Abstract
Human stem cell research represents an exceptional opportunity for regenerative medicine and the surgical reconstruction of the craniomaxillofacial complex. The correct architecture and function of the vastly diverse tissues of this important anatomical region are critical for life supportive processes, the delivery of senses, social interaction, and aesthetics. Craniomaxillofacial tissue loss is commonly associated with inflammatory responses of the surrounding tissue, significant scarring, disfigurement, and psychological sequelae as an inevitable consequence. The in vitro production of fully functional cells for skin, muscle, cartilage, bone, and neurovascular tissue formation from human stem cells, may one day provide novel materials for the reconstructive surgeon operating on patients with both hard and soft tissue deficit due to cancer, congenital disease, or trauma. However, the clinical translation of human stem cell technology, including the application of human pluripotent stem cells (hPSCs) in novel regenerative therapies, faces several hurdles that must be solved to permit safe and effective use in patients. The basic biology of hPSCs remains to be fully elucidated and concerns of tumorigenicity need to be addressed, prior to the development of cell transplantation treatments. Furthermore, functional comparison of in vitro generated tissue to their in vivo counterparts will be necessary for confirmation of maturity and suitability for application in reconstructive surgery. Here, we provide an overview of human stem cells in disease modeling, drug screening, and therapeutics, while also discussing the application of regenerative medicine for craniomaxillofacial tissue deficit and surgical reconstruction.
Introduction
H
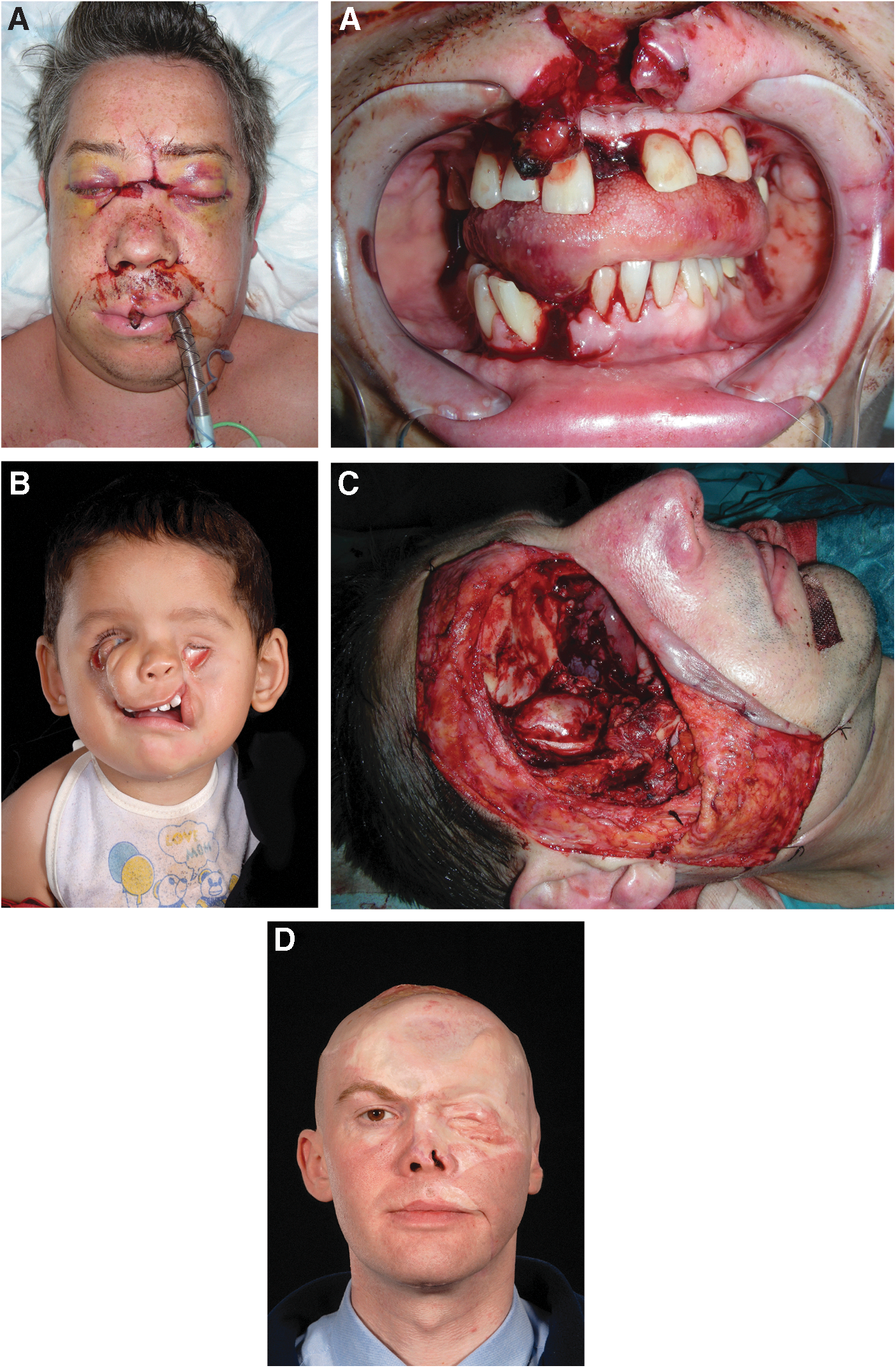
Patients with craniomaxillofacial tissue deficit.
