Abstract
Reciprocal signaling between the lung mesenchyme and epithelium is crucial for differentiation and branching morphogenesis. We hypothesized that the combination of signaling pathways comprising early epithelial–mesenchymal interactions and a 3D spatial environment are necessary for an efficient induction of embryonic and induced pluripotent stem cells (ESCs and iPSCs) into a lung cell phenotype with hallmarks of the distal niche. Aggregating early, but not late, embryonic lung mesenchyme with endoderm-induced mouse ESCs and iPSCs for 6 days resulted in organization into tubular structures and differentiation of the tubular lining cells to an NKX2-1+/SOX2−/SOX9+/proSFTPC+ lineage. Over 80% of the endoderm-induced cells committed to an NKX2-1+ lineage. Electron microscopy analysis demonstrated numerous multivesicular bodies and glycogen deposits in the tubular lining cells, characteristic features of type II epithelial cell progenitors. Using soluble FGFR2 receptor antagonists, we demonstrate that reciprocal fibroblast growth factor (FGF) 2, 7, and 10 signaling is essential for differentiation of endoderm-induced cells to an NKX2-1+/proSFTPC+ phenotype within 3D aggregates. Only FGF2 was able to commit endoderm-induced cells in monolayer cultures to an NKX2-1+ lineage, however with a significant lower efficiency (∼16%) than seen with mesenchyme. Thus, while FGF2 signaling alone can induce a primed population of ESCs and iPSCs, the cells do not differentiate to distal lung epithelial progenitors with the same efficiency and level of maturity that is achieved when the complex tissue and 3D environment of the developing lung is more accurately recapitulated.
Introduction
T
To gain further insight into the mechanisms of commitment from endoderm to early lung epithelial progenitor cells, we used ESCs and iPSCs to model lung development. We took advantage of the epithelial–mesenchymal interactions that are crucial for lung morphogenesis and differentiation by using early embryonic lung mesenchyme, producer of various growth and matrix factors implicated in early lung development, to differentiate ESCs and iPSCs to a lung epithelial cell phenotype. We demonstrate that early (E13), but not late (E19), distal embryonic lung mesenchyme of mouse and rat possesses all the instructive cues necessary to drive the majority of endoderm-induced mouse ESCs and iPSCs to an early proSFTPC+ lung epithelial phenotype in a 3D culture environment. Growth factor receptor inhibition studies revealed that signaling via FGFR1c and/or FGFR2c induces early lung epithelial (NKX2-1+) differentiation in the 3D aggregates while further advancement to a NKX2-1+/pro-SFPTC+ phenotype requires signaling via FGFR2b. Initial commitment of endoderm-induced mouse ESCs to an NKX2-1+ lineage was achieved with fibroblast growth factor-2 (FGF2), but not FGF9 treatment; however, the differentiated cell population was immature and less numerous compared with that generated using lung mesenchyme in 3D culture.
Materials and Methods
ESC/iPSC maintenance
The double-reporter mouse ESC line Foxa2/hCD4;Bry/GFP was obtained from Dr. Gordon Keller and was maintained as described previously [6]. The mouse iPSC line iPS EOS3F-29 was obtained from Dr. James Ellis and was maintained as described [26]. Additional ES and iPS cell lines tested produced similar results (Supplementary Fig. S2D; Supplementary Data are available online at
ESC/iPSC endoderm differentiation
ESCs were trypsinized using TrypLE express (Gibco, Burlington, ON, Canada) for 4 min at 37°C and cultured at 20,000 cells/mL in low-adherence six-well plates (Nunc, Roskilde, Denmark) to allow for embryoid body (EB) formation. EBs were collected and dissociated after 48 h and reaggregated with 50 ng/mL activin A (R&D Systems, Minneapolis, MN) for an additional 48 h. EBs were harvested for flow cytometry or fluorescence-activated cell sorting (FACS) analysis. iPSCs were differentiated in a similar manner with one additional day of activin A treatment. To achieve anterior definitive endoderm differentiation, sorted endoderm cells were seeded on a monolayer and treated with 200 ng/mL NOGGIN (BMP signaling inhibitor) and 10 μM SB-431542 (pharmacological inhibitor of TGFβ/nodal signaling) for 24 h [31].
FACS and flow cytometry
Day-4 (ESCs) or day-5 (iPSCs) EBs were dissociated and the single-cell suspensions were labeled with antibodies in Hank's balanced salt solution (HBSS) supplemented with 2% (v/v) fetal bovine serum (FBS) and 10 mM HEPES buffer (Invitrogen, Burlington, ON, Canada) for 30 min on ice. Antibodies used are summarized in Supplementary Table S1. Cells were analyzed using the Galios flow cytometer and data analysis was carried out using Kaluza software (Beckman Coulter, Mississauga, ON, Canada). Cell sorting was carried out on AriaII-RITT BRV (Becton Dickson, Franklin Lakes, NJ) and MoFlo BRU cell sorter (Beckman Coulter).
ESC/iPSC-lung mesenchyme aggregate cultures
All animal experiments were approved and carried out in accordance with the animal care committee guidelines of the Hospital for Sick Children. CD1 mice were time-bred on-site overnight while timed-pregnant Wistar rats were ordered from Charles River (St. Constant, QC, Canada). On day 11.5 (mouse) or 13 (rat) of gestation (day 19 for late mesenchyme), dams were euthanized and fetuses were delivered by caesarean section. Lungs were dissected from embryos and transferred to HBSS and treated with 20% (v/v) dispase and 1.5% (v/v) DNase (Invitrogen, Mississauga, ON, Canada) for 45 min at 37°C. Cultures were neutralized with Dulbecco's modified Eagle's medium+10% (v/v) FBS (Invitrogen) and the distal mesenchymal layer was separated manually from the endoderm using 27-gauge needles. Distal mesenchyme was transferred to Nucleopore membranes (8-μm pore size; Whatman, Piscataway, NJ) with 10–20 activin-induced EBs, resulting in a ratio of 1:4 EBs to mesenchyme. These aggregates and controls (EBs without mesenchyme) were cultured in serum-free differentiation medium, at air–liquid interface (Supplementary Fig. S1). Cells were maintained in culture for a total of 6 days with two media changes. To confirm absence of endodermal carryover during mesenchyme dissection, aggregates using ES-EBs and mesenchyme from E11.5 enhanced green fluorescent protein (EGFP) mice were generated and cultured. This resulted in no EGFP expression in epithelial tubular structures, consistent with no endodermal carryover during the dissection (Supplementary Fig. S2A). Alternatively, ES-EB-expressing RFP variant, DsRed.T3 (gift of Dr. A. Nagy, Lunenfeld Research Institute, Toronto, Canada), was aggregated and cultured with E13 rat mesenchyme. Coexpression of this RFP variant with early lung endodermal marker NKX-2.1 confirmed absence of endodermal carryover (Supplementary Fig. S2B). For the inhibition studies, soluble receptor-Fc chimeras were added to mesenchyme and EBs on day 0 of culture for 1 h prior to aggregation in the following concentrations: 100 ng/mL sFGFR2c, 650 ng/mL sFGFR2b, and 500 ng/mL sNGFR (R&D Systems, Burlington, ON, Canada). These were the lowest dosages to elicit a response. On day 6, the aggregates and controls were removed from culture and analyzed. Soluble receptor neutralization experiments using FGFs were carried out in the same manner as described previously, with the preincubation of soluble receptors with either 500 ng/mL FGF2, FGF10, or FGF7 to ensure complete saturation.
Immunofluorescence
Immunofluorescence analysis was performed as described previously [32]. Details of antibodies are provided in Supplementary Table S1. Whole lungs of E16 mouse embryos were used as positive controls. Images were captured with a Leica CTRMIC 6000 confocal microscope in conjunction with a Hamamatsu C910013 spinning-disc camera (Leica Microsystems, Inc., Concord, ON, Canada).
