Abstract
Human mesoangioblasts are vessel-associated stem cells that are currently in phase I/II clinical trials for the treatment of patients with Duchenne muscular dystrophy. To date, little is known about the effect of mesoangioblasts on human immune cells and vice versa. We hypothesized that mesoangioblasts could modulate the function of immune cells in a similar manner to mesenchymal stromal cells. Human mesoangioblasts did not evoke, but rather potently suppressed human T-cell proliferation and effector function in vitro in a dose- and time-dependent manner. Furthermore, mesoangioblasts exert these inhibitory effects uniformly on human CD4+ and CD8+ T cells in a reversible manner without inducing a state of anergy. Interferon (IFN)-γ and tumor necrosis factor (TNF)-α play crucial roles in the initial activation of mesoangioblasts. Indoleamine 2,3-dioxygenase (IDO) and prostaglandin E-2 (PGE) were identified as key mechanisms of action involved in the mesoangioblast suppression of T-cell proliferation. Together, these data demonstrate a previously unrecognized capacity of mesoangioblasts to modulate immune responses.
Introduction
H
A significant hurdle in all allogeneic cell therapies is immune rejection. Indeed, it appears that different cell therapies evoke varying immune responses which may be due to the immunogenicity of the cells in question [7 –9]. Although allogeneic cell therapies are administered in conjunction with immunosuppression, a greater understanding of the immunogenicity of cell therapies and their influence on the immune response may allow a more tailored therapy that is geared toward enhancing cell therapeutic efficacy and minimizing the immunosuppressive regimen.
DMD is caused by mutations in the dystrophin gene that lead to muscle fiber degeneration followed by inflammation, which, in turn, plays a role in disease progression [10]. DMD onset and progression has been associated with the up-regulation of inflammatory genes, both in patient samples [11,12] and in the MDX mouse model [13,14]. Thus, it seems clear that cell therapies applied to DMD patients will encounter a hostile inflammatory environment. In particular, previous studies have shown that the pro-inflammatory cytokine tumor necrosis factor (TNF)-α is elevated in DMD patients [15] and also in MDX mice [16]. The presence of such pro-inflammatory cytokines as well as immune cells present in damaged and regenerating muscles may directly affect the therapeutic ability of mesoangioblasts.
A considerable body of data has accumulated, suggesting that most stem cells display some level of immune privilege [7,17 –19] and in some cases, immunosuppressive capacity [20 –25]. Mesoangioblasts are pericyte-derived cells [1,26] that share a number of surface markers with mesenchymal stromal cells (MSC) [27] which are also thought to be of a perivascular origin [28]. Recently, a study comparing mesoangioblasts, MSC, and multipotent adult progenitor cells [27] has revealed that while mesoangioblasts and MSC share a number of surface markers, the 2 cell populations are distinct and have differential transcriptomic gene signatures that are specific to their functional properties [27]. While these are important data, this study did not compare the immunosuppressive functional capacity of these different cell types. In this study, we sought to examine the immunogenicity of mesoangioblasts in vitro and to characterize the effects that pro-inflammatory cytokines and exposure to activated T cells have on the function of mesoangioblasts and vice versa. Here, we present novel findings which demonstrate that human mesoangioblasts do not evoke but rather significantly inhibit T-cell proliferation through a cell-contact independent mechanism involving indoleamine 2,3 dioxygenase (IDO) and prostaglandin E-2 (PGE-2).
Experimental Procedures
Cell culture
Human mesoangioblasts were isolated from adult skeletal muscle as previously described [2]. Human mesoangioblasts were cultured in MegaCell Dulbecco's modified Eagle medium (Sigma) containing 5% fetal bovine serum (FBS; PAA), 2 mM glutamine (PAA), 0.1 mM β-mercaptoethanol (Gibco-Invitrogen), 1% nonessential amino acids (NEAA; Sigma), 5 ng/mL human bFGF (Peprotech, UK), 100 IU mL−1 penicillin and 100 mg/mL streptomycin (PAA), and 1% insulin transferrin selenium X (ITSX) supplement (Gibco-Invitrogen). Alternatively, the same cells were cultured in Iscove's modified Dulbecco's medium (PAA) containing 10% FBS, 2 mM glutamine, 0.1 mM β-mercaptoethanol, 1% NEAA, 5 ng/mL human bFGF, 100 IU mL−1 penicillin, 100 mg/mL streptomycin, 1% ITSX supplement, 0.5 μM oleic and linoleic acids (Sigma), 1.5 μM iron [II] chloride tetrahydrate (Fe++; Sigma-Aldrich), and 0.12 μM iron [III] nitrate nonahydrate (Fe+++; Sigma-Aldrich).
Human peripheral blood mononuclear cells (PBMCs) were isolated from buffy coats. All PBMC cultures were carried out in RPMI 1640 (Sigma-Aldrich) supplemented with 10% FBS, 1% v/v L-glutamine, 0.05 mM β-mercaptoethanol, 100 IU mL−1 penicillin, and 100 mg/mL streptomycin.
T-cell proliferation assays
The proliferation of PBMCs was measured using the 5,6 carboxyfluorescein diacetate succinimidyl ester (CFSE) dilution assay. PBMCs were labeled with 10 μM CFSE in warm phosphate-buffered saline (PBS) at room temperature. After 10 min, cells were washed in ice-cold PBS. CFSE-labeled PBMCs (2.5×105/mL) were then seeded into 96-well round-bottom plates with or without human CD3/CD28 beads (Dynal-Life Technologies) (0.5×105/mL) in a total volume of 0.2 mL of complete RPMI 1640. For suppressor assays, mesoangioblasts were seeded overnight in 96-well round-bottom plates in mesoangioblast cell culture medium. Medium was then replaced with complete RPMI 1640 containing PBMC and anti-CD3/CD28 beads where appropriate. Cells were harvested on days 2, 3, 4, 5, or 6, and CFSE dilution was analyzed by flow cytometry. The actual numbers of CFSE divided cells were calculated using counting beads (BD Biosciences).
