonatal CPCs derived from four unique neonates (1 day–1 month) were seeded into eight polystyrene Biocells (BioServe Space Technologies) containing 20 mL of growth media. Biocells were loaded into self-contained environments with 5% CO2 and 95% air, and flown aboard SpaceX CRS-11 to the United States National Laboratory on the ISS. Three days after launch, the Biocells arrived at the National Laboratory, where they received fresh growth media and were placed in an incubator containing 5% CO2 and 95% air.
Thereafter, fresh media were provided every 4–5 days, while aboard the ISS. Cells were either fixed with RNAProtect (Qiagen, Valencia, CA) after 12 days of culture on the ISS and stored at −80°C or fed and returned live to Earth after 30 days on the ISS. Cells that were returned live were immediately processed. Clone- and passage-matched ground controls were fed and treated in parallel with the feeding schedule and activities performed by our astronaut collaborator aboard the ISS.
Postflight sample processing
Upon landing and retrieval of the payload, live cells were trypsinized, counted, and used to generate protein lysates. After 30 days, Biocells cultured aboard the ISS or on the ground contained 1,126,000 ± 176,000 or 152,300 ± 46,700 CPCs, respectively. Biocells containing cells fixed and frozen in RNA protect were thawed at room temperature. The RNA protect was removed and centrifuged at 10,000 g at 4°C for 10 min. Biocells were disassembled and the culture membranes were then rinsed with TRIzol® reagent (Life Technologies). RNA was purified from RNA protect samples using the RNeasy Mini Kit (Qiagen), as per the manufacturer's instructions, while total RNA was purified from TRIzol reagent using isopropanol- and ethanol-based precipitation. cDNA was generated and reverse transcription–polymerase chain reaction (RT-PCR) was performed as described below.
Quantitative RT-PCR
cDNA was prepared using 2 μg of RNA with Superscript III (Life Technologies). Quantitative real-time polymerase chain reaction was performed using Go-Taq® qPCR Mastermix (Promega, Madison, WI) and the iCycler iQ™5 PCR Thermal Cycler (Bio-Rad, Hercules, CA) following a protocol of 94°C for 10 min followed by 45 cycles of 94°C for 15 s, 52–68°C (depending on the primer) for 60 s, and 72°C for 30 s. RT-PCR products were visualized using 1%–2% agarose gel electrophoresis and low mass DNA ladder (Invitrogen, Carlsbad, CA). Primers were designed using the National Center for Biotechnology Information Primer-BLAST program and obtained from Integrated DNA Technologies (Coralville, IA). Primers used in experiments are listed in Table 2.
RT2 Profiler Array for SMG- and ISS-cultured CPCs
Gene expression changes in neonatal CPCs cultured under SMG were broadly assessed using the RT2 profiler PCR array for the human epidermal growth factor/platelet-derived growth factor signaling pathway array plate (PAHS-040Z) following the manufacturer's instructions (Qiagen). We used custom array plates (CLAH22469A; Qiagen) as per the manufacturer's instructions to analyze gene expression changes in ISS-cultured neonatal CPCs that were relevant to Wnt, ERK, BMP/Smad, and Notch signaling; cytoskeletal maintenance; calcium handling; apoptosis and cell cycle; cardiac development and stemness; and regeneration.
For both array signaling plates, RNA was isolated and purified, of which 2 μg of RNA was reverse transcribed into cDNA as described above. cDNA was thoroughly mixed with 2 × RT2 SYBR Green Mastermix and RNase- and DNase-free water before being loaded into the profiler array plate. The array plate was placed in the iCycler iQ™5 PCR Thermal Cycler (Bio-Rad) and underwent a protocol of 95°C for 10 min and 40 cycles of 95°C for 15 s and 60°C for 1 min. Threshold cycle values were then analyzed for each individual clone using the Qiagen Data Analysis Center (
Since this analysis center performs only a two-tailed Student's t-test to calculate P-values, all fold changes for individual clones were exported to Prism and analyzed, as described below.
Western blot with protein simple
Following detachment, CPCs were homogenized using RIPA buffer containing phosphatase inhibitor cocktail (Millipore, Temecula, CA), followed by centrifugation at 14,000 g for 15 min at 4°C and collection of the supernatant for analysis. Total protein concentrations were determined using the Pierce Micro BCA Protein Assay Kit (Thermo Scientific, Rockford, IL). A capillary-based western blotting system (ProteinSimple Wes, San Jose, CA) was used to assess protein expression. All procedures were completed according to the manufacturer's instructions and default settings. The concentration of protein lysates, antibodies used in experiments, and antibody dilutions are indicated in Table 1. A positive control for phosphorylated Akt (Cell Signaling Technology, Danvers, MA; catalog number 9273S, lot number 20) was diluted 1:2.
The anti-rabbit and anti-mouse secondary antibodies included in the Wes Detection Module kit (ProteinSimple Wes) were used. All data were analyzed with the Compass Software associated with the Wes instrument (ProteinSimple Wes). Data were exported to Prism for further analysis, as described below.
Wnt5a treatment
Neonatal CPCs were grown until approximately 85% confluent and treated with 100 ng/mL recombinant human/mouse Wnt5a (R&D Systems, Minneapolis, MN) in CPC growth media for 1 h, a concentration and duration that were previously shown to induce protein kinase C activity [19]. Cells were then washed with phosphate-buffered saline and placed in TRIzol reagent or formed into protein lysates, as described by Abrahamsen and Lorens [20]. RT-PCR and western blotting were performed, as described above.
Statistical analysis
The Shapiro–Wilk test for normality was used to test the normality of data distribution. Student's t-test was used to compare the mean of all normally distributed data. Non-normally distributed data were compared using a Wilcoxon matched-pairs signed-rank text. For protein expression analysis, either Student's t-test or Mann–Whitney U test was used to compare the mean of normally or non-normally distributed data, respectively. All data are reported as the mean ± the standard error of the mean. Prism 7 version 7.02 (GraphPad, La Jolla, CA) was used for all statistical analyses. P values <0.05 were assumed to indicate statistical significance.
Results
CPCs derived from human neonates express markers of precardiovascular mesoderm
Neonatal CPCs express SSEA1 (Fig. 1A), PDGFRα (Fig. 1B), and Mesp1 (Fig. 1C). These clonal, early cardiovascular progenitors coexpress both Isl1 and cKit (Fig. 1D) and, as previously reported, can differentiate into all cardiovascular lineages [21]. Viability of CPCs was assessed using the Viobility 405/520 dye (Fig. 1E).
