Abstract
Retinoic acid (RA) signaling is important to normal development. However, the function of the different RA receptors (RARs)—RARα, RARβ, and RARγ—is as yet unclear. We have used wild-type and transgenic zebrafish to examine the role of RARγ. Treatment of zebrafish embryos with an RARγ-specific agonist reduced somite formation and axial length, which was associated with a loss of hoxb13a expression and less-clear alterations in hoxc11a or myoD expression. Treatment with the RARγ agonist also disrupted formation of tissues arising from cranial neural crest, including cranial bones and anterior neural ganglia. There was a loss of Sox 9-immunopositive neural crest stem/progenitor cells in the same anterior regions. Pectoral fin outgrowth was blocked by RARγ agonist treatment. However, there was no loss of Tbx-5-immunopositive lateral plate mesodermal stem/progenitor cells and the block was reversed by agonist washout or by cotreatment with an RARγ antagonist. Regeneration of the caudal fin was also blocked by RARγ agonist treatment, which was associated with a loss of canonical Wnt signaling. This regenerative response was restored by agonist washout or cotreatment with the RARγ antagonist. These findings suggest that RARγ plays an essential role in maintaining stem/progenitor cells during embryonic development and tissue regeneration when the receptor is in its nonligated state.
Introduction
R
The specific roles of each of the different isoforms of RARs in regulating the response of stem/progenitor cells to RA during development remain largely unknown. Despite potential for functional redundancy, there is likely to be significant differences in RAR function because expression levels of RAR isoforms vary both temporally and spatially [9,10]. Gene-knockout experiments of RARs in mice support differential function of RARs. For example, RARα-knockout mice exhibit no discernable phenotype [11] and RARβ-knockout mice have ocular defects and reduced body weight [12], while knockout of RARγ results in several severe developmental defects, including growth deficiency, cartilage dysmorphogenesis, and vertebrate malformations [13]. However, as RARs can function both to repress as well as activate targeted gene expression through respective engagement by RAR of corepressor or coactivator proteins, the use of gene-knockout methods to determine the physiological role of RARs is problematic. For example, the deletion of RAR may result in release of corepressor proteins bound to the RAR when non-RA ligated and the activity of these released corepressors may in themselves produce developmental defects, at least in mammals [14]. Moreover, Williams et al. (2009) suggested that the ability of RARγ, in particular, to repress gene expression is required for normal skeletal development of long bones. Therefore, in order to fully understand the importance of RAR in regulating gene activity, there are advantages to the use of specific agonists and antagonists for each of the different RARs [14].
In zebrafish, there are two major RARs, RARα and RARγ [10]. RARγ expression is restricted in early zebrafish embryos to mesodermal and neural crest stem/progenitor cells, in the head area, in the lateral plate mesoderm, and in the presomitic mesoderm of the tail bud [9,10]. Retinaldeyhyde dehydrogenase (RALDH), which is responsible for synthesizing RA, is not expressed within these areas [16,17]. Further, a transgenic reporter line indicating the presence of endogenous RA has shown that RA is not present in the tail bud, which is one of the locations for RARγ expression [18]. These nonoverlapping expression patterns of RARγ and RALDH suggest that RARγ does not function during development to activate RA-responsive genes. Instead, a lack of RARγ-dependent transcriptional activity may be important and RARγ may play a key role in repressing gene expression in its nonligated state.
We hypothesized that if RARγ functions in its nonligated state to repress gene expression, then engaging an RARγ-specific ligand will result in aberrant transcriptional activity of genes that by default should be switched “off.” In other words, cells or tissues expressing RARγ that are affected by treatment with an RARγ agonist may be those wherein RARγ plays an important gene repressive activity. To investigate this possibility during embryonic development, this study has examined the effects of RARγ-specific agonist treatments in zebrafish larvae [15]. Our findings have shown that treatment with an RARγ agonist has major deleterious consequences on the development of tissues that derive from neural crest and mesoderm, and also on caudal fin regeneration. The effects of the RARγ agonist treatments on the pectoral and caudal fin were reversed by washout of the agonist or cotreatment with an RARγ-specific antagonist. We have also used transgenic reporter zebrafish lines to identify downstream pathways regulated by RARγ activity, which paradoxically showed decreased expression of some hox genes and canonical Wnt signaling. These findings provide evidence that RARγ plays a critical role to maintain stem/progenitor cells during normal embryonic development and tissue regeneration, which depends on its ligation status.
Materials and Methods
Zebrafish husbandry
Stocks of the wild-type AB strain of zebrafish were maintained in the fish unit of the Biomedical Research facility in Aston University according to U.K. Home Office guidelines. The unit had a 14/10 h day/night cycle according to zebrafish husbandry guidelines [19]. Following breeding, zebrafish embryos were kept in an incubator at 28°C in petri dishes (SLS, Hessle, UK) in a 4 mL solution of Hank's solution (Molekula, Dorset, UK) or E3 fish media. Experiments using transgenic reporter fish were performed at the National Institute of Genetics (NIG) in Mishima, Japan, with husbandry according to NIG institutional guidelines [20]. Two transgenic hox gene reporter fish lines of the Gal4-UAS system were used, namely, the hoxc11a and hoxb13a lines [21]. In these transgenic fish, the gene trap and enhancer trap constructs are integrated within hoxc11a and hoxb13a genes, respectively. Therefore, Gal4FF expression is likely to recapitulate that of the endogenous genes [22]. The transgenic zebrafish reporter line HGn39D was used to visualize the lateral line [23]. The transgenic zebrafish reporter line Tcf:mini-p was used to visualize canonical Wnt signaling [24], which was kindly provided by Dr. Ishitani (Kyushu University). For Gal4-UAS breeding, either male or female adult zebrafish of the Gal4 line was crossed with UAS-GFP reporter zebrafish as pair-wise crossings in breeding tanks. The male and female zebrafish were separated using a barrier the evening before the actual day of breeding. The barrier was removed on the following morning to allow male and female mating and fertilized eggs were collected within 30–60 min of this using a strainer.
RARγ reagents: preparation and treatment protocols
The RARγ-specific agonist (AGN205327) and antagonist (AGN205728) [15] were kept as 10 mM stock solutions dissolved in 50% dimethyl sulfoxide (DMSO) and 50% methanol. These stocks were diluted with 100% DMSO to give a working stock solution at 100 μM according to previously published studies [25]. These working stocks were then further diluted with Hank's solution or E3 media to the final doses used for the treatments of zebrafish embryos as given in “Results” section. A control solution of a 50%:50% DMSO:methanol-carrier-alone stock further diluted into 100% DMSO and then into Hank's or embryo media was prepared using the exact dilutions that were used for the RARγ reagents. Following treatment of zebrafish embryos by immersion in E3 media supplemented with the RARγ reagents or carrier alone at 4 hours postfertilization (hpf) (or at other times as indicated in “Results” section), the treated and control embryos were incubated at 28°C [19]. Zebrafish embryos were treated with the RARγ agonist at 4 hpf because the agonist is not subjected to degradation and RARγ expression is detected at the 40% epiboly stage, which is around 5 hpf [10].
In situ hybridization and immunolocalization protocols
In situ hybridization for myoD expression was performed according to previously published methods [26]. In brief, zebrafish embryos treated with the RARγ agonist (at 4 hpf) and control embryos were harvested at 22 hpf and fixed in 4% paraformaldehyde (PFA) at 4°C overnight, before dehydrating in 100% methanol, also at 4°C overnight. The embryos were then serially rehydrated through decreasing alcohols (100%–25% methanol) finally to phosphate-buffered saline (PBS) alone. The rehydrated embryos were digested with proteinase K (10 μg/mL; Sigma, St. Louis, MO) for 10 min. In situ hybridization was performed by incubating the embryos in hybridization mix solution at 70°C for 5 h, prior to a further overnight incubation at 70°C with the myoD probe (ZFIN:ZDB-GENE-980526-561). Following a series of washes in sodium citrate solutions and then PBS, the embryos were incubated overnight at 4°C with anti-DIG-alkaline phosphatase antibody specific for the myoD probe (Invitrogen, Carlsbad, CA), and, following further washes in PBS, immunopositivity was revealed using staining solutions of nitro blue tetrazolium (Sigma) and 5-bromo 4-chloro 3-indolyl phosphate (Sigma). The stained embryos were then transferred to glycerol (Molekula) for microscopy and imaging.
