Abstract
Our objective was to facilitate ligament tissue reconstruction by characterizing the mechanism of expression of ligament tissue. To accomplish this, we searched for proteins specific to the tissue and introduced them into mesenchymal stem cells. In the two-dimensional phosphorescent gel electrophoresis, the spots in common with the normal human ligament tissue were selected after removing the spots of the normal bone tissue from those of the ossified tissue in the spinal ligament. Proline/arginine-rich end leucine-rich repeat protein (PRELP) was identified in ligament-specific locations by liquid chromatography–tandem mass spectrometry. Transfection of PRELP into mouse mesenchymal stem cells yielded ligament-like connective tissue comprised of parallel fibers. Thus, expression of the PRELP protein could reconstruct the ligament tissue. Since zinc-related proteins were found with high incidence as a result of an array analysis of PRELP's ProtoArray, it was considered that there is a relationship to the zinc metabolism. Tissue induction was mediated by the tumor necrosis factor (TNF)-α via the zinc pathway. PRELP may be a useful gene in syndesmoplasty, provided zinc is present for tissue reconstruction. Chromosome division becomes active with the addition of zinc, and rapid tissue induction takes place in the presence of zinc and TNF-α. Currently, the reconstruction of a ruptured ligament tissue is difficult, but we expect that the PRELP protein expression may facilitate this process. This study describes the discovery of the gene responsible for the differentiation of stem cells into ligament tissue. This important finding may lead to treatments for gonarthrosis, cruciate ligament, and periodontal ligament ruptures, and ossification of the posterior longitudinal ligament.
Introduction
L
Ossification of the posterior longitudinal ligament (OPLL) is a challenging disease that involves ossification of the ligament that stabilizes the spinal column; its cause is unknown. In Japan, this disease has been investigated as part of a national research initiative on intractable diseases organized by the Ministry of Health, Labour and Welfare [1,2]. A proteome analysis, in which normal human bone proteins were subtracted from those detected in ossified ligaments, resulted in the identification of the proline/arginine-rich end leucine-rich repeat protein (PRELP), which was confirmed by two-dimensional (2D) electrophoresis and a comparison with a healthy human ligament tissue.
Introduction of PRELP genes into mesenchymal stem cells induced the formation of ligament-like tissue. Fluorescence in situ hybridization (FISH) analysis confirmed synostosis [3], and ligament reconstruction was achieved by introducing a pPyCAG-cHA-IpacflexEGFP-PRELP vector into ruptured chicken ligaments.
PRELP is strongly expressed in the ligaments of transgenic mice, and stretching experiments confirmed these ligaments are sturdier than those of wild-type mice. High levels of atelocollagen (atelopeptide type 1 collagen) were also observed in the urine of transgenic mice.
When the PRELP ProtoArray and RNA microarray analysis of stem cell-derived ligament-like tissue, transgenic mouse ligament, and wild-type mouse ligament were entered into the Ingenuity Pathway Analysis (IPA) software, the PRELP pathway was associated with mutations in the collagen and matrix that caused the reduced muscle strength associated with distal muscular dystrophy and atrophy in ataxia telangiectasia, Parkinson's disease, and OPLL, thus connecting the PRELP pathway to new diseases.
We wondered whether PRELP was also related to muscle function due to its cross-reactivity with the β-actin control in PRELP western blots. Cross expression with fetal bovine serum (FBS) in the culture fluid and PRELP suppression by small hairpin RNA (shRNA)-PRELP yielded high expression with pCAGGS-PRELP, based on the appearance of platelet-derived growth factor (PDGF) receptors. Matrix metalloproteinase (MMP)-13 and type I collagen expression followed similar patterns.
The ligament tissue, besides being an organ of limb movement, supports the teeth and eyes, and is deeply involved in supporting quality of life through movement and eating. However, overcoming ligament damage remains an unresolved issue. We have demonstrated that the ligament-like tissue derived from mesenchymal stem cells is, in fact, the ligament tissue.
Johnson's report on the sclera of the eye described PRELP expression [4], while the Tenon's capsule, which envelops the outer sclera in the connective tissue, is supported by many ligaments that maintain the position of the eye, as it is fixed firmly to the sclera anterior to the attachment of the extraocular muscles, and partially becomes a cheek ligament continuous with the periosteum of the orbital bone. We analyzed PRELP protein interactions to characterize the connections between ligament tissue proteins. In this article, we describe the restoration of ligament tissue by PRELP and propose clinical applications of PRELP in plastic surgery and dentistry.
Materials and Methods
This study was approved by the ethics committees of the collaborating institutions as part of the research on intractable diseases under the Ministry of Health, Labour and Welfare of Japan. Individual information systems based on FreezerWorks (FDA validation) were used.
Sample preparation
OPLL tissues were collected from 38 of 52 cases of ligament ossification squamous tissues, which were surgically resected in 1999 and 2014 at the Fukui University, Hirosaki University, Kudanzaka Hospital, and Kurume University. Ossified ligament tissues were extracted from specimens of vertebral body tissues obtained from patients with OPLL during a surgery that involved drilling a hole in the anterior part of the vertebral body to broaden the range of motion. The extracted ossified ligament tissues were as small as 5 mm2. All components other than the ossified ligaments were removed from the extracted tissues. The resected tissues were snap-frozen in liquid nitrogen and stored at −80°C. Proteome analysis, in which normal human bone proteins were subtracted from the proteins detected in the ossified ligaments, resulted in the identification of PRELP; these proteins were confirmed by 2D electrophoresis using healthy human ligament tissues. Specimens of human normal ligament tissue were collected from patients undergoing total knee arthroplasty, with their consent, after obtaining approval of the ethics committee.