Electron microscopy
For transmission electron microscopy (TEM), specimens were fixed in 2.5% (w/v) glutaraldehyde in 0.1 M phosphate buffer (pH 7.4) for 4 h and processed as previously described [13]. For immunogold EM, specimens were fixed in 4% (v/v) paraformaldehyde and 0.1% (v/v) glutaraldehyde in 0.1 M phosphate buffer (pH 7.4) for 4 h and processed as previously described [33]. Cryosections were incubated with primary antibodies for 1 h (Supplementary Table S1) followed by incubation with 10-nm gold-goat anti-rabbit IgG complexes (Amersham, Life Sciences, Arlington Heights, IL) for an additional hour at room temperature. Controls included omission of the primary antisera. All EM samples were examined on a JEOL JEM 1011 transmission electron microscope (JEOL USA, Peabody, MA).
Quantitative polymerase chain reaction
RNA was isolated from ESC/iPSC-mesenchyme aggregates using the Arcturus Picopure RNA isolation kit (Life Technologies, Burlington, ON, Canada) using an RNase-free DNase I treatment step (Qiagen, Toronto, ON, Canada). One microgram of total RNA was reverse transcribed with random hexamers using the Superscript III enzyme according to the manufacturer's specifications (Invitrogen). Ten micrograms of template cDNA (1 ng for 18S) was used for real-time polymerase chain reaction (PCR) using SYBR GreenER quantitative PCR (qPCR) SuperMix in conjunction with murine-specific primers sets (Supplementary Table S2). qPCR was performed using OneStepPlus qPCR (Applied Biosystems, Burlington, ON, Canada). Fold change was calculated with normalization to 18S, according to Livak and Schmittgen [34].
Immunoblotting
Immunoblotting was performed as described previously [32]. Antibodies used are listed in Supplementary Table S1.
Quantification of structures
Luminal area of the tubular structures was measured using Image J version 1.44p (National Institute of Health, Bethesda, MD). Five randomly selected nonoverlapping fields from sections obtained from four aggregates per group were counted and total luminal epithelial area per aggregate consisted of these pooled counts normalized against total field area. Each field was viewed at 200 times magnification.
Statistical analysis
Data are expressed as means±SEM. Student's unpaired t-test and one-way analysis of variance with Holm-Sidak post hoc analysis were performed using SigmaPlot version 11 software (Systat Software, San Jose, CA). P<0.05 was deemed statistically significant.
Results
Embryonic lung mesenchyme directs ESCs and iPSCs to an early distal lung phenotype
Crosstalk between the mesenchyme and the endoderm is critical for establishing cell fate and maturation in the developing lung [3]. While lung mesenchyme has been previously used to induce ESCs to a lung cell phenotype [9], its induction potential has not been tested on a pure endoderm population derived from ESCs, nor has it been attempted with iPSCs. To determine the mesenchymal signals necessary for commitment to lung, we first combined endoderm-induced EBs derived from ESCs (ES Foxa2/hCD4;Bry/GFP) or iPSCs (iPS EOS3F-29) with early distal lung mesenchyme to assess its induction capacity (Fig. 1A and Supplementary Fig. S1). Following activin A treatment, ∼50% of ES-EB and iPS-EB cultures coexpressed definitive endoderm markers CXCR4 and cKIT. Roughly half of the ES-EB cells (∼51%) also coexpressed GFP and hCD4, surrogate markers for BRY and FOXA2 in this ESC line, respectively (data not shown). To obtain a pure population of endodermal cells devoid of remaining pluripotent cells and nonendoderm lineages, ES-EBs and iPS-EBs were sorted for CXCR4 and EPCAM following activin treatment, prior to aggregation with mesenchyme (P4 population, Fig. 1B). CXCR4 is expressed in the embryonic endoderm and mesoderm [35], while EPCAM is expressed in embryonic endoderm and extraembryonic endoderm [36]. Sorted cells form endoderm cell clusters when cultured under nonadherent conditions and express foregut endodermal markers FOXA2 and SOX2 following 6 days of culture at an air–liquid interface, but do not express lung markers NKX2-1 and proSFTPC (Fig. 1C). When sorted ES-EBs and iPS-EBs were combined with early lung mesenchyme, both cell lines organized into tubular structures with the lining cells coexpressing NKX2-1 and SOX9, characteristic of the distal tip progenitors that generate alveolar epithelial lineages, reminiscent of SOX9 expression in natural E14.5 mouse lung tissue [37]. Lack of SOX2 and Club/Clara cell marker SCGB1A1 expression indicates absence of proximal airway lineage within the aggregate cultures [38] (Fig. 1C). Coexpression of proSFTPC and SOX9 provides further evidence that a distal lung progenitor population has been established [21] (Fig. 1D).
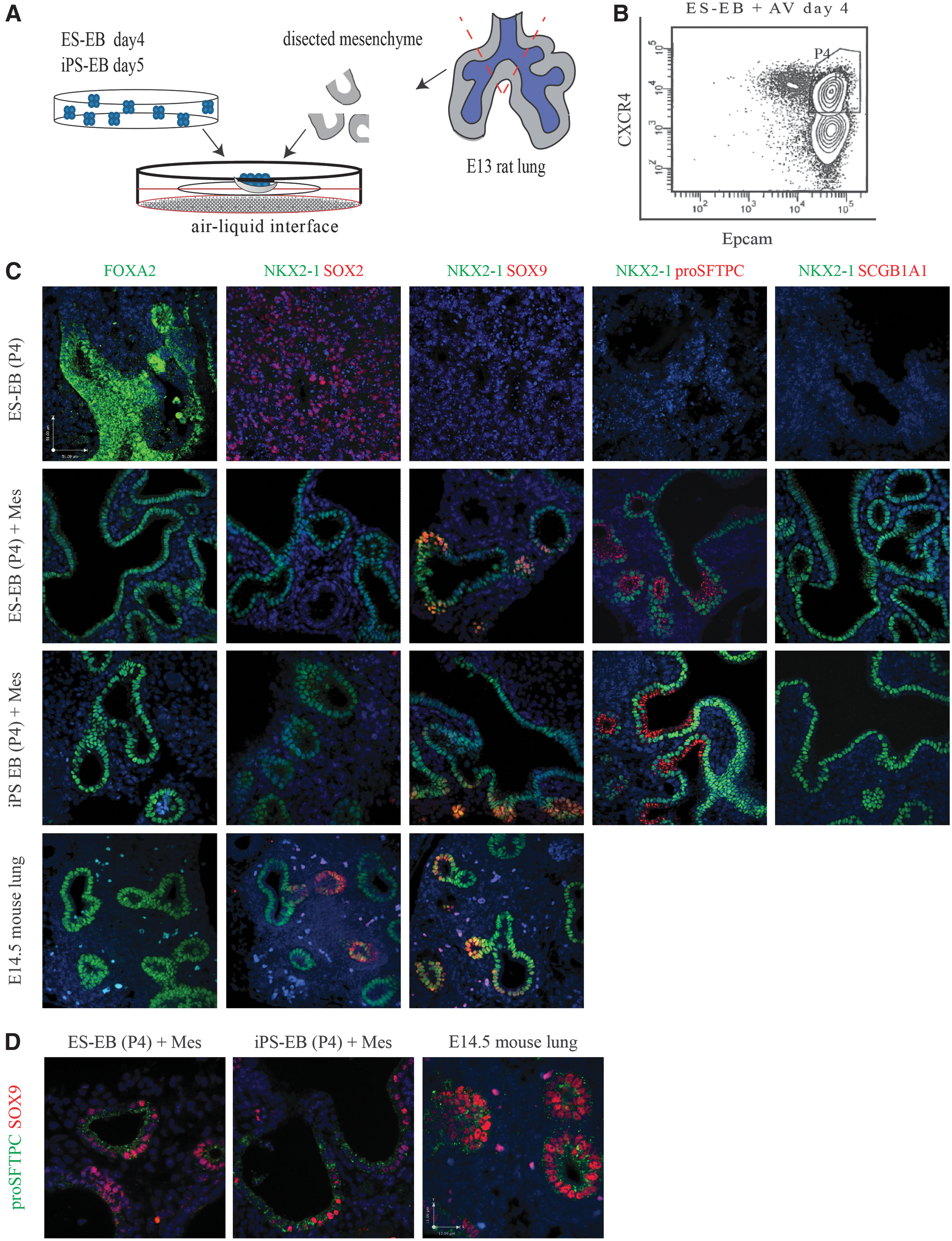
Embryonic lung mesenchyme stimulates differentiation of ESC- and iPSC-derived endoderm to an NKX2-1/proSFTPC-positive lineage.