The correct architecture and function of the vastly diverse tissues of this important anatomical region are critical for life supportive processes such as breathing and eating. The face is also central to aesthetics, facial expression, and social interaction, in addition to the delivery of senses such as sight, smell, and sound [3]. Craniomaxillofacial tissue loss is commonly associated with significant scarring, disfigurement, and psychological sequelae as an inevitable consequence [4]. Physical deformity caused by tissue deficit and scar contractures can be painful and disabling, while psychological impairment and diminished quality of life related to anxiety, depression, disruption of activities of daily living, and loss of self esteem may also ensue [5]. Physical and psychosocial implications can mean individuals are unfit for work and thus add to the financial burden of craniofacial trauma and disease such that it impacts not only healthcare systems but also society at large.
Since both maxillofacial trauma and head and neck cancer remain significant health problems, it is vital to seek new opportunities to optimize care for individuals suffering with complex craniofacial tissue loss [6 –9]. hPSCs represent an unparalleled opportunity for the development of novel tissue-regenerative therapeutics and could allow the production of infinite quantities of specific cell types for replacement of skin, muscle, cartilage, bone, and neurovascular tissues, which have been subject to congenital and acquired disease or traumatic injury.
While advances and innovation in modern day craniofacial surgery continue to improve patient outcomes, complications related to graft or flap failure, scarring, and infection remain problematic and may be overcome with the use of stem cell-derived replacement tissues. Good progress has been made over the past decade in the development of microvascular free tissue transfer and bone grafting techniques for conditions of the craniomaxillofacial complex, however, hurdles related to donor site morbidity and satisfactory restoration of form and function remain significant challenges. The difficulty lies in the diversity and intricacy of structures present in this anatomical region and our current inability to adequately restore hard and soft tissues. Patients who suffer from functional and aesthetic compromise of the craniomaxillofacial complex have at present therefore limited scope for full recovery. The unmet need for regenerative therapies for individuals with congenital anomalies and acquired craniomaxillofacial defects persists and must be addressed by the field as a priority.
Depending on the cell type of origin, hPSCs are either human embryonic stem cells (hESCs) or human induced pluripotent stem cells (hIPSCs). The former cells are derived from the inner cell mass (ICM) of the blastocyst at early embryonic development, while the latter are produced by the process of epigenetic reprogramming of somatic cells (Fig. 2). Ever since the discovery of hIPSCs by Takahashi and Yamanaka in 2006 [10 –12], the human stem cell field has been advancing at a staggering rate and this work has given increased vitality to a clinically relevant and rapidly progressive area of basic science, which holds tremendous hope for translation to patient care.
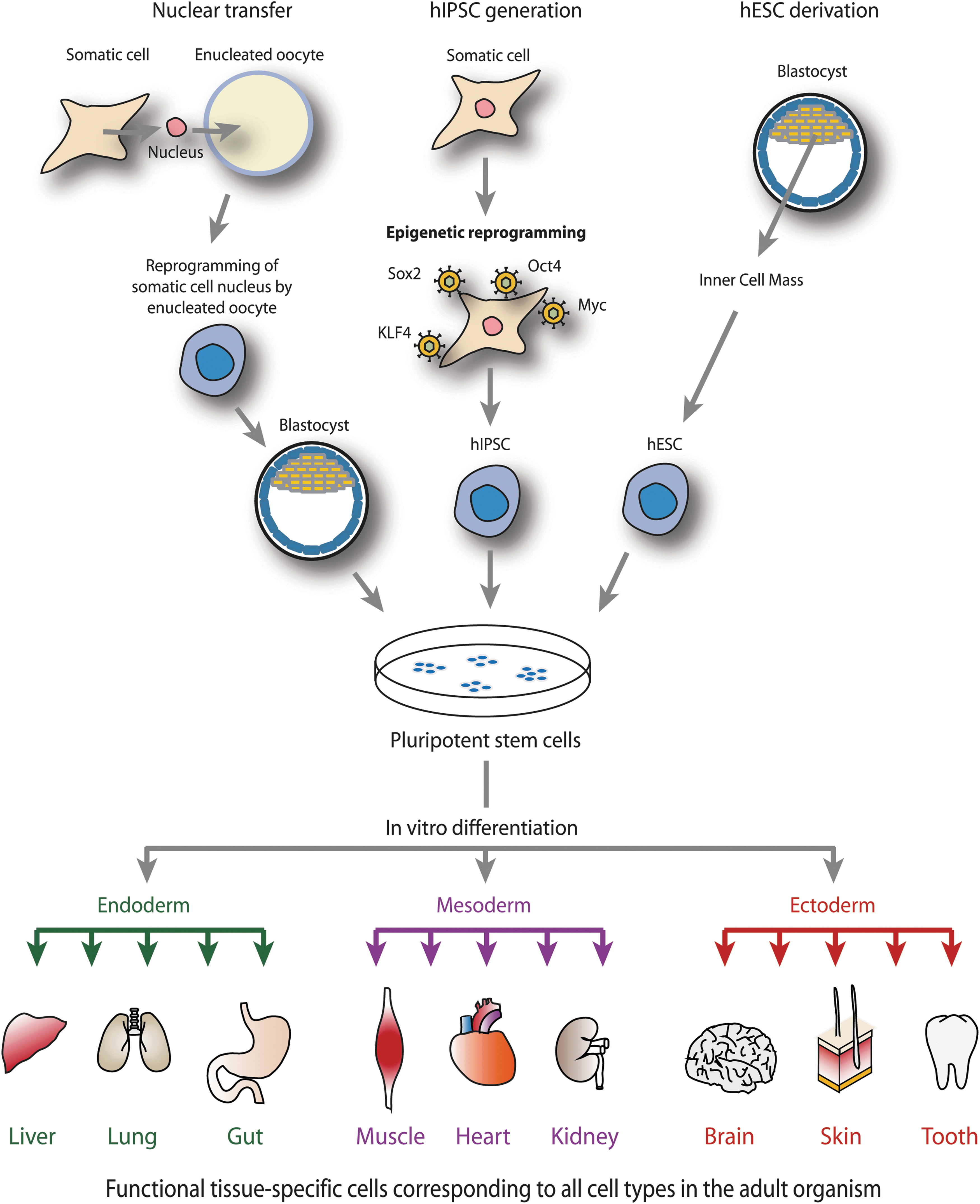
Schematic overview of human pluripotent stem cell (hPSC) derivation. hPSCs are obtained either from inner cell mass cells of the blastocyst, nuclear transfer, or epigenetic reprogramming of adult cells into induced pluripotent stem cells. Pluripotent cells can be differentiated in vitro into the three primary germ layers endoderm, mesoderm, and ectoderm, and further to functional tissue-specific cells.