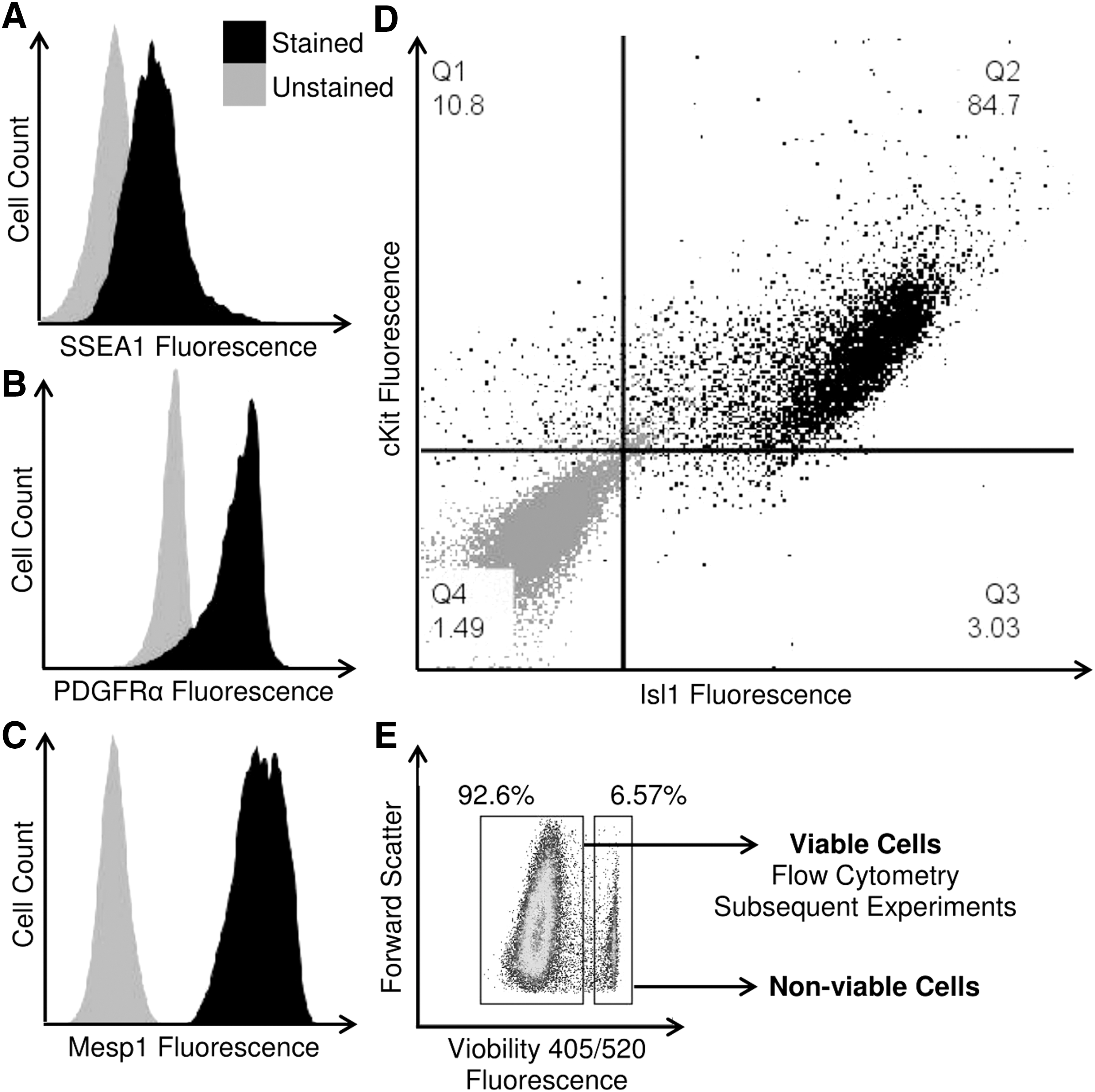
CPCs exhibit early cardiovascular developmental markers. Cells isolated from human tissue were screened for the expression of SSEA1
Neonatal CPCs express markers of an earlier developmental state after SMG and spaceflight
Following culture under SMG for 6–7 days and aboard the ISS for 12 days, cardiogenesis-related gene expression was assessed in neonatal CPCs derived from human heart tissue and analyzed in the context of a modified version of a previously reported cardiac development profile [22] (Fig. 2).
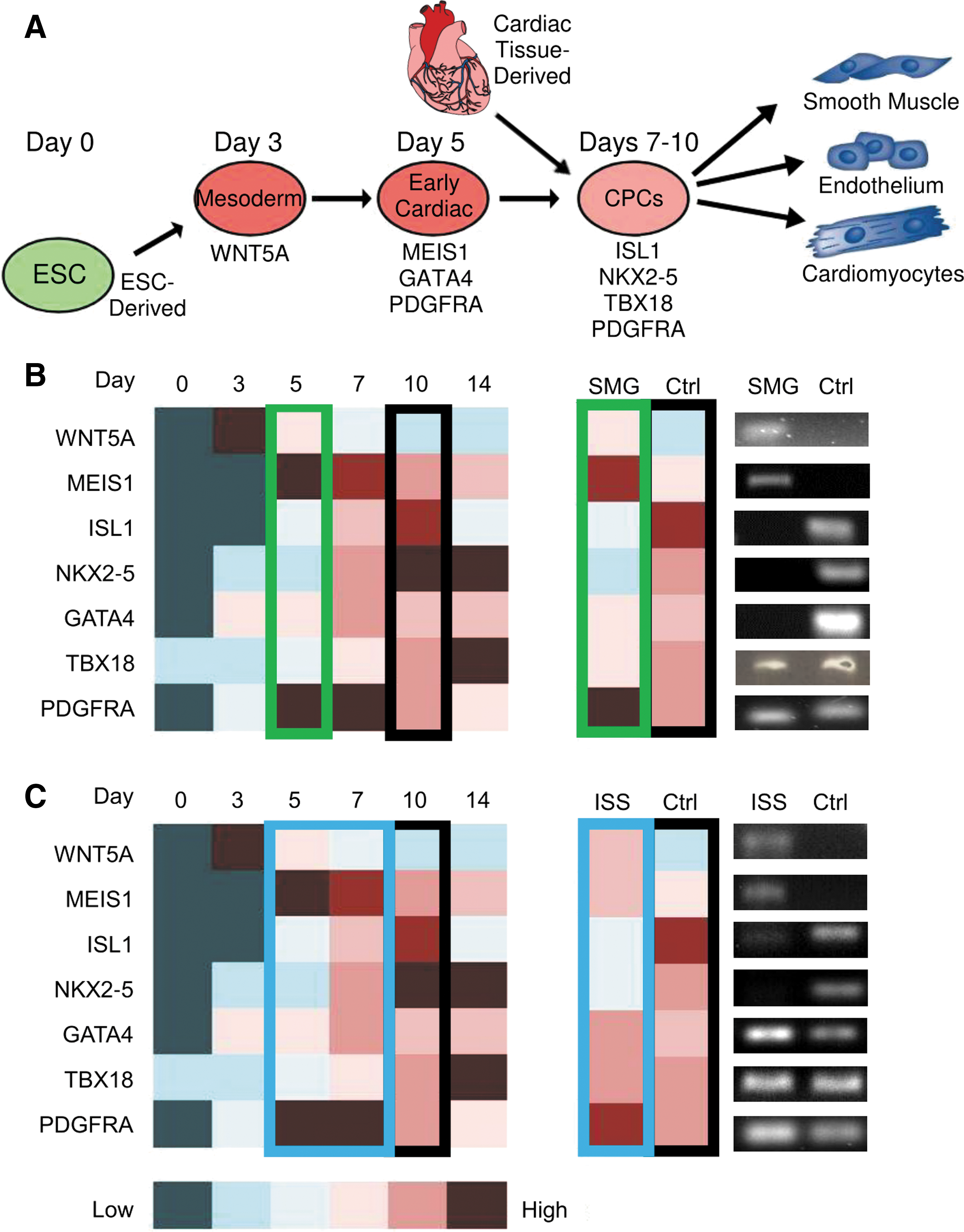
SMG and spaceflight induce the expression of markers of an earlier developmental state in neonatal CPCs. Neonatal CPCs were cultured under SMG for 6–7 days and aboard the ISS for 12 days before being fixed in RNA protect, screened for the expression of markers of early development, and compared to a day 0–14 molecular cardiogenesis profile adapted from den Hartogh et al. [22]
We observed increased expression of WNT5A, MEIS1, and PDGFRA, but decreased expression of ISL1, NKX2-5, GATA4, and TBX18 (Fig. 2B) in SMG-cultured CPCs. The black box indicates the control profile of gene expression, whereas the green box indicates the approximate SMG profile of gene expression. Meanwhile, we observed similar changes in WNT5A and ISL1 expression under MG, but less pronounced changes in the remaining markers (Fig. 2C). This led to a modestly dedifferentiated cardiac developmental gene expression profile in ISS-cultured CPCs (blue box) compared to SMG-cultured CPCs (green box).