Colocalization of Foxa2-hCD4 and NKX2-1 in the ESC-mesenchyme aggregates demonstrated that the tubular epithelial lining cells were derived from the ESC population (Supplementary Fig. S2C). In addition to the distal lung markers, the aggregate cultures were stained for keratin 5 (basal cell marker), β tubulin IV (ciliated cell marker), and Pgp9.5 (neuroepithelial cell marker), all of which are proximal lung epithelial markers [39 –41]. None were found to be present in the ESC- or iPSC-mesenchyme aggregate cultures. Positive controls included lung samples from various developmental gestations (Supplementary Fig. S3).
TEM analysis was performed to characterize the organization and differentiation of the ESCs and iPSCs within the aggregates at the ultrastructural level. Endoderm-induced cells cultured with mesenchyme oriented into tubular structures with a clear lumen, surrounded by mesenchymal cells (Fig. 2A). Cells lining the tubules appeared columnar, polarized, and with microvilli on the apical surface. Extracellular matrix was found on the basal surface of the structures, typical of basement membrane (Supplementary Fig. S4E). Many of the cells in the tubules contained large glycogen deposits (Supplementary Fig. S4D), structures reminiscent of multivesicular bodies (Fig. 2B, left panel and Supplementary Fig. S4A, B) and prelamellar bodies (Fig. 2B, right panel), characteristic features of immature distal type II epithelial cells. Immunogold labeling of these aggregates (Fig. 2C) verified that proSFTPC localized within the multivesicular structures of the epithelial lining cells, in line with reported localization of proSFTPC in maturing alveolar type II epithelial cells [42]. Immunogold labeling of TJP1 (also known as ZO-1) demonstrated the presence of tight junctions between the epithelial cells, hallmark of a functional epithelial lining (Fig. 2C). In contrast, EBs cultured without mesenchyme at an air–liquid interface contained poorly organized cells with little or no tubule formation. Many of the cells had apoptotic nuclei and displayed poor contact with one another (Fig. 2A, inset).
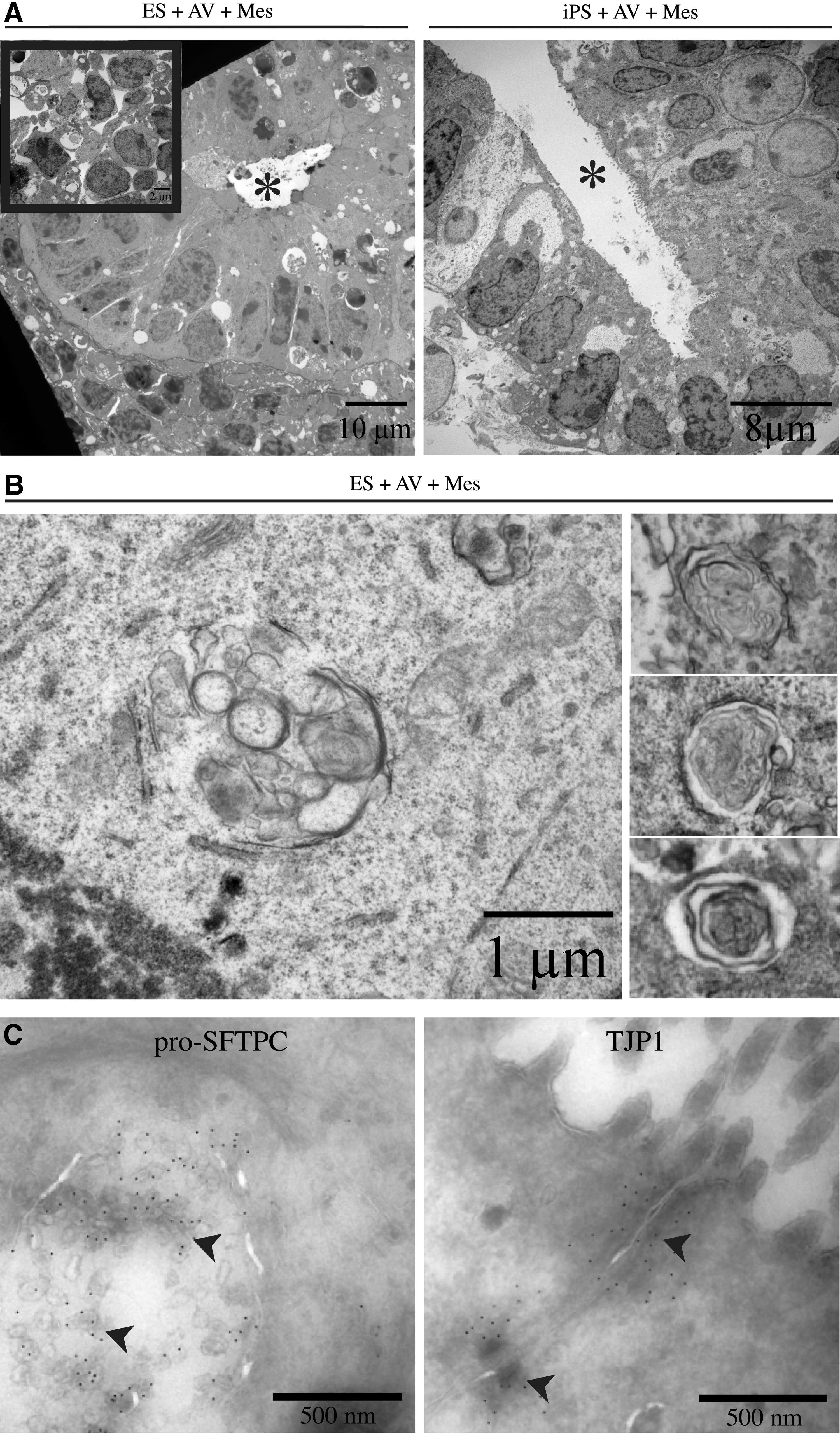
Ultrastructural detail of ESC- and iPSC-derived NKX2-1/proSFTPC-positive lineage.
Markers of other endodermal organ lineages as well as Oct4, a pluripotency marker, and Olig2, a neuronal marker that is coexpressed with Nkx2-1+ cells in the developing forebrain, were examined in the aggregates to determine whether the combination with early lung mesenchyme prevented the formation of other lineages. Real-time PCR analysis showed absence of expression of other endoderm lineage markers (Pax8 for thyroid, Pdx1 for pancreas, and Albumin for liver) in EB-mesenchyme aggregates using sorted endoderm-induced ESCs or iPSCs when compared with respective tissues or stem cell controls (Supplementary Fig. S5A). Further, sorting of iPSCs for an enriched endoderm population prior to combination with mesenchyme increased the expression of both Nkx2-1 and surfactant protein B (Sftpb), compared with nonsorted aggregate cultures. Aggregates exhibited significantly greater expression of Nkx2-1, Sftpb, and Sftpc than E13 lung controls, suggesting that they comprise a higher proportion of distal lung epithelial cells or they represent a later stage of differentiation (Supplementary Fig. S5B, C).
As a surrogate assessment of epithelial branching, we measured the luminal area of the airways and tubular structures per unit field in the developing lung (E13, E16, and E18) and EB+Mes and iPS+Mes aggregates, respectively. The measured luminal epithelial area of both ESC- and iPSC-derived mesenchyme aggregates at day 6 resembled that of E18 mouse lungs (Supplementary Fig. S5D), suggesting extensive epithelial surface expansion by tubular branching in the aggregates. Immunostaining at several time points reveals the presence of NKX2-1- and proSFTPC-positive tubular structures in day-4 aggregates resembling that of the developing lung (Supplementary Fig. S5E). The increase in number of positive tubular structures at day 6 compared with day 4 suggests ongoing branching morphogenesis with extended time culture.