Immunolocalization was performed for the muscle marker α-actinin, the neural crest marker Sox9, and the lateral plate mesoderm marker Tbx5, by adapting previously published methods [27]. In brief, RARγ-agonist-treated and control embryos were fixed in 4% PFA at 4°C overnight at the time points indicated in “Results” section and then washed repeatedly in PBS-Tween (PBS-T; 90%:10%) before an enzymatic digestion with collagenase (1 mg/mL; Sigma) for 75 min at 37°C. The embryos were then incubated with antibodies specific for α-actinin (1/100 dilution in PBS-T; Sigma) or Sox9 (1/100 dilution in PBS-T, Clone ab76997; Abcam Ltd., Cambridge, UK) or Tbx5 (Genetix, Irvene, CA) at 4°C overnight. Embryos were then washed repeatedly in PBS-T prior to incubation with Alexa 594-conjugated anti-mouse antibodies for α-actinin or Sox9 (1/250 dilution in PBS-T; Invitrogen Ltd.) at 4°C overnight, washing again in PBS-T, and then mounted in glycerol for microscopy and image capture. For Tbx5 immunolocalization, embryos were similarly washed repeatedly in PBS-T after incubation with the primary antibodies, after which immunopositivity was revealed using biotin-anti-rabbit secondary antibodies (1/400 in PBS-T; Vector Labs, Peterborough, UK) followed by further washes and incubation with streptavidin-linked fluorescein isothiocyanate (FITC) (1/50 in PBS-T; Vector Labs), adapting methods previously described [28].
Alizarin red staining for bone formation
Alizarin red staining was performed to examine the presence of bone by adapting previously published methods [25]. In brief, RARγ-agonist-treated and control embryos were euthanized at 5 days postfertilization (dpf) and fixed in 4% PFA at 4°C overnight and then washed in PBS-T prior to staining in a solution of alizarin red (96% of 0.5% potassium hydroxide:4% of 0.1% alizarin red S; Molekula) for 3 h at room temperature. After staining, the embryos were washed and mounted in glycerol prior to microscopy and image capture.
Image capture and analysis
Phase-contrast or bright-field digitized images were captured using a Nikon DXM1200 camera attached to a Nikon SMZ745T stereomicroscope, a Leica DFX 300FX camera attached to a Leica MZ 16FA stereomicroscope, or a Jenoptik camera attached to a Ceti inverted microscope. A Leica DFX 300FX camera, attached to a Leica MZ 16FA stereomicroscope, was also used to capture fluorescence images of the transgenic zebrafish embryos and of α actinin-immunostained embryos. Laser scanning confocal microscopy was performed with the Leica Microsystems DM6000B-SP57CS confocal system to generate z stacks of Sox9- and Tbx5-immunostained zebrafish embryos. These were then converted into 3 dimensional (3D) projected images of the entire ventral-dorsal or lateral aspects. The projected images of Sox9-immunostained zebrafish embryos were used to quantify the number of Sox9-immunopositive cells present in the total head region that was anterior from the anterior aspect of the otic vesicle. Phase-contrast digitized images were captured at low magnification (4× lens) and these images were analyzed to determine the embryo length along the antero-posterior axis using Image J software.
Caudal fin transection
The AB strain or Tcf:mini-p transgenic zebrafish embryos at 2 or 3 dpf, respectively, were anesthetized with 0.4% Tricaine (Sigma) and then placed on a glass slide and the caudal fin was transected using a thin scalpel blade (SLS). The embryos were then transferred to fresh Hank's saline or E3 media in an incubator at 28.5°C for those periods of fin regrowth indicated in “Results” section. The embryos were treated with the RARγ agonist (10 nM) or carrier alone immediately following caudal fin transection. In some experiments, the AB strain embryos were treated with an RARγ-specific antagonist at 3 dpf (following RARγ agonist treatment at 2 dpf) or subjected to washout of the RARγ agonist at 3 dpf.
Statistical analysis
Data were evaluated for normal distribution using the D'Agostino and Pearson omnibus normality test. One-way or two-way analysis of variance (ANOVA) was used to assess the relationships between treatment of zebrafish embryos with different concentrations of the RARγ agonist and fish length or the growth of treated versus control embryos over time, with post hoc analysis. Student's paired t-tests or Mann–Whitney U tests were used to examine differences between treatment versus control groups for hox gene expression or the prevalence of Sox9-immunopositive cells, according to whether the data were normally or not normally distributed, respectively. All statistical analyses were performed using GraphPad Prism software. Values were considered statistically significant, as indicated in “Results” section, at P≤0.05 (*). Unless otherwise indicated all data have been presented as means±standard deviations where a minimum of n=3 procedures were performed for all experiments. The total number of embryos in these experiments that were pooled and analyzed have been shown in figure legends.
Results
Treatment of zebrafish embryos with the RARγ agonist inhibited antero-posterior growth and decreased somite formation
Treatment of zebrafish embryos at 4 hpf with the RARγ agonist was associated with clear morphological differences during development, with the main difference being a reduced antero-posterior axis length (Fig. 1A). The reduction in fish length was RARγ agonist dose dependent. Treatment of embryos with 1 nM RARγ agonist had no obvious effects on zebrafish growth while treatments with the RARγ agonist at 80 nM (or above) caused severe truncation (Fig. 1B). The RARγ-agonist-treated embryos also developed cardiac edema and, beyond 3 dpf, embryos treated with the higher doses of the RARγ agonist lost viability (data not shown). Therefore, all further analyses were performed using the RARγ agonist at 10 nM, which was not lethal. Changes in the antero-posterior length of embryos treated with 10 nM RARγ agonist were most markedly affected from 2 dpf onward, such that the treated embryos were significantly shorter than the control embryos at 3–5 dpf (Fig. 1C).
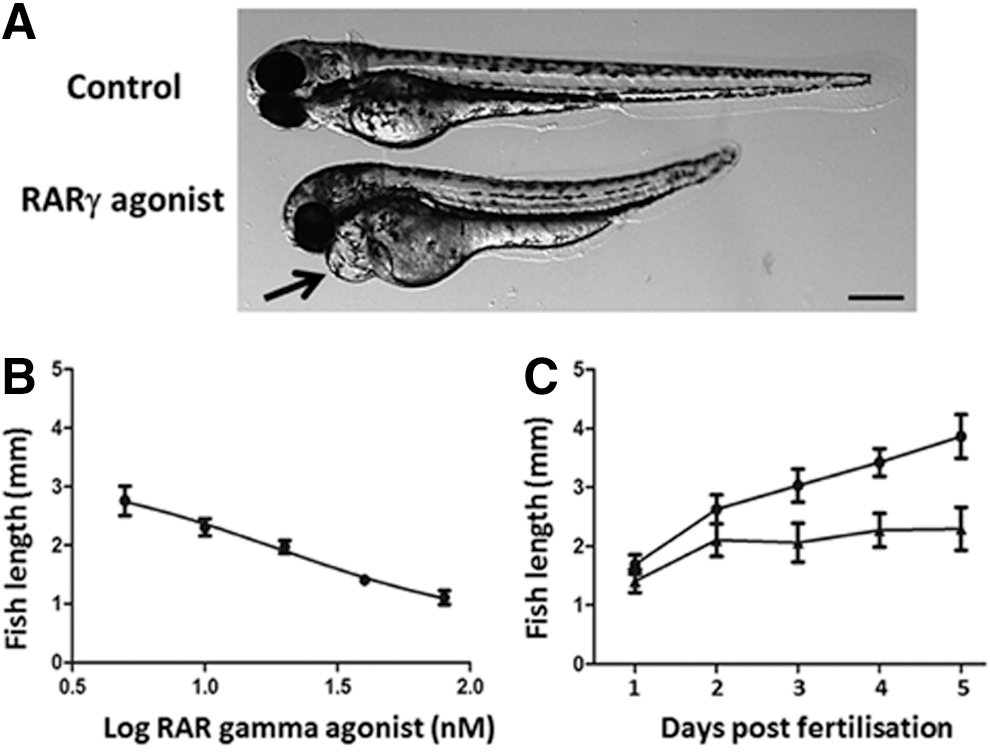
Treatment of embryonic zebrafish at 4 h postfertilization (hpf) with a retinoic acid receptor γ (RARγ) agonist was associated with a dose-dependent decrease in the antero-posterior length and slower axial growth.
