Abstract
Evidence has accumulated that both murine and human adult tissues contain early-development stem cells with a broader differentiation potential than other adult monopotent stem cells. These cells, being pluripotent or multipotent, exist at different levels of specification and most likely represent overlapping populations of cells that, depending on the isolation strategy, ex vivo expansion protocol, and markers employed for their identification, have been given different names. In this review, we will discuss a population of very small embryonic-like stem cells (VSELs) in the context of other stem cells that express pluripotent/multipotent markers isolated from adult tissues as well as review the most current, validated working criteria on how to properly identify and isolate these very rare cells. VSELs have been successfully purified in several laboratories; however, a few have failed to isolate them, which has raised some unnecessary controversy in the field. Therefore, in this short review, we will address the most important reasons that some investigators have experienced problems in isolating these very rare cells and discuss some still unresolved challenges which should be overcome before these cells can be widely employed in the clinic.
Introduction
T
Since a VSEL-specific marker has not yet been identified, these developmentally early cells are currently isolated by a multiparameter sorting strategy employing a cocktail of antibodies and proper gating [17,19 –22]. Both murine and human purified VSELs express several early-development markers, including Oct-4, Nanog, SSEA-1 (mouse), and SSEA-4 (human) [1,8,23] and are highly quiescent [24,25]. Despite their small size, similar morphological features, and phenotype, VSELs are, to some degree, heterogenous. In particular, cDNA libraries created from purified, double-sorted VSELs (20 cells/library) revealed that these cells, while having a characteristic morphology, small size, and overlapping molecular signature, still differ slightly in the expression of some genes [26]. The best method for assessing the quality of sorted VSELs is the combination of ImageStream analysis, which enables the identification of real, nucleated cell events and excludes those from cell debris, with 7AAD staining, which excludes cells that become damaged during the sorting procedure [19,27].
The best-characterized VSELs at the molecular level using microarray analysis are murine BM-derived VSELs [23,26,28] and small SSEA-4+ cells corresponding to murine VSELs isolated from human gonads [14,29,30]. Therefore, more work is needed to characterize molecular signature of VSELs isolated from other murine organs (eg, brain, heart, and skeletal muscles) and, in particular, the phenotypically corresponding populations of human VSELs in BM, UCB, and mPB. The crucial question to ask is whether VSELs are precommitted to monopotent tissue committed stem cells (TCSCs) in the tissues of their residence.
In this short editorial review, we will address the current validated working criteria for how to properly isolate these rare small cells. This is an important issue, because, as was recently observed, by changing the well-established isolation protocols, an incorrect population of cells was isolated and misidentified as VSELs [31 –33]. We will also discuss the relationship of VSELs to other potential pluripotent stem cells (PSCs) and multipotent stem cells (MPSCs), which have been isolated from adult tissues by several other investigators. We noticed that very often cells which correspond to VSELs are given different names [5,6,14,34 –38]. Finally, we will also try to justify why we named these cells “embryonic-like,” despite the fact that they are isolated from adult tissues.
Multiparameter Sorting of VSELs–How to Avoid Sorting the Wrong Cell Populations
The current validated strategy for the isolation of cells enriched for VSELs from BM, UCB, or mPB is based on multiparameter sorting of viable small cellular events. Murine VSELs can be isolated as small Sca-1+Lin−CD45− cells, and this approach has been successfully employed not only by us but also by other independent groups [9,39 –41] who followed our detailed sorting protocols [17,19,20,22]. As reported, the highly quiescent populations of VSELs sorted from murine BM in appropriate experimental settings may give rise to hematopoietic stem cells (HSCs) [42], mesenchymal stem cells (MSCs) [40], lung alveolar type II pneumocytes [39], cardiomyocytes [43,44], and gametes and VSELs isolated from rat BM have been shown to give rise to cardiomyocytes and endothelial cells [45]. Moreover, murine VSELs may support the development of stroma in growing tumors [46]. In parallel, human VSELs have been already shown to become specified into HSCs [47] and MSCs [41].
The detailed protocol on how to sort VSELs by FACS has been described in detail in a chapter in Current Protocols in Cytometry [19]. Unfortunately, a few other groups significantly modified this protocol, which, instead of VSELs, resulted in an incorrect population of sorted cells [31 –33]. This has already been the subject of an extensive analysis that we recently published [21]. However, here, we will address some of the most important issues on how to avoid such mistakes in the future.