While rat mesenchyme was used in the experiments represented here, these results were reproducible using mouse lung mesenchyme (Supplementary Fig. S6A, B). This underlies the conserved nature of the signaling pathways involved in early lung commitment. In addition to culturing EB alone as control, aggregates were generated using E13 rat skin mesenchyme. The absence of lung lineage differentiation in these aggregates provided evidence for the specificity in the process of lung epithelial induction by lung mesenchyme (Supplementary Fig. S6C).
Highly efficient induction of an NKX2-1+ population is achieved within the epithelial cell compartment
To test the efficiency of NKX2-1 induction by early lung mesenchyme, we used an Nkx2-1mCherry ES cell line to quantify NKX2-1+ progenitors generated in the mesenchyme aggregates [30]. These ESCs were induced as described before and sorted for dual expression of CXCR4 and EPCAM (Fig. 3A). As expected aggregation with early lung mesenchyme resulted in organization into tubular structures with lining cells coexpressing NKX2-1 and proSFTPC (Fig. 3B). To quantify the proportion of cells committed to a lung epithelial cell lineage, we first examined the population that expressed EPCAM. Following 6 days in culture, 20.85%±1.8% (mean±SE, n=7) of the total cells in the 3D aggregates were EPCAM+ by flow cytometry, retaining the initial fraction of around 25% endodermal cells used to generate the aggregates (Fig. 3C). Flow cytometry was performed for mCherry expression and was found to be 16.5%±1.6% (mean±SE, n=7) of the total cells, suggesting that the aggregation of endoderm-sorted ESCs with early lung mesenchyme had resulted in ∼80% of the EPCAM+ epithelial cells to commit to an NKX2-1+ lineage (Fig. 3C). To corroborate these findings, we repeated the experiment with Foxa2/hCD4;GFP/Bry ES cells to track the portion of EPCAM+ cells that were also expressing hCD4, a surrogate marker for FOXA2 in this cell line. Following 6 days of culture at air–liquid interface, we found 28% of the total cells in the EB-mesenchyme aggregates to coexpress EPCAM and hCD4. Since 35% of the total cell population was EPCAM+, these results demonstrate that about 80% of the epithelial cells were positive for FOXA2 (assessed by hCD4 expression) (Fig. 3D). Immunostaining of these aggregate cultures also demonstrated colocalization of EPCAM and NKX2-1 in the epithelial cells lining the tubule structures (Fig. 3E). Overall, these results demonstrated that the three-dimensional aggregation system leads to the rapid generation of organized pulmonary epithelial cells with high efficiency.
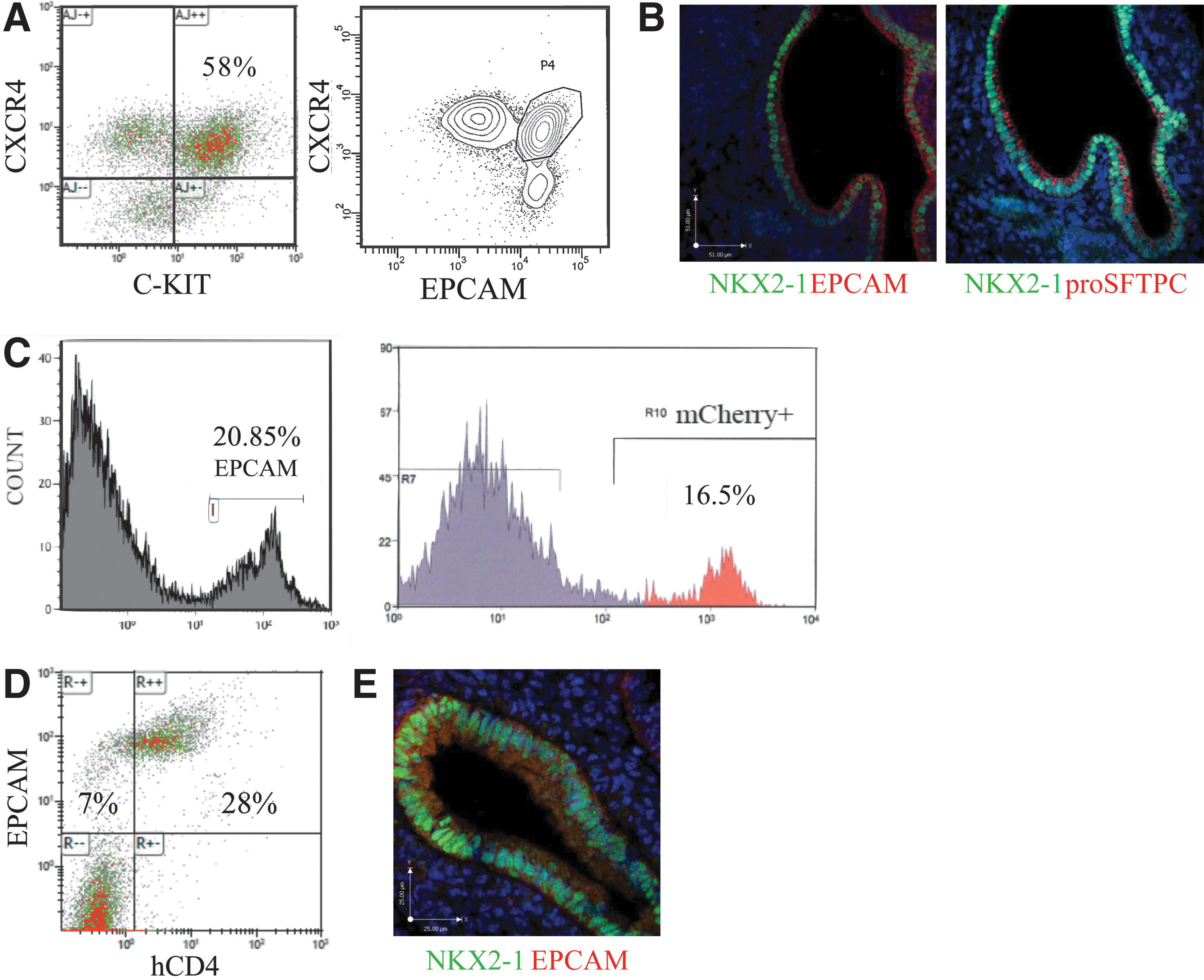
Quantification of NKX2-1mCherry-positive ESCs in aggregate cultures.
Late lung mesenchyme is unable to commit ESCs or iPSCs to a lung cell phenotype
Previously we have shown that late (E19) rat lung mesenchyme is a stronger morphogenic inducer of E13 rat lung epithelium compared with early (E13) mesenchyme [32]. To determine whether late mesenchyme has similar inductive effects on endoderm-induced ESCs and iPSCs, aggregate cultures were generated using late mesenchyme [32]. Immunostaining of these cultures showed no evidence of NKX2-1 or proSFTPC at the protein level (Fig. 4A). Recombinants, composed of E13 lung epithelium and E19 mesenchyme, were generated as previously described [32] and functioned as controls. As expected, results from these control experiments showed that combining late mesenchyme with early epithelium produces structures with saccular, lung-like morphology coexpressing NKX2-1 and proSFTPC (Fig. 4B) [32]. To elucidate the difference between early and late mesenchyme that is accountable for this contrast in induction potential of endoderm-induced ESCs and iPSCs, we examined the expression of selected FGFs implicated in lung development [1,43] between the E13 and E19 mesenchyme. Real-time PCR analysis revealed that expression of Fgf2 and Fgf10 mRNA was significantly greater in early versus late mesenchyme (Fig. 4C). Conversely, Fgf7 mRNA levels dramatically increased with advancing gestation in accordance with previously published results [44]. This suggests that FGF2 and/or FGF10 in early mesenchyme are candidate mediators of commitment to an NKX2-1+ phenotype.