Proteomics
Ligament tissues from patients with OPLL were analyzed. Proteins were extracted from tissue and healthy bone tissue, and then visualized by two-dimensional difference gel electrophoresis (2D-DIGE) (Supplementary Fig. S1A; Supplementary Data are available online at
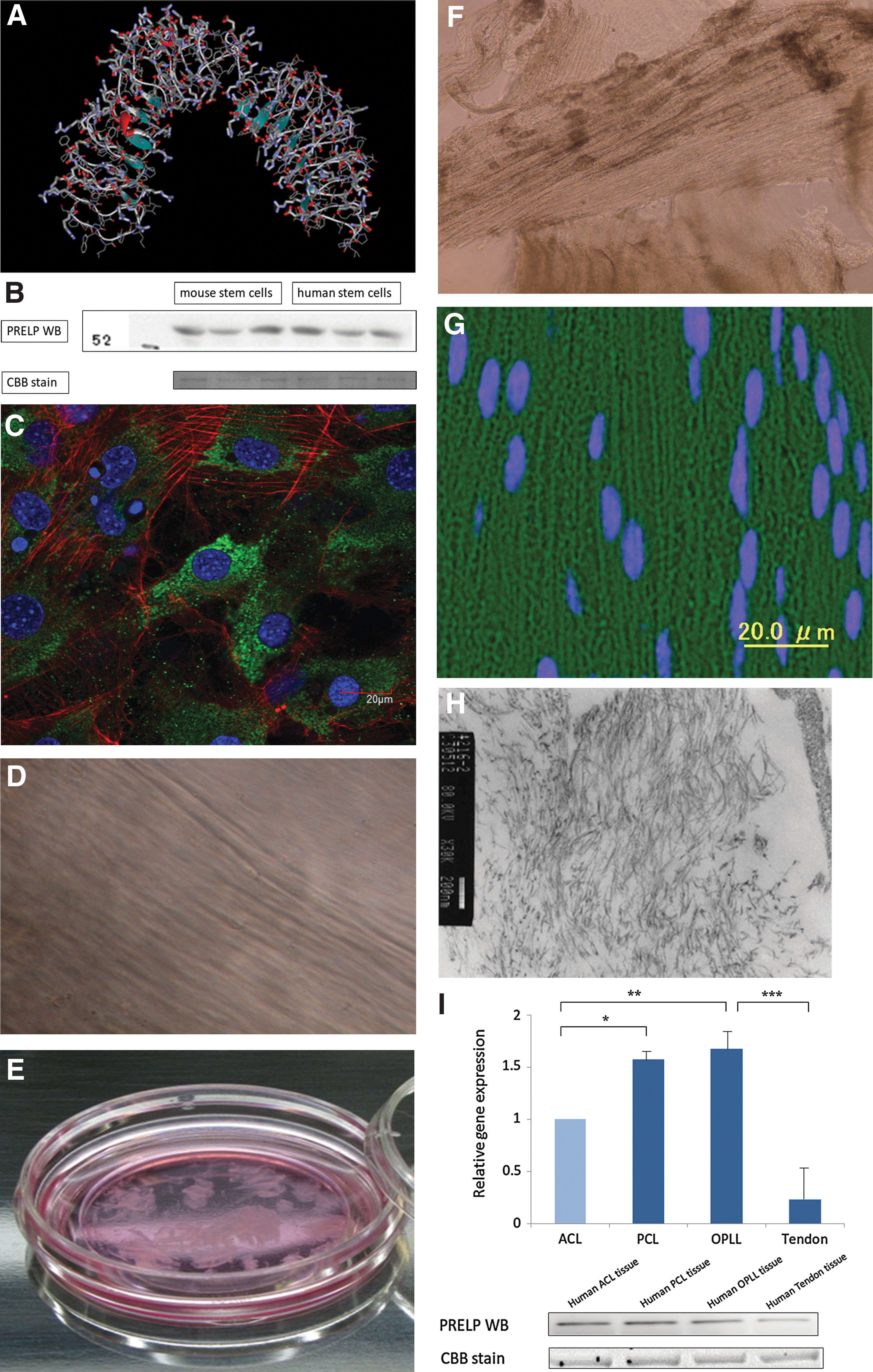
Generation of ligament-like cells from mesenchymal stem cell cultures and the predicted target protein structure.
Preparation of protein expression plasmids
pCAGGS was provided by Professor Junichi Miyazaki (Osaka University). pPyCAG-cHA-IpacflexEGFP was provided by Professor Hitoshi Niwa, (RIKEN Centre for Developmental Biology) [7].
Mesenchymal stem cells derived from mouse embryos (C3H/10T1/2-clone 8; JCRB0003, MC3T3-E1; JCB1126) [8] were obtained from the RIKEN Bioresource Center Cell Bank in Japan. To express murine proteins, the synthesized mouse PRELP sequence was inserted in the XhoI site of pCAGGS, then cloned, and transfected into mouse mesenchymal stem cells. Protein expression in the culture supernatant was confirmed by western blotting (Fig. 1B).
Method for use with PuraMatrix peptide hydrogel
The casting frame was placed at room temperature in a 1×phosphate-buffered saline (PBS) bath to initiate gelation. After 5 min, the gel slab was transferred to a Petri dish containing a 4°C medium and subsequently placed in a 37°C incubator. Peptide gels were cultured in a 10% FBS-supplemented feed medium. The slabs were seeded at a cell density of 15×106 cells/mL. Each slab was fed 12 mL of the medium every other day. A peptide solution was added to a final peptide concentration of 0.5% and a cell density of 15×106 cells/mL. We washed the cells and the peptide gel with a saccharose solution. At this time, the sucrose solution was used to maintain the physiological osmotic pressure.
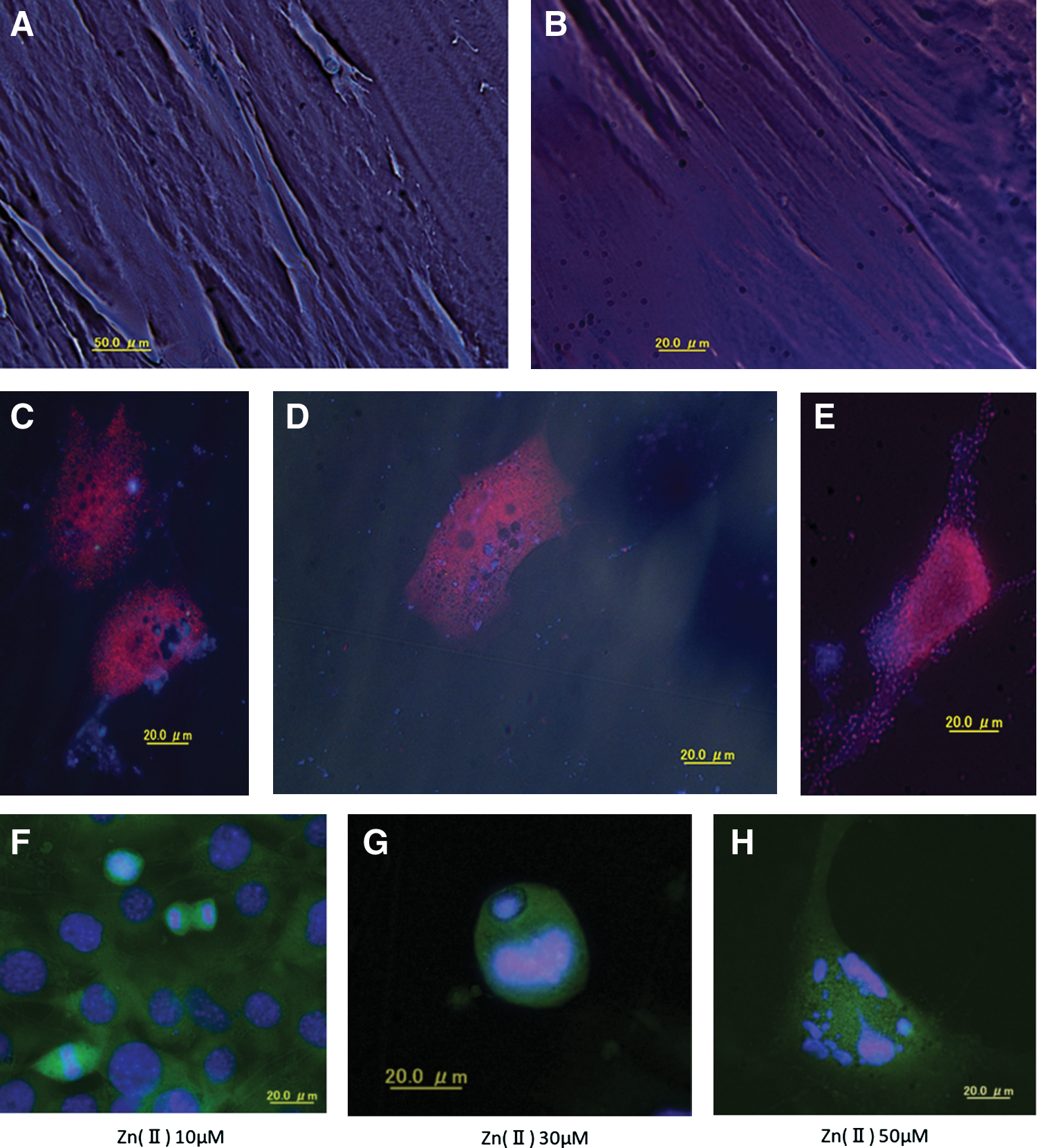
The culture medium was removed from stem cell-derived tissues cultured in a glass-bottom dish. The cells were washed twice with cold PBS and fixed in 4% paraformaldehyde in PBS. The rabbit PRELP antibody was used as the primary antibody and Alexa Fluor 488 goat anti-rabbit immunoglobulin G (IgG) was used as the secondary antibody. Nuclei were stained with 4′,6-diamidino-2-phenylindole (DAPI) and the actin cytoskeleton was visualized with Alexa Fluor phalloidin. The tissues were deparaffinized, washed twice in cold PBS, and fixed in 4% paraformaldehyde in PBS. A rabbit anti-PRELP (Sigma Aldrich) antibody was used as the primary antibody, and Alexa Fluor 488 goat anti-rabbit IgG was used as the secondary antibody. DAPI was used to stain the nuclei. After staining, the tissues were observed under CSLM (Olympus FluoView FV 1000-D) (Fig. 1C). A ligament-like connective tissue composed of parallel fibers is observed (Fig. 1D). Images of the ligament-like tissue that was cultured in a three-dimensional (3D) gel are shown (20×objective). The tissue was exfoliated with a spatula, and the cells floated up from the bottom of the culture dish, which permitted us to observe their fibrous morphology. In this state, we were able to hold the tissue using the dressing forceps (Fig. 1E). Cell counts were performed with the automated cell counter TC10 (Bio Rad, Inc.).