Somite formation was observed using an indicator transgenic fish line (hspGFF55B), which has green fluorescent protein (GFP) expression in its somites and heart [21]. At 3 dpf, the number of somites present in RARγ agonist (10 nM)–treated embryos in this transgenic fish line was consistently observed to be 25, which is 5 fewer than the 30 somites that were observed in the control transgenic embryos at the same stage of development (and is the normal somite number in adult zebrafish) [29] (Fig. 2A). In situ hybridization for myoD, which specifies myocyte differentiation, as well as immunolocalization for the myocyte marker α-actinin, was performed to further assess somite formation. Although a somewhat weaker level of myoD expression was seen in the anterior regions of the RARγ-agonist-treated embryos compared with control embryos (Fig. 2B, C), this was not associated with a clear inhibition in myocyte formation as delineated by α-actinin immunoreactivity (Fig. 2D, E). Further, the pattern of α-actinin immunoreactivity confirmed that there was the same reduced number of somites (ie, 25 somites at 3 dpf) in the RARγ-agonist-treated embryos compared with control embryos (ie, 30 somites at 3 dpf). Therefore, treatment with an RARγ agonist was associated with decreased axial length and a specific reduction in the formation of somites without a uniform inhibition of myocyte differentiation.
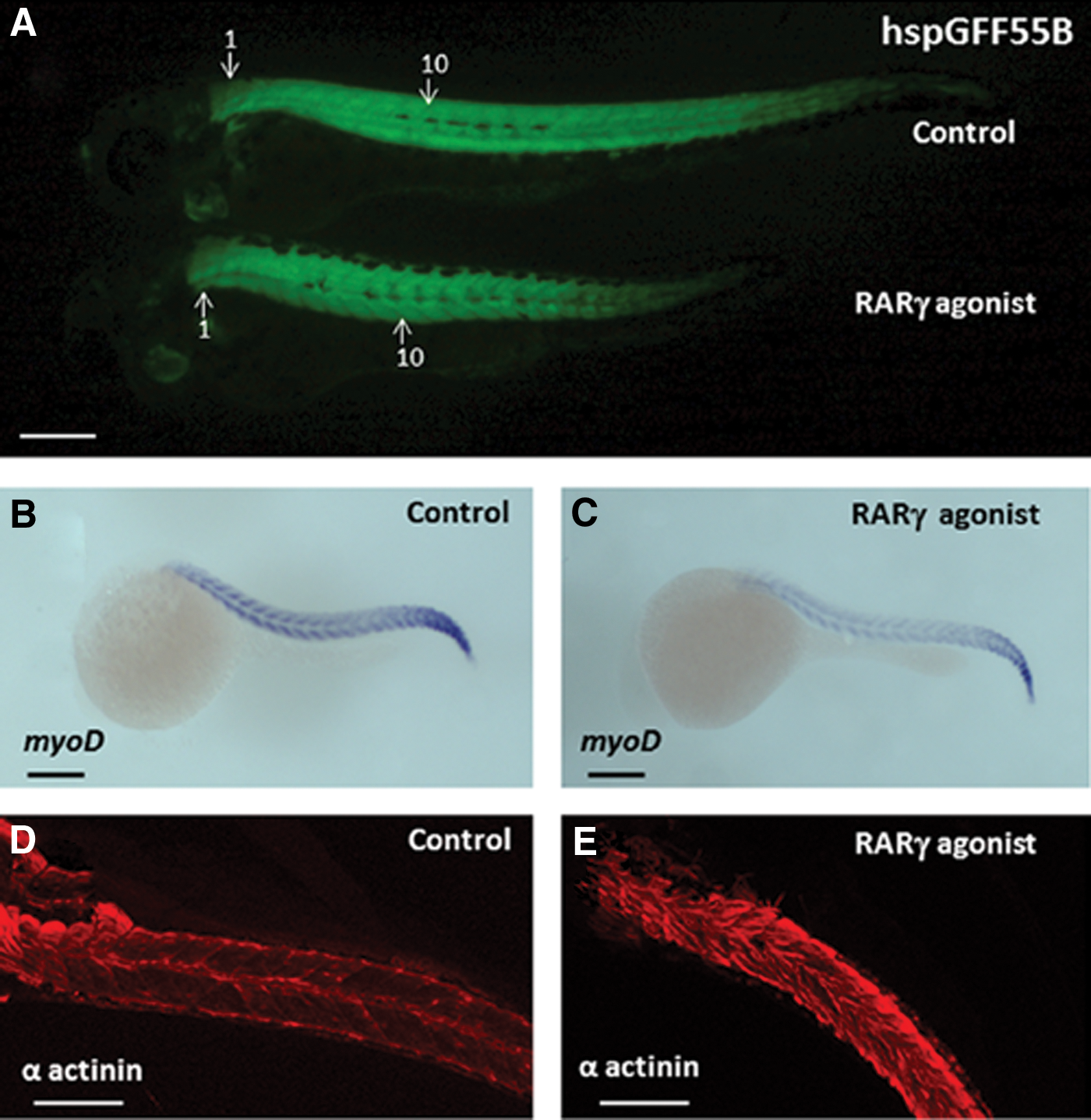
Treatment of embryonic zebrafish with a RARγ agonist was associated with a reduction in somite formation.
Differential hoxc11a and hoxb13a expression patterns demonstrated that the reduced antero-posterior length in RARγ-agonist-treated zebrafish embryos was associated with a loss of posterior tissues
Precise expression of the hox gene family is necessary for normal antero-posterior axis formation in vertebrates [30]. Transgenic fish lines, SAGFF155A and hspGFFDMC28B, were used to indicate the expression patterns of hoxc11a and hoxb13a in RARγ-agonist-treated versus control embryos (Fig. 3). As shown, hoxc11a was expressed from near the anal region to the caudal regions in the RARγ-agonist-treated and control embryos at 2 dpf (Fig. 3A), although the cranial start of hoxc11a expression was three to four somites more posterior in the RARγ-agonist-treated embryos. In sharp contrast, the expression of hoxb13a at 2 dpf (Fig. 3B) was only seen in the most posterior caudal region of the control embryos, as well as in the anal region, whereas no hoxb13a expression was seen in the RARγ-agonist-treated embryos. These patterns of expression were consistent in all treated and control zebrafish embryos throughout their development. The distance from the anterior end of the head to the anal regions of either the wild-type (AB strain; see Fig. 1A) or the hox gene reporter fish lines was similar in the RARγ-agonist-treated and control embryos. Conversely, there was a marked decrease in the distance from the anal regions to the most caudal end of the RARγ-agonist-treated embryos compared with control embryos. Hence, these differential patterns of hox gene expression indicate that RARγ agonism adversely prevented the formation of the most posterior somites and caudal fin, which was associated with a complete loss of hoxb13a expression.