The major concern with the work of some groups which reported negative data [31 –33] is that, despite their claims, they did not follow our published protocol for isolating VSELs. However, we have to admit that these are not trivial sorting strategies, and failure to isolate these rare cells occasionally happens even in our experience in VSELs sorting hands.
The most critical step in VSEL sorting is proper setting of the gate to exclude contamination by cell debris and small erythroblasts. Enrichment in undesired objects may have a critical impact on subsequent genetic and in vitro functional characterization of these cells. Of great importance, despite the presence of anti-glycophorin antibodies (anti-Ter119) employed in the lineage cocktail to deplete murine lineage-positive cells [19], if the wrong gating strategy is employed, it may result in unwanted enrichment of the sorted cells for erythroblasts [32]. In brief, one of the groups [32] failed to isolate murine VSELs because of (i) setting up an enlarged input gate on the FSC versus SSC plot that included granulocytes and apoptotic cells resulted in enrichment of sorted fractions with artifacts, which were reflected in their further analyses (eg, Annexin V binding) and excluded some of the critical very small objects from further sorting; (ii) additional loss of very small objects by their exclusion of VSELs by gating for “singlets”; (iii) employing some selection markers that are unproved as VSEL markers (eg, c-kit); and (iv) focusing on some populations and discarding other fractions (potentially containing VSELs) based on results such as Annexin V binding. For instance, the entire fraction of CD45−/Lin−/Sca-1+/c-kit−/KDR− cells was excluded by these authors from further sorting [32], because it was deemed “apoptotic,” while it most likely contained not only real Annexin V+ FSClow SSCdim/hi apoptotic objects, but, in addition, VSELs. Importantly, we have reported [48] that healthy normal cells (including VSELs and HSCs) may bind Annexin V after lysis of RBCs due to microvesicle/microparticle release and posphatydylserine transfer to the membranes of the normal cells. Thus, not all Annexin V+ objects should be interpreted as “apoptotic,” as these may represent normal, functional cells [48]. Figure 1 shows a simulation of sorting strategy employed by one of the groups that failed to sort VSELs [32] to demonstrate how important it is to set a proper gate during sorting of murine BM-residing VSELs because of the possibility of contamination by small CD45-negative erythroblasts.