After 12 h, a BioStation ID cell analyzer (GE Healthcare) indicated the cells had transformed into a ligament fiber-like structure (Supplementary Video SV1). A 3D culture showed that histogenesis was induced by introduction of the PRELP gene. Figure 1F shows the tissue at 7 days. The fibrous morphology was observed by causing the cells to float from the bottom of the culture dish, as followed by confocal scanning laser microscopy (CSLM) with the laser on the Z-axis. The stem cells in the upper layer of the ligament-like tissue were 15 μm thick. Figure 1G shows the stem cell-derived ligament-like tissues cut into 3-μm slices and labeled with rabbit anti-PRELP (Sigma Aldrich) and Alexa Fluor 488 goat anti-rabbit IgG antibodies. Nuclei were stained with 4′,6-diamidino-2-phenylindole and visualized by CSLM. The ligament-like fibrous tissue was fixed with a 2% glutaraldehyde/0.1 M phosphate buffer (pH 7.4) and observed by transmission electron microscopy (Fig. 1H).
Methods for use without PuraMatrix peptide hydrogel
Transfected cells were cultured in 30 μM ZnCl2 and 10 ng/mL tumor necrosis factor-α (TNF-α) to stem cells carrying the PRELP gene.
Western blotting
Western blotting of the PRELP antibody (Sigma Aldrich; SAB1100370) with the C3H10T1/2 culture fluid showed a reaction with FBS, so the nonreactive Opti-MEM I Reduced Serum Medium (Life Technology) was used. The doubling time of C3H10T1/2 cells in 10% FBS Eagle's minimal essential medium (Wako) was 18 h, but the doubling time of the Opti-MEM I Reduced Serum Medium (Life Technology), which does not contain FBS and does not cross with PRELP expression, was 27 h. For shRNA-PRELP, four genes were cloned into the pRNAi/LV-RNAi vector (Biosettia, Inc.); after confirming suppression of PRELP expression, sh-NM_054077 5′-AAAAGGATTAGGCGTAAACCCAATTGGATCCAATTGGGTTTACGCCTAATCC-3′, which suppressed collagen and PDGF receptor expression in the supernatant, was used. shRNA-lacZ, AAAAGCAGTTATCTGGAAGATCAGGTTGGATCCAACCTGATCTTCCAGATAACTGC, and sh-scramble, AAAAGCTACACTATCGAGCAATTTTGGATCCAAAATTGCTCGATAGTGTAGC, were created for the control. For transfection, Lipofectamine 2000 (Life Technology) was used according to the manufacturer's protocol for overexpression, and Gene Porter (Genlantis) was used for shRNA. The plate was shaken to uniformly distribute the vector and incubated for 4 h at 37°C in 5% CO2. The culture medium was replaced with a fresh medium, and the mixture was cultured for another 96 h. PRELP expression in the supernatant was confirmed by western blotting, and secretion of the product in the culture fluid was confirmed every 2 days against the desired sequence. The culture medium was replaced every 2 days. After 1 week, the cells had transformed into ligament fiber-like structures. Transfected cells were then cultured in PuraMatrix Peptide Hydrogel. After 24 h, images obtained with a BioStation ID (GE Healthcare) revealed the cells had transformed into ligament fiber-like structures. Cultures were performed in Opti-MEM without FBS for western blot experiments. Cell lysates or nuclear proteins extracted from cultured cells or tissues were used as samples. Each sample was separated by sodium dodecyl sulfate–polyacrylamide gel electrophoresis in 12% or 8% gels and transferred to polyvinylidene fluoride (PVDF) membranes (0.4 Å for 1 h at 10°C). After transfer, the PVDF membranes were incubated in a blocking solution at room temperature for 1 h and probed with primary antibodies for 16 h at 4°C. After washing in 0.05% Tween-20 in PBS, the membrane was probed with secondary antibodies for 1 h at room temperature. After the PVDF membrane was washed again in 0.05% Tween-20 in PBS, the membrane was imaged using an enhanced chemiluminescence (ECL) western blotting analysis system (GE Healthcare). The LAS-4000 Bioimaging Analyzer (GE Healthcare) was used to detect the ECL reactions.
The PRELP (Sigma Aldrich) antibody western blotting analysis confirmed the presence of protein in the control sh-Lac Z and sh-scramble, and that shRNA-PRELP-1 resulted in PRELP knockdown. Therefore, the nonreactive Opti-MEM I Reduced-Serum Medium (Life Technology) was used instead.
Relative quantification using real-time polymerase chain reaction analysis
RNA was extracted using the AllPrep DNA/RNA mini kit (Qiagen) and RNA integrity was analyzed (Agilent 2100 Bioanalyzer). RNA with an integrity value of 9.0 or higher was reverse transcribed (Superscript VILO cDNA Synthesis Kit; Life Technologies) with a StepOnePlus real-time polymerase chain reaction (PCR) analysis system (Applied Biosystems) using TaqMan PCR (sequences from mouse collagen, type I, alpha 1Col1a1: Mm00801666_g1; human collagen, type I, alpha 2: Hs00164099_m1; mouse 18S ribosomal RNA, hypothetical LOC790964: Mm03928990_g1; human eukaryotic 18S rRNA: Hs99999901_s1; human proline/arginine-rich end leucine-rich repeat protein: Hs01941580_s1; matrix metallopeptidase 13 [collagenase 3]: Hs00233992_m1; mitogen-activated protein kinase kinase kinase 7: Mm00554514_m1; TNF receptor-associated factor 6: Mm00493836_m1; mitogen-activated protein kinase kinase kinase 3: Mm00803725_m1 and nuclear factor of kappa light polypeptide gene enhancer in B-cells inhibitor, alpha: Mm00477800_g1) (Applied Biosystems). The reaction conditions were as follows: 1 cycle at 95°C for 3 min followed by 40 cycles of 95°C for 15 s and 60°C for 1 min. The threshold cycle (Ct) was selected as the first PCR cycle in which the desired PCR products were detected. Quantitative analysis was performed using the comparative Ct method [9]. This technical note demonstrates the utility of a ΔΔCq method for calculating the relative gene expression and percent knockdown from quantifying cycle (Cq) values that were obtained by quantitative real-time PCR analysis in an RNA interference (shRNA) experiment. To determine the relative gene expression, probe-based real-time PCR was performed using Thermo Scientific Solaris real-time PCR Gene Expression Assays with cDNA synthesized from the total RNA that was harvested from the cell culture. Here, a ΔΔCq method is demonstrated for the normalized determination of gene knockdown, and the necessary experimental controls have been previously described (Supplementary Fig. S2).