SMG promotes expression of genes involved in noncanonical Wnt/Ca2+ signaling
Since the induction of early cardiac genes under reduced gravity conditions may represent enhanced stemness, we sought to identify the mechanism by which such changes occurred. Previous reports of RhoA as a gravity-sensing molecule led us to assess the involvement of small RhoGTPases in mediating the effects of SMG on neonatal CPCs.
We observed significantly decreased expression of RHOA (0.041 ± 0.014-fold change, P < 0.001, n = 4) and CDC42 (0.074 ± 0.001-fold change, P < 0.05, n = 3) (Fig. 3A). Given the role of small RhoGTPases in planar cell polarity Wnt signaling, we evaluated the expression of both canonical and noncanonical Wnt ligands in neonatal CPCs cultured under SMG. In doing so, we identified no change in the expression of the canonical Wnt ligand WNT3A (2.260 ± 0.154-fold change, P = 0.063, n = 3) or of GSK3B (4.308 ± 1.599-fold change, P = 0.131, n = 4), which sequesters β-catenin for degradation. Meanwhile, we identified a significant decrease in the transcript level of the canonical Wnt effector CTNNB1 (0.017 ± 0.002-fold change, P < 0.01, n = 3) (Fig. 3B).
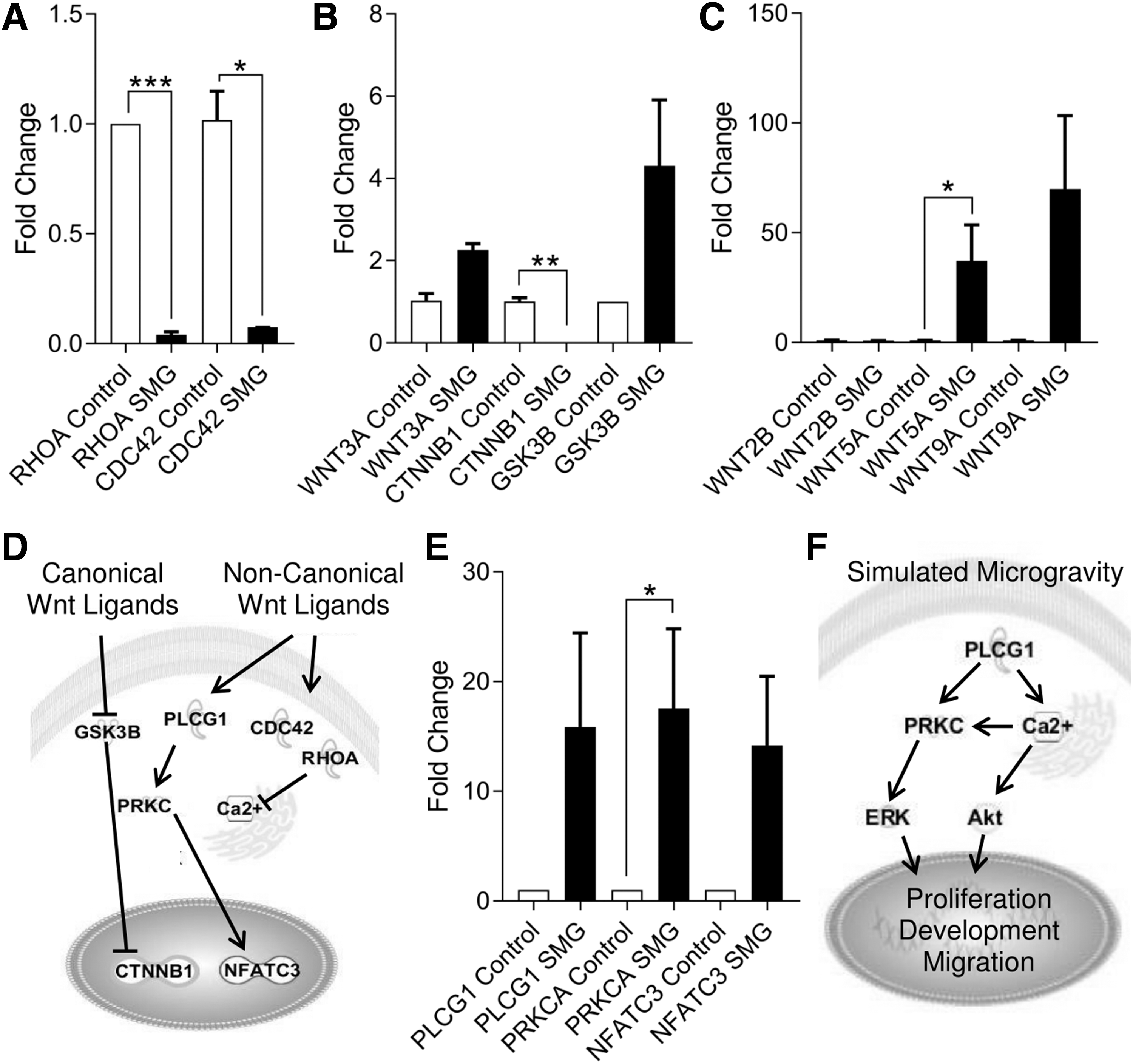
SMG induces noncanonical Wnt/Ca2+ signaling gene expression. The reduced expression of small RhoGTPases under SMG
In the absence of canonical Wnt signaling, we examined the expression of noncanonical Wnt ligands and found an induction in WNT5A expression (37.190 ± 16.330-fold change, P < 0.05, n = 6) and a general increase in WNT9A expression (69.910 ± 33.430-fold change, P = 0.063, n = 6) (Fig. 3C). Given the relationship between intracellular calcium handling and planar cell polarity signaling (Fig. 3D), we evaluated changes to the transcript levels in the noncanonical Wnt/Ca2+ pathway (Fig. 3E): PLCG1 (15.870 ± 8.571-fold change, P = 0.092, n = 3), PRKCA (17.560 ± 7.246-fold change, P < 0.05, n = 3), and the transcription factor NFATC3 (14.180 ± 6.318-fold change, P = 0.055, n = 3).