Ethical and moral debate surrounding the controversial destruction of in vitro fertilized human embryos [13] for the creation of hESCs is circumvented by the utilization of hIPSCs, which can be generated from skin-derived fibroblasts or other adult cell types. Additionally, the predicted avoidance of immune response and rejection when hESCs are transplanted into unmatched individuals makes hIPSCs an appealing alternative source of human stem cells (Fig. 2). These stem cells are able to give rise to all tissues of the adult organism, including the diverse cell types in the craniomaxillofacial region.
Development of the Craniomaxillofacial Compartment
Mammalian development begins with the formation of a fertilized ovum (or zygote). Embryogenesis is the process by which a zygote undergoes rapid mitotic division (embryonic cleavage) and cellular differentiation to form an embryo. Following the formation of the morula (at the 32-cell stage), a hollow cavity is formed in the cell mass to create a blastocyst. The blastocyst consists of the ICM and the outer layer of cells known as the trophoblast, which forms the extraembryonic tissues. Following implantation of the blastocyst into the uterine lining, reorganization into a three-layered structure known as the gastrula occurs, in a process known as gastrulation. These three layers are the germ layers of endoderm, mesoderm, and ectoderm, which will give rise to all tissues of the adult [14,15]. So-called “neuralation” of the ectoderm layer produces an outer-positioned epidermis, inner neural tube (as precursor to the central nervous system), and the migratory neural crest cells [16].
The mammalian craniomaxillofacial compartment is served by cranial neural crest cells, which give rise to both mesodermal (comprising osteoblast, adipocyte, and smooth muscle) and ectodermal (comprising neuron, melanocyte, and Schwann cell) cell types [17]. Cranial neural crest cells originate at the neural tube and go on to populate the frontonasal skeleton and first, second, third, and fourth pharyngeal (or branchial) arches, forming the jaw among other structures. The neural tube goes on to divide into four regions namely the prosencephalon (forebrain), mesencephalon (midbrain), the rhombencephalon (hindbrain), and the spinal cord (Fig. 3) [18,19]. Aberrations in the tightly coordinated spatial and temporal signaling network, which orchestrates the directed migration of cranial neural crest cells or their subsequent developmental stages, can lead to craniofacial abnormalities (Fig. 1B).
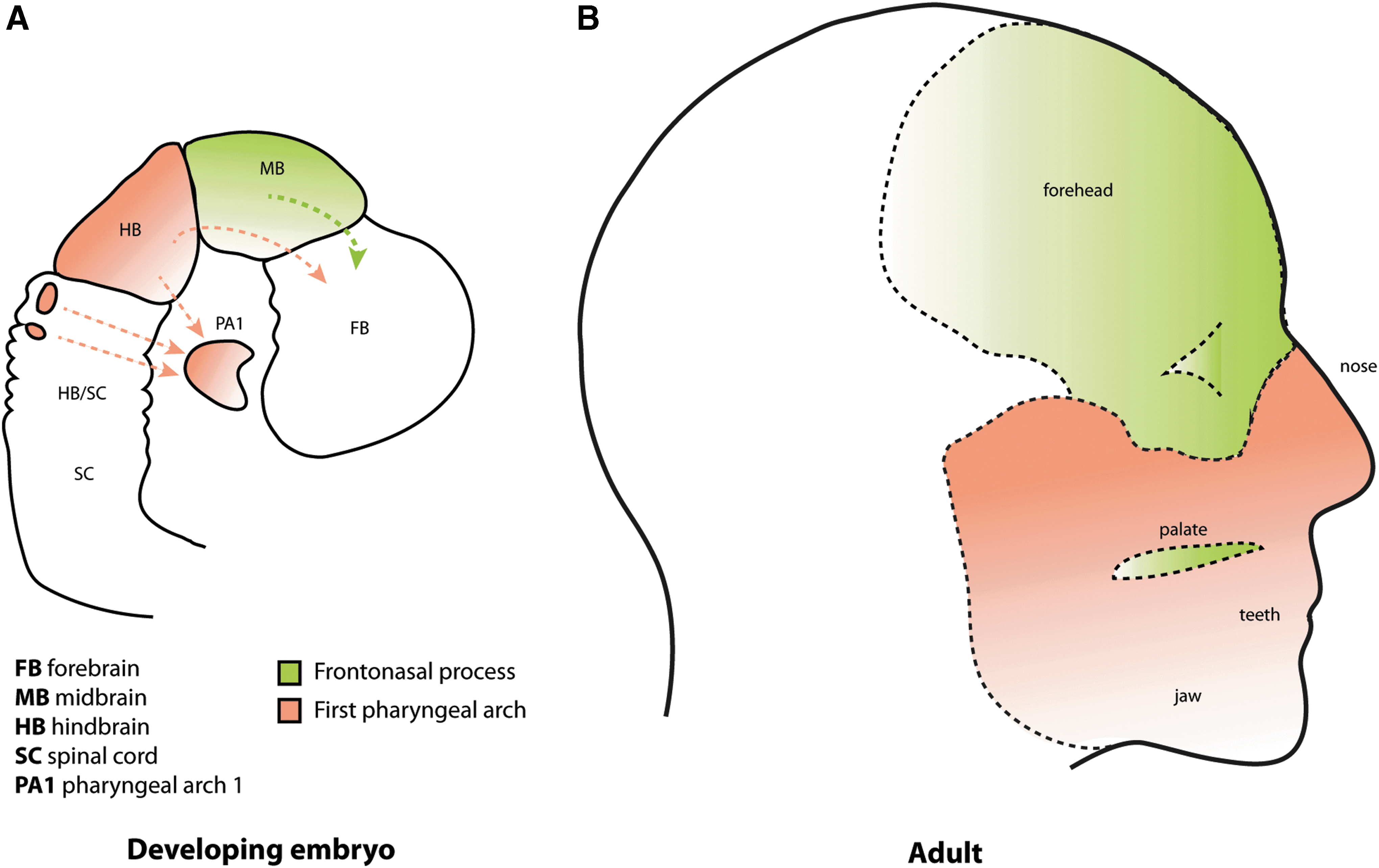
Embryonic origin of craniofacial tissues.