Transwell assays
Transwell assays were carried out in 24-well tissue-culture grade plates (Corning). Mesoangioblasts were seeded out at 6.25×104/well overnight as earlier. The medium was then carefully removed and replaced by complete RPMI 1640. 0.4 μM transwell inserts (Corning) were gently inserted into the wells, and PBMCs (2.5×105/well) +/− anti-CD3/CD28 beads (0.5×105/well) were added directly to the insert. As a control, PBMC +/− anti-CD3/CD28 beads were also added directly to the wells without transwells at the same concentrations as described earlier.
Flow cytometric analysis
Mesoangioblasts were characterized for the following phenotypic surface markers: HLA-ABC, HLA-DR (BD Biosciences), CD105, CD73, CD49b (eBioscience), and CD146 (Biocytex). In addition, mesoangioblast expression of CD40, CD80, CD86, CTLA-4 (BD Biosciences), and PD-L1 (eBioscience) as well as the HLA molecules were analyzed before and after stimulation (24 h) with the pro-inflammatory cytokines interferon (IFN)-γ, TNF-α, or interleukin (IL)-1β (Peprotech). The effect of mesoangioblasts on the expression of early activation markers on T cells was analyzed by flow cytometry. Cells were harvested on days 3, 4, 5, and 6 and stained with anti-CD3, anti-CD25, anti-CD69, and 7AAD.
Intracellular cytokine staining was used to determine the number of cells producing cytokines in the presence or absence of mesoangioblasts. On days 3, 4, 5, or 6, cells were stimulated with phorbol myristate acetate (PMA) (Sigma-Aldrich) (100 ng/mL) and ionomycin (Sigma-Aldrich) (1 μg/mL) in the presence of golgi stop (eBioscience) (3 μL/mL) to block cytokine secretion for 4 h. Cells were then harvested and stained for surface marker expression of CD3 and 7AAD for 30 min at 4°C before washing. Subsequently the cells were fixed/permeabilised using a commercial kit (eBioscience) for 45 min at 4°C. After washing, the cells were blocked in 2% rat serum for 15 min followed by staining with anti-IFN-γ and anti-IL-2 antibodies (eBioscience) for 30 min at 4°C. The cells were washed and analyzed by flow cytometry using 7AAD and CD3 to gate on live T cells. The controls consisted of isotype control antibodies matched for fluorochrome.
Real-time PCR
Total RNA was isolated from mesoangioblast samples using an Absolute RNA miniprep kit (Stratagene), and reverse transcribed by the AffinityScript Multiple Temperature Reverse Transcriptase (Agilent Technologies) as previously described [28]. Real-time quantification was performed using the ABI Prism 7700 Sequence Detection System (Applied Biosystems) using either the fluorogenic probe [PTGS2 (prostaglandin-endoperoxide synthase 2)]_Taqman Gene Expression Assay from Life Technologies, hypoxanthine phosphoribosyl transferase (HPRT), or the SYBR Green technology (IDO and HPRT) as previously described [29]. Samples were standardized for HPRT, and quantification of the gene of interest was given by 2−ΔCT, where CT is obtained by calculating the difference between CT of the gene of interest and HPRT.
ELISA
IFN-γ, IL-2 (eBioscience) ELISA and PGE-2 competitive ELISA (R&D Systems) were performed according to the manufacturer's instructions.
Neutralizing and blocking studies
Proliferation assays in the form of [3H]-thymidine incorporation or CFSE dilution were carried out as described earlier in the presence or absence of neutralizing antibodies to IFN-γ (R&D Systems), TNF-α (eBioscience), isotype control (R&D Systems), or recombinant IL-1 receptor antagonist (IL-1RA) (eBioscience) at concentrations of 0.5, 1.0, and 2.0 μg/mL. Similarly, inhibitors of IDO; -1-Methyl-L-tryptophan (1MT) (Sigma-Aldrich) (0.5 mM), Cox-2; indomethacin (Indo) (Sigma-Aldrich) (10 μM), NS-398 (NS3) (Calbiochem) (0.1, 1.0 μM), inducible nitric oxide synthase (iNOS); L-NMMA (L-NM) (Sigma-Aldrich) (1 mM) and matrix metalloproteinases (MMP)-2/9; and SB-3CT (SB3) (Calbiochem) (6 μM) were utilized in these assays.
Statistical methods
Data were analyzed using the statistical software Prism (version 5; GraphPad Software) and are reported as mean±standard error (SE). The unpaired Student's t-test was performed to compare 2 mean values. Otherwise, data were analyzed using a paired Student's t-test or a two-way ANOVA with Bonferonni's post–test, and p-values <0.05 were considered statistically significant.