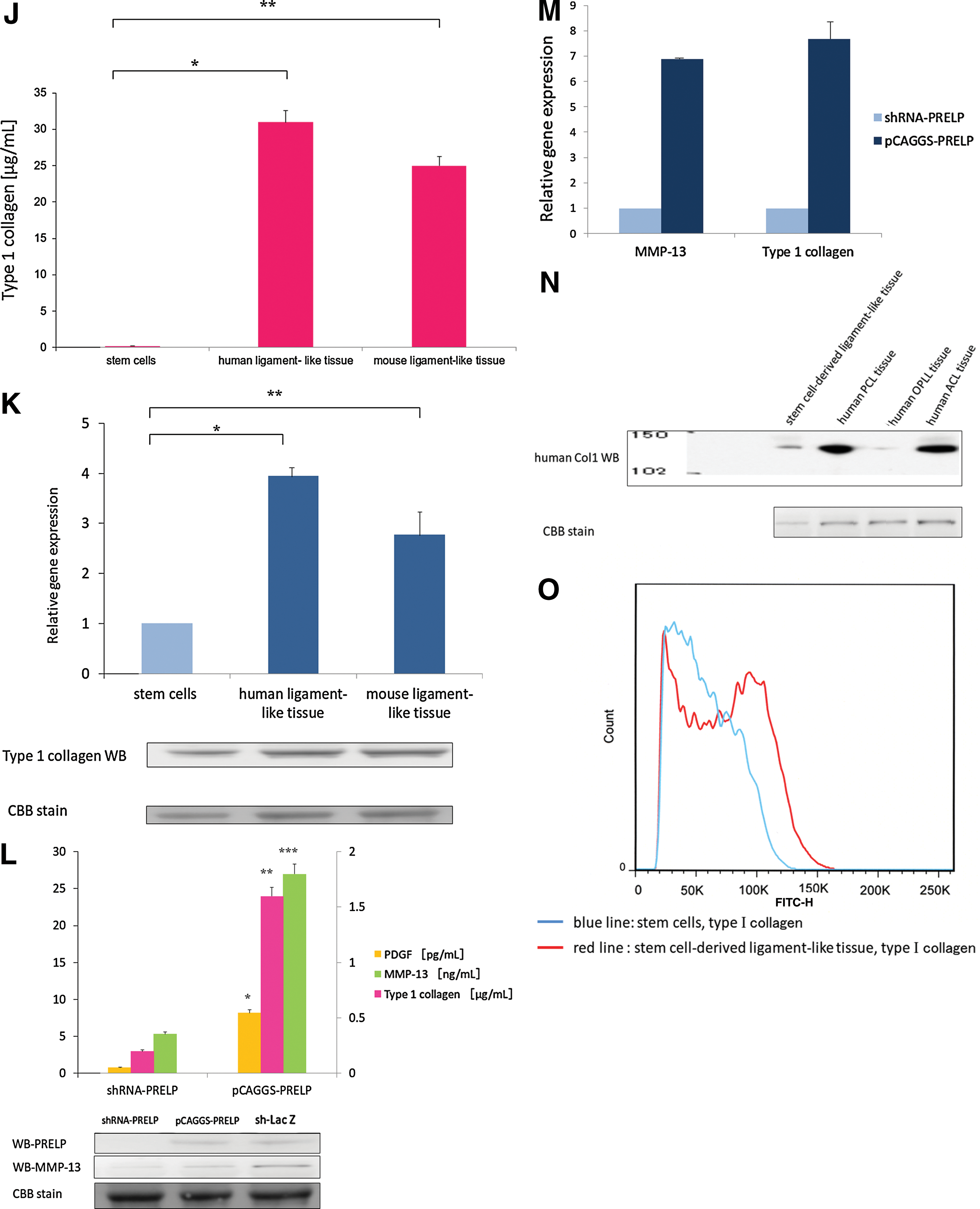
We conducted the transfection of the shRNA-PRELP vector and the pCAGGS-PRELP vector into the stem cells to produce the cell in which the PRELP protein was completely knocked down and the cell in which it was overexpressed. Then, we examined the expression of MMP-13 and type 1 collagen in each cell type (Fig. 1L, M).
PRELP protein array and gene array interactions, and statistical analysis
Gene expression analysis was performed with microarrays of stem cells, ligament tissue from wild-type mice, ligament tissue from transgenic mice, and stem cell-derived ligament tissue.
We used ProtoArray Human Protein Microarrays (Invitrogen) to analyze PRELP protein interactions. We confirmed that the control array had no background. In the negative control, reaction of PRELP antibodies and Alexa Fluor 647 secondary antibodies were detected by Gene Pix 4000B. Expression was individually analyzed after PRELP primary and Alexa Fluor 647 secondary antibody reactions. The box plot, like the normal plot, is frequently used in statistics (Supplementary Fig. S1). The Z-factor >0.5 is a measure of statistical effect size. It has been proposed for use in high-throughput screening to determine whether the response in a particular assay is large enough to warrant further attention [10]. A panel of values was calculated for each protein array, including the Z score, the Chebyshev inequality precision (CIP) value, and the coefficient of variation (CV). The Z score is the signal value for a given spot minus the mean signal value from all human proteins in the array, divided by the standard deviation of the signal values for all human proteins in the array. The CIP value evaluates the signal strength for a given spot relative to all negative control spots, calculating the probability that the observed signal may come from a negative control distribution. The CV evaluates the standard deviation between duplicates. A Z score >3, a CIP value <0.05, and a CV <0.5 define a positive spot. Signals for each individual protein across all samples from a given population were aligned for downstream analysis (Supplementary Table S1).
Using the IPA tool, genetic networks were constructed using the PRELP ProtoArray for 53 genes that exhibited high expression in the ligament tissue of transgenic and wild-type mice and in the ligament-like tissue. These networks revealed functional and genetic relationships. The IPA tool yielded no connections to known biologic pathways. This study revealed a new network significantly correlated with PRELP.
All statistical analyses were performed with SAS9.2 software.
Accession codes
Primary accessions: GEO array, GSE35269.
Results
Characteristics of the new ligament tissue protein
The stem cell-derived ligament-like tissue and human ligament tissue from the anterior cruciate ligament (ACL), posterior cruciate ligament (PCL), and OPLL tissue were probed for the PRELP protein by western blotting. The PRELP protein and real-time PCR confirmed PRELP expression in stem cell-derived ligament-like tissue and in ACL, PCL, OPLL tissue, and tendon (Fig. 1I). We investigated the expression of type I collagen, which is highly expressed in the ligament tissue. Western blotting revealed type I collagen expression in stem cell-derived ligament tissue and in human PCL, OPLL, and ACL. After confirmation with the secondary antibody Alexa Fluor 488, type I collagen expression in stem cell-derived ligament-like tissue was investigated. Mesenchymal stem cells and induced ligament cells were analyzed by immunohistochemistry and flow cytometry with the CD34-fluorescein isothiocyanate (FITC) antibody. We previously demonstrated that type I collagen antigen is differentially expressed in developing ligament tissue (Fig. 1O). CD34 expression was high in the stem cells of the lower layer of the stem cell-derived tissue, and as the cells differentiated to form ligament-like tissue, CD34 expression decreased and type I collagen surface antigens increased.