Obtained from the ICM, explanted developing blastocysts form embryonic stem cell colonies when allowed to proliferate in specific cell culture conditions [20]. Embryonic stem cells were first derived by Cambridge scientists Evans and Kaufman in 1981, and later that year Martin coined the term “embryonic stem cell” [21,22]. Seventeen years later, this murine work was followed by Thomson et al., who derived embryonic stem cells from human blastocysts and demonstrated their capacity of long-term proliferation in culture, and their differentiation into multiple lineages [20,23,24].
Human Pluripotent Stem Cells
hPSCs are characterized by two fundamental stem cell properties: self-renewal and pluripotency. Self-renewal is the ability of cells to proliferate indefinitely while maintaining their cellular identity [25 –28] and pluripotency is the ability of the ICM or its derivative embryonic stem cells to give rise to all cells of the embryo. However, pluripotency is distinct from totipotency because pluripotent cells cannot produce extraembryonic tissues, whereas totipotent cells such as those of the zygote can give rise to all cells of an organism, including embryonic and extra-embryonic tissues [29].
Early work on murine chimeras identified the ICM of the blastocyst as a source of pluripotent cells [30 –32] and paved the way for the isolation of mouse embryonic stem cells and their propagation in culture. The maintenance of pluripotency and the inhibition of differentiation in embryonic stem cells involves three core transcription factors, namely Oct4 (Pou5F1), Sox2, and Nanog. Oct4 is critical in the development [33] and maintenance of pluripotent cell identity [34 –36]. Sox2 acts to ensure cells maintain the correct level of Oct4 expression and regulates self-renewal [34,37]. Nanog is known to be critical for the acquisition of the pluripotent state but its loss is associated with variable effects dependent on the origin and developmental stage of the pluripotent cells [38 –41]. These three transcription factors maintain the expression of a broad range of pluripotency genes by cooperative binding to their loci [42].
Pluripotent cells can adopt either an ICM-like pluripotent state or an epithelial epiblast-like pluripotent state that corresponds to the postimplantation blastocyst [43 –47]. Since ICM-like pluripotent cells represent a developmentally earlier stage, they have a broader developmental capacity. Although it has been unclear whether human cells can be captured at an ICM-like pluripotent state in vitro, a recent study established defined culture conditions that allow hESCs and hIPSCs to adopt a pluripotent state that retains the growth characteristics, molecular circuits, chromatin landscape, and signaling pathway dependence that are similar to ICM cells [48]. This finding may be relevant for the molecular study of early lineage commitment and for applications in regenerative medicine research.
Epigenetic Reprogramming
Epigenetics is the study of how gene function and cellular phenotype is altered by changes such as chromatin reorganization, DNA methylation, and histone modification, which do not directly relate to alterations in DNA sequences. Reprogramming involves both functional and molecular changes to a cell when it undergoes cell fate alteration. Functional changes are the conversion of one differentiated cell type into another, while molecular changes comprise demethylation of DNA and remodeling of histone modifications [49 –51]. By forced expression of the transcription factors described, it is possible to reprogram somatic cells (Fig. 4).