Results
Mesoangioblasts fail to induce T-cell proliferation in vitro
Mesoangioblasts from 3 individuals were characterized by flow cytometry. All 3 donors shared a similar pattern of expression of cell surface molecules that was typical of human mesoangioblasts (Supplementary Fig. S1; Supplementary Data are available online at
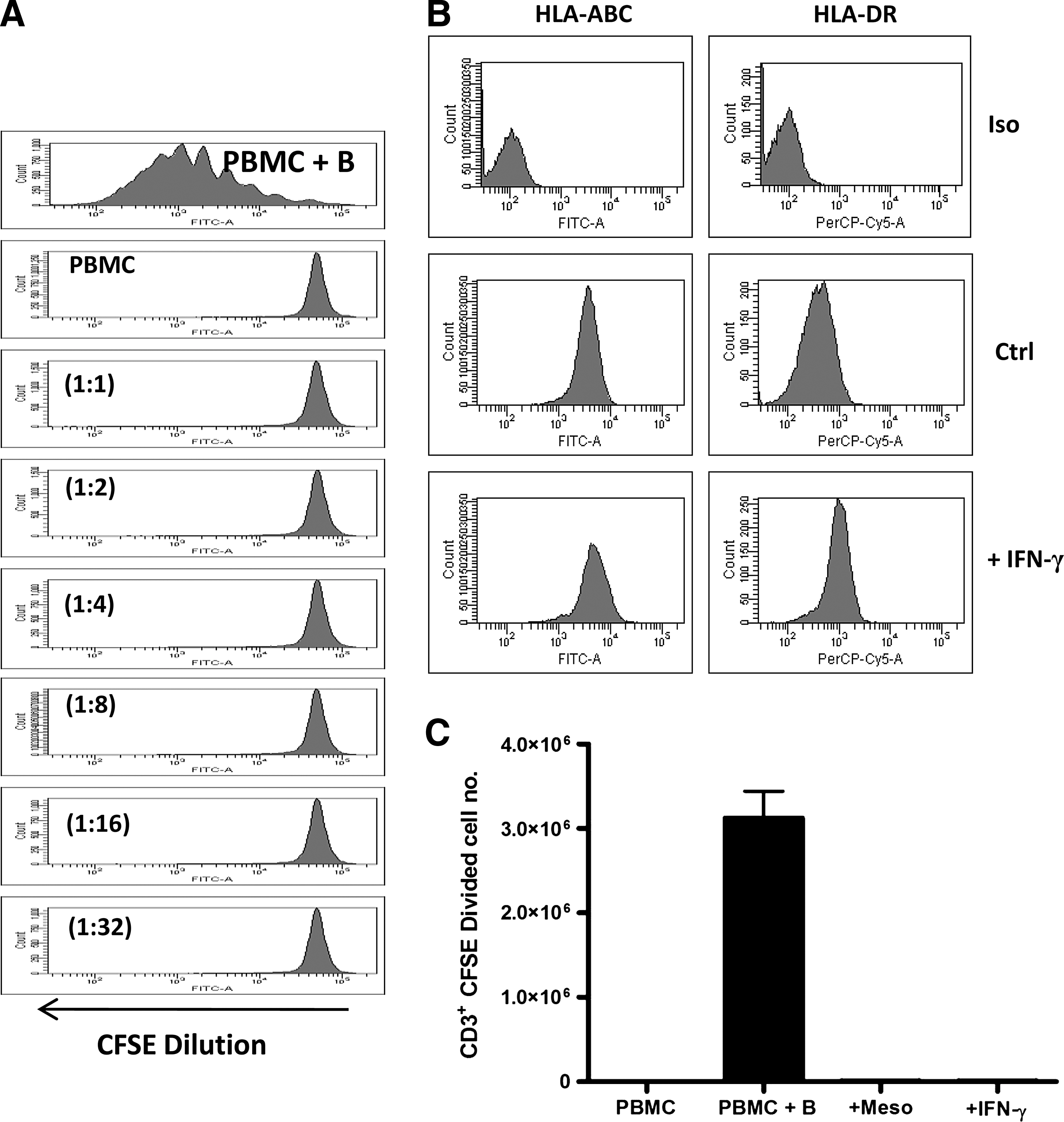
Allogeneic mesoangioblasts fail to induce T-cell proliferation in vitro. 5,6 carboxyfluorescein diacetate succinimidyl ester (CFSE)-labeled peripheral blood mononuclear cells (PBMCs) were cultured in the presence or absence of mesoangioblasts at varying ratios [Mesoangioblast: PBMC (1:1 to 1:32)]. CFSE-labeled PBMCs were stimulated with anti-CD3/CD28 beads (PBMC+B) as a positive control. On day 6, cells were harvested and analyzed for CFSE dilution gating in CD3+ 7AAD− cells
Characterization of mesoangioblast expression of HLA, costimulation, and coinhibitory molecules
Efficient activation of T cells requires the expression of costimulatory molecules. A number of nonprofessional antigen presenting cells that express costimulatory as well as coinhibitory molecules, and pro-inflammatory cytokines are known to facilitate the up-regulation of such molecules. Mesoangioblasts constitutively express neither the costimulatory molecules CD40, CD80, and CD86 nor the coinhibitory molecules CTLA-4 or PD-L1 (Supplementary Fig. S2 and Fig. 2A). However, the expression of HLA-ABC, HLA-DR, and PD-L1 and low levels of CD40 were either increased or induced in mesoangioblasts after stimulation with IFN-γ, but not TNF-α or IL-1β or a combination of the latter. In addition, combinations of these cytokines did not significantly increase the expression of these molecules compared with stimulation with IFN-γ alone (Fig. 2). Thus, it seems that under noninflammatory conditions, mesoangioblasts express a hypoimmunogenic phenotype, while in the presence of inflammatory mediators, they display a more immunogenic phenotype, although induced expression of PD-L1 suggests that mesoangioblasts have the potential for suppression of immune responses under inflammatory conditions.