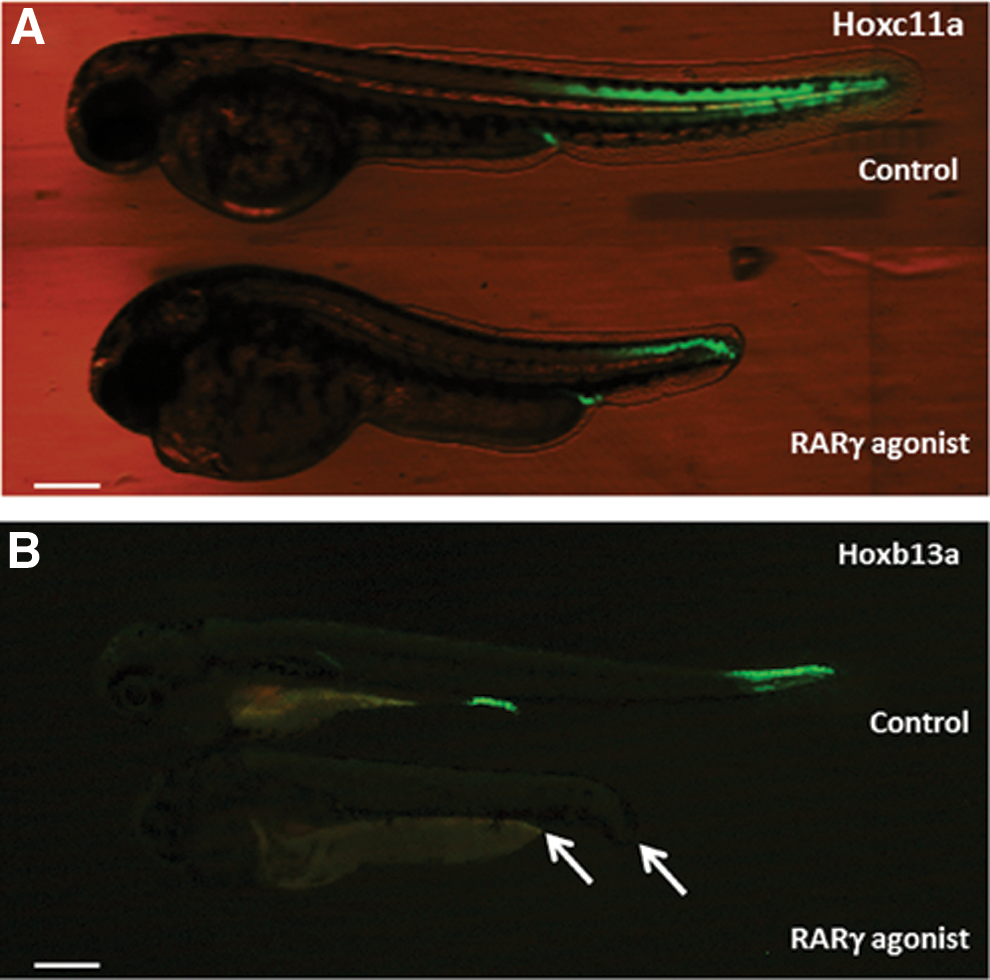
Differential patterns of hoxc11a and hoxb13a expression in transgenic reporter zebrafish embryos demonstrated that the reduced axial length of RARγ-agonist-treated embryos was associated with loss of their most posterior regions.
Hoxb13a expression and growth of the caudal fin were rescued by washout of the RARγ agonist or additional treatment with the RARγ antagonist
To examine whether the loss of hox gene expression after RARγ agonist treatment may have been due to irreversible teratogenic effects early in the development of the zebrafish, rather than being targeted toward the hox gene pathway, we examined whether hoxb13a expression returned following treatment with the RARγ agonist at 4 hpf and then either washout of the RARγ agonist at 23 hpf or cotreatment with the RARγ antagonist, also at 23 hpf. As shown (Fig. 4), no hoxb13a expression was observed in the anal region and the most posterior region of the tail or caudal fin in RARγ-agonist-treated embryos at 5 dpf (Fig. 4A). In contrast, in the embryos that were treated with the RARγ agonist at 4 hpf and then subsequently treated with the RARγ antagonist at 23 hpf, increased hoxb13a expression was seen in both anal and the tail regions at 5 dpf (Fig. 4B, C). This pattern of elevated hoxb13a gene expression at 5 dpf was also evident in embryos in which the RARγ agonist was washed out at 23 hpf (data not shown). Moreover, data analysis of caudal fin length also showed that both washout and RARγ antagonist treatment caused significant increase in caudal fin length at 5 dpf compared with RARγ-agonist-treated embryos (Fig. 4D, E). Hence, these data suggest that the inhibitory effects of the RARγ agonist on hoxb13a expression and caudal fin formation were not due to early irreversible teratogenic wherein hox gene expression was not possible.
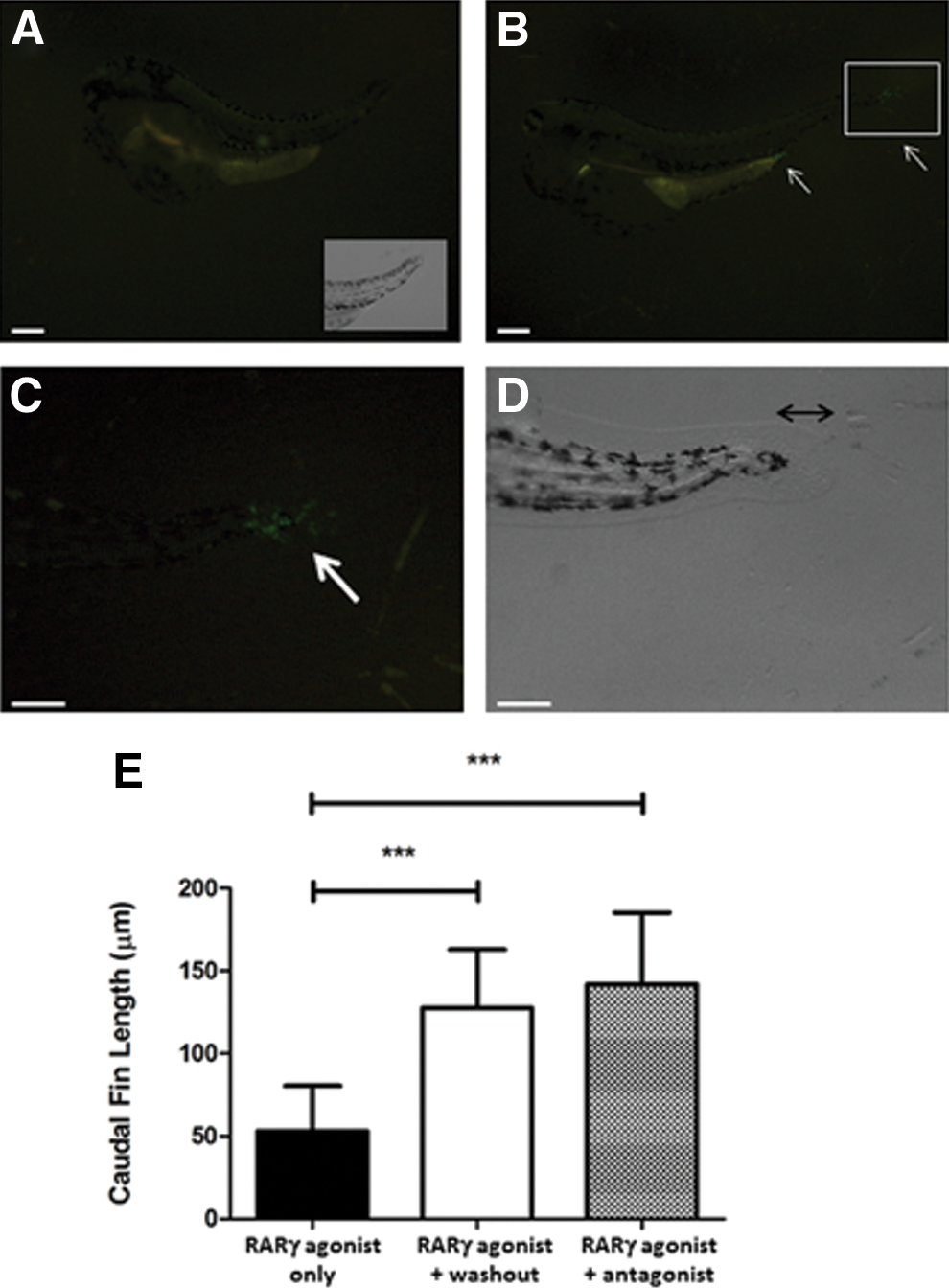
The effects of a RARγ agonist on hoxb13a and caudal fin outgrowth were reversed by agonist washout or cotreatment with a RARγ antagonist. Representative fluorescence microscopy image showing GFP expression in the hspGFFDMC28B transgenic zebrafish reported line for hoxb13a expression at 5 dpf, in which the embryos were treated with RARγ agonist at 4 hpf