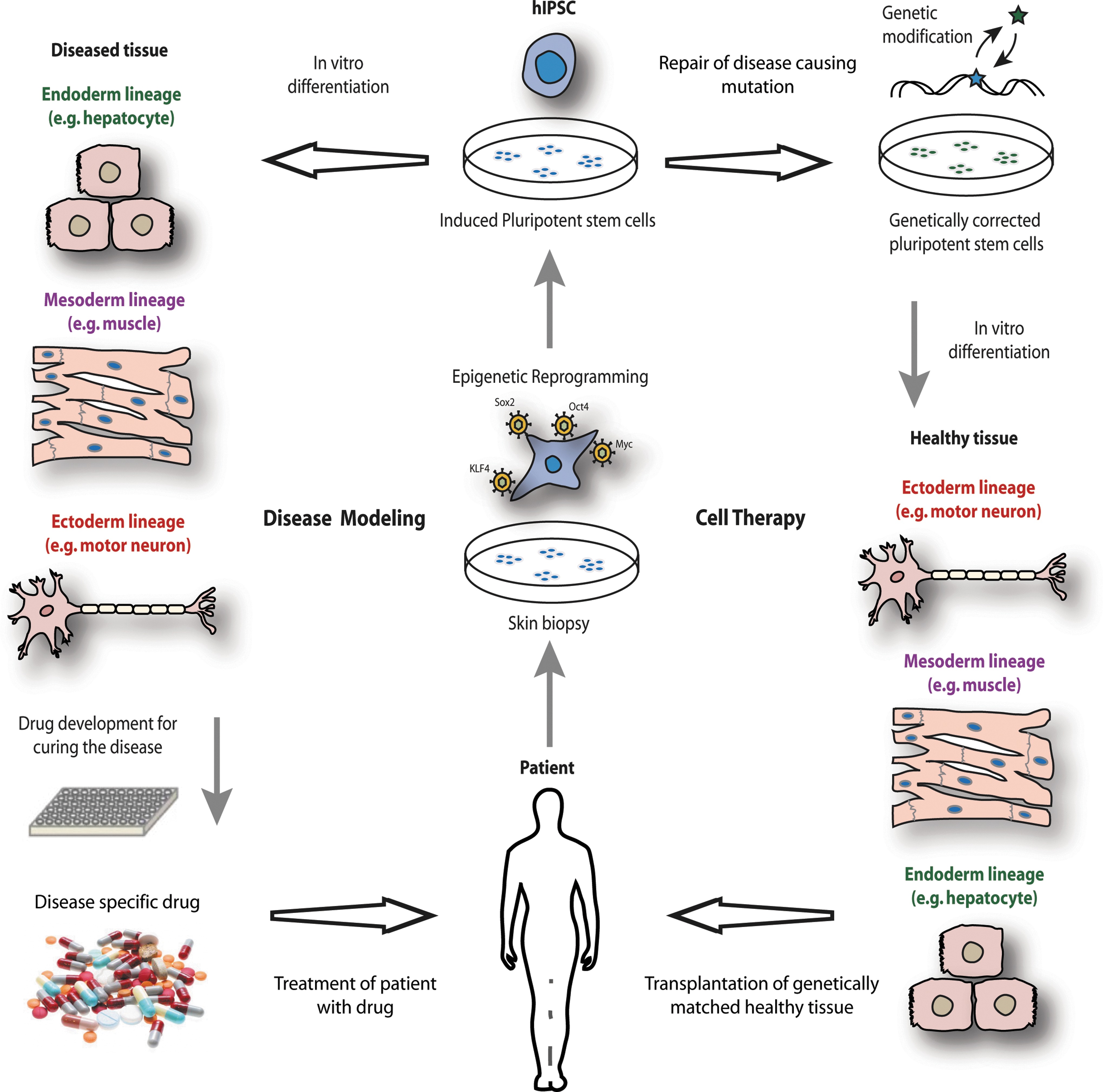
Schematic overview of the application of human induced pluripotent stem cells (hIPSCs). hIPSCs generated from patient tissue biopsy can be utilized for disease modeling or cell therapy.
It was long thought that the differentiated state was synonymous with irreversible cell fate specification and somatic cell type. In 1958, Cambridge developmental biologist Sir John Gurdon demonstrated the plasticity of the differentiated state by reprogramming somatic cells, by transfer into enucleated Xenopus oocytes [52]. This was later shown to be also possible with ectopic transcription factor expression. Induced pluripotency, first described by Takahashi and Yamanaka [10,11], is the epigenetic reprogramming of adult somatic cells to an embryonic pluripotent state by retroviral transduction of four transcription factors, namely Oct4, Sox2, Kruppel-like factor 4 (Klf4), and c-Myc. Since the initial reprogramming of human fibroblasts by the Yamanaka group it has become clear that all of the above transcription factors do not need to be present for reprogramming to occur. While Oct4, Sox2, and Nanog help to maintain pluripotency in these cells, it is also critical that lineage-specifying genes and exogenous transgenes are repressed, and this is carried out by the concurrent regulation of DNA methyltransferases and microRNAs [53,54].
Due to its stochastic nature, the method of epigenetic reprogramming has been time consuming and inefficient, producing induced pluripotent stem cells with less than 1% efficiency and requiring several weeks of gradual changes in the epigenetic landscape of the somatic cell. The utilization of specific “path-breaker” proteins such as CCAAT/enhancer-binding protein alpha (C/EBPα) make the chromatin more accessible and thus significantly speed up and improve the efficiency of epigenetic reprogramming [55]. Furthermore, depletion of the Mbd3/nucleosome remodeling and deacetylation (NuRD) repressor complex allows for epigenetic reprogramming as a deterministic process with high efficiency [56]. The generation of induced pluripotent stem cells from mouse somatic cells by small-molecule compounds [57] suggests that this will soon be achieved in human cells, thus avoiding the danger of introducing genetic aberrations. Epigenetic reprogramming has also been demonstrated by so-called stimulus-triggered acquisition of pluripotency (STAP) using transient low-pH as an external stressor to drive reprogramming. This allows the acquisition of pluripotency within a few days of exposure to such stimuli and thus speeds up the process of producing pluripotent cells [58]. Other methods of experimental reprogramming namely nuclear transfer and cell fusion are discussed elsewhere [59].
hPSCs in Disease Modeling
The utilization of hIPSCs for patient-specific disease modeling in vitro, pharmacological agent testing and obtaining further insight into human development and disease pathogenesis, is now possible. hPSCs can be utilized to replicate various diseases in vitro, with clear advantages over doing so in animals related to accurate recapitulation of human disease and varied response to pharmacologic agents compared to humans [2,60]. This approach relies on the generation of hIPSCs of a patient who is known to suffer from a particular disease, and the in vitro differentiation of hIPSCs into particular cell types that are mostly affected by the disorder. The genetic abnormalities causing the disease will therefore also be present in the derived cell types, making the investigation of the molecular mechanisms responsible for pathogenesis more accessible. The limitless supply of disease-affected cells from patient-specific hIPSCs also allows for large-scale screening of molecular compounds that aim to alleviate the pathological phenotype and speed up the process of drug development (Fig. 4).