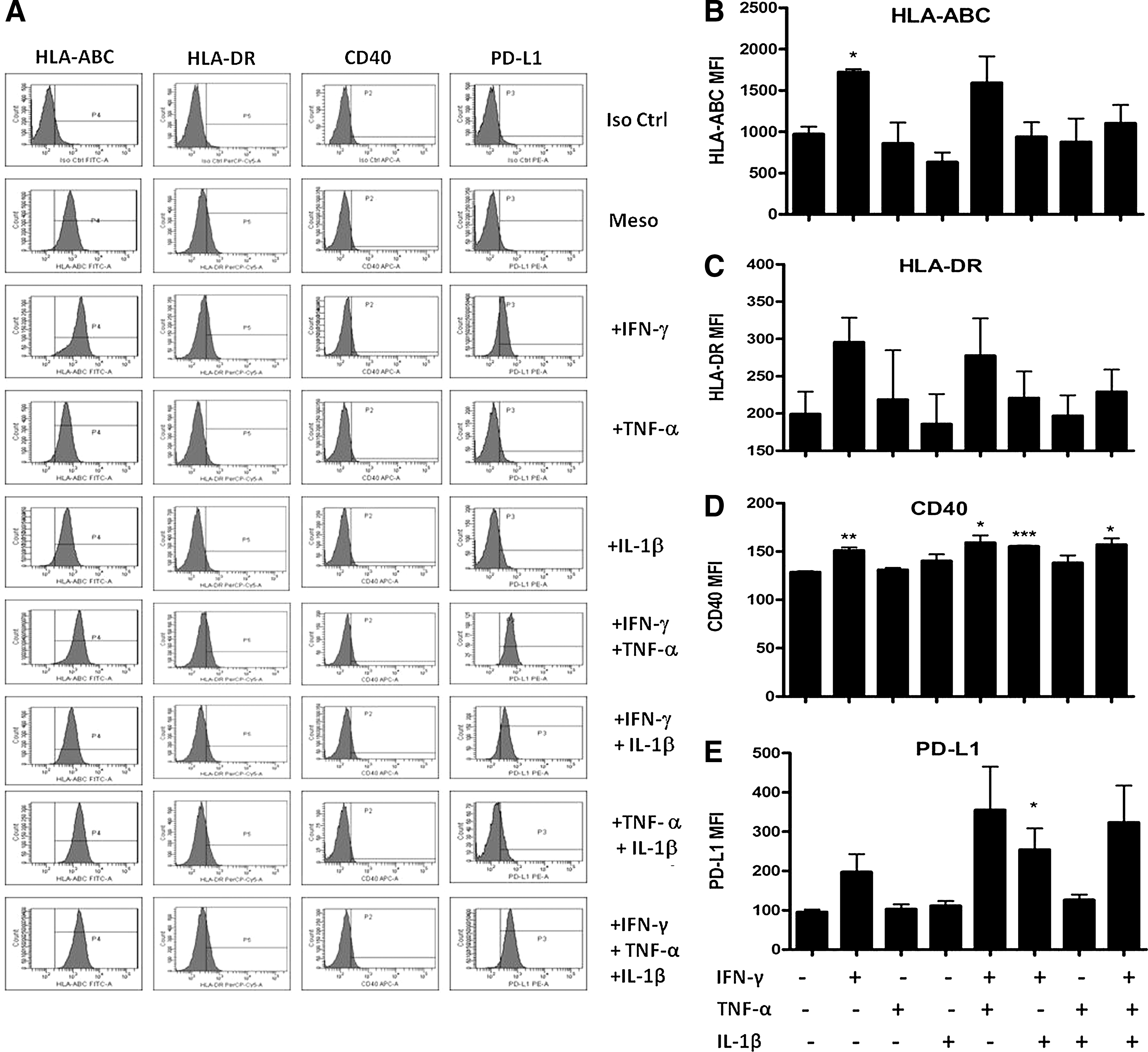
Characterization of mesoangioblast expression of HLA and costimulatory/inhibitory molecules. Mesoangioblasts were stimulated with IFN-γ, tumor necrosis factor (TNF)-α, or IL-1β (20 ng/mL) for 24 h. Cells were trypsinised and washed, followed by surface staining for HLA-ABC, HLA-DR, CD40, PD-L1, or fluorochrome-matched isotype controls and analysis by flow cytometry
Mesoangioblasts suppress T-cell proliferation in a dose- and time-dependent manner
Since mesoangioblasts share a similar cell surface phenotype and immunogenicity with MSC, we next sought to determine whether or not mesoangioblasts could exert immune suppressive effects similar to those reported for MSC. Mesoangioblasts potently suppressed CD3+ T-cell proliferation in vitro in a dose-dependent manner with significant inhibition of T-cell proliferation at ratios of 1:1 to 1:4 (mesoangioblast: PBMC) (Fig. 3A). Robust suppression of T-cell proliferation independent of the source of the mesoangioblasts was observed at a ratio of 1:4, and, therefore, this ratio was selected for further experiments.
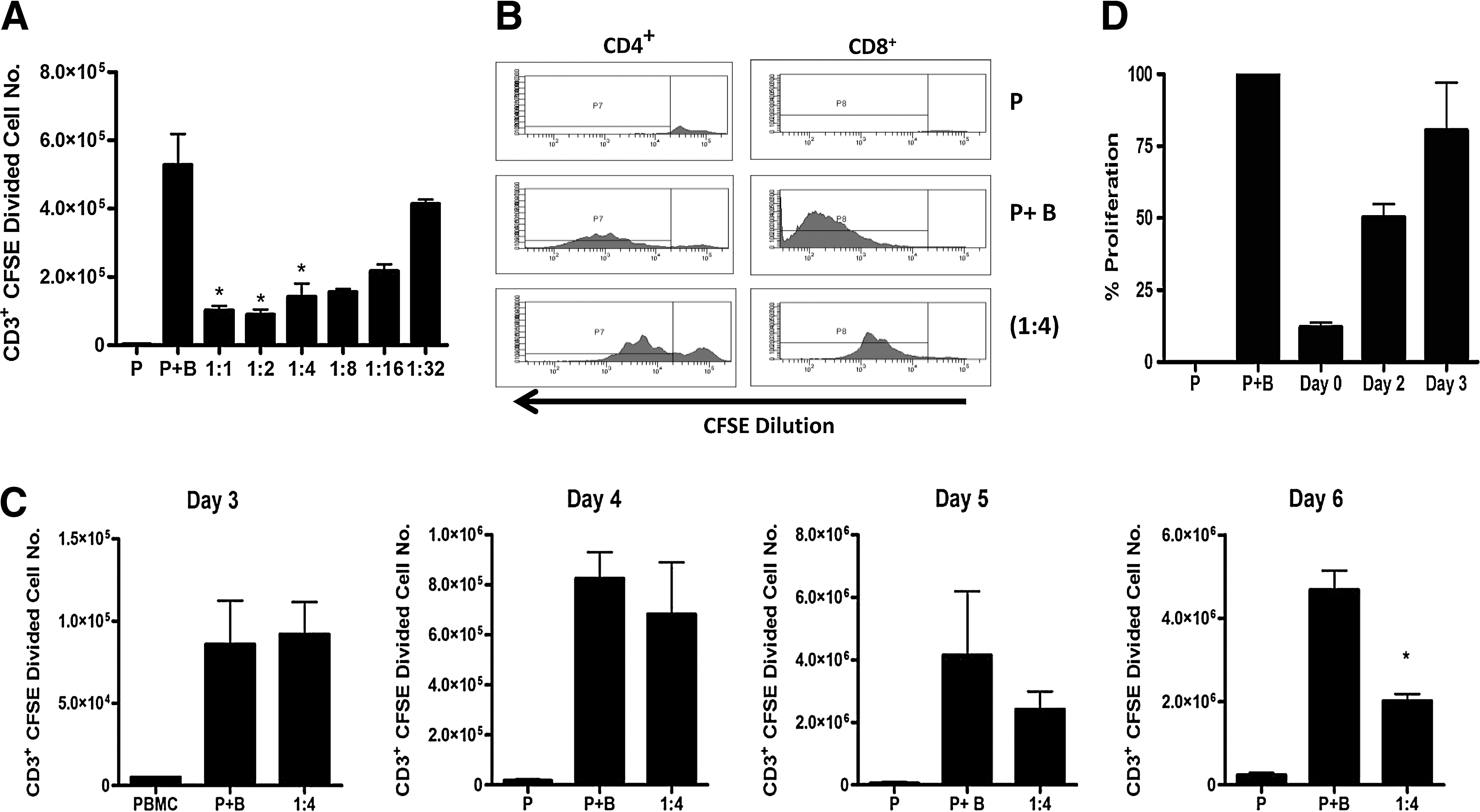
Mesoangioblasts suppress T-cell proliferation in a dose- and time-dependent manner. CFSE-labeled PBMCs (5×104/well) were stimulated with anti-CD3/CD28 beads (1×104/well) (P+B) in the presence or absence of mesoangioblasts at decreasing ratios (mesoangioblast:PBMC). On day 6, cells were harvested and stained with anti-CD3, anti-CD4, anti-CD8, and 7AAD and analyzed by flow cytometry. CFSE dilution was analyzed in gated CD3+ 7AAD− cells, and the absolute number of CD3+CFSE dividing cells was enumerated using counting beads
Significantly, mesoangioblasts suppressed both CD4+ and CD8+ T-cell proliferation (Fig. 3B). In addition, the mesoangioblast inhibition of T-cell proliferation was time dependent with suppression evident on day 4/5 but most significant on day 6 (Fig. 3C). To determine whether mesoangioblasts exert their effects immediately or slowly over time, we carried out a CFSE dilution assay with mesoangioblasts added to the PBMC cultures on days 0, 2, or 3. While mesoangioblasts added to the cultures on day 0 reduced the level of T-cell proliferation to an average of 12%, mesoangioblasts added at later times on days 2 and 3 reduced proliferation to 50% and 80%, respectively (Fig. 3D). Together, these results reveal that mesoangioblasts suppress both CD4+ and CD8+ T-cell proliferation in a dose- and time-dependent manner.