Treatment with the RARγ agonist caused changes in head morphology, loss of anterior cranial bones, and loss of anterior lateral line ganglia, but did not affect the prevalence of Sox 9-immunopositive neural crest cells
The head morphology of RARγ-agonist-treated embryos was markedly different from that of control embryos. At 3 dpf, there was a clear loss of the most anterior parts of the head, that is, the primordia of the mouth and jawbone region, as well as an apparent loss of pharyngeal arches in the RARγ-agonist-treated embryos compared with control embryos (Fig. 5A, B). Alizarin red staining of embryos at 5 dpf confirmed a complete lack of anterior cranial bones, including the maxillary, the hyomandibular, and the opercle bones, as well as reduced formation of the cerebrobrachial bones in RARγ-agonist-treated embryos compared with control embryos (Fig. 5C, D). The HGn39D transgenic line was used to examine neural tissues of the lateral line system within the anterior regions of the head [23]. This analysis demonstrated a loss of anterior lateral line ganglia in the RARγ-agonist-treated embryos at 3 dpf compared with controls at the same time point (Fig. 5E), but no clear differences were seen in either the posterior lateral line ganglia or the lateral line itself. Immunolocalization of Sox9, which is a marker for neural crest cells [31], showed that there was no clear difference in the prevalence of Sox9-immunopositive cells between RARγ-agonist-treated and control embryos at 25 hpf, but that Sox9-immunopositive cells then decreased in number in the anterior regions of the head in the RARγ-agonist-treated embryos only by 60 hpf (Fig. 6). Hence, RARγ agonist treatment was associated both with a loss of cranial neural crest stem/progenitor cells as well as loss of the most anterior cranial tissues that these cells form.
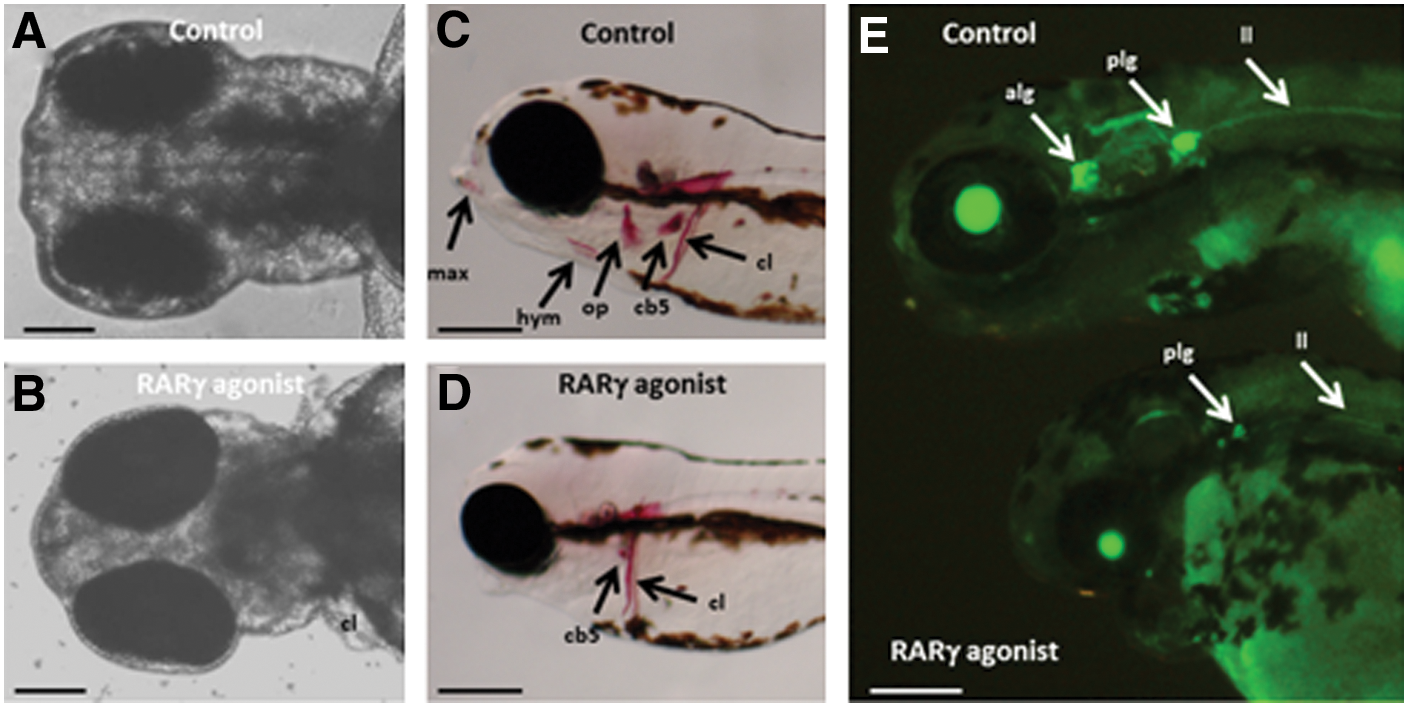
Treatment of zebrafish embryos with a RARγ agonist was associated with loss of cranial tissues derived from the neural crest.
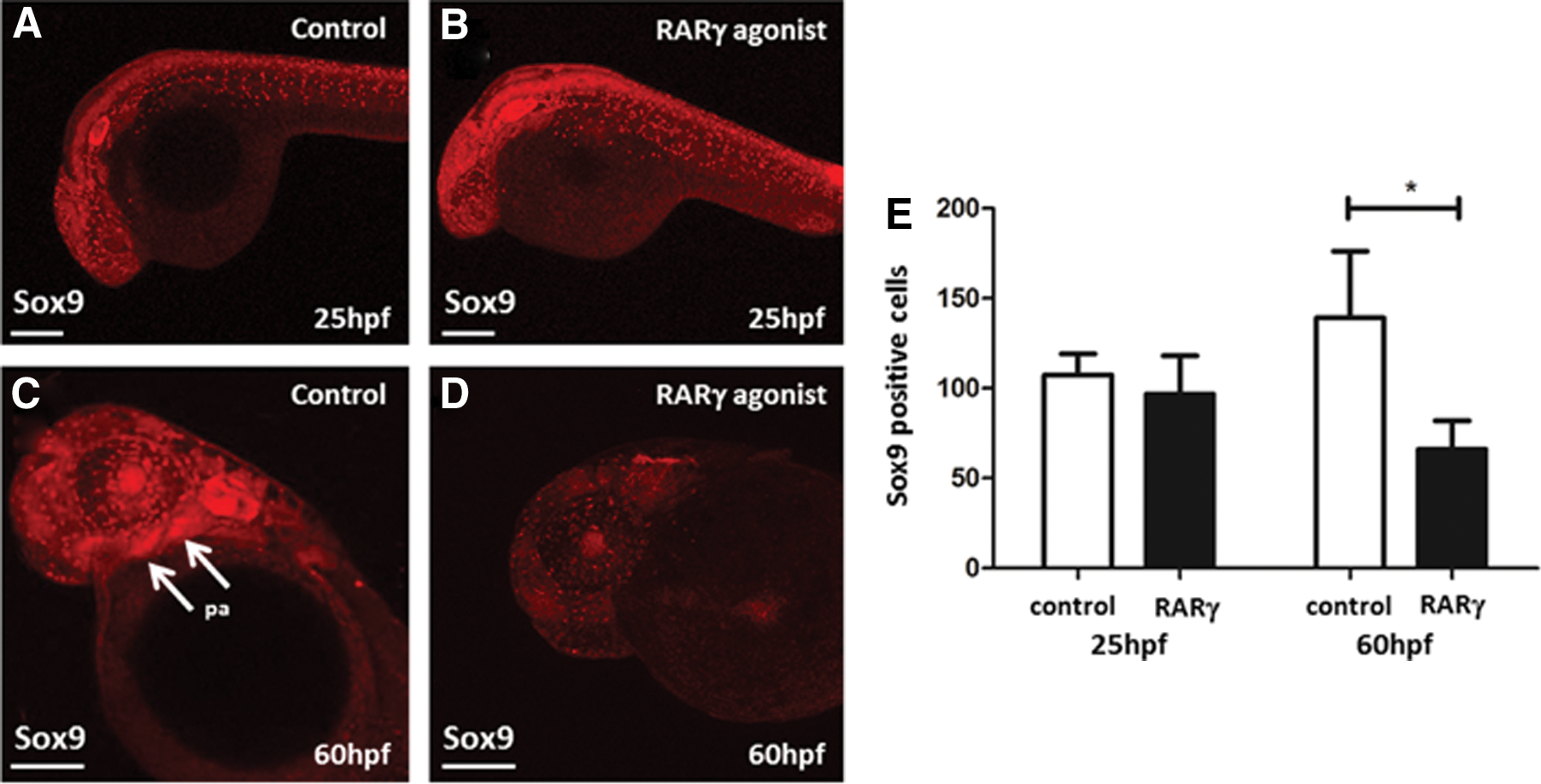
Treatment of zebrafish embryos with a RARγ agonist was associated with loss of Sox9-immunopositive cranial neural crest stem/progenitor cells.