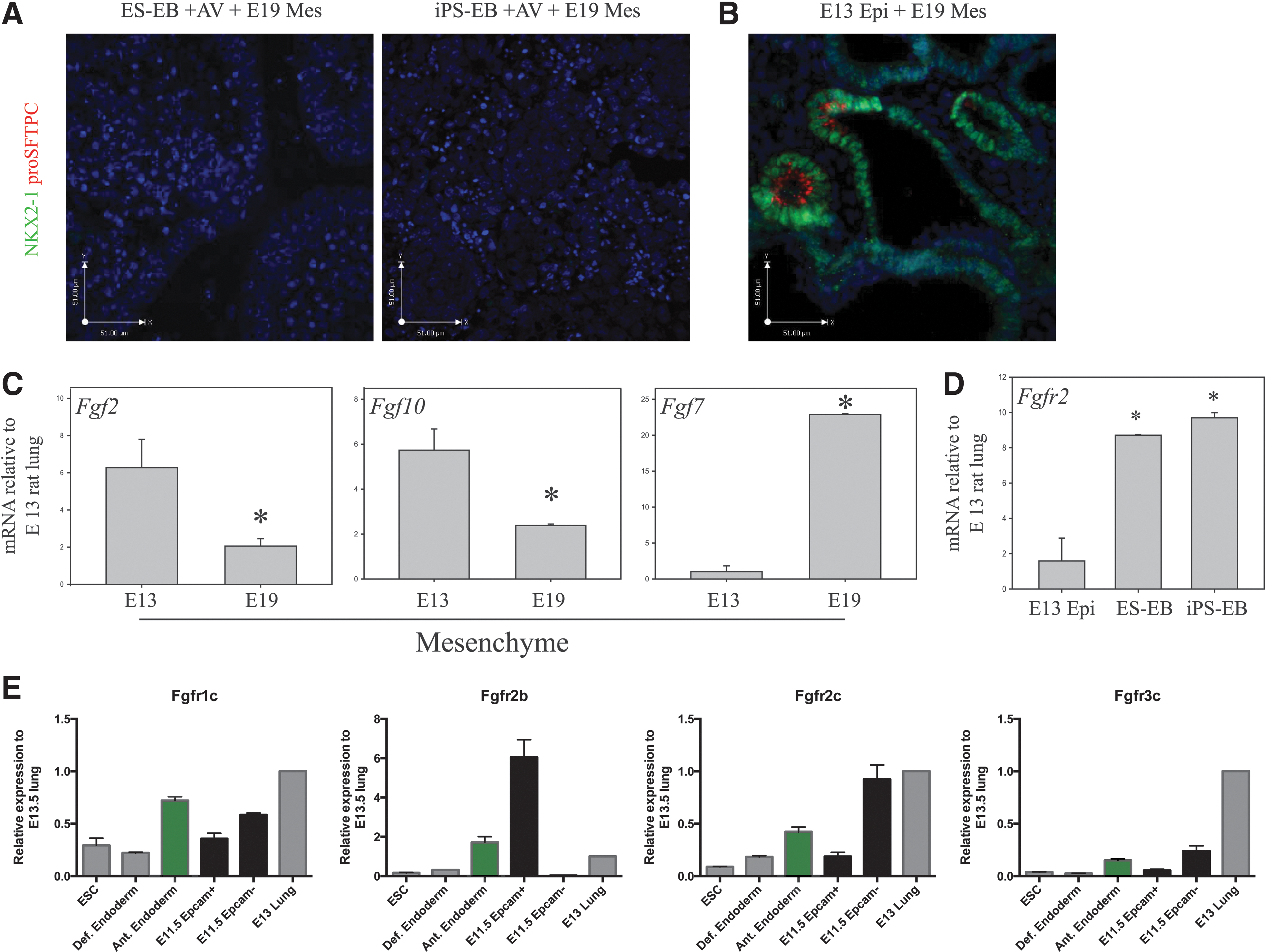
Fgf expression in early and late-gestation lung mesenchyme.
The interaction between ESCs and iPSCs with early lung mesenchyme was further characterized by evaluating whether the mesenchyme continued to develop alongside the endoderm-induced cells for the duration of aggregate culture. We observed that the level of Fgf2 mRNA in both ES and iPS aggregates peaked at day 4 and declined thereafter to baseline levels (Fig. 5A), while Fgf10 mRNA expression was highest at day 2 and declined thereafter. This expression pattern is similar to the lung mesenchyme in vivo (Fig. 4C). In contrast, Fgf7 mRNA expression increased over time in the EB-mesenchyme aggregates (Fig. 5A), a pattern that also mimics what occurs in the lung mesenchyme in vivo (Fig. 4C). Nkx2-1 mRNA expression appears to follow the expression pattern of Fgf7, as it steadily increased between days 2 and 6 in both ESC and iPSC aggregate cultures (Fig. 5B).
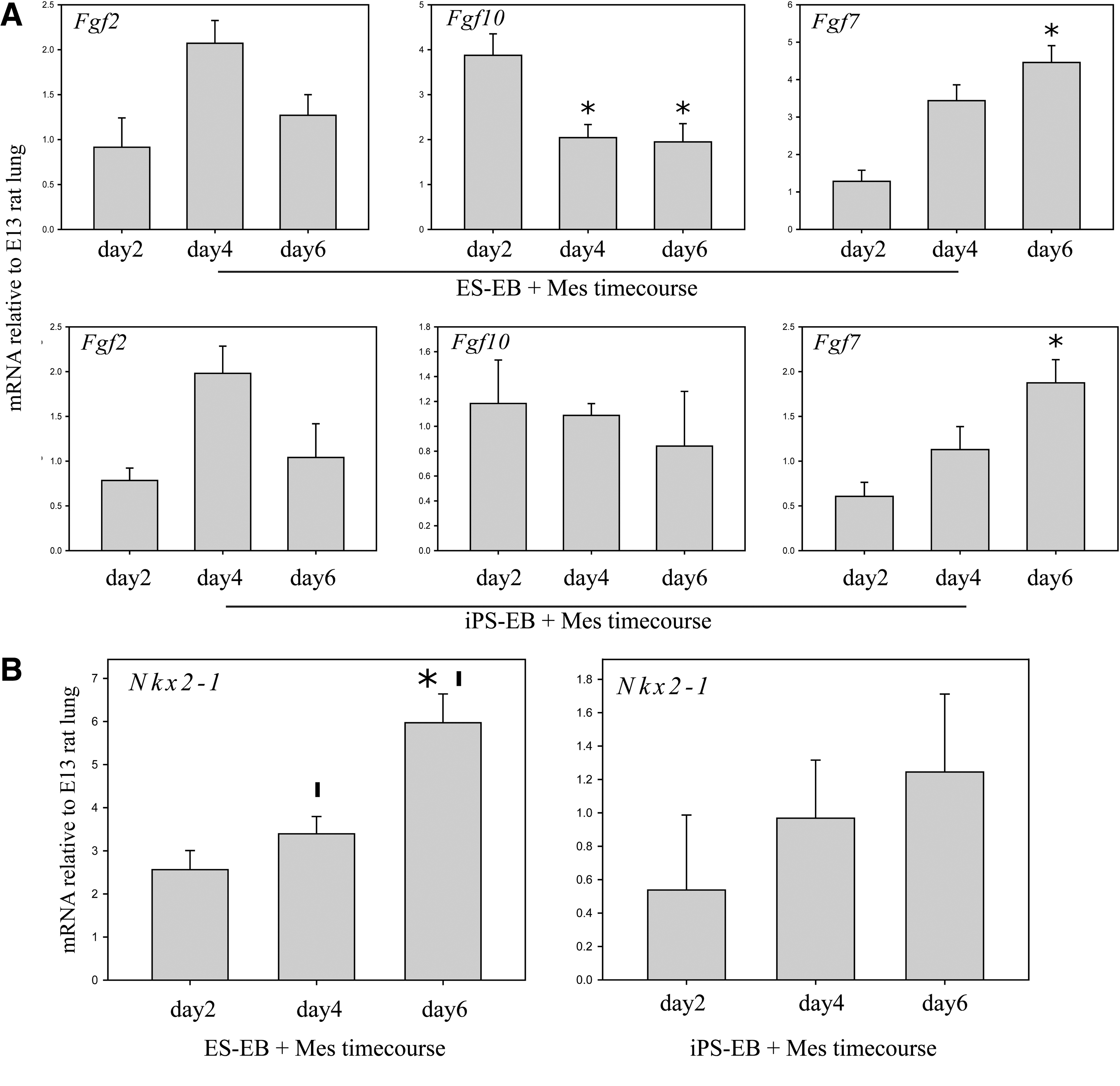
Temporal expression of Fgfs and Nkx2-1 in mesenchyme-EB aggregates. Expression was measured by qPCR of EB-mesenchyme aggregates at days 2, 4, and 6 in culture.