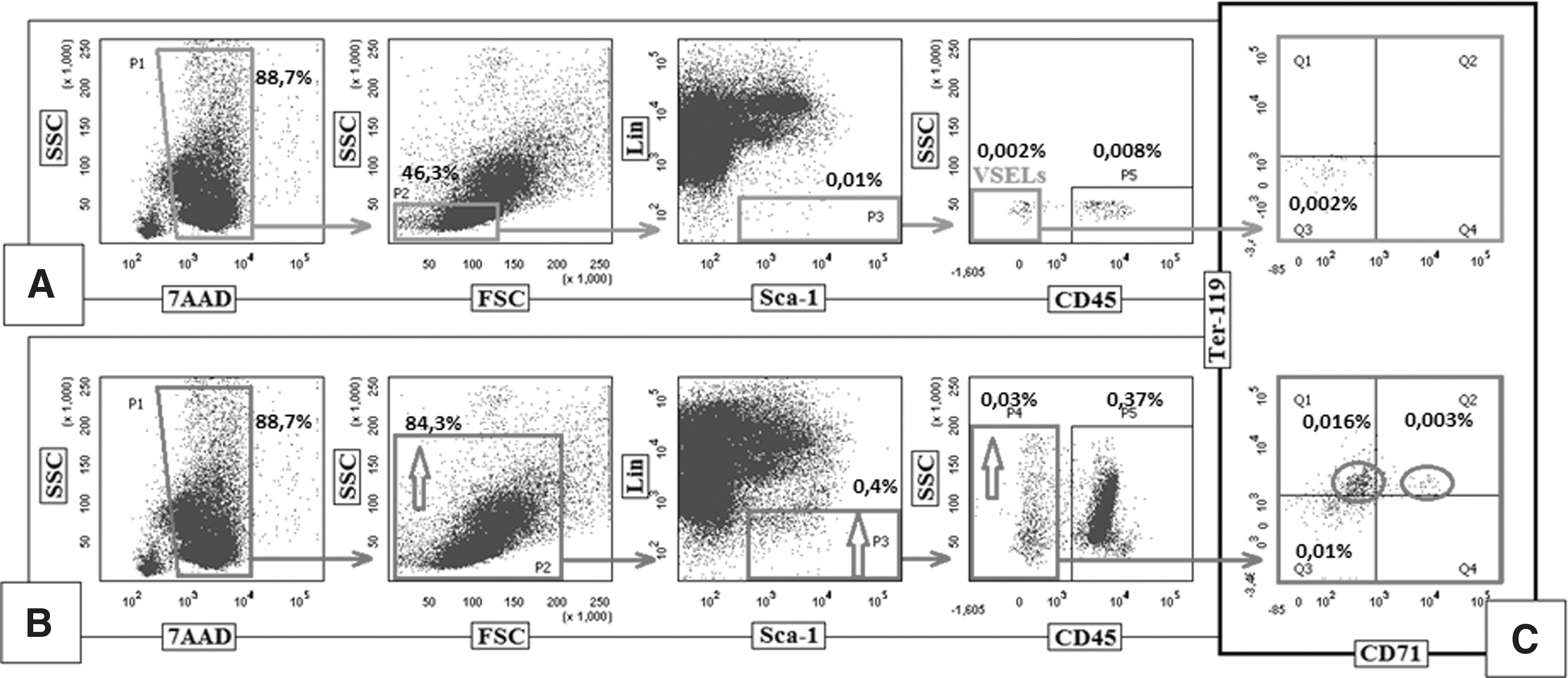
Comparison of a correct and incorrect gating strategy for murine BM very small embryonic-like stem cells (VSELs).
Unfortunately, the second group [33] also did not avoid several major technical mistakes that could result both in VSEL loss/exclusion and dilution with extraneous objects. The reason for this was (i) expanding the gating for VSEL isolation to include extraneous objects (including Lindim and CD45dim cells), resulting in VSEL dilution and enrichment in erythroblasts and (ii) relying on inexact Syto-16 staining as the main indicator of “VSEL candidates,” which, in fact, are an incorrect, non-VSEL fraction [33]. Together, these approaches led to both VSEL dilution and loss and had a critical effect on the subsequent gene expression and functional analysis of these cells.
In contrast to murine VSELs, phenotypically corresponding human VSELs are isolated as a population of small CD133+Lin−CD45− cells. We reported that expression of CD133 antigen is, so far, the most important positive marker of human early-development VSELs [49]. However, human VSELs may also co-express CXCR4, CD34, and SSEA-4, the rarest population of CD133+Lin−CD45− cells is highly enriched for small Oct-4+ VSELs [8]. If a sorted population of human cells does not express CD133, it cannot be considered to be enriched for VSELs [31]. This can be observed in one report where [31] an FACS-sorted population of CXCR4+Lin−CD45− cells that lacked expression of CD133 was mistakenly identified as UCB-derived VSELs [31]. Furthermore, this group not only replaced a reliable anti-CD133 antibody clone with one that is less effective for CD133 detection, but, in addition, “proved” the absence of a CD133+Lin−CD45− population of VSELs in UCB by employing histograms in their paper instead of dot plots [31]. It is well known that since VSELs are very rare, dot plot analysis is required to show the presence of these cells. Figure 2 demonstrates an example of the analysis of UCB-derived CD133+lin−CD45− cells by histogram as was performed by this group [31] versus a dot plot, as they should be presented. Thus, based on histogram data, this group [31] isolated an incorrect population of cells for further analysis, and what is even more interesting, failed to realize that, in fact, on the original histogram curves in their paper, small “teeth” corresponding to rare VSELs are visible, and not displaying their data in a dot plot [31].