Treatment with the RARγ agonist reversibly blocked pectoral fin outgrowth
Although the pectoral fin bud was clearly present, RARγ-agonist-treated embryos did not grow the pectoral fin (Fig. 7A, B). Immunolocalization for the transcription factor Tbx5a, which is a marker of the lateral plate mesoderm stem/progenitor cells that form the pectoral fin as well as the heart [32,33], showed that Tbx-5-immunopositive cells were present in these regions in both the RARγ-agonist-treated and the control embryos at 27 hpf (Fig. 7C, D), which is the time point when pectoral fin outgrowth commences [33]. Hence, there was no loss of lateral plate mesodermal stem/progenitor cells associated with the effects of the RARγ agonist on the pectoral fin, or, indeed, the development of cardiac edema. Embryos were treated with the RARγ agonist from 4 hpf and then the medium containing the RARγ agonist was either replaced with control media (ie, washout) or the same concentration (10 nM) of the RARγ antagonist was added at 27 hpf. In both of these experiments, subsequent pectoral fin outgrowth was observed at 3 dpf (Fig. 7E, F). In contrast, when the control embryos were treated with the RARγ agonist at 27 hpf, pectoral fin outgrowth was subsequently completely blocked (Fig. 7G). Therefore, treatment with the RARγ agonist was associated with a reversible block in pectoral fin formation, which was independent of Tbx5 expression.
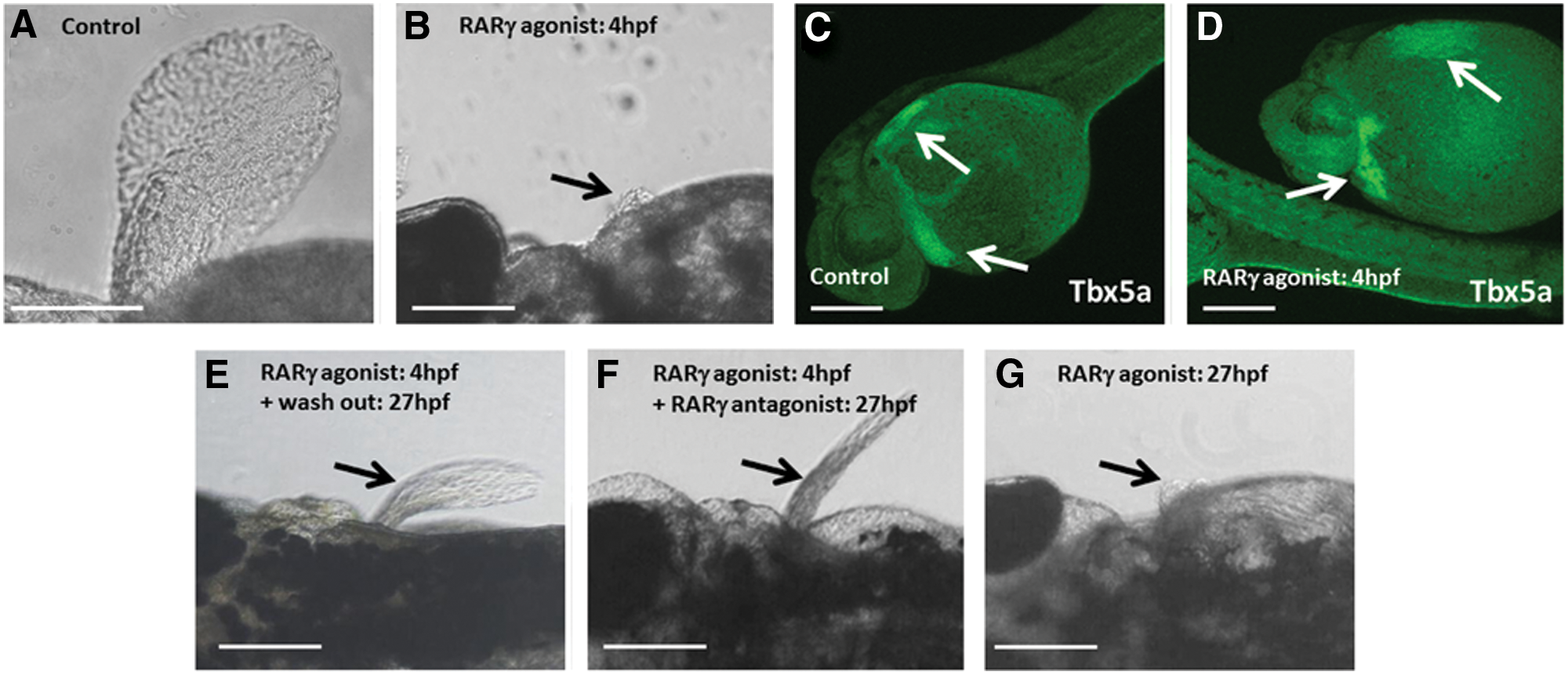
Treatment of zebrafish embryos with a RARγ agonist was associated with a reversible block in pectoral fin outgrowth with no evident changes in loss of Tbx5 expression in the lateral plate mesoderm at 27 hpf.
Treatment with the RARγ agonist reversibly blocked caudal fin regeneration
RARγ is expressed in the caudal regions of the developing zebrafish [9,10] and again during adult caudal fin regeneration after transection [34]. The caudal fin in larval zebrafish can regenerate within 3 days after fin transection at 2 dpf [35]. Therefore, we performed experiments to examine whether RARγ agonist treatment also affected caudal fin regeneration in zebrafish larvae. Caudal fin regeneration, following transection at 2 dpf, was observed in control embryos at 5 dpf (Fig. 8A). In contrast, the regeneration process was completely blocked when the embryos were treated with the RARγ agonist immediately following fin transection (Fig. 8B). When treatment with the RARγ agonist immediately following fin transection at 2 dpf subsequently followed with the addition of the RARγ antagonist (10 nM) or by washout of the RARγ agonist at 24 h post-transection, then fin regeneration was again observed at 5 dpf (Fig. 8C, D).