Activation of ERK and AKT pathways in association with SMG signaling in CPCs
The pleiotropic nature of calcium signaling can impact other signaling pathways (Fig. 3F). Therefore, we sought to identify additional pathways that might be affected following exposure to SMG. MAPK/ERK-related transcripts were expressed at higher levels (Fig. 4A, B). In particular, MAPK/ERK pathway inducer [TGFB1 (29.130 ± 3.637-fold change, P < 0.05, n = 3)] and transducer genes [MAPK3 (12.050 ± 8.644-fold change, P < 0.05, n = 3), MAPK8 (14.320 ± 6.291-fold change, P < 0.05, n = 3), and MAPK9 (4.624 ± 0.571-fold change, P < 0.001, n = 3)] were all expressed at elevated levels. However, targets of MAPK/ERK signaling were not significantly altered: GRB2 (9.395 ± 5.715-fold change, P = 0.280, n = 3), RAF1 (18.750 ± 13.840-fold change, P = 0.290, n = 3), and ELK1 (9.105 ± 4.203-fold change, P = 0.194, n = 3).
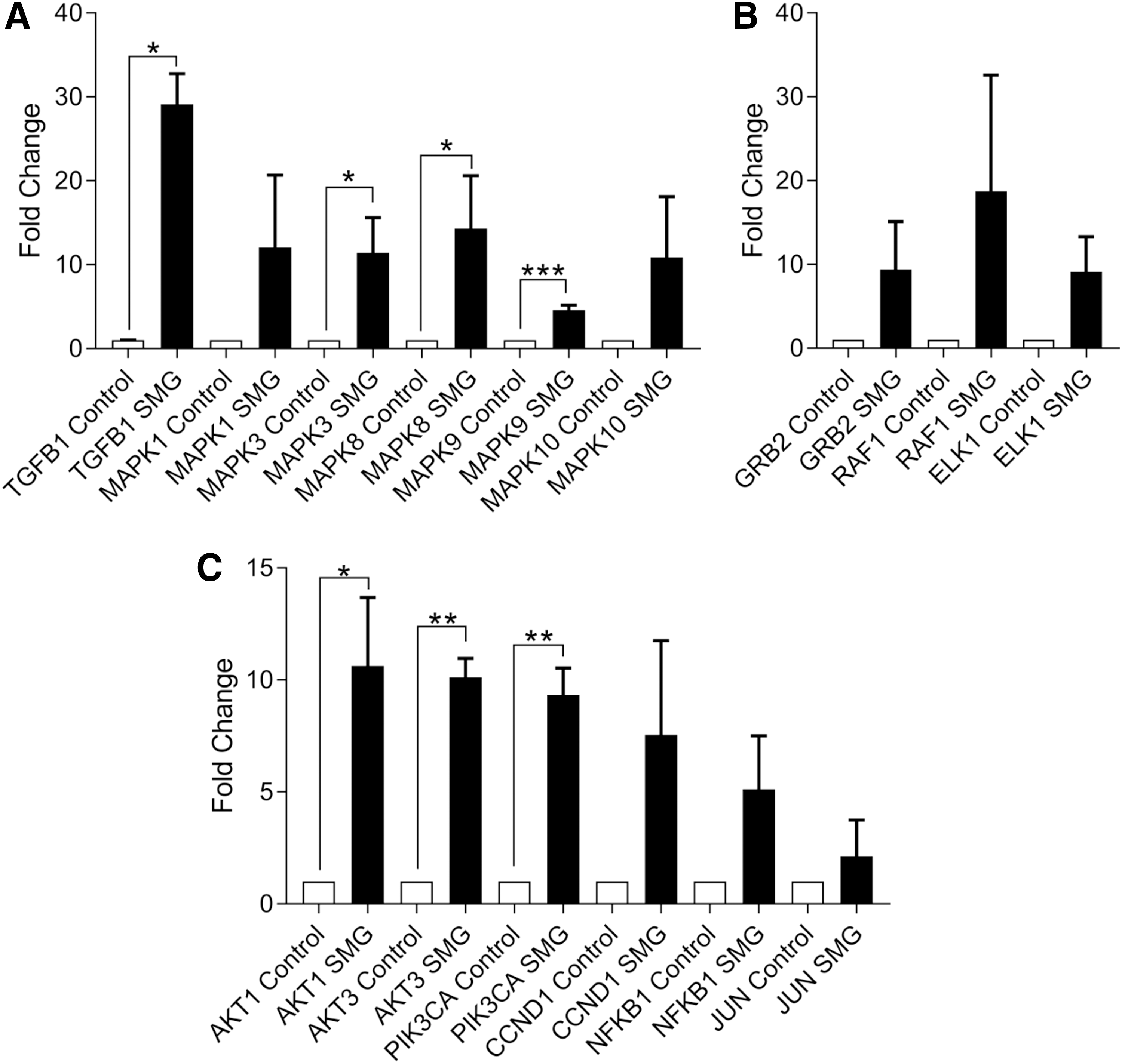
AKT and ERK/MAPK signaling pathway constituent genes induced by SMG. Transcripts of genes involved in ERK/MAPK signaling activation and transduction
Similarly, AKT-related signaling genes were induced, including AKT1 (10.620 ± 3.065-fold change, P < 0.05, n = 4), AKT3 (10.120 ± 0.839-fold change, P < 0.01, n = 3), and PIK3CA (9.334 ± 1.196-fold change, P < 0.01, n = 4), which promote Akt phosphorylation (ie, activation). AKT/PI3K pathway activation also enhances the stability of cyclin D1 (CCND1) and c-Jun, which facilitate G1-to-S cell cycle transition, while also targeting NFκB during antiapoptotic signaling. We then assessed changes in the levels of transcripts for Akt pathway targets (Fig. 4C).
Flight aboard the ISS activates PKCα in neonatal CPCs
These observations in SMG prompted us to determine whether calcium handling and signaling were altered by spaceflight. We evaluated the expression of genes involved in regulating intracellular calcium levels and subsequent signaling events in neonatal CPCs after flight aboard the ISS and observed significantly elevated levels of RYR2 (5.265 ± 1.508-fold change, P < 0.05, n = 3), CACNA1S (8.059 ± 1.771-fold change, P < 0.05, n = 3), and CAMK2A (5.979 ± 1.289-fold change, P < 0.05, n = 3) (Fig. 5A).