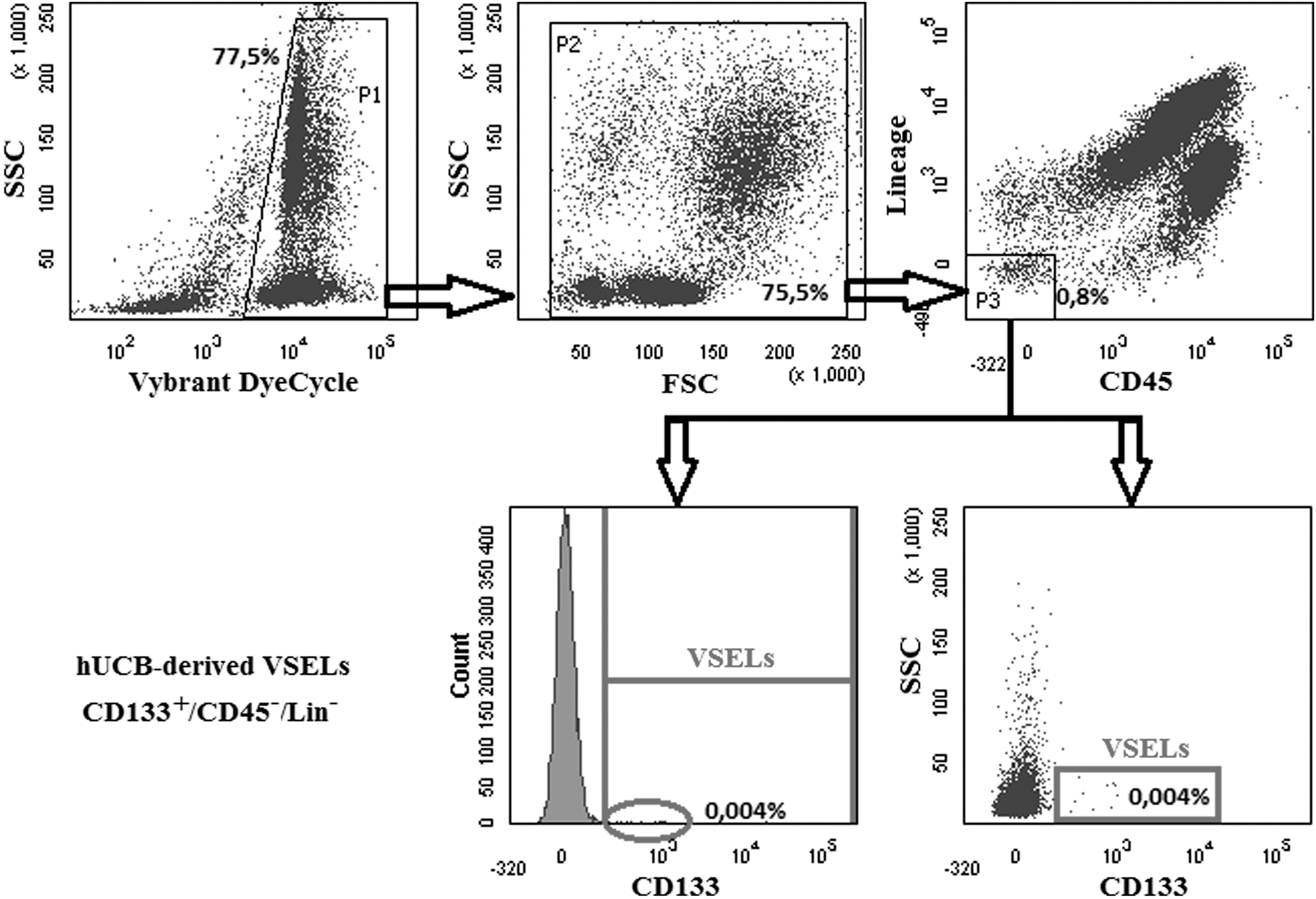
FACS analysis of umbilical cord blood (UCB) CD133+Lin−CD45− VSELs. UCB VSELs are very rare cells, and their content varies, sometimes significantly, between UCB units. Upper panel–P1–shows DNA positive events shown in P2 as SSC versus FSC dot plot. P3–includes Lin− CD45− cells. These human UCB Lin−CD45− cells were subsequently evaluated by FACS for the expression of CD133 antigen. The lower left panel illustrates an analysis for the presence of CD133+ cells in cells from P1/P2/P3 by employing a histogram, and the right panel is a visualization of these rare cells by dot plot. Danova-Alt et al. [31], in their recent studies, concluded that CD133+CD45−Lin− cells as well as CD34+CD45−Lin− cells do not exist in UCB. In fact, one of the reasons that these rare stem cell populations were overlooked by Danova-Alt and colleagues is they employed histograms instead of dot plot cytograms [31].