Induced pluripotency has been used to derive cells from numerous tissue types including skin fibroblasts [11,61], pancreatic beta cells [62], hepatocytes and gastric cells [63], umbilical cord blood [64,65], and peripheral blood both directly [66] and indirectly [67]. This technology has also been applied to derive disease-specific lines for the purposes of modeling. Models of human disorders using hIPSCs have recently been demonstrated including neurodegenerative disease [68 –70], neurodevelopmental disease [71], type 1 diabetes [72], heart disease [73 –76], pancreatic cancer [77], and in our laboratory, liver disease [78]. Problems related to authentic disease replication and cell identity following reprogramming however, must be borne in mind. Concerns have been raised regarding genetic and epigenetic stability following reprogramming [79], including erosion of X chromosome inactivation [80] and changes in the copy number of cell proliferation genes, such as tumor suppressor genes [81].
While monogenic disease models with faithful recapitulation of disease phenotype have been successful, the next step for disease modeling with hIPSCs is the modeling of complex (multigenic) genetic disorders and the use of hIPSCs to investigate the role of genetic modifiers in disease severity. To achieve this we require large-scale generation of hIPSCs for banking and experimentation—such operations are ongoing in centers around the world [82].
Therapeutic Use of hPSCs
Following the restrictions placed on hESC research [13], limited numbers of patient-unmatched hESC lines were made available to scientists and thus hampered any efforts to pursue therapeutic application of these cells [83]. Nevertheless clinical trials using hESC-derived retinal pigmented epithelial cells to treat macular degeneration and macular dystrophy in two patients are ongoing [84,85], with encouraging early reports of some improvement of visual acuity in both individuals and no hyperproliferation or immunological rejection in either patient [86]. Ethical controversy and concerns over tumorigenicity [28] and immunocompatibility [87] have impeded the progress of translational application of hESCs to date.
hIPSCs hold the hope of the production of patient specific cells for cell-based therapies, for individuals with incurable disease that is currently resistant to pharmacological therapy (Fig. 4). In 2007, the application of induced pluripotency technology to a mouse model of sickle-cell anemia proved to be successful and heralded the beginning of studies employing this novel tool for disease correction in regenerative medicine [88]. Similarly, Parkinsonian symptomology was improved in a rat model of the disease [89], murine hemophilia A phenotype correction achieved [90] and cardiac contractility improved in a murine model of acute myocardial infarction [91], with hIPSC use. Our laboratory has demonstrated the ability to correct disease phenotype by targeted gene correction [92]. We have also shown the generation of cells of clinical interest in hepatocyte-like cells [93,94], as have other groups in the production of glucose-responsive insulin-secreting cells [95] and intestinal cells [96].
Tissue Reconstruction of the Craniomaxillofacial Compartment
It is anticipated that hPSCs will someday provide unlimited quantities of replacement cells for patients with craniomaxillofacial tissue loss due to a multitude of pathologies (Fig. 5). The restoration of appearance and function, the pillars on which the medical specialty of plastic and reconstructive surgery is based, may be taken to new heights with the advent of regenerative medicine in this way. It is conceivable that one day reconstructive surgeons will have the option of a plethora of tissue types derived from their patients' own cells, with which to treat individuals affected by traumatic or disease induced compromise of the craniomaxillofacial complex.
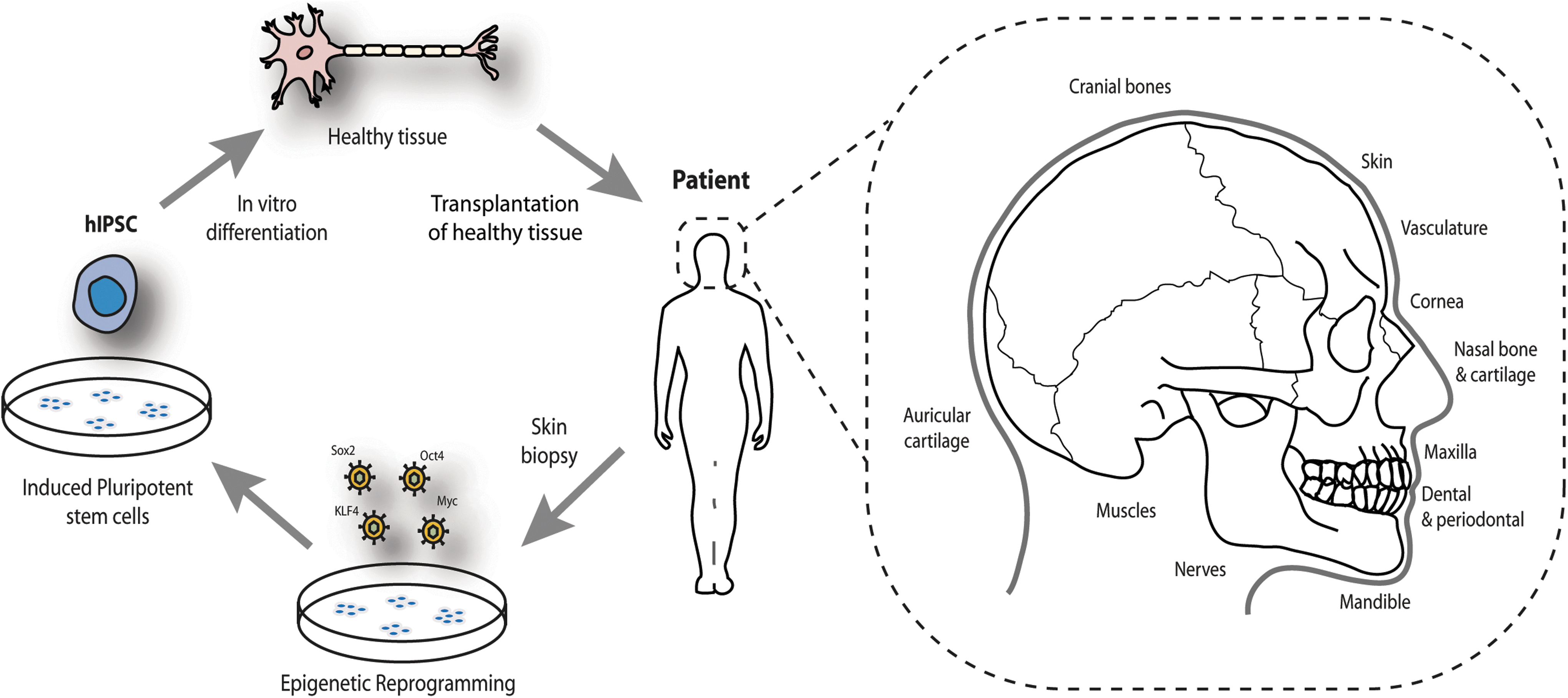
Application of patient-specific induced pluripotent stem cells for craniofacial reconstruction. Induced pluripotent stem cells can be differentiated into all cell types necessary for reconstructing the craniomaxillofacial complex.