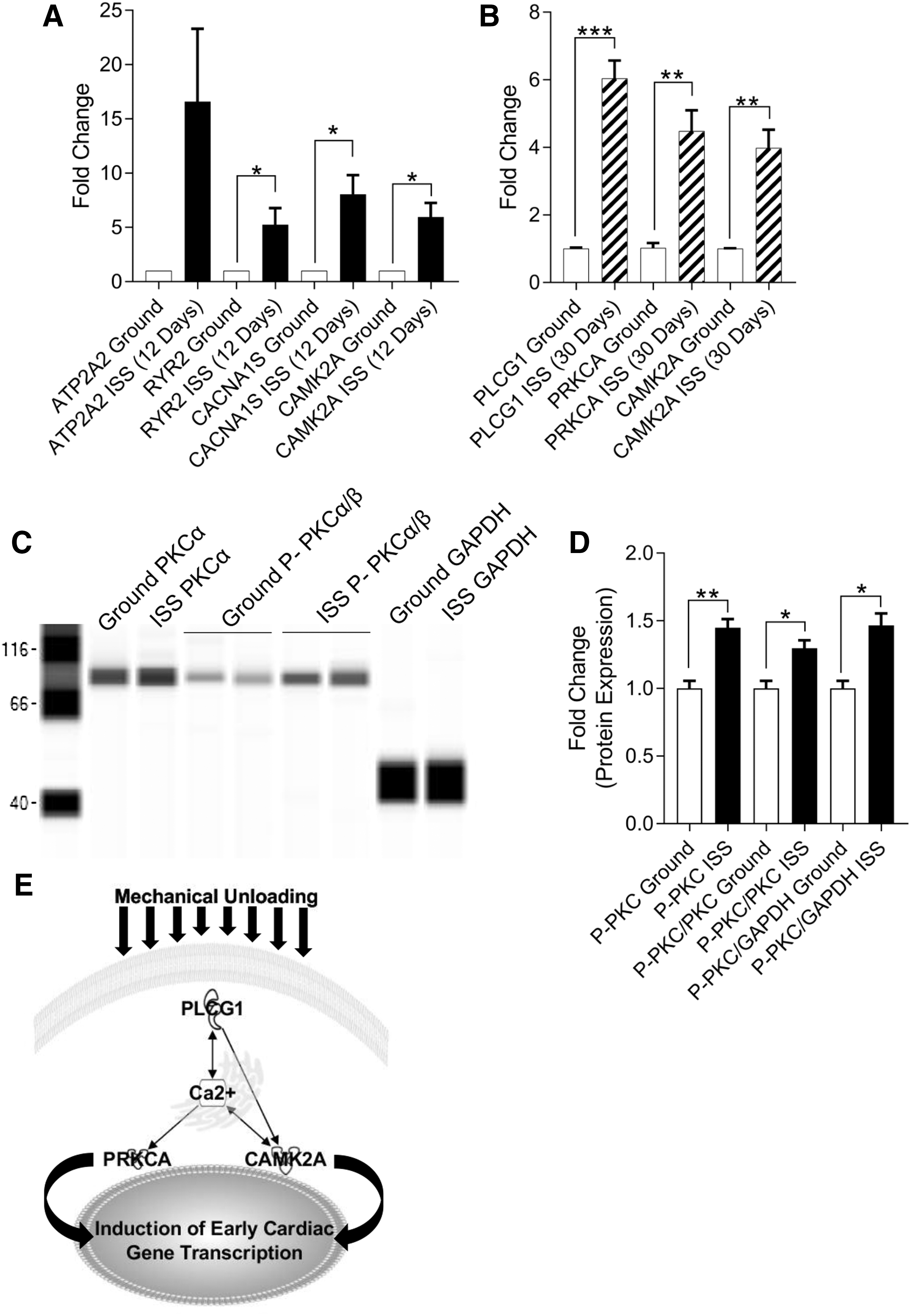
Spaceflight activates PKCα in neonatal CPCs after 30 days. After 12 days aboard the ISS, neonatal CPCs were fixed and induction of genes involved in calcium handling was measured by RT-PCR
Changes in calcium handling at 12 days suggested an ultimate induction of calcium signaling at 30 days, which was supported by significantly elevated levels of PLCG1 (6.041 ± 0.527-fold change, P < 0.01, n = 3), PRKCA (4.482 ± 0.617-fold change, P < 0.01, n = 3), and CAMK2A (3.982 ± 0.540-fold change, P < 0.01, n = 3) transcripts (Fig. 5B). Moreover, we assessed a significant increase in phosphorylated protein kinase C alpha (PKCα), a calcium-dependent protein kinase C (P-PKCα: 1.448 ± 0.065-fold change, P < 0.01; P-PKCα/PKCα: 1.298 ± 0.058-fold change, P < 0.05; P-PKCα/GAPDH: 1.466 ± 0.088-fold change, P < 0.05; n = 3–4) after 30 days of ISS culture (Fig. 5C, D). Such ISS-mediated changes to calcium signaling may have a direct impact on cardiogenesis, as shown in the schematic in Fig. 5E.
Flight aboard the ISS activates Akt in neonatal CPCs
These findings of increased expression of calcium-related signaling prompted us to assess the role of calcium-sensitive pathways that are pertinent to stem cell physiology (ie, ERK and Akt). We assessed the expression of Akt-related signaling genes in neonatal CPCs after 12 and 30 days of flight aboard the ISS. At 12 days, we observed significantly elevated levels of PIK3CA (2.216 ± 0.343-fold change, P < 0.01, n = 9), cMYC (4.811 ± 1.341-fold change, P < 0.05, n = 9), and NFκB1 (2.704 ± 0.528-fold change, P < 0.05, n = 9). At 30 days, we observed significantly elevated levels of PIK3CA (7.764 ± 0.472-fold change, P < 0.001, n = 3), CCNDI (3.257 ± 0.195-fold change, P < 0.01, n = 3), cMYC (3.297 ± 0.309-fold change, P < 0.01, n = 9), JUN (4.997 ± 0.213-fold change, P < 0.001, n = 3), NFκB1 (5.935 ± 0.830-fold change, P < 0.01, n = 3), and RELA (2.799 ± 0.291-fold change, P < 0.01, n = 3) (Fig. 6A).