VSELs Express Several PSCs Markers and Exhibit the In Vitro Criteria of PSCs But Do Not Complete Blastocyst Development and Do Not Grow Teratomas
Several stringent in vitro and in vivo criteria for defining stem cells as PSCs have been proposed by embryologists who are working with embryonic stem cells (ESCs) isolated from embryos or induced PSCs (iPSCs), and we reviewed these criteria in our recent publications [50,51].
In brief, recent experimental data showed that murine BM VSELs fulfill all the in vitro criteria expected for PSCs. Specifically, they possess the primitive morphology of early-development cells (high nuclear/cytoplasmic ration, the presence of euchromatin in nuclei, and a few mitochondria) and express markers typical of PSCs (eg, Oct-4, Nanog, and Rex-1). More importantly, we recently demonstrated the presence of an open-type chromatin in the Oct-4 promoter in murine BM VSELs. Specifically, molecular analysis revealed its hypomethylation and association with transcription-permissive histones (indicated by a high H3Ac/H3K9me2 ratio) [24]. Moreover, the promoter of another core transcription factor, Nanog, despite having a higher level of methylation in VSELs (∼50%) than the Oct-4 promoter, is also transcriptionally active and has a high H3Ac/H3K9me2 ratio that favors transcription [24]. Based on these results, we conclude that murine VSELs truly express both Oct-4 and Nanog [24]. With regard to the other in vitro criteria of pluripotency, murine VSELs also possess bivalent domains in promoters that encode developmentally important homeobox-containing transcription factors, such as Sox21, Nkx2.2, Dlx1, Lbx1h, Hlxb9, Pax5, and HoxA3 [26]. Furthermore, VSELs derived from female mice reactivate the X-chromosome [51]. Finally, we and other groups have succeeded in differentiating VSELs in vitro into cells of all three germ layers [4,36,39,40,42,52].
Nevertheless, in contrast to pluripotent ESCs and iPSCs, murine VSELs do not complete blastocyst development and do not grow teratomas in immunodeficient mice [24,50]. This discrepancy between in vitro and in vivo pluripotency criteria can be explained by epigenetic changes in the expression of some paternally imprinted genes [24] that govern quiescence of these rare cells. Specifically, VSELs, similar to primordial germ cells (PGCs), erase imprinting in regulatory regions for paternally imprinted genes at the Igf2-H19 and RasGRF1 loci and increase imprinting at some regulatory regions for maternally imprinted genes such as Igf2R and KCNQ1. Thus, murine BM VSELs, by epigenetic modulation of imprinted genes (Igf2-H19, RasGRF1, and IGF2R) that play an important a role in IIS, remain resistant to stimulation by insulin, IGF-1, and IGF-2 and to additional modulation of expression of the KCNQ1 locus, which regulates expression of the cell cycle inhibitor p57Kip2. As a result of this resistance, VSELs remain in adult tissues as a population of highly quiescent cells [24], and, therefore, these epigenetic changes explain their lack of ex vivo expansion.
Considering the in vivo criteria of stem cell pluripotentiality, one has to consider that while all these in vivo criteria apply very well to ESCs and iPSCs [53,54], they are not always applicable for other pluripotent stem cells such as epiblast stem cells (EpiSCs) [55 –58] or PGCs [59 –61]. In particular, PGCs, by changes in expression of imprinted genes, remain quiescent, do not proliferate, do not complement blastocyst development, and do not form teratomas [62 –65]. However, they may be converted by appropriate manipulation to embryonic germ cells (EGCs), but this requires appropriate changes in the expression of the imprinted genes mentioned earlier [24,66]. This well-known fact has also important practical implications. As previously reported, since murine BM VSELs express several markers of migrating PGCs and undergo similar (but not identical) epigenetic changes as PGCs in the expression of imprinted genes [24], it might be possible by manipulating the expression of imprinted genes to reprogram them to a proliferation-permissive state and expand them ex vivo for therapeutic purposes. This is one of the current challenges in our laboratory.
In Table 1, we have summarized the most important characteristics of murine and human VSELs.
The best characterized so far at molecular level are murine VSELs purified from BM. More work is needed to compare VSELs isolated from other tissues with BM-purified ones.