FGF receptor expression in endoderm-induced ESCs
Five FGF receptors have been identified to date but only the FGFR2 receptor has been shown to be crucial for lung development [45]. The FGFR2 receptor has two primary isoforms. FGFR2b is expressed primarily in the epithelium and binds mesenchymal-derived FGF7 and FGF10, which are key regulators of lung branching, proliferation, and differentiation throughout development [32,43,46]. FGFR2c is expressed in the mesenchyme and epithelial cells in early lung development [47] and binds FGF2, which is involved in early specification of endoderm and lung cell lineages [1], and FGF9, which is important for expansion of the mesenchymal and epithelial cell compartments [48]. We first confirmed the mRNA expression profile of both Fgfr2b and Fgfr2c in sorted EPCAM+ (epithelial) and EPCAM− (mesenchymal) cells of E11.5 mouse lungs (Fig. 4E); the epithelial compartment showed high expression of Fgfr2b and relatively low expression of Fgfr2c. Transcripts for Fgfr2c were detected mostly in the mesenchyme. Both definitive endoderm-induced ESCs and iPSCs expressed total Fgfr2 mRNA (Fig. 4D). Additional real-time PCR using isoform-specific primers revealed that anterior-endodermal cells derived from ESCs expressed significant levels of Fgfr2b mRNA. Expression levels of Fgfr2c and Fgfr1c were much lower while that of Fgf3c were negligible (Fig. 4E). These data suggest that anterior endoderm-induced ESCs and iPSCs are capable of responding directly to various FGF signaling, including FGF2, FGF7, FGF9, and FGF10.
FGFR2b signaling is not crucial for differentiation into early lung progenitor cells
Having confirmed Fgfr1c, Fgfr2b, and Fgfr2c expression in anterior endoderm-induced ESCs and iPSCs, and that of Fgf2, 7, and 10 in early lung mesenchyme, we next examined the contribution of these FGFs for initial commitment to NKX2-1/proSFTPC-positive cells using truncated soluble receptors. We treated the aggregates with a high concentration of truncated soluble FGFR2b (sFGFR2b) to determine whether disruption of signaling through this receptor isoform will affect differentiation. This resulted in limited differentiation to NKX2-1+/proSFTPC+ populations in small regions of the ESC- and iPSC-derived aggregates (Fig. 6A) that lacked tubular organization when compared with nontreated controls. Soluble NGFR, a structural analogue to the soluble FGF receptors, was used as a control as it is not known to play a role in distal lung epithelial differentiation. Immunoblotting provided further evidence as a weaker positive signal for both NKX2-1 and proSFTPC was detected in the aggregates treated with sFGFR2b (Fig. 6B). Treatment with truncated soluble FGFR2c (sFGFR2c) completely eliminated the NKX2-1+/proSFTPC+ populations as demonstrated by immunostaining and immunoblotting in both ESC- and iPSC-derived aggregates (Fig. 6A, B). The addition of both truncated soluble receptors together showed a similar result, with no expression of any lung cell markers and an overall lack of organization within the cultures. To test the specificity of the blocking receptor isoforms, we out-competed the soluble FGFR receptors with exogenous FGFs. Addition of FGF2, which signals through FGFR1c and FGFR2c, restored differentiation to an NKX2-1+/proSFTPC+ phenotype in aggregate cultures treated with sFGFR2c (Fig. 6C). In contrast, addition of FGF7, which has negligible binding affinity for FGFR1c and FGFR2c [49], did not reverse the negative effects of sFGFR2c on NKX2-1/proSFTPC expression (Fig. 6C). Both FGF7 and -10 bind with high affinity to FGFR2b [49] and, indeed, their addition to the aggregate cultures treated with sFGFR2b enhanced the expression of NKX2-1/proSFTPC and restored the organization into tubular structures. Together, this data suggests that the mesenchymal induction of ESC- and iPSC-derived definitive endoderm toward NKX2-1+/proSFTPC+ lung progenitor cells likely involves FGFR1c and/or FGFR2c, but not FGFR2b, signaling.
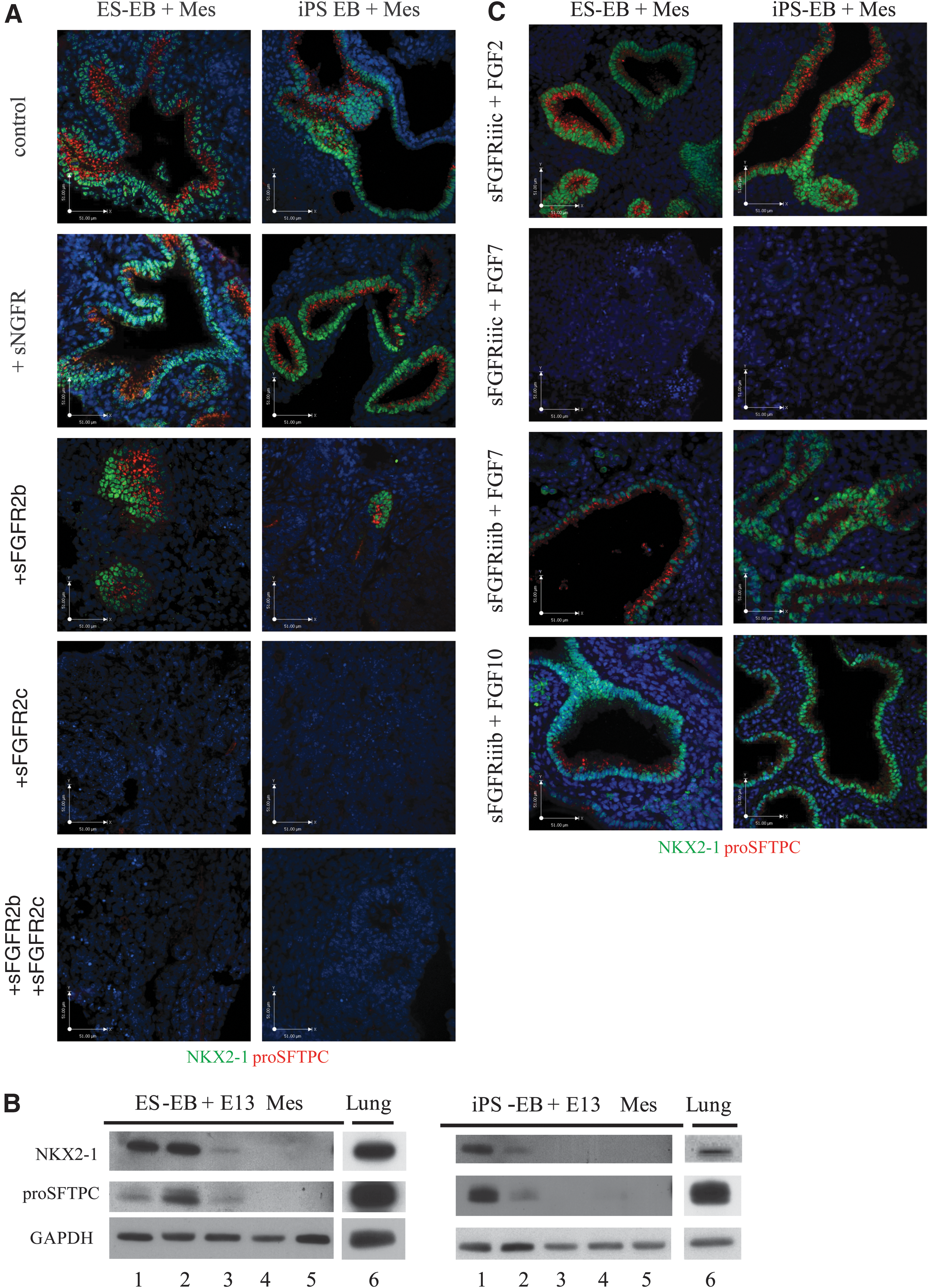
Blockage of signaling through FGFR2c, but not FGFR2b, inhibits ESC and iPSC differentiation to an NKX2-1/proSFTPC-positive lineage.