Type I collagen gene expression was higher in induced ligament tissue than in mesenchymal stem cells (Fig. 1J, K). *P<0.05, **P<0.05 TaqMan PCR (sequences from mouse collagen, type I, alpha 1Col1a1: Mm00801666_g1; human collagen, type I, alpha 2: Hs00164099_m1; mouse 18S ribosomal RNA, hypothetical LOC790964: Mm03928990_g1; human eukaryotic 18S rRNA: Hs99999901_s1). Real-time PCR was performed using a TaqMan Gene Expression Assay kit with a StepOnePlus real-time PCR system. The amount of mouse 18S ribosomal RNA (Mm03928990_g1) or human eukaryotic 18S rRNA (Hs99999901_s1) mRNA in each sample was used to standardize the quantity of the following mRNAs: mouse Col1a1 (n=21) *P<0.001, and human Col1a1 (n=24) **P<0.001.
Transfection of a shRNA-PRELP vector into shRNA-PRELP mesenchymal stem cells was followed by enzyme-linked immunosorbent assay (ELISA) measurements of PDGF [11], MMP-13 [12], and Type 1 collagen in the culture supernatant; levels of each protein increased in the presence of pCAGGS-PRELP (Fig. 1L). Furthermore, after comparing the test results in which the expression of the PRELP protein was suppressed by shRNA and the test results in which the protein was overexpressed by pCAGGS, we found the expression of MMP-13 and type 1 collagen to have also been suppressed by shRNA.
PRELP transgenic mouse
PRELP transgenic mice were developed using the microinjection method. PRELP immunohistochemical staining of all body tissues of transgenic and wild-type mice showed high expression in the spinal (Fig. 2F, J), knee (Fig. 2G, K), periodontal ligament (Fig. 2H, L), and skin tissues of the transgenic mouse (Fig. 2I, M).
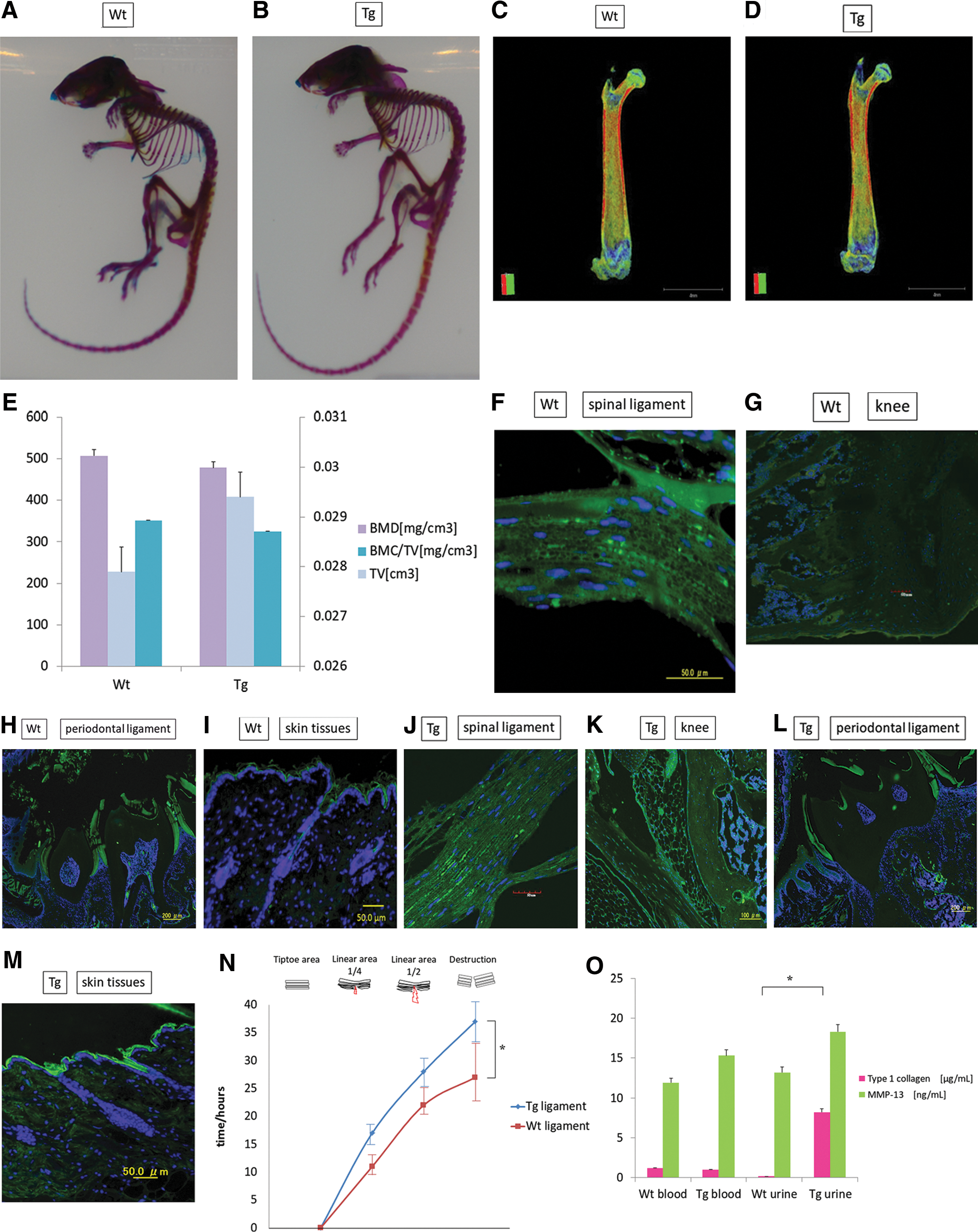
Preparation and genotyping of transgenic mice. The PRELP gene (3.4 kb) was ligated into the XhoI site of pCAGGS, and DNA was injected into the pronuclei of fertilized C57BL/6 mouse eggs with microcapillaries and an inverted microscope. DNA extracted from the tail tissue of the resulting newborn mice was PCR-amplified. Genotyping confirmed that transgenic mice had been produced. The PRELP gene was synthesized using the phosphoramidite method and ligated into the XhoI site of pCAGGS-XhoI. The expression vector was digested with XhoI and the electrophoretic pattern was confirmed. Sequencing of the inserted cDNA region confirmed that the inserted region matched the intended sequence (GenBank accession number: EAW91481.1). Sections of tail tissue were cut from 20 of the resulting newborn mice and DNA was extracted using the DNA Mini Kit (Qiagen). PCR was performed to determine whether the samples contained the exogenous gene using the following primer set: R5′-GTCGAGGGATCTCCATAAGAGAAGAGGGACA-3′ and F5′-GTCGACATTGATTATTGACTAGTTATTAA TAGTAATC-3′. The following PCR conditions were used: 30 cycles of 94°C for 2 min, 98°C for 10 s, and 68°C for 4 min. Please refer to the Supplementary Data for protocol details.