Recent data reported on VSELs-like cells isolated from human ovaries indicate their molecular similarity to murine BM-purified VSELs [14,15,18,68,117,118].
VSEL, very small embryonic-like stem cells; UCB, umbilical cord blood; BM, bone marrow.
Why “Small Embryonic-Like” Stem Cells?
The name “embryonic” has been proposed historically, based initially on the morphology of these cells and transmission electron microscope images that revealed a similar chromatin structure as the chromatin in ESCs [3,67]. Furthermore, molecular analysis of gene expression performed later revealed that VSELs express not only Oct-4, Nanog, and Rex-1 but, in addition, also several markers characteristic of EpiSCs and epiblast-derived migratory PGCs [23,28]. We are aware that the name “embryonic-like” may create some confusion, in particular, when size and morphology of VSELs are compared with that of established immortalized ESC lines. However, one should take into consideration that stem cells in the preimplantation blastocyst or epiblast are very small and, in addition, we have to consider that quiescent cells residing in adult tissues have reduced cytoplasm and a high nuclear/cytoplasmic ratio.
In fact, several investigators have described populations of very small stem cells in adult murine [15,34 –37,45,68], and most likely, some of these cell populations that possess broader differentiation potential across germ layers could be related to the VSELs (Table 2).
The Biological Function of VSELs in BM and Other Adult Tissues
We and others postulate that in BM, VSELs are a dormant population of stem cells which serve as precursors for long-term repopulating HSCs (LT-HSCs) [50,69,70] and MSCs [40,41]. By contrast, VSELs residing in the gonads give rise to gametes [15,68,71]. However, more studies are required to see whether VSELs in other organs play a similar role as precursors for monopotent TCSCs. Evidence has accumulated that VSELs are activated and mobilized into PB during tissue or organ injuries in murine and human models of heart infarct [10,44,72], stroke [11], skin burns [13], colitis ulcerosa [12], and tumor expansion [46] and in several of these pathologic situations, the number of VSELs circulating in peripheral blood may be of some prognostic value.
The number of VSELs in adult murine tissues reportedly decreases with age [73 –75], and the number of these cells also correlates with lifespan in mice. Mice that live longer (eg, Laron and Ames dwarfs) maintain higher numbers of these cells in BM [73]. Preliminary not published data also indicate a positive effect of physical activity and calorie restriction in maintaining a high number of VSELs in adult murine tissues.
Based on this evidence, VSELs could be a back-up population of dormant stem cells in adult tissues that plays, on the one hand, an important role in organ regeneration during tissue injuries, and on the other hand, is involved in rejuvenation of the tissues. Since the number of these cells decreases with the age [73 –75], this could help explain both the impaired regeneration and tissue rejuvenation observed in older individuals.
What Is the Rationale for Why PSCs/MPSCs Reside in Adult Tissues?
A decade ago, the concept of stem cell plasticity was proposed, based on the assumption that adult monopotent TCSCs (eg, HSCs) may trans-dedifferentiate into cells from other germ layers (eg, neural cells) [76 –80]. This concept is currently rejected by a majority of the scientific community, and alternative explanations for the “phenomenon of stem cell plasticity” have been proposed, such as cell fusion [81 –83] or the presence of heterogeneous populations of stem cells, for example, in BM, UCB, or mPB, including some rare stem cells endowed with broader differentiation potential [1,5,6,36,47,84,85]. Moreover, in parallel, cumulative evidence from several laboratories shows that in the adult tissues may reside cells which express some early development embryonic markers [5,6,30,86 –95], and some of these cells may even possess germ line potential [96 –103]. Some of these intriguing cells are listed in Table 3. It is important to emphasize that murine BM-purified VSELs are enriched as mentioned earlier in cells which express several markers characteristic for migrating PGCs [23].