Anergy is a state of unresponsiveness that can be reversed by the addition of IL-2. To test the hypothesis that mesoangioblasts induce T-cell anergy, PBMCs were stimulated with anti-CD3/CD28 beads in the presence or absence of mesoangioblasts separated by a transwell. After 6 days, anti-CD3/CD28 beads were removed, and PBMCs were CFSE labeled and cocultured with either 1×104 or 5×104 allogeneic dendritic cells (DC) in the presence or absence of exogenous IL-2. PBMCs were harvested on day 5 after the second-round stimulation and analyzed for CFSE dilution. PBMCs proliferated in response to allogeneic DC at both concentrations, regardless of whether or not PBMC were exposed to mesoangioblasts in the first round (Supplementary Fig. S3). The addition of exogenous IL-2 resulted in further increased proliferation of T cells again, irrespective of the presence of mesoangioblasts in the first round (Supplementary Fig. S3), demonstrating that mesoangioblasts do not induce a state of anergy in T cells.
Mesoangioblasts inhibit T-cell production of pro-inflammatory cytokines, but do not interfere with T-cell activation
The activation markers CD25 and CD69 are rapidly up-regulated on the surface of antigen-stimulated T cells. To directly test the effect of mesoangioblasts on the expression of these early activation markers, PBMCs were stimulated with anti-CD3/CD28 beads in the presence or absence of mesoangioblasts. In the presence of mesoangioblasts, while the number of proliferating cells decreased, the proportion of cells expressing CD25 and CD69 remained higher relative to that observed in PBMC stimulated in the absence of mesgoangioblasts (Fig. 4A, B). In addition, mesoangioblasts did not reduce the median fluorescence intensity of CD25 or CD69 in proliferating T cells, a finding that is consistent throughout the 4 time points examined (Fig. 4C).
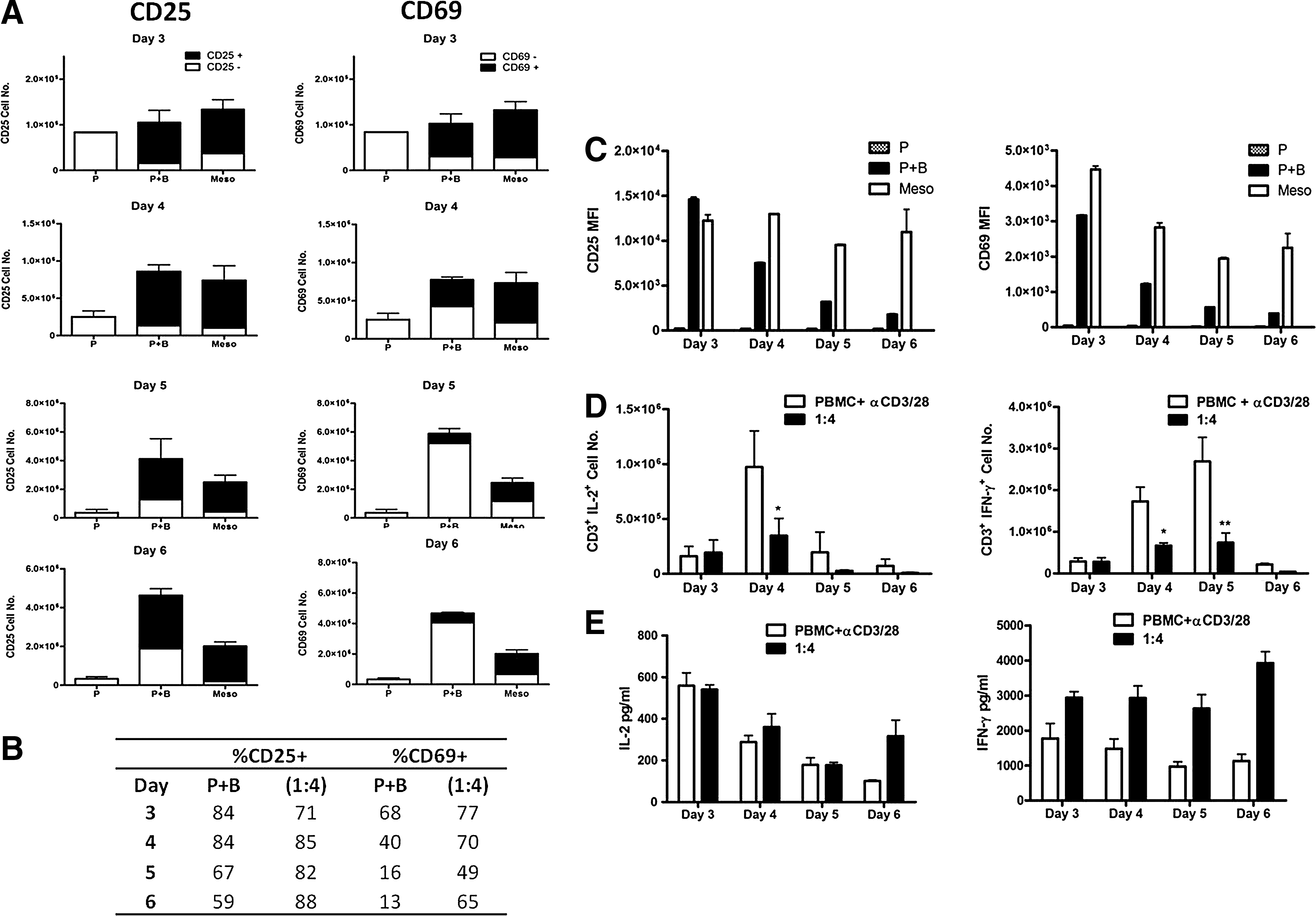