Blood and urine analysis revealed equal amounts of type I collagen in the blood of transgenic and wild-type mice, but high levels of atelocollagen in the urine of transgenic mice. This was likely due to PRELP overexpression, which led to high collagen expression and excretion in the urine. Further, although MMP-13 is decomposed into type I collagen, its concentrations were higher in the blood and urine of transgenic mice (Fig. 2O) [11].
Ligament and osseous adhesion
Because ligaments join bony tissue and form bone-to-bone connections, we investigated synostosis of the PRELP-derived ligament tissue by combining the mouse stem cell-derived ligament-like tissue with normal human osteoblasts for 24 h. FITC staining for chromosomes showed fusion of human (rhodamine: red) (Fig. 3A) and mouse chromosomes (FITC: green) (Fig. 3B). Karyotyping confirmed that the mouse stem cell-derived ligament-like tissue was fused with human osteoblasts.
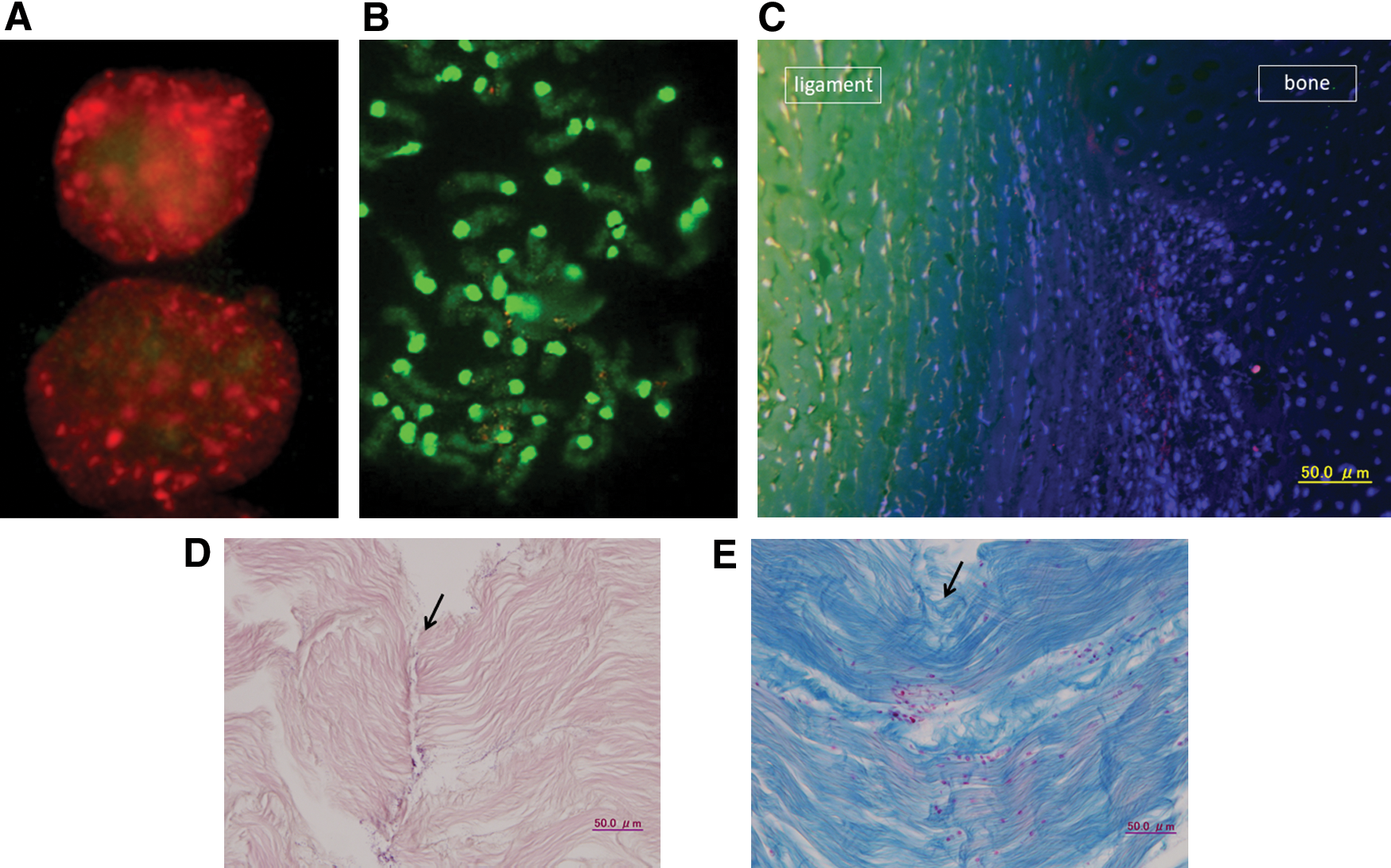
Osseous adhesion and ligament reconstruction. Because ligaments join bony tissue and form bone-to-bone connections, we investigated the synostosis of PRELP-derived ligament tissue by combining mouse stem cell-derived ligament-like tissue with human osteoblasts. FITC chromosome staining showed fusion of human (rhodamine: red) and mouse chromosomes (FITC: green). Karyotyping confirmed that the mouse stem cell-derived ligament-like tissue was fused with human osteoblasts. Probes were labeled with digoxigenin, rhodamine spectrum orange, human Cot-1DNA, biotin, FITC Spectrum Green, and mouse Cot-1DNA. Interphase
Manifestation of protein at the junction of ligament and bone tissue
A shallow cut was made at the juncture of chicken ligament and bone tissue. Fluorescence staining of PRELP (Alexa Fluor 488) and type II collagen (Alexa Fluor 555) showed PRELP expression only in the ligament tissue (Fig. 3C), and clear expression of type II collagen between it and the bone.
Experimental reconstruction of ruptured ligament tissue
The ACL was obtained from a chicken and cut in several places (Fig. 3D). After incubation in a culture fluid containing pPyCAG-cHA-IpacflexEGFP-PRELP, the ruptured ligament tissue was restored. Green fluorescent protein (GFP) expression in the restored tissue was confirmed by fluorescence microscopy. One week later, collagen fiber connections were observed by azan staining (Fig. 3E). GFP fluorescence of a new ligament tissue after restoration revealed stem cells carrying pPyCAG-cHA-IpacflexEGFP-PRELP.
Based on these results, we concluded that the tissue derived from the transfected mesenchymal stem cells was the ligament tissue and PRELP could reconstruct ligament tissue.
PRELP protein and gene interaction
RNA microarray analyses showed a statistically significant similarity between the stem cell-derived ligament-like tissue and mouse ligament tissue (Fig. 4A). Analysis of all genes showed homologous changes in wild-type mouse ligament tissues, transgenic mouse ligament tissues, and stem cell-derived ligament tissues vs. stem cells from the extracellular matrix (Fig. 4B). A list of useful proteins was generated through ProtoArray analysis with PRELP protein interactions (Supplementary Table S1). The new pathway revealed using the IPA software was based on RNA expression and PRELP protein interactions (Fig. 4C).