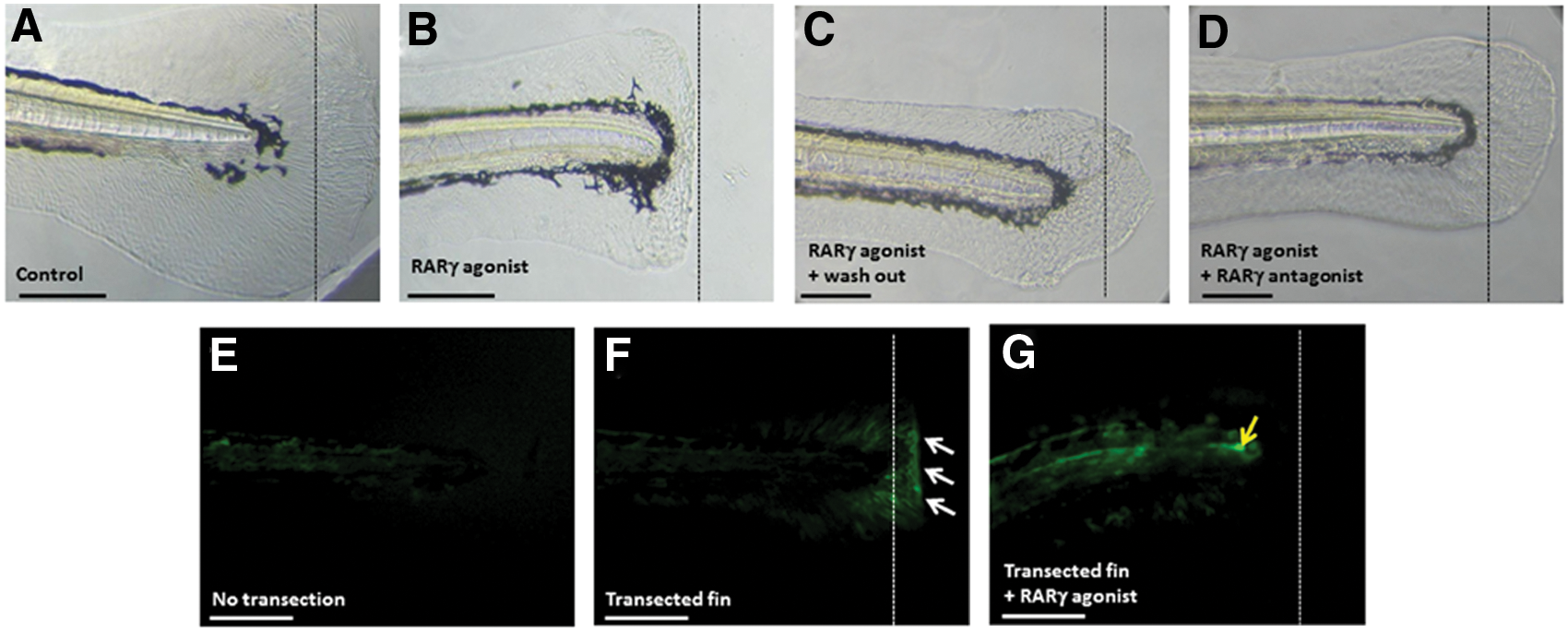
Treatment of zebrafish embryos with a RARγ agonist was associated with a reversible block in caudal fin regeneration and decreased canonical Wnt signaling.
Canonical Wnt signaling has been reported as important to blastema formation during zebrafish caudal fin regeneration [36]. The upregulation of canonical Wnt signaling during adult caudal fin regeneration is observed in the Tcf:mini-p reporter zebrafish line, in which GFP expression is driven by the engagement of β catenin with the Tcf reporter [24]. However, the involvement of canonical Wnt signaling in larval fin regeneration has not been reported. Canonical Wnt signaling in caudal fin rays during normal development was not evident using the reporter fish line by 4 dpf (Fig. 8E). In contrast, following caudal fin transection at 3 dpf, increased canonical Wnt signaling was observed in the regenerating blastema at 4 dpf (Fig. 8G). This increased Wnt signal at 4 dpf was not observed in zebrafish embryos that were transected at 3 dpf and immediately treated with the RARγ agonist (10 nM), although there was an evident increase in signal in the notochord (Fig. 8F). Therefore, treatment with the RARγ agonist was associated with a reversible block in caudal fin regeneration, which was associated with decreased canonical Wnt signaling.
Discussion
This study was performed to examine the potential role of RARγ in regulating embryonic development and tissue regeneration using wild-type and gene reporter embryonic zebrafish as model systems. We demonstrated that treatment of embryonic zebrafish with a RARγ-specific agonist had profound developmental consequences, including decreased antero-posterior growth, inhibition of cranial bone and neural tissue formation, and loss of growth of the pectoral and caudal fins, as well as abrogation of caudal fin regeneration following fin transection. We also noted that RARγ-agonist-treated fish exhibited cardiac edema. These findings strongly suggest that RARγ activity must be tightly regulated during normal embryonic development and tissue regeneration.
The tissues that were affected by treatment with the RARγ agonist derive from those stem/progenitor cell populations in which RARγ expression is restricted during early embryonic development, that is, in the cranial neural crest, in the lateral plate mesoderm, and in the presomitic mesoderm of the tail bud [9,10]. In addition, the tissues affected by the RARγ agonist also mirror those that are similarly affected by morphilino gene knockdown of RARγ (a and b), namely, the pharyngeal arches and pectoral and caudal fins [37]. Further, this phenotype is similar to the effects of RA treatment on embryonic development [38]. Taken together, this suggests that the agonist used in the current study was specific for RARγ in zebrafish, as it has been conclusively demonstrated in mammalian reporter cell lines [15].
The mechanisms involved in the developmental changes observed have been examined using a combination of transgenic reporter fish lines, in situ hybridization and immunohistology for several important genes, and appropriate stem/progenitor cell markers. The shorter body axis formation seen in RARγ-agonist-treated embryos was associated with decreased somite formation, as revealed in hspGFF55B transgenic embryos and α-actinin immunolocalization. The differential expression of the hox gene family plays an essential role in formation of the antero-posterior axis during vertebrate embryonic development [30]. Hence, we examined the effects of the RARγ agonist on transgenic hox gene reporter fish lines. Analysis of the hoxc11a and hoxb13a reporter lines demonstrated that the shorter body axis was largely the result of loss of the most posterior region of the embryos. Hoxc11 is developmentally expressed in the posterior region of the mouse embryo [39] and homozygous knockout of the hoxc cluster results in loss of caudal vertebrae [30,40]. In mice, hoxb13 expression occurs in the tail bud area around E9 [41]. Hoxb13 expression is also found in the developing tail of axolotl and re-expressed in the regenerating tail after transection [42]. These expression patterns suggest that hoxb13 plays an important role in the development and growth of the tail. Conversely, a heterozygous knockout of hoxb13 in mice was shown to cause overgrowth of the tail [43]. Our observation of a complete loss of hoxb13a expression in RARγ-agonist-treated zebrafish supports the hypothesis that this gene is essential for the formation of the posterior regions of developing embryos and may be regulated by RARγ. Extra-somitic expression of hoxb13a was also seen in the anal fin area of the control embryos, which was completely lost in the RARγ-agonist-treated embryos. Other researchers have similarly reported Hoxb13 expression in the hindgut and urogenital area of mice [41]. Hence, our observation further suggests that treatment with the RARγ agonist targeted the expression of hox genes in the posterior regions of the zebrafish embryos, particularly hoxb13a expression, in that no expression was seen following such treatment. We cannot exclude the possibility that hoxb13a expression may have been absent from the RARγ-agonist-treated embryos because the tissues in which hoxb13a is normally expressed during development, including the anal area, were not formed. However, the re-expression of hoxb13a in both the anal region and tail region in embryos that were treated at 4 hpf with the RARγ agonist, but then subsequently were the RARγ agonist was washed out or the embryos were additionally treated with the RARγ antagonist, which was also associated with evident renewed growth of the caudal fin, would suggest that at least such a loss of tissue formation is acutely responsive to the presence of reagents that affect RARγ. The loss of hox gene expression in response to treatment with a RAR agonist is certainly paradoxical as RARE is located within the regulatory region of hox gene clusters [44], which are normally upregulated in response to RA [45 –47]. Such a paradox may be resolved if RARγ does not require RA or indeed does not function through RARE to regulate target gene expression.