Mesoangioblasts inhibit T-cell production of pro-inflammatory cytokines without interfering with T-cell activation. CFSE dilution assays were performed as earlier in the presence or absence of mesoangioblasts (1:4). Cells were harvested on days 3, 4, 5, or 6 and analyzed for CFSE dilution and expression of CD25 and CD69. The numbers of CD3+7AAD− cells expressing CD25 or CD69 denoted CD25+ or CD69+ (■) or CD25− or CD69− (□) were calculated using counting beads
The production of pro-inflammatory cytokines by T cells is an important effector function. We, therefore, examined the effect of mesoangioblasts on the intracellular production of IL-2 and IFN-γ by stimulated (anti-CD3/CD28) CD3+ T cells. Mesoangioblasts significantly inhibited the production of both IL-2 and IFN-γ by CD3+ T cells at the peak of cytokine production on days 4 and 5, respectively (Fig. 4D). However, mesoangioblasts did not reduce the total production of IL-2 or IFN-γ, as measured in the supernatants of the cocultures by ELISA, and, in fact, increased IFN-γ concentrations (Fig. 4E). These findings highlight the importance of examining the actual number of cells actively producing cytokines versus measuring the sum total of cytokine being produced over time. Taken together, these data suggest that mesoangioblasts modulate some but not all of the cells present within the PBMC population.
Cell contact is not required for mesoangioblast suppression of T-cell proliferation in vitro
Mesoangioblast suppression of T-cell proliferation may be mediated either through a cell-contact-dependent mechanism or via the production of an immune modulatory soluble factor or factors. Cell contact was not required for mesoangioblast suppression of T-cell proliferation in vitro (Fig. 5A, B), and mesoangioblasts that were separated by a transwell were almost as effective at suppressing T-cell proliferation as those in direct contact with PBMCs. These data provide strong evidence for an important role of a soluble factor in the mechanism of action of mesoangioblasts.
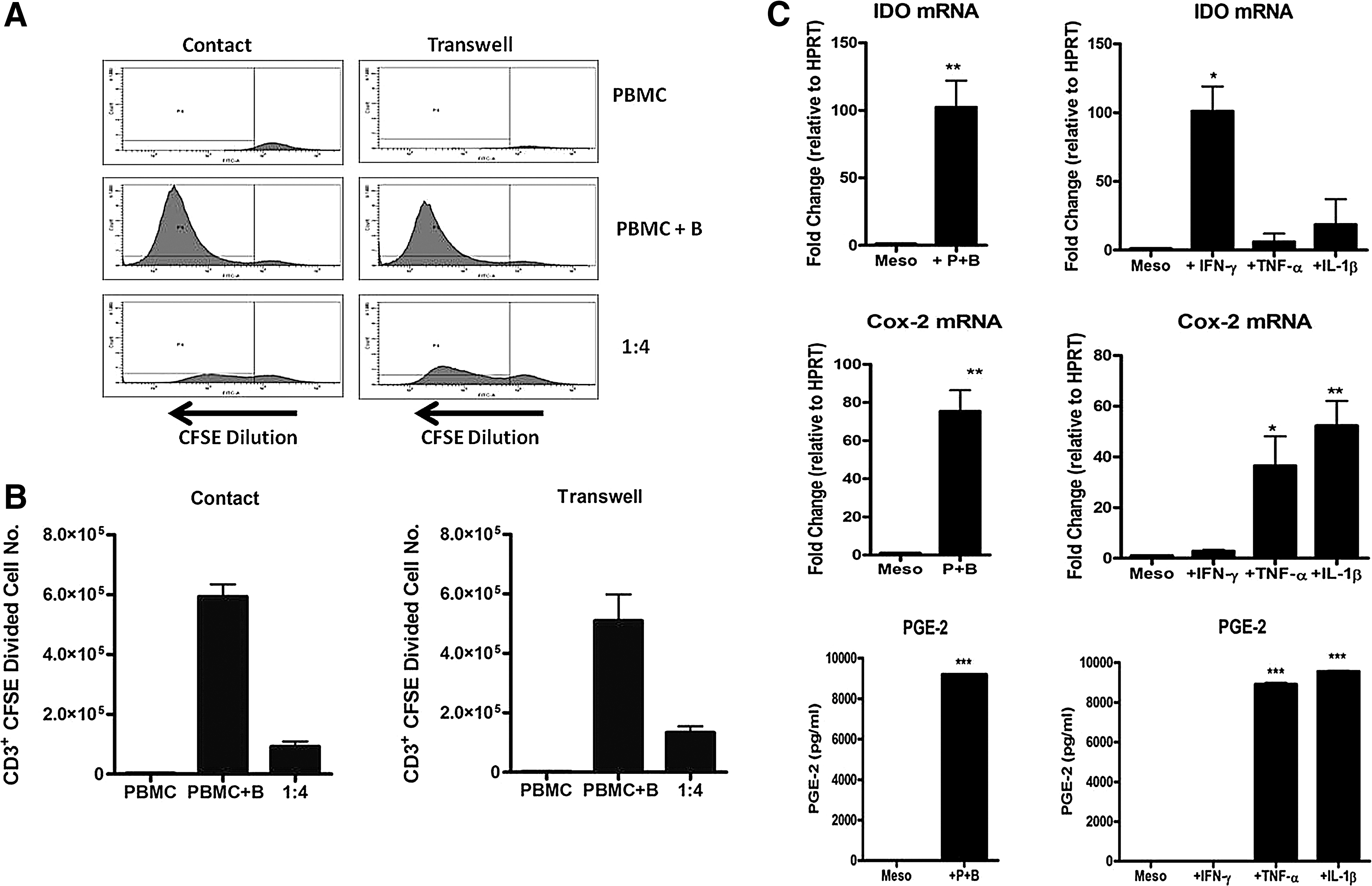
Cell contact is not required for mesoangioblast suppression of T-cell proliferation. Transwell assays were set up in 24-well plates with mesoangioblasts seeded overnight. 0.4 μM inserts were gently inserted into wells containing mesoangioblasts or medium only followed by the addition of CFSE-labeled PBMCs +/− anti-CD3/CD28 beads. For contact assays, CFSE-labeled PBMCs +/− anti-CD3/CD28 beads were added directly to wells containing mesoangioblasts or medium only. Cells were harvested on day 6, and CFSE dilution was analyzed by flow cytometry gating in CD3+7AAD− cells