FGF2 is unable to mimic the full effects of early lung mesenchyme
Concurrent with our soluble FGFR2c findings, other studies [12,31] have demonstrated that FGF2 (a FGFR1c/FGFR2c ligand) is responsible for commitment of endoderm-induced cells to an NKX2-1+ lineage. To examine the induction capacity of FGF2 alone in our culture system, we sorted activin A-treated iPSCs and ESCs for EPCAM+/CXCR4+ cells, which were then plated at air–liquid interface on floating membranes, without mesenchyme. Sorted cell clusters cultured without any factors did not express NKX2-1 or proSFTPC, nor did the addition of FGF7 or FGF10 (alone or together) induce lung marker expression. Addition of FGF2, however, resulted in large clusters of NKX2-1+ cells, although lacking organization. Further addition of FGF7 and FGF10 after 3 days of FGF2 exposure resulted in expansion of the NKX2-1+ cell population. ProSFTPC expression was not present at any concentration of FGF2 or in combination with FGF7 and FGF10 (Fig. 7A). Only cells treated with FGF2 expressed levels of NKX2-1 (∼16%) above untreated controls quantified with flow cytometry using Nkx2-1mCherry ES cells. FGF9, which exhibits a high affinity for FGFR3c but also binds (although with lower affinity) to FGFR2c and FGFR1c [49], was unable to induce any significant levels of NKX2-1 (Fig. 7B). Overall, this provides evidence that complex FGF signaling is required to commit endoderm-induced pluripotent cells to an NKX2-1+ lineage, but is not sufficient to efficiently induce a distal lung phenotype characterized by the coexpression of NKX2-1 and proSFTPC.
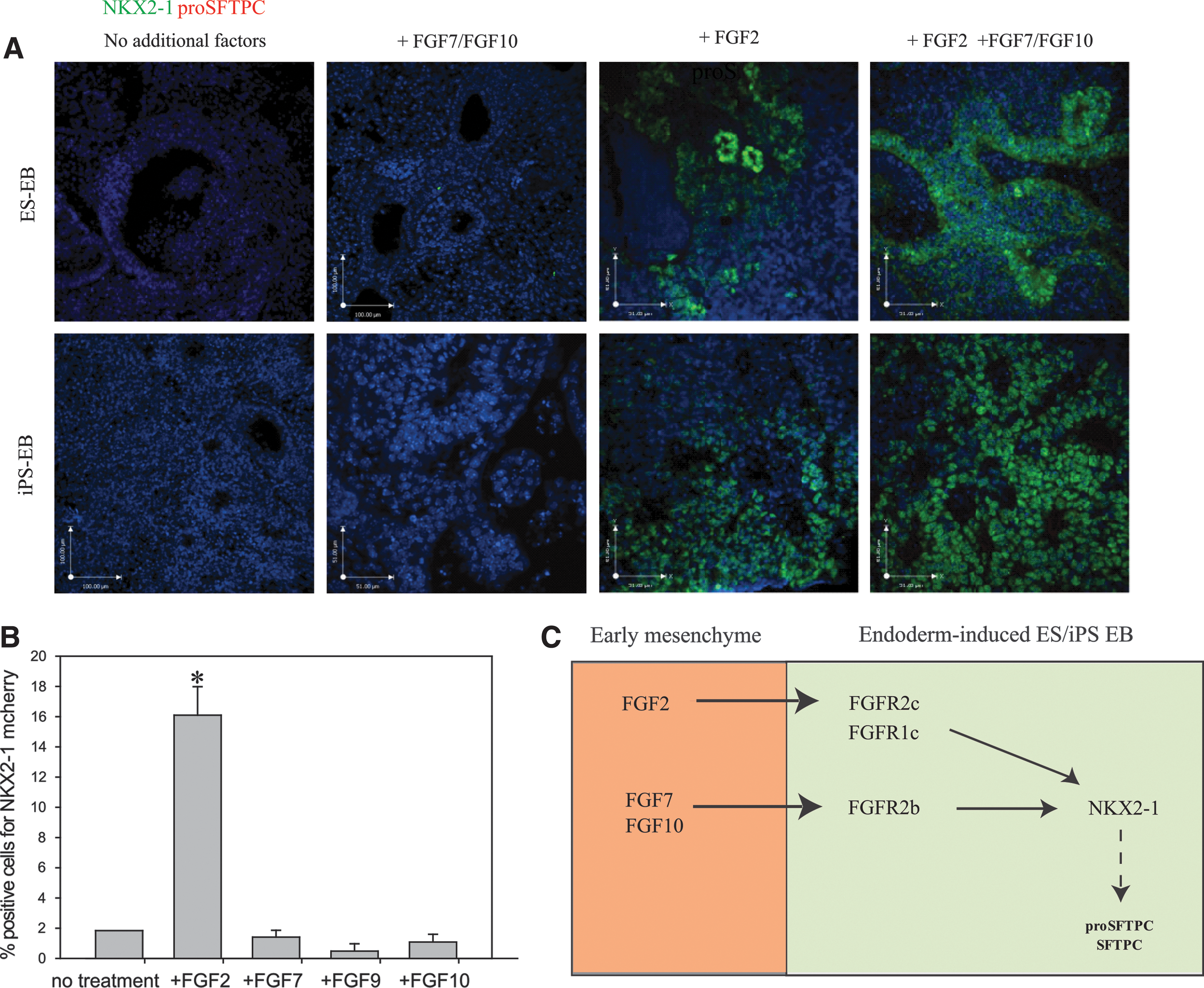
Addition of FGF2 to endoderm (EPCAM+/CXCR4+)–sorted cells induces an NKX2-1+ phenotype.
Discussion
In this study, using lung mesenchyme, we were able to rapidly induce ESCs and iPSCs to a distal lung epithelial cell phenotype with high efficiency. We built on the initial ESC work by Bishop and colleagues [9] by utilizing the known inductive capabilities of the embryonic mesenchyme [2 –4]. We first treated ESCs and iPSCs with activin A, which were then sorted to obtain a highly enriched endoderm population. The sorted cells were subsequently combined and cultured as 3D aggregates with embryonic (E13) lung mesenchyme. Within 10–11 days we were able to generate a population of polarized lung epithelial cells from ESCs and iPSCs that were ∼80% positive for NKX2-1 and largely expressing proSFTPC, making this to our knowledge, one of the most rapid and efficient inductions of pluripotent cells into distal lung epithelial progenitor cells to date [10,12,13]. We found coexpression of hCD4 and EPCAM with a separate ES cell line at similar levels. The starting ratio of endoderm-sorted stem cells to mesenchyme remained roughly 30% following 6 days in aggregate culture, despite a 5-fold increase in total cell number. This suggests that the endoderm-induced stem cells and mesenchymal cells proliferate at similar rates and the majority of the endoderm-induced stem cells retain EPCAM expression following sorting, aggregation, and culture with mesenchyme.