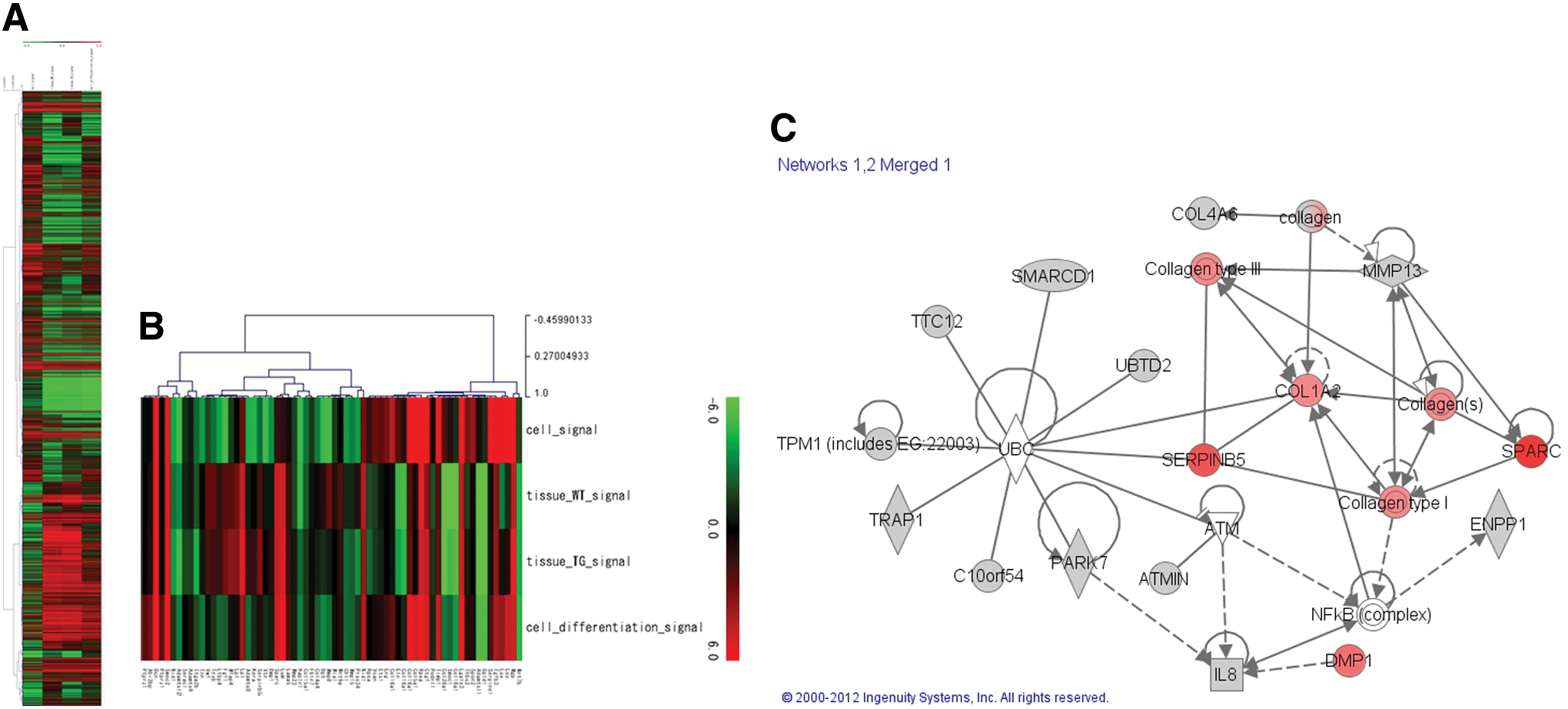
Expression route of PRELP
The apparent relationship with zinc revealed by the ProtoArray analysis prompted us to measure p65, p52, and p50 of NF-κB, and c-Rel. The PRELP gene was introduced into stem cells, and ZnCl2 and LY294002 (a PI3 kinase inhibitor) were introduced without a 3D gel, yielding high values of ZnCl2-induced NF-κB-p65 and very little influence of LY294002 (Fig. 5K, L). Introduction of TNF-α resulted in ligament tissue formation after 24 h (Fig. 5A); adding LY294002 also resulted in ligament tissue transformation (Fig. 5B). Other cellular changes were observed by immunofluorescence tissue staining with Alexa Fluor 555 for nuclear factor-kappa B (NFκB) and Alexa Fluor 350 for PRELP. After 3 h, the intracellular endoplasmic reticulum was demarcated (Fig. 5C); after 6 h, PRELP expression was confirmed in the endoplasmic reticulum (Fig. 5D). After 12 h, a fibrous transformation had taken place and PRELP expression was confirmed (Fig. 5E). Intranuclear transition of NF-κB p65 was also verified by fluorescence staining in the presence of ZnCl2 (Fig. 5F–H) [13]. Aggregation of the intranuclear pathway was induced by the addition of (ZnCl2 10, 30, and 50 μM) and Zn(II) influx through the ligament tissue. Zinc induced nuclear fission and apoptosis by nuclear explosion occurred at the high concentration of 50 μM (Fig. 5H), but nuclear import occurred at less than 30 μM (Fig. 5F, G), and PRELP synthesis was confirmed in the cytoplasmic membrane (Fig. 5I, J).
Expression route of PRELP.
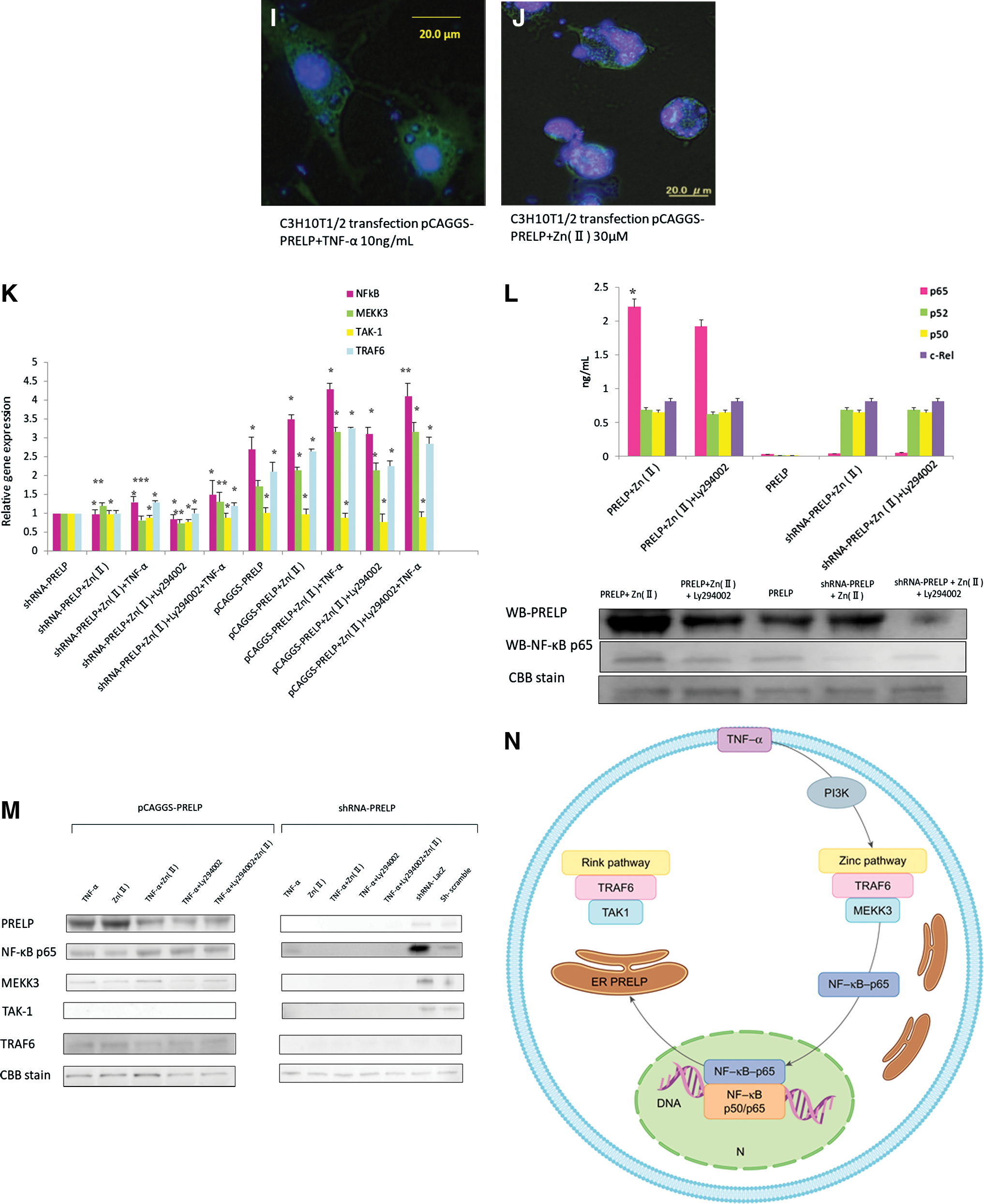
Western blotting of TAK1, TRAF6, MEKK3, and NF-κB p65 revealed that the intranuclear transition is mediated by a zinc pathway via TNF-α (10 mg/mL) (Fig. 5M) [14 –17]. In addition, NF-kBIA, MEKK3, TAK-1, and TRAF6 expression in human stem cell-derived ligament-like tissue transfection by pCAGGS-PRELP was higher than in the case of 30 μM ZnCl2 or TNF-α (Fig. 5K).