Surgical reconstruction will be aided by the production of replacement tissues, of which bone is of crucial importance to both form and function. The replacement of soft tissues in areas of trauma or disease such as skin, muscle, and neurovascular structures has been achieved with patients' own tissues from local or distant anatomical sites, using techniques such as split or full-thickness skin grafting, local flaps, and free microneurovascular tissue transfer, for large composite tissue defects. However, complications causing flap failure (due to thrombus, hematoma, or seroma formation), scarring, infection, and donor site morbidity [97], continue to plague the outcome of many procedures. Furthermore, the replacement of hard tissue such as bone and cartilage with autologous transplantation is more challenging by virtue of their inflexible rigid structures in an anatomically complex area. Numerous bony structures in the head and face such as the cranial bones, mandible, maxilla, and zygomatic arch may be subject to trauma or disease and demand reconstruction or perhaps one day replacement with stem cell-derived therapies seeded onto biomimetic scaffolds or indeed bony templates grown in vitro [98,99]. They serve critical roles in the craniomaxillofacial complex such as load bearing, generating forces required for mastication, and protection of the central nervous system.
Currently, bone-grafts utilized can be autografts, allografts, synthetic, or xenografts. Autologous bone grafts are the preferred source of bone for craniofacial reconstruction, avoiding the risk of transmissible disease and immunological rejection, while providing a biological and metabolically active bone substitute. Sourced most commonly from the iliac crest, calvarium, fibula, scapula, or rib [100], autologous bone grafts do, however, come with complications such as donor site morbidity [97] and unavoidable limitations due to restricted availability and the necessity for prolonged hospitalization and rehabilitation of patients postoperatively. Furthermore, the achievement of good form and function following transplantation is challenging in view of craniofacial architecture.
It has therefore been the focus of translational scientists to seek new avenues for bone replacement therapy for patients with craniofacial tissue deficit. Bioactive, biodegradable acellular scaffolds, which act to recruit native cells and provide an attachment surface for bone growth, and cellular scaffolds, which are seeded with previously derived osteoblasts from an autologous source such as bone marrow, continue to be areas of interest for scientists and engineers alike [98]. An alternative lies in the use of stem cell-derived autologous bone grafts for craniofacial tissue replacement.
Mesenchymal Stem Cells for Craniomaxillofacial Reconstruction
Craniomaxillofacial tissue replacement requires tissues originating from either mesoderm-derived cells or ectoderm-derived neural crest cells during development. First described in the 1960s by Friedenstein et al. [101], mesenchymal stem cells have been shown to be a source of adipocytes, chondrocytes, and osteocytes [102], originating from the neural crest in the vertebrate embryo. Mesenchymal stem cells can be sourced from a multitude of adult and perinatal tissues [103]. The spotlight of interest has been on bone marrow-derived stem cells [104], adipose-derived stem cells [105], and dental stem cells [106,107] for reasons of osteogenic differentiation capacity and bone formation. To date, bone marrow-derived stem cells have been used most extensively for clinical applications [108].
Bone marrow-derived mesenchymal stem cells, themselves the progenitors of osteoblast cells, are obtained from marrow tissue in the medullary cavity of bone and differentiate to form muscle, cartilage, bone, and adipose tissue. Grayson et al. showed the generation of anatomically shaped bioreactor scaffolds from decellularized trabecular bone, which were then seeded with mesenchymal stem cells and allowed fully viable large bone formation, permitted by the controlled interstitial flow of nutrients and waste materials between the cells and culture medium [109]. This approach may have the ability to surmount the challenges that come with bone autografts (donor site morbidity), allografts (incompatibility and disease transmission), and the production of large anatomically accurate bone structures. Applications of mesenchymal stem cells in craniomaxillofacial reconstruction have been demonstrated for skull defects [110,111], for maxillary reconstruction using adipose-derived stem cells [112], and mandibular reconstruction using both bone marrow-derived stem cells [113] and adipose-derived stem cells [114]. The production of chondrocytes from adipose-derived stem cells has also been demonstrated [115], and differentiation into auricular cartilage [116] in animals. Adipose-derived stem cells promote wound repair by enhancing reepithelialization and angiogenesis [117,118], and also peripheral nerve repair [119] although much is yet to be uncovered about how they function. This will necessitate further clarification of their characteristics.
Mesenchymal Stem Cells from hPSCs
An alternative route to derive mesenchymal progenitor cells is via hPSCs [120 –122], which represents a theoretically inexhaustible source of cultured mesechymal stem cells, without the risks of teratoma formation on implantation. Furthermore, mesenchymal stem cells derived from hPSCs exhibit less senescence than those derived from adult stem cells [123]. While more work clearly remains to be done, the potential of hPSC-derived mesenchymal stem cells for clinical application remains indubitably attractive due to its benefits of not evoking an immune response [124].