In support of the presence of early development stem cells in postnatal life, several types of putative PSCs or MPSCs have been described and isolated, primarily from hematopoietic tissues and that are able to give rise to cells from more than one germ layer [36,87,104 –109]. These cells were isolated by employing various strategies, such as ex vivo expansion of partially purified immunomagnetic- or FACS-based sorted cells [35,36,39,45,87,104,107,108]. Nevertheless, in most of the expansion cultures, those rare cells that were able to initiate expansions and cross germ-layer commitment were not characterized at the single-cell level [78,87,105,106,108], and in most of these cases, the phenotype of the putative stem cell with PSC/MPSC properties was described “post factum,” after phenotyping clones of already differentiated, in vitro-expanded cells [87,104,108,110]. Nevertheless, many of the investigators would agree that if early-development stem cells endowed with broader differentiation potential reside in adult tissues, they are probably closely related and exist at different levels of tissue specification. Most likely, they represent overlapping populations of early-development stem cells that, depending on isolation strategy, ex vivo expansion protocol, and markers employed for their identification, have been given different names [87,104,105,107,108,110 –112]. Figure 3 shows a hypothetical relationship to other populations of multi/pluripotent stem cells described in adult BM, PB, and UCB such as, for example, Multipotent Adult Stem Cells (MASC) [104], Multilineage-differentiating stress-enduring cells (Muse) [110,111], MSCs [112], Multipotent Adult Progenitor Cells [106], Unrestricted Somatic Stem Cells [107], Marrow-Isolated Adult Multilineage-Inducible Cells [108], and Multipotent Progenitor Cells [87,104].
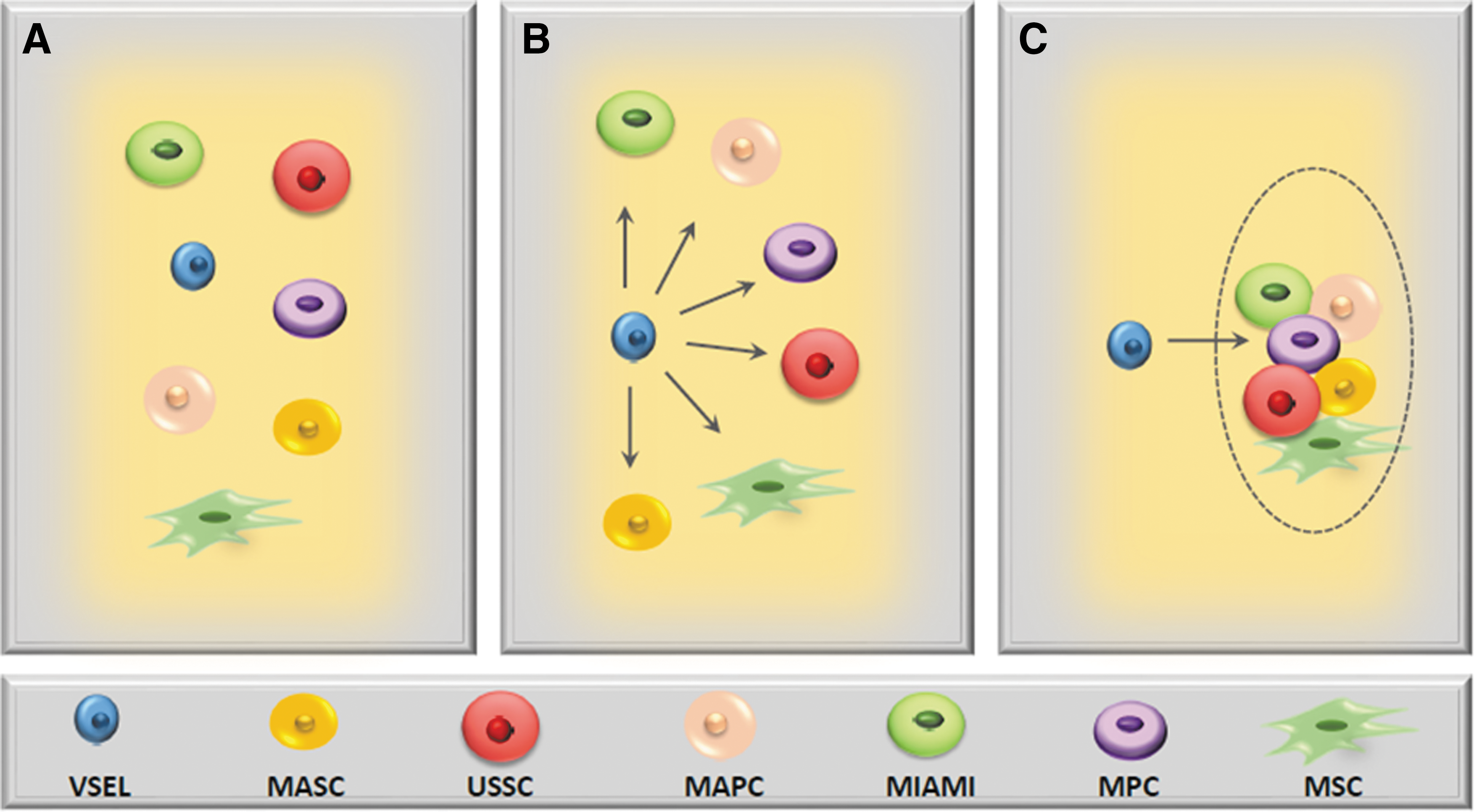
Hypothetical relation of VSELs to other multi/pluripotent stem cells identified in adult bone marrow (BM), peripheral blood (PB), and UCB.