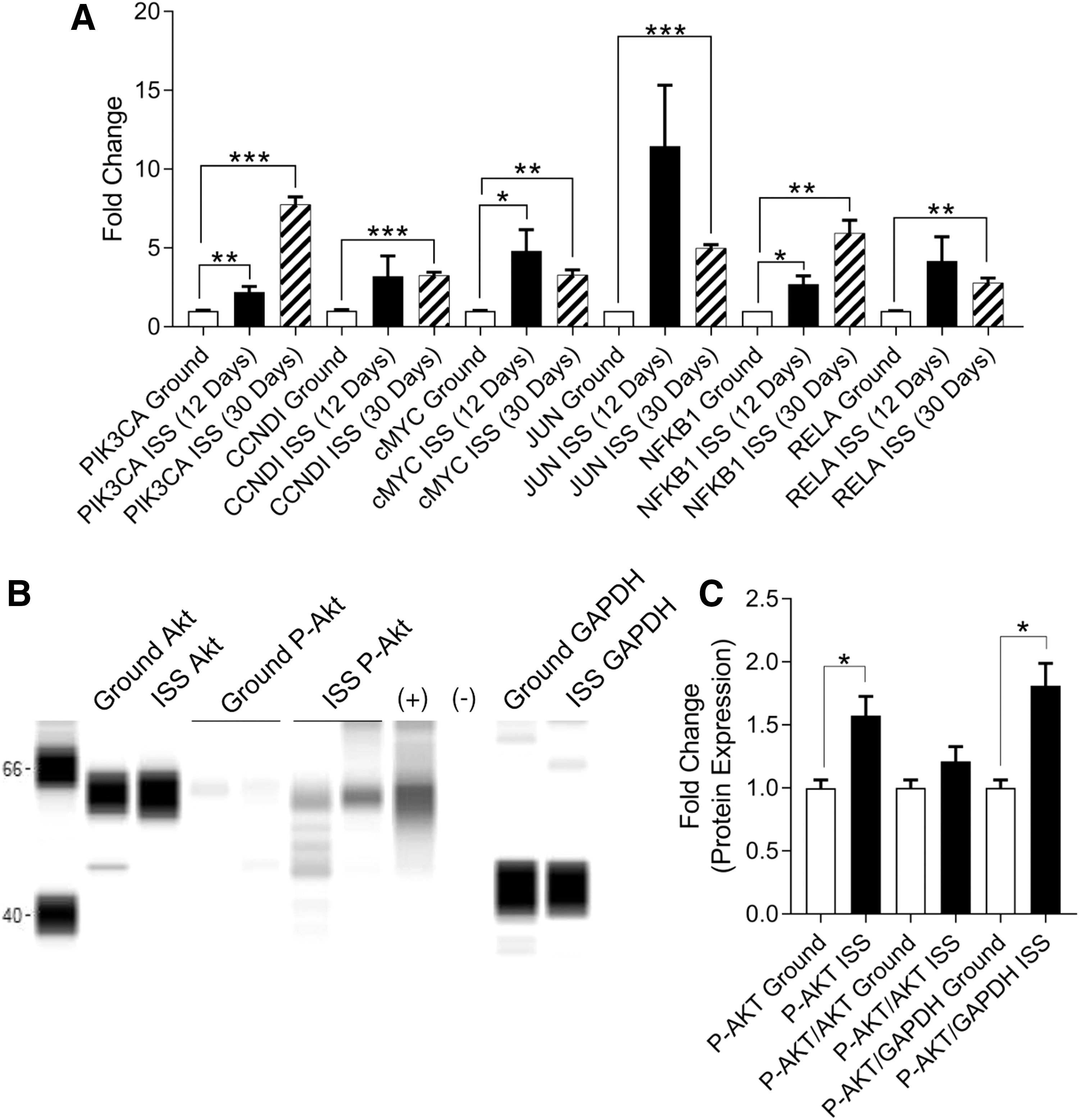
Spaceflight activates Akt signaling in neonatal CPCs after 30 days. After 12 and 30 days aboard the ISS, neonatal CPCs were fixed and gene expression along the Akt pathway was measured by RT-PCR
Protein expression analysis revealed an induction of phosphorylated Akt following spaceflight when normalized to GAPDH or analyzed as total substrate (P-AKT: 1.573 ± 0.152-fold change, P < 0.05; P-AKT/GAPDH: 1.812 ± 0.176-fold change, P < 0.05; n = 3) after 30 days of ISS culture. Notably, Akt phosphorylation only trended toward an increase when normalized to pan-Akt (P-AKT/AKT: 1.211 ± 0.117-fold change, P = 0.189, n = 3) (Fig. 6B, C).
ERK/MAPK is not activated in neonatal CPCs by spaceflight
This change in Akt activity prompted us to investigate whether ERK/MAPK signaling was altered by spaceflight. We evaluated the expression of ERK/MAPK-related signaling genes in neonatal CPCs after 12 and 30 days of flight aboard the ISS (Fig. 7A). At 12 days, we observed significantly reduced levels of DUSP3 (0.280 ± 0.126-fold change, P < 0.01, n = 3), which encodes a phosphatase involved in the deactivation of ERK/MAPK signaling. Interestingly, the targets of ERK/MAPK signaling were also elevated: GRB2 (11.660 ± 2.709-fold change, P < 0.05, n = 3) and RAF1 (5.230 ± 0.762-fold change, P < 0.01, n = 3) (Fig. 7B). However, at 30 days, the induction of genes along this signaling pathway was muted. Moreover, we observed no increase in phosphorylated ERK after 30 days of ISS culture (Fig. 7C, D).
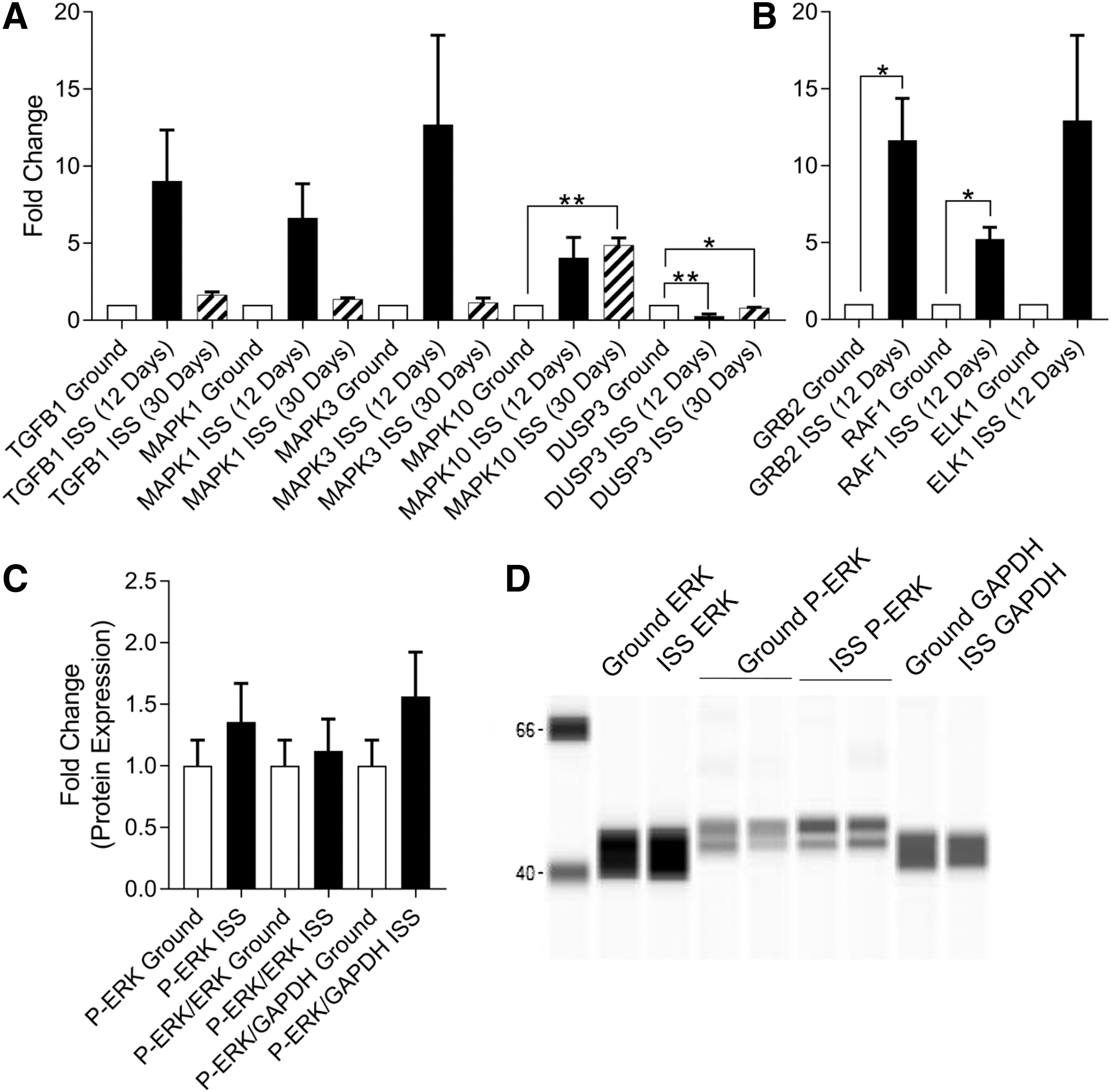
Spaceflight does not activate Erk signaling in neonatal CPCs after 30 days. After 12 and 30 days aboard the ISS, neonatal CPCs were fixed and gene expression along the ERK/MAPK pathway was measured by RT-PCR. At 12 days, the ERK/MAPK pathway
Ca2+ signaling contributes to induction of genes involved in early cardiogenesis
Given the role of calcium signaling in mediating the effects of spaceflight in neonatal CPCs, we sought to assess the effects of such signaling on the ground. Neonatal CPCs were treated with recombinant hWnt5a (100 ng/mL) for 1 h, which is a concentration and duration that were previously shown to induce PKC activity [19]. We then investigated the induction of noncanonical Wnt ligands (Fig. 8A) and calcium signaling genes (Fig. 8B): WNT5A (3.027 ± 0.087-fold change, P < 0.01, n = 3), WNT9A (4.587 ± 0.485-fold change, P < 0.05, n = 3), WNT11 (3.713 ± 0.297-fold change, P < 0.05, n = 3), PLCG1 (4.609 ± 0.544-fold change, P < 0.05, n = 3), PRKCA (28.190 ± 2.658-fold change, P < 0.01, n = 3), and CAMK2A (342.700 ± 37.760-fold change, P < 0.05, n = 3).