The marked morphological changes seen in the head of the RARγ-agonist-treated embryos were associated with loss of cranial bones and the anterior lateral line ganglia as revealed by alizarin red staining and the HGn39D transgenic fish line, respectively. These tissues form from the neural crest, as well as the placode for the lateral line [48]. This suggests that RARγ agonist treatment may have targeted neural crest stem/progenitor cells, which are known to express RARγ [9,10]. The fact that we saw significantly fewer Sox9-immunopositive cells in the cranial region of the RARγ-agonist-treated embryos might be considered to support this hypothesis. However, this difference was only seen in later time points, that is, at 60 hpf but not at 25 hpf, even though RARγ is expressed at the earlier time point. Further, even at 60 hpf, Sox9-immunopositive cells were still observed in the cranium. Therefore, it is also possible that fewer Sox9-immunopositive cells were prevalent within the anterior cranial regions of the RARγ-agonist-treated embryos simply because the anterior tissues did not form. Further research is required to examine whether the loss of Sox9 neural crest stem/progenitor cells following treatment with the RARγ agonist is causal to the loss of anterior cranial tissues observed. However, it is noteworthy that Sox9a or Sox9b morphilino knockdown and Sox9a mutant zebrafish embryos were shown to lack cranial skeletal structures [49,50]. These developmental defects were associated with Sox9 aberrant neural crest stem/progenitor cell differentiation, rather than any changes in neural crest formation or cell migration. Similarly, we conclude that treatment with the RARγ agonist did not markedly affect the formation of neural crest stem/progenitor cells, as depicted by Sox9-immunopositive cells at 25 hpf, but may well have influenced their survival, proliferation, migration, or differentiation thereafter, to adversely affect the development of cranial tissues.
Although almost all of the neural-crest-derived cranial bones were absent or greatly decreased in the RARγ-agonist-treated embryos, there were still fully intact cleithrum bones, which also are of neural crest origin [51]. There is no clear explanation for this difference. However, it is interesting that cleithrum bones undergo a process of dermal ossification, unlike other affected cranial bones that undergo endochondral ossification [51,52].
An additional major phenotype seen following treatment with the RARγ agonist was the loss of pectoral fin outgrowth, despite formation of the fin bud. Other studies have reported that the interaction of RA signaling with expression of Tbx5a transcription factor is essential for pectoral fin formation. Increased raldh1a2 expression and its localized synthesis of RA in the region of somites two to six, where Tbx5a expression is induced in response to RA, is required for formation of the pectoral fin [32]. Both the Nls (neckless)–mutant zebrafish, which carries a mutation in raldh1a2 [16], and embryonic zebrafish treated with the RA synthesis inhibitor diethylaminobenzyldehyde (DEAB) do not express Tbx5a and do not form pectoral fins [53]. Moreover, loss-of-function Tbx5a mutations block pectoral fin formation, as well as causing heart defects [54]. Zebrafish Tbx5a expression can be seen from 14 hpf in the lateral late mesoderm, which is the common stem/progenitor cell population for development of the heart and pectoral fins and separate from 24 to 27 hpf [33]. Because the pectoral fins did not form in the RARγ-agonist-treated embryos in our study, Tbx5 was considered a potential target gene for RARγ agonism. However, we found no marked differences in the presence of Tbx5a-immunopositive stem/progenitor cells in the RARγ-agonist-treated or control embryos, suggesting that Tbx5a expression was unaffected. Further, RARγ-agonist-washout experiments or cotreatment with a RARγ-specific antagonist (as well as the agonist) completely abrogated the block of pectoral fin formation, demonstrating that Tbx5 was functional. Finally, treatment with the RARγ agonist at 27 hpf, which we had confirmed was when Tbx5 was present in the lateral plate mesoderm, was also found to completely block pectoral fin outgrowth. Therefore, we conclude that the effect of RARγ agonist treatment on pectoral fin outgrowth was independent of Tbx5. Potential targets that lie downstream of Tbx5 activity include fgf family genes, that is, fgf8, fgf10, and fgf24 [55,56], as well as sall4 [57], blimp-1 [58], prdm1 [59], beta-CaMK-II [60], and ndrg4 [61]. In addition, hox gene expression was also associated with pectoral fin formation, including hoxa [62], hoxb [63], and hoxd [64,65]. Further study will elucidate whether RARγ agonist treatment alters the expression of each of these genes, but given our finding that hoxb13a was completely inhibited by such treatment, this family is a clear target.
Lastly, we tested whether the RARγ agonist affected tissue regeneration as well as development using transection of the caudal fin as a model because RARγ is expressed in blastemal cells at least during adult zebrafish caudal fin regeneration [34]. Similar to our findings with pectoral fin outgrowth, we found that RARγ agonist treatment was associated with a complete, but reversible, block on caudal fin regeneration. Canonical Wnt signaling is known to play a major role in caudal fin regeneration [36]. Therefore, we investigated whether this pathway was affected by treatment with the RARγ agonist using the transgenic reporter line Tcf:mini-p [24]. As we hypothesized, the block in caudal fin regeneration following RARγ agonist treatment was associated with an observed reduction in Wnt signaling, suggesting that the Wnt pathway is a target for RARγ. However, similarly to our observations of Sox9-immunopositive cells in the cranial neural crest, there is also the possibility that Wnt signaling was not seen following transection of the caudal fin and treatment with the RARγ agonist because the regenerating tissues did not form. Therefore, further study is required to determine whether the evident loss of Wnt signaling following RARγ agonist treatment is causal to the lack of a regenerative response.
In summary, our results have shown that treatment of zebrafish embryos with a RARγ-specific agonist adversely affected the development and growth of tissues that form from stem/progenitor cells that express RARγ [9,10,34]. These stem/progenitor cell zones [4,32,66] do not express RA-synthesizing enzymes [16,17] and are not exposed to intrinsic RA [18]. This suggests that RARγ functions in the absence of RA ligation. The function of non-RA-ligated RARs is not clear. In mice, RARs were considered to repress gene expression through corepressor activity in the absence of RA ligation [8,14]. However, it has been reported that RARs in zebrafish may not have such corepressor activity [67]. What is clear is that RARγ plays an essential role during embryonic development and likely regulates stem/progenitor cell populations. Similar to our results using a RARγ agonist, RARγ morphilinos were found to adversely affect the formation of cranial tissue, pectoral fin outgrowth, and the caudal tail [37]. Moreover, in somatic cell reprogramming, RARγ overexpression was related to the rate and efficiency of reprogramming to induced pluripotency [68]. This was suggested to indicate that RARγ functions to maintain pluripotent stem cell populations. Although the exact function of RARγ is still not clear, based on our current findings we concur with this hypothesis and further suggest that such activity is seen only when RARγ is nonligated.
Footnotes
Acknowledgments
The authors are grateful to Dr. Tohru Ishitani (Kyushu University) for the provision of the transgenic zebrafish Tcf:mini-p reporter line. We are grateful to Charlotte Bland (Aston University) for confocal microscopy and to Anjana Patel, Georgiana Absoud, and Nisha Patel (all from Aston University) for assistance with other experimental procedures. We are grateful to the Charles Wallace Trust for their financial support of H.A.W. The research leading to these results has received funding from the People Programme (Marie Curie Actions) of the European Union's Seventh Framework Programme FP7/2007–2013/under REA grant agreement number 315902. G.B. and W.E.B.J. are partners within the Marie Curie Initial Training Network DECIDE (decision-making within cells and differentiation entity therapies).
Author Disclosure Statement
No competing financial interests exist.