Surfactant protein B expression was detected in both ESCs and iPSCs containing aggregates at greater levels than E13 lung. Although Rippon et al. have examined Sftpb expression, the data were normalized to ESCs and, therefore, it is unknown how this expression relates to the early lung [13]. Huang et al. recently reported directed differentiation of human ESCs in distal lung cells with ∼50% of cells expressing SFTPB [11]. However, only 5% of the cells expressed SFTPC, the typical marker for type II cells [42], indicating that no fully differentiated alveolar epithelial phenotype was achieved. No expression of SCGB1A1 (Club/Clara cell marker) or SOX2 (proximal lung progenitor marker when coexpressed with NKX2-1) was found in any of our culture conditions, in line with previous studies that used recombinants of early distal lung mesenchyme with embryonic lung epithelium [32]. The observed colocalization of SOX9 expression (distal lung progenitor marker when coexpressed with NKX2-1) with proSFTPC and NKX2-1 underscores the distal inductive capacity of the mesenchyme [3,32]. Expression patterns of Fgfs in the EB-mesenchyme aggregate cultures mimicked those of the developing lung [43,44]. The temporal expression pattern of mesenchymal-derived Fgfs when cultured with ESC- and iPSC-derived endoderm suggests that the mesenchyme is not simply acting as a static provider of signals, but that communication in the form of crosstalk occurs between the two tissue layers in a three-dimensional setting. Interestingly, while developmental studies that used late embryonic (E19) mesenchyme found that these cells can support early embryonic epithelial cell growth and differentiation [32], late mesenchyme was unable to specify ESCs or iPSCs to an NKX2-1+ lineage. This suggests that early lung epithelium is already more lineage committed than the endoderm-induced ESCs and iPSCs and is better primed to respond to factors present in the late lung mesenchyme.
We provide evidence that signaling through FGFR1c and/or FGFR2c is critical for commitment of ESCs and iPSCs to an NKX2-1+ phenotype, and that FGF2 can initiate this event. Previous studies that used Fgfr2b-null mice, with intact FGFR2c function, show that the mice are viable until birth; however, they suffer from severe defects of the limbs, lungs, and the posterior pituitary gland [50]. In these mice, the development of the lung is initiated, as a primitive lung bud can be found, but fails to continue to develop and differentiate appropriately, eventually undergoing extensive apoptosis. This finding is consistent with our results, as signaling via FGFR2c (and/or FGFR1c) seems to be critical for the initial induction of NKX2-1 expression, whereas FGFR2b appears to play a role in organization, and further proliferation and differentiation. FGFR1c, the functionally active isoform of FGFR1 [51], is expressed throughout the epithelium and mesenchyme of the early chick lung [52], in agreement with our Fgfr1c expression findings in E11.5 EPCAM+ (epithelium) and EPCAM− (mesenchymal) cells. Fgfr1-null mice have demonstrated essential functions in embryonic development prior to early lung formation [51,53]. Although a role for FGFR1c in the developing lung mesenchymal has been demonstrated [48,54], the importance of epithelial FGFR1c is unknown.
Our observation that FGF2, but not FGF7, is able to “rescue” the NKX2.1/proSFTPC induction in 3D aggregates treated with sFGFR2c shows the specificity of the receptor isotypes and suggests that FGF2 is a key molecule responsible for initial commitment to an early lung epithelial cell phenotype. In vitro studies have shown that FGF2 signaling from the primitive foregut mesenchyme is able to induce endoderm organ formation at varying concentrations, with higher concentrations specifying lung and lower concentrations specifying pancreas and liver [1]. Further support for our findings comes from early tracheal epithelial cultures in the absence of mesenchyme that assessed the potential of various FGFs to reprogram the proximal epithelium to a distal phenotype expressing SFTPC [55]. FGF2 was able to induce an SFTPC phenotype. Studies with ESCs have shown conflicting results on the effects of FGF2 on distal lung cell differentiation with some showing that it decreases alveolar type II cell markers [56] while others have reported that it is necessary for commitment to a proSFTPC+ population [10]. Fgf2-null mice have a relatively mild phenotype [57,58], which has led to speculation that there is compensation for FGF2 functions by FGF1; however, Fgf1;Fgf2 double knockouts produce a similar mild phenotype [59]. This suggests that either other factors (including other FGFs) are taking over the roles of FGF1 and FGF2, or that they play limited role in lung development and homeostasis. When we examined the inductive capacity of FGF2 alone on endoderm-induced ESCs and iPSCs, limited patches of NKX2-1+ cells lacking proSFTPC expression were observed. The overall percentage of cells expressing NKX2-1 in these monolayer cultures was much lower than those of cells cultured as 3D aggregates with early lung mesenchyme (16% vs. 80%). This confirms that while FGF2 is important for the initial specification, it is not sufficient. Additional FGFs are required (see schematic depicted in Fig. 7C). Moreover, numerous other factors, including matrix proteins, aid in the polarization, differentiation, and maturation to mature lung epithelia [60]. Interestingly, FGF9 was unable to induce an NKX2-1 lineage in endoderm-induced ESCs. FGF9 signaling via mesenchymal FGFR1c and FGFR2c has been shown to be important for lung mesenchymal proliferation [48,61]. However, treatment of the 3D aggregates with sFGFR2c did not result in significantly smaller aggregates with less mesenchyme, suggesting that FGF9 signaling in the aggregates did not occur or was not completely abolished by sFGFR2c. Fgf9 mRNA expression increased in the 3D aggregates with duration in culture (not shown); thus, the amount of sFGFR2c used may not have inhibited FGF9 signaling. Other studies have suggested that FGF9 acts on epithelial proliferation via FGFR2b [62]; however, FGF9 addition to aggregates treated with sFGFR2b did not the overcome the antiproliferative action of sFGFR2b (not shown). While the precise binding site of the ligands to FGFRs has yet to be characterized, there is evidence that cofactors, including heparin, and the formation of heterodimers between FGFs and their receptors can alter binding specificity and activation of downstream pathways in vivo [44,63], which may explain in part why FGF2 and FGF9 did not produce the same result in the endoderm-induced pluripotent cells.
The importance of crosstalk between tissue layers for proper lung morphogenesis and differentiation is well accepted. During early lung development, the mesenchyme has been shown to be the instructive tissue layer [2 –4,22,32], and in this study we have attempted to build on this knowledge and translate it to the differentiation of ES and iPS cells. While mesenchyme has been used previously to differentiate ES cells [9], it had not been fully characterized with respect to differentiation potential or efficiency, nor has it been done using an optimized starting material. The underlying hypothesis is that early lung mesenchyme has the ability to differentiate ES and iPS cells to an early lung cell phenotype, and that this lineage commitment is due to FGF signaling. As expected, early lung mesenchyme supported the differentiation of pluripotent cells into early lung progenitor cells and this effect was mediated through the FGFR1c and/or FGFR2c receptors. Our success in differentiating mouse ESCs and iPSCs using both rat and mouse lung mesenchyme illustrates the conserved nature and importance of the FGF pathway in early cell fate decisions [64,65]. While others have attempted to differentiate ESCs [10 –12] or iPSCs [20] in a monolayer in culture, such an approach does not take into account the complexities of the cell–cell and cell–matrix interactions as well as the 3D spatial environment. Moreover, since all of the nuances of lung development have yet to be characterized, including cell intermediates and selection of the compliment of factors, their concentration and optimal timing of administration is a matter of trial and error. Aggregate cultures with mesenchyme overcome many of these issues as the appropriate factors and dosages are provided, including the matrix proteins, reciprocal signaling, and a 3D environment, which contribute to an optimal induction milieu. The rapid and efficient differentiation as well as the morphogenesis seen in this culture system demonstrates that generating lung epithelial cells from a pluripotent starting material requires a system that can temporally adapt to the needs of the differentiating cells. Our current findings highlight the need for more complex protocols that focus on matrix interactions, growth factors, and reciprocal signaling of the differentiating cells in place of archaic monolayer culture systems.
Footnotes
Acknowledgments
The authors wish to thank Dr. Ellis for supplying the iPS cells, Dr. Keller for the Foxa2/hCD4;Bry/GFP ESC, and Dr. Rossant for the Nkx2-1mcherry ESCs. FACS was performed in The SickKids-UHN Flow Cytometry Facility. This work was supported by operating grants (MOP-77751 and RMF-92088) from the Canadian Institutes for Health Research and an infrastructure grant (CSCCD) from the Canadian Foundation of Innovation. E.F. was the recipient of a CIHR Banting and Best doctoral award. M.B. received a CIHR/Canadian Lung Association/GlaxoSmithKline partnership fellowship and a fellowship from the Fonds de la Recherche en Santé du Québec. M.P. is the holder of a Canadian Research Chair in Fetal, Neonatal and Maternal Health.
Author Disclosure Statement
No competing financial interests exist.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