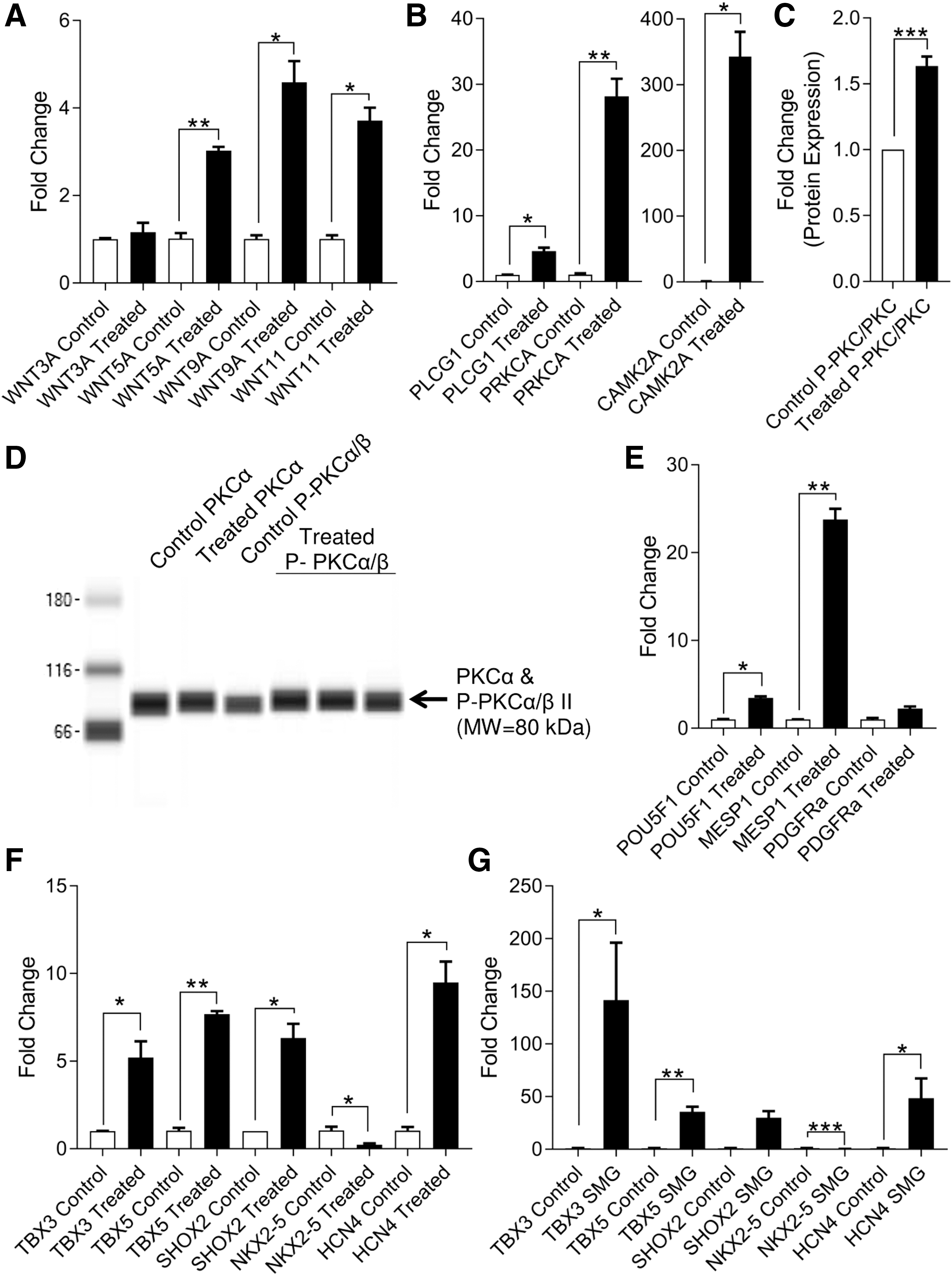
Noncanonical Wnt/Ca2+ signaling impacts the developmental phenotype of neonatal CPCs. Wnt5a treatment of neonatal CPCs for 1 h induces expression of noncanonical Wnt ligands
The induction of PKCα phosphorylation indicated the presence of calcium activity [(P-PKC/PKC: 1.635 ± 0.072-fold change, P < 0.05, n = 3; (Fig. 8C, D)]. This occurred concomitantly with the induction of early developmental genes (Fig. 8E), including the pluripotency marker POU5F1 (3.453 ± 0.194-fold change, P < 0.05, n = 3) and the mesodermal marker MESP1 (23.760 ± 1.230-fold change, P < 0.01, n = 3).
Sinoatrial nodal genes and Hcn4 are induced by calcium pathway activity
Increasing evidence indicates that sinoatrial nodal cells represent a population of embryonic myocardium that retains its primitive phenotype [24]. For this reason, we assessed the relationship between primordial cardiogenesis gene induction and that of the sinoatrial nodal gene program in Wnt5-treated CPCs (Fig. 8F). In doing so, we observed significant increases in the expression of TBX3 (5.212 ± 0.917-fold change, P < 0.05, n = 3), TBX5 (7.678 ± 0.178-fold change, P < 0.01, n = 3), SHOX2 (6.318 ± 0.813-fold change, P < 0.05, n = 3), and HCN4 (9.489 ± 1.191-fold change, P < 0.05, n = 3) along with decreased expression of NKX2-5 (0.237 ± 0.044-fold change, P < 0.05, n = 3).
A similar induction of the sinoatrial nodal gene program was observed in SMG-treated CPCs: TBX3 (141.600 ± 54.550-fold change, P < 0.05, n = 3), TBX5 (35.620 ± 4.667-fold change, P < 0.05, n = 3), HCN4 (48.510 ± 18.710-fold change, P < 0.05, n = 3), and NKX2-5 (0.146 ± 0.042-fold change, P < 0.001, n = 3) (Fig. 8G).