While cell types of interest have been generated from mesenchymal stem cells, which themselves have been derived from hPSCs, cells generated directly from hPSCs have been shown for a variety of cell types and would represent a more plentiful supply of transplantable cells with faster proliferation rates compared with mesenchymal stem cells, for expansion in vitro.
hPSCs for Craniomaxillofacial Reconstruction
Both hESCs and hIPSCs have been used to derive tissues suitable for potential use in craniomaxillofacial reconstructive surgery. Replacement of bone in this anatomical region is particularly challenging, however, some progress has been made in deriving this tissue type directly from hPSCs. Since the achievement of osteogenic differentiation of hESCs in 2003 [125], there have been subsequent similar studies with [126,127] and without [128] an embryoid body intermediary step (with trilaminar germ layer formation) [129], the former resulting in more efficient osteogenesis. Bone tissue has also been generated from hIPSC-derived mesenchymal stem cells by culturing mesenchymal progenitors on osteoconductive scaffolds in bioreactors [130], although further validation for implantation in humans is necessary. The production of cartilage from hESCs [131,132] (summarized in Toh et al. [133]) and hIPSCs [134] has been demonstrated, however, any differences between that derived from hIPSCs and hESCs remain to be fully investigated. Encouraging work by Toh et al. showed the repair of osteochondral defects using rodent models, which closely resembled that of native cartilaginous structures many months after engraftment [135].
Considerable progress has also been made with the derivation of human vascular smooth muscle and endothelial cells from hPSCs, including the generation of functional vascular endothelium [136,137] and vascular grafts [138,139]. A platform for proof of principle experimentation with functional smooth muscle cells of specific embryological origin is described in [140,141]. Skeletal muscle cells have been developed from hESCs [142,143] and implanted in rodent models demonstrating engraftment with existing muscle tissue. In addition, keratinocytes [144], and three-dimensional skin equivalents [145] have been generated from hIPSCs. These skin equivalent tissues may be used in future as a three-dimensional structure for implanting vasculature, muscle tissue, and neural tissue concomitantly. While the majority of neural applications from hPSCs have been for central nervous system disorders and neurodegenerative disease, both sensory [146] and motor [147] neurons have successfully been derived from hIPSCs. Furthermore, the production of clinical (Good Manufacturing Practice) grade neurons has also been achieved [148], which will be a necessary criterion for further applications in reconstructive surgery. Meanwhile, it is important to note that the field of hPSC biology continues to broaden [149] and fundamental characteristics of these cells require further elucidation, prior to safe use in patients.
Safety Concerns and Current Barriers for hPSC Clinical Translation
The translation of hPSC technology for human benefit faces several hurdles that need to be solved. Two main challenges are the tumorigenic capacity of hPSCs and the generation of functional cell types. Initial concerns regarding reprogramming with genome-integrating viruses have now been resolved with the advent of virus/vector free hIPSC technology [150 –152]. However, hPSCs share cellular and molecular phenotypes with cancer cells and their defining properties carry with them the risk of contamination by hIPSCs on transplantation [28,153 –155].
As such their inherent traits render them potentially tumorigenic and thus essentially too dangerous for therapeutic use, with our current level of understanding. Tumorigenicity remains a major problem, which must be overcome if we are to realize the potential of therapeutic hPSC use. The formation of tumors from pluripotent stem cells is well documented [28,156,157]; indeed the current functional gold standard for the identification of hIPSCs is their ability to form teratomas in immunodeficient mice [158]. It is also true that hIPSCs carry the risk of transferring any genetic or epigenetic abnormalities due to the reprogramming process or originating from the somatic cells from which the hIPSCs were derived. Indeed a donor-derived brain tumor following neural stem cell transplantation has been described [159]. There have been recent studies that attempt to reduce the tumorigenicity of hIPSCs [160,161]. The efficiency of the reprogramming process is another important hurdle for hPSC translation, although there has been recent progress on this front [55,56,162 –164].
One of the major advantages of utilizing hIPSCs is that they are “self” cells and consequently overcome the challenges associated with donor grafts and tissues such as immune rejection and lifelong immunosuppression, as seen with the advent of face and limb transplantation [165]. Reassuringly, the immunogenicity of hIPSCs has been described as negligible [124,166].
Conclusions and Perspectives in Human Stem Cells for Craniomaxillofacial Surgery
Human stem cells provide an unrivaled opportunity for scientific discovery and innovation in healthcare. The challenges faced by craniomaxillofacial reconstructive surgeons revolve around the replacement of major tissue loss in an anatomically complex area of multiple tissue types. Although research in regenerative medicine holds the potential for translation to clinical practice, the biology of hPSCs and in vitro derived cell types and tissues for the craniomaxillofacial region must be further elucidated, to permit safe use in patients. Despite this, the future remains bright for stem cell use in craniomaxillofacial reconstructive surgery and well-deserved excitement continues to encircle this rapidly progressive field of science and translational medicine.
Footnotes
Acknowledgments
We thank Professor Roger Pedersen for helpful discussions and proofreading of the article. M.J. is the recipient of a Cambridge Cancer Centre Clinical Research Training Fellowship award for doctoral (PhD) study undertaken at Emmanuel College, Cambridge. S.P. is funded by a Federation of European Biochemical Societies (FEBS) Long-Term Fellowship. L.V. is supported by a Medical Research Council (MRC) Senior Non-Clinical Fellowship award and the National Institute for Health Research (NIHR) Cambridge Biomedical Research Centre.
Patient Consent for Medical Photography
Informed written consent to publish patient photographs (in print and online) was obtained from all persons (and from parents or legal guardians for minors).
Author Disclosure Statement
The authors declare no competing financial interests.