Overall, the presence of PSCs/MPSCs in adult tissues can be explained by the possibility that early, during embryogenesis, not all of the earliest-development stem cells disappear from the embryo after giving rise to TCSCs, but some may survive in developing organs as “a dormant back-up population of more primitive stem cells” [113]. These cells could give rise to monopotent TCSCs and, thus, be involved in tissue/organ rejuvenation and in organ regeneration after organ injury. In support of this notion, evidence has accumulated that adult murine tissues, in fact, contain, in addition to rapidly proliferating stem cells, a back-up population of more primitive dormant stem cells [3,4]. We have proposed as mentioned earlier that these most-primitive dormant stem cells are kept quiescent in adult tissues by changes in the expression of imprinted genes which mostly regulate insulin/insulin like growth factor-1 and -2 signaling (IIS) [24,66]. Interestingly, our proposal that the most-primitive stem cells in adult tissues which give rise to BM LT-HSCs [42,47] follow this mechanism has been recently somehow confirmed by another group [114]. Thus, we envision that VSELs, or stem cells very closely related to them, could fulfill the criteria for such dormant stem cells in adult tissues [113]. This, however, requires further experimental evidence, in particular for the most-primitive stem cells residing in other extra-hematopoietic tissues.
Future Directions
Despite significant progress in the field, there are still many problems with VSELs that should be solved. First, most of the data has been generated so far in murine BM-derived VSELs. We do not know whether phenotypically similar VSELs residing in other murine organs are regulated in the same way. Second, we are aware that we may be purifying a collection of small cells which are at different levels of tissue specification and development. Third, we also do not know whether human VSELs have a same molecular signature as their murine counterparts. Finally, as mentioned earlier, expansion in vitro is still a problem for both murine and human VSELs. We believe that the most important reason for these obvious obstacles is epigenetic modification of some imprinted genes in these cells [24] and unfortunately, our in vitro models which we applied so far did not provide optimal signals and microenvironment to reverse this phenomenon. Therefore, we need to explore the possibility that modification of imprinted genes in VSELs, as reported for PGCs [62 –64], could help expand these cells. Another strategy would be to find a proper scaffold or supportive microenvironment that will force expansion and differentiation of VSELs, similarly as has been recently reported for other types of stem cells [115,116].
We expect that the next few years will bring answers about the developmental origin and biological role of this distinct and intriguing population of stem cells residing in adult tissues. We should also seriously consider a new hierarchy for the stem cell compartment not only in adult BM but also in other tissues and try to investigate the mutual relationship between VSELs and PSCs and MPSCs described by different investigators in adult organs [5,15,35,36,41,85,88,109].
Finally, we ask the scientific community to follow our well-described isolation protocols [19] and directly contact our group if there are problems with gating and sorting of these very rare cells. This will avoid confusion in the field and situations in which cells are identified as VSELs but lack a true VSEL phenotype [31 –33]. While this paper was prepared for print two recent reports confirmed presence of VSELs in adult human and murine tissues [121, 122].
Footnotes
Acknowledgments
This study was supported by UE structural funds, the Innovative Economy Operational Program POIG.01.01.02-00-109/09 grant, NIH grants 2R01 DK074720 and R01HL112788, and the Stella and Henry Endowment to MZR.
Author Disclosure Statement
The University of Louisville is the owner of patents on VSELs and some areas of VSEL technology are licensed to Neostem, Inc., New York. None of the authors have any stock in Neostem or any other biotechnological stem cell company.