Two key soluble factors known to play a role in immune modulation and suppression of T-cell proliferation are IDO and PGE-2 [30,31]. We, therefore, examined the magnitude of expression of IDO and Cox-2 (an enzyme required for the production of PGE-2) by mesoangioblasts under basal conditions and after coculture with activated PBMCs or stimulation with IFN-γ, TNF-α or IL-1β. Mesoangioblasts do not constitutively express IDO or Cox-2 mRNA; however, the expression of both enzymes could be dramatically increased after coculture with activated PBMC (Fig. 5C). IFN-γ, TNF-α, and IL-1β differentially regulated the expression of IDO and Cox-2 by mesoangioblasts with IDO induced by IFN-γ but not TNF-α or IL-1β and Cox-2 induced by TNF-α or IL-1β but not IFN-γ (Fig. 5C). The production of PGE-2 by mesoangioblasts was measured by competitive ELISA and followed a similar pattern to Cox-2 (Fig. 5C). Significantly, other pro-inflammatory cytokines, such as IL-6 and TGF-β1, had no effect (data not shown). Together, these data demonstrate that direct cell-cell contact is not required for mesoangioblast suppression of T-cell proliferation and provide evidence for a pivotal role of pro-inflammatory cytokine induction of the immunomodulatory molecules IDO and PGE-2 by mesoangioblasts.
IFN-γ and TNF-α play important roles in the suppression of T-cell proliferation by human mesoangioblasts
Since IFN-γ, TNF-α, and IL-1β have been identified as important regulators of the immunomodulatory molecules IDO and PGE-2 (Fig. 5), we next sought to determine the importance of these cytokines in mesoangioblast suppression of T-cell proliferation in vitro. Neutralizing antibodies against IFN-γ and TNF-α at concentrations of 1.0 and 2.0 μg/mL but not 0.5 μg/mL partially but significantly abolished the inhibitory effect mediated by mesoangioblasts (Fig. 6A). IL-1RA (1.0 μg/mL) also partially restored proliferation, although the difference was not significant. In addition, combinations of IFN-γ and TNF-α or all 3 also partially restored T-cell proliferation, but the effect was no greater than when either anti-IFN-γ or TNF-α (2.0 μg/mL) was used alone (Fig. 6A).