We have demonstrated that PRELP is a ligament-forming gene, and our findings suggest that even proteins can be used as ligament tissue markers.
Ligaments are connective tissues that link bones to each other and maintain joint strength and stability, and in this study, we investigated synostosis by using FITC (Fig. 3A, B). PRELP expression was observed in ligament tissue at the junction of the ligament and bony tissue, and type II collagen expression was observed between bony tissues (Fig. 3C). Ligament ruptures healed after 7 days in animal experiments (Fig. 3D, E) and stretching experiments in PRELP transgenic mice showed that the ligament tissue was sturdy (Fig. 2N). Investigation of the intracellular signaling pathways showed that PRELP nuclear transcription is mediated by a zinc pathway via TNF-α and transforms into ligament-like tissue as it is secreted from the endoplasmic reticulum (Fig. 5).
Discussion
This study describes the discovery of the gene responsible for the differentiation of stem cells into ligament tissue. This important finding may lead to treatments for gonarthrosis, cruciate ligament and periodontal ligament ruptures, and OPLL.
Johnson's report on the sclera of the eye described PRELP expression [4], while Tenon's capsule, which envelops the outer sclera in the connective tissue, is supported by many ligaments that maintain the position of the eye, as it is fixed firmly to the sclera anterior to the attachment of the extraocular muscles, and partially becomes a cheek ligament continuous with the periosteum of the orbital bone. The levator muscle of the upper eyelid originates from the annular tendon, divides into multiple aponeuroses, and attaches to the anterior surface of the tarsal plate and around the orbicular muscle. Some of these aponeuroses attach medially to the trochlea and laterally to the tear ducts, and the apical aponeuroses form a swing shape to become the superior transverse ligament (Whitnall's ligament) [18]. Both apical aponeuroses attach to the medial and lateral palpebral ligament, as well as to the orbital septum. Thus, the superior transverse ligament and orbital septum form a continuous structure, while the superior transverse ligament functions as a pulley to change the direction of action at the levator attachment. The fascia of the inferior rectus and inferior oblique muscles join and continue to the Tenon subcapsular region, and the fascia that spreads from the lower fornix to the tarsal plate is the Lockwood's ligament. Laterally, fibers from the lateral palpebral ligament connect directly to Whitnall's ligament and the levator aponeurosis. Fibers from the medial palpebral ligament attach to the cheek ligament and the levator aponeurosis. The orbital septum is a sheet of connective tissue that connects the orbital margin and tarsal plate, and divides the eyelids into anterior and posterior lobes. The inner upper eyelid is integrated with the aponeurosis of the upper palpebral levator deep in the orbicular muscle. The upper and lower orbital septum join and become the medial and lateral palpebral ligaments, which also attach to the orbicular muscle to support the eyelid. So, the eye position is maintained through many ligaments, which are continuously changing from fascia to tendon to ligament. PRELP suggests that it develops in ligament.
Fluorescence images of the tissue where the ligament adheres to bone showed PRELP expression in ligament tissue and type II collagen expression between the ligament tissue and the bony tissue (Fig. 3C). The ligament tissue was sturdy under strong stretching in transgenic mice (Fig. 2N). High levels of atelocollagen in the urine of these mice were likely due to strong PRELP expression and urinary excretion of excess collagen (Fig. 2O).
Because synostosis was demonstrated by FISH analysis (Fig. 3A, B) and reconstruction of ruptured chicken ligaments was achieved (Fig. 3D, E), we determined the substance generated to be ligament tissue. The pathway identified by mRNA microarray of ligament-like tissue and a PRELP ProtoArray (Fig. 4C) revealed a link to gene mutation associated with reduced strength in distal muscular dystrophy. Because the ProtoArray results suggest PRELP is affected by zinc bonding, a zinc experiment was conducted with stem cell-derived ligament tissue. PRELP expression in the ligament tissue was measured by NFκB-p65, NFκB-p52, NFκB-p50, and c-Rel ELISA; ZnCl2 increased p65, TRAF6, and MEKK3 expression and eliminated TAK1 expression, as demonstrated by western blotting. Transformation into ligament tissue was induced by TNF-α after 24 h via intranuclear transition from a zinc pathway and PRELP secretion from the endoplasmic reticulum (Fig. 5N). Furthermore, because of the presence of NfκB expression, PRELP was determined to function as an intranuclear transcription factor influenced by zinc. For intracellular information transmission, PRELP enters a zinc pathway from TNF-α, transitions inside the nucleus, and is transformed into a ligament-like tissue, while it is secreted from the endoplasmic reticulum. In many countries with aging societies, numerous people have knee pain and an impaired ability to walk, which is an inherently human behavior. We are hopeful that our finding will eventually help people to maintain or regain their ability to walk. We believe this discovery may also be useful in restoring periodontal ligaments. Tooth loss affects an individual's eating habits. In addition, we believe that the results of this study may contain a clue to the conquest of locomotorium incontinence. All of these issues are of immense importance in an individual's life.
Footnotes
Acknowledgments
We thank our collaborators for their cooperation in research on intractable diseases at the Ministry of Health, Labour and Welfare. The authors thank Prof. H. Baba, Dr. K. Uchida (University of Fukui), Prof. S. Toh, Dr. A. Ono (Hirosaki University Graduate School of Medicine), Prof. Emeritus H. Umeyama (Pharmacy, Kitasato University, Laboratory of Biomolecular Design), K. Yoshida (Kurume University), K. Yasuda, and A. Doi (Cell Innovator Co., Ltd.), H. Nakamura (Ecogenomics Co., Ltd.), Y. Ishimatsu (Koken Service Engineering Co., Ltd.), I. Sugimura, R. Hachisu, and I. Tenmoto (Hokkaido System Science Co., Ltd.). We are also indebted to Prof. J. Miyazaki (Osaka University) and Prof. H. Niwa (RIKEN Centre for Developmental Biology), whose comments contributed enormously to our work. Financial support was provided by a grant for research on intractable diseases from the Japanese Ministry of Health, Labour and Welfare.
Author Disclosure Statement
The authors report no conflict of interests.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