Discussion
Both SMG and culture aboard the ISS induced the expression of markers of early cardiovascular development along with genes involved in calcium signaling, although to different extents. While WNT5A and PRKCA induction occur along with that of ERK and Akt in SMG, neonatal CPCs that were cultured aboard the ISS exhibited increases in calcium-dependent protein kinase C (PKCα) activation along with only increased Akt activation. This increase in calcium signaling was modeled in vitro on Earth using hWnt5a, a noncanonical ligand that promotes protein kinase C activation. In doing so, we observed an important role in calcium signaling in accounting for the effects, at least in part, of spaceflight within neonatal CPCs.
Across models and culture systems, molecular biologists have increasingly identified the role of small RhoGTPases in mediating the response of cells to a reduced gravity environment [7,8]. RhoA and CDC42 are small RhoGTPases that associate with the actin cytoskeleton [25], participate in the noncanonical Wnt planar cell polarity pathway [26], and modulate intracellular calcium potentiation [27]. Other experiments have identified alterations to small RhoGTPases, such as the suppression of RhoA in mesenchymal stem cells (MSCs), in response to SMG [28]. Elsewhere, researchers have reported the involvement of the primary cilium, which coordinates the early molecular events of cardiogenesis [29], as a sensor of cell–cell contact and altered mechanical stress [30]. Interestingly, RhoA activity was found to be related to ciliogenesis [31], further supporting the hypothesis that small RhoGTPases have an important role in cardiac development
Ultimately, this reduction in RHOA expression logically follows from the reduced mechanical stress that is expected under MG conditions. Accordingly, mechanotransduction-induced changes in RhoA expression would be expected to impact calcium handling. Indeed, substrate rigidity (or the lack thereof in this context), has been shown to directly impact calcium oscillations within MSCs. Using fluorescence resonance energy transfer, Kim et al. [27] observed changes to Ca2+ oscillation in accordance with changes to the stiffness of the MSC culture environment.
Similarly, we found that calcium pathway genes and those related to calcium handling were expressed at higher levels following culture aboard the ISS as well as under SMG, although to a more modest extent. These changes in calcium extend beyond human CPCs. During spaceflight, calcium loss is widely observed in astronauts and usually linked to changes in bone metabolism [32]. Yet, in one animal model of SMG, mice were observed to exhibit an increased incidence of arrhythmias along with alterations in intracellular calcium handling, including ryanodine receptor (RyR2) phosphorylation [33]. Moreover, short-duration atrial fibrillation, premature ventricular contractions, and ventricular tachycardia have all been reported in astronauts during spaceflight [34]. Importantly, while the etiology of such cardiac events during spaceflight remains unknown, disturbances in calcium handling may be an important contribution to cardiac abnormalities in space.
Meanwhile, the modification of signaling pathways related to calcium, either as an important secondary or mediating molecule, directly impacts the ability of CPCs to differentiate or maintain pluripotency [35,36]. The effect of calcium signaling is dependent both on the pathway in which it is involved as well as the species under study, with mESCs exhibiting far different responses to calcium-mediated activity compared to human ESCs (hESCs). For example, calcium signaling promotes pluripotency in hESCs, while being affiliated with differentiation in mESCs [35].
In the context of cardiogenesis, studies performed in both embryos and ESC-derived cardiomyocytes have shown a critical role of Ca2+ in regulating multiple steps of heart formation [12]. For example, induction of Ca2+ oscillation promotes proliferation and, upon transplantation, enhances engraftment and expansion [37]. In this way, our observed shift in expression of markers of early cardiac development is likely the result, at least in part, of modified calcium signaling activity within neonatal CPCs. Furthermore, research into Mesp1-expressing precardiac mesoderm derived from hESCs exhibited enriched activity along the calcium, extracellular matrix receptor, and Wnt signaling pathways at day 5 of development [22], which is supported by our observed increase in markers of such a state of development after culture aboard the ISS and using SMG (Fig. 2).
Meanwhile, Akt signaling by calcium activation has been well documented to promote the maintenance of pluripotency in hESCs [35]. In addition, Akt signaling exerts a critical role in several cell functions that are relevant to stem cell transplantation, migration, and cytokine expression [38]. Therefore, manipulating calcium signaling on Earth and promoting Akt activation present a novel therapeutic opportunity for cell-based cardiac repair.
In addition to modifying intracellular calcium signaling, the expression of noncanonical Wnt ligands has been observed in association with the cryoinjury response of the neonatal rodent heart [39]. Since WNT5A and PRKCA gene expression induction is involved in the response of neonatal CPCs to SMG, we sought to assess the potential role of Wnt5a in promoting enhanced stemness in CPCs. While preliminary, these early results indicate a relationship between enhanced intracellular calcium signaling as well as an increased expression of markers of early cardiogenesis in neonatal CPCs. The expression of early cardiogenic mesoderm markers indicates a potentially earlier developmental state.
In addition to being linked to enhanced therapeutic potential following transplantation [40 –43], such an early developmental state may also provide the appropriate cell source for biological pacemaker development. In an avian model, Bressan et al. used fate mapping to determine that sinoatrial nodal cells were observed to already be specified shortly after gastrulation before the onset of cardiogenesis within a region that was observed to be Nkx2-5 and Isl1 negative [44]. Meanwhile, the Keller group recently reported a method of generating sinoatrial nodal-like pacemaker cells in Nkx2-5-negative cardiomyocytes [45]. Similarly, we observed decreased NKX2-5 and ISL1 expression under both SMG and MG.
When considered together, our findings using simulated and real MG support the emerging hypothesis that sinoatrial nodal cells represent a dedifferentiated/undifferentiated state of cardiac development [24]. Moreover, our observation that calcium signaling has an integral role in this process is mirrored by observations that Wnt7a, which mobilizes intracellular calcium [46], has been observed in the developing cardiac conduction system [47]. Given the observed changes to the expression of calcium handling genes and proteins, future studies should assess the effect of SMG and ISS culture on intracellular Ca2+ transients and the electrophysiological properties of CPCs.
Importantly, many of these processes observed under SMG and MG are dependent upon the developmental status of the cell. Indeed, later stages of cardiogenesis and the incipient developmental cues of ESCs respond differently to calcium and Wnt signaling [48 –50]. Therefore, understanding the effects of mechanical transduction and altered intracellular calcium signaling within the myriad cell types that constitute the human body will help inform medical interventions that will be necessary to sustain deep space missions. Meanwhile, the molecular events that constitute MG sensing can be manipulated on Earth to facilitate regeneration. In doing so, MG-inspired, cell-based therapies can be developed.
Footnotes
Acknowledgments
We thank Peggy Whitson for performing cell culture experiments aboard the ISS. We thank Carla V. Hoehn, Stephanie Countryman, and BioServe Space Technologies for their assistance with cell culture hardware development, preparation, and deployment. Funding for this experiment was provided by the Center for Advancement of Science in Space (grant no. GA-2014-130 to M.K.J.).
Prior Conference Presentations: Simulated microgravity data were presented, in part, at the 2016 International Space Station Research and Development Conference (San Diego, CA) and at the 2015 and 2016 World Stem Cell Summits (Atlanta, GA and West Palm Beach, FL).
Author Disclosure Statement
No competing financial interests exist.