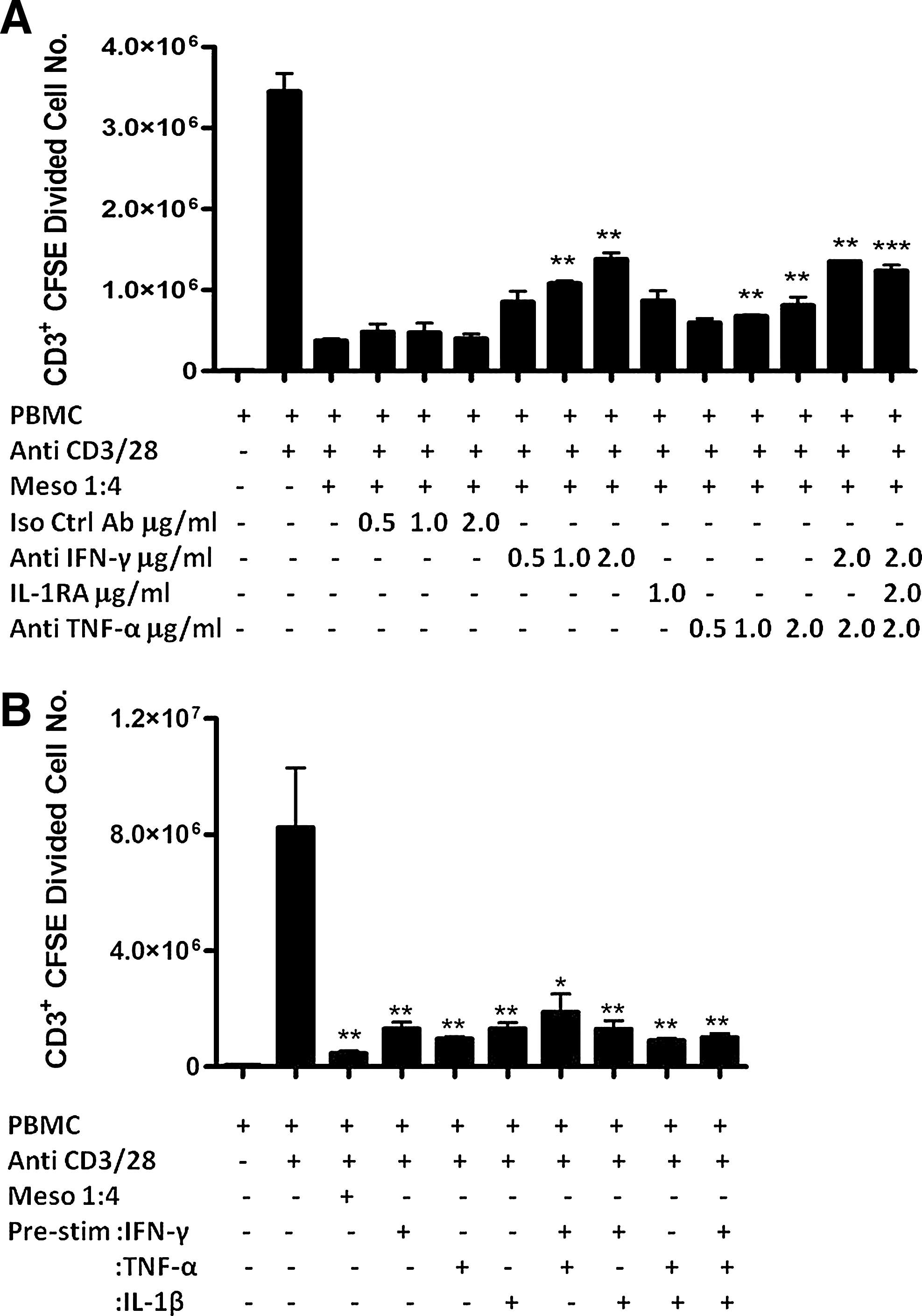
IFN-γ and TNF-α play important roles in mesoangioblast suppression of T-cell proliferation, but prestimulation with these cytokines does not enhance their effect. CFSE-labeled PBMCs were stimulated with anti-CD3/CD28 beads in the presence of mesoangioblasts (1:4) and neutralizing antibodies against IFN-γ and TNF-α or irrelevant isotype control antibody (0.5, 1.0, and 2.0 μg/mL) or recombinant IL-1 receptor antagonist (IL-1RA) (1.0 μg/mL). Cells were harvested on day 6 and stained with anti-CD3 and 7AAD. After gating on CD3+7AAD−, the numbers of CFSE diluting cells were enumerated using counting beads. Data are represented as mean CD3+ CFSE diluted cell number±SE
IFN-γ is a key cytokine that is required for the suppression of T-cell proliferation which is mediated by MSC, and studies have demonstrated the importance of IFN-γ in the activation of MSC to exert their immunosuppressive effects [32 –34]. Moreover, it has been shown that prestimulation of MSC with IFN-γ enhances their immune modulatory capacity both in vitro and in vivo [33,35]. Given our data demonstrating that mesoangioblasts require activation by the pro-inflammatory cytokines IFN-γ, TNF-α, and IL-1β to produce immunomodulatory molecules and to suppress T-cell proliferation, we hypothesized that prestimulation with these cytokines would enhance mesoangioblast suppression of T-cell proliferation in vitro. To test our hypothesis, we stimulated mesoangioblasts with IFN-γ, TNF-α, IL-1β, or combinations of these cytokines for 24 h before adding them to CFSE dilution assays to examine their immunosuppressive capacity. Prestimulation did not promote the suppressor capacity of mesoangioblasts, compared with unstimulated controls, but slightly increased the level of proliferation by CD3+ T cells (Fig. 6B). However, there was no significant loss of suppressor capacity by mesoangioblasts after stimulation with pro-inflammatory cytokines.
IDO and PGE-2 play nonredundant roles in mesoangioblast suppression of T-cell proliferation
Although cell-cell contact was not required for mesoangioblast suppression of T-cell proliferation, the significant increase in PD-L1 expression on mesoangioblasts after stimulation with IFN-γ (Fig. 2) warranted an examination of the importance of PD-L1 in the mechanism of action of mesoangioblasts. Neutralizing PD-L1 had no effect on mesoangioblast suppression of T-cell proliferation (Supplementary Fig. S4).
A number of soluble immunosuppressive factors, including IDO [31], PGE-2 [30], iNOS [36], and MMPs [37], have been demonstrated to modulate T-cell proliferation. To investigate the relevance of these immunomodulatory soluble factors in mesoangioblast suppression of T-cell proliferation, chemical antagonists were added to CFSE dilution assays in the presence of mesoangioblasts. The addition of 1-Methyl-L-tryptophan (an inhibitor of IDO) or indomethacin (a Cox-1 and 2 inhibitor) to cultures containing mesoangioblasts partially but significantly restored proliferation, with 1MT having the most potent effect (Fig. 7A). Importantly addition of these inhibitors to PBMC in the absencse of mesoangioblasts did not interface with PBMC proliferation (Supplementary Fig. S5). The specific Cox-2 inhibitor NS-398 (1.0 μM) restored proliferation to the same extent as indomethacin at a concentration of 10 μM (Supplementary Fig. S6); however, antagonists against iNOS (L-NMMA) and MMP-2/9 (SB-3CT) had no effect (data not shown). Earlier, we demonstrated that mesoangioblasts modulate both CD4+ and CD8+ T-cell proliferation in vitro. To further characterize mesoangioblast immunomodulation, we examined the mechanisms of action involved in the suppression of CD4+ and CD8+ T cells. Indeed, 1MT and indomethacin restored the proliferation of both T-cell subsets in part, while the addition of these antagonists in concert bestowed an additive effect and completely abolished mesoangioblast suppression of proliferation in both CD4+ and CD8+ T cells (Fig. 7A, B).
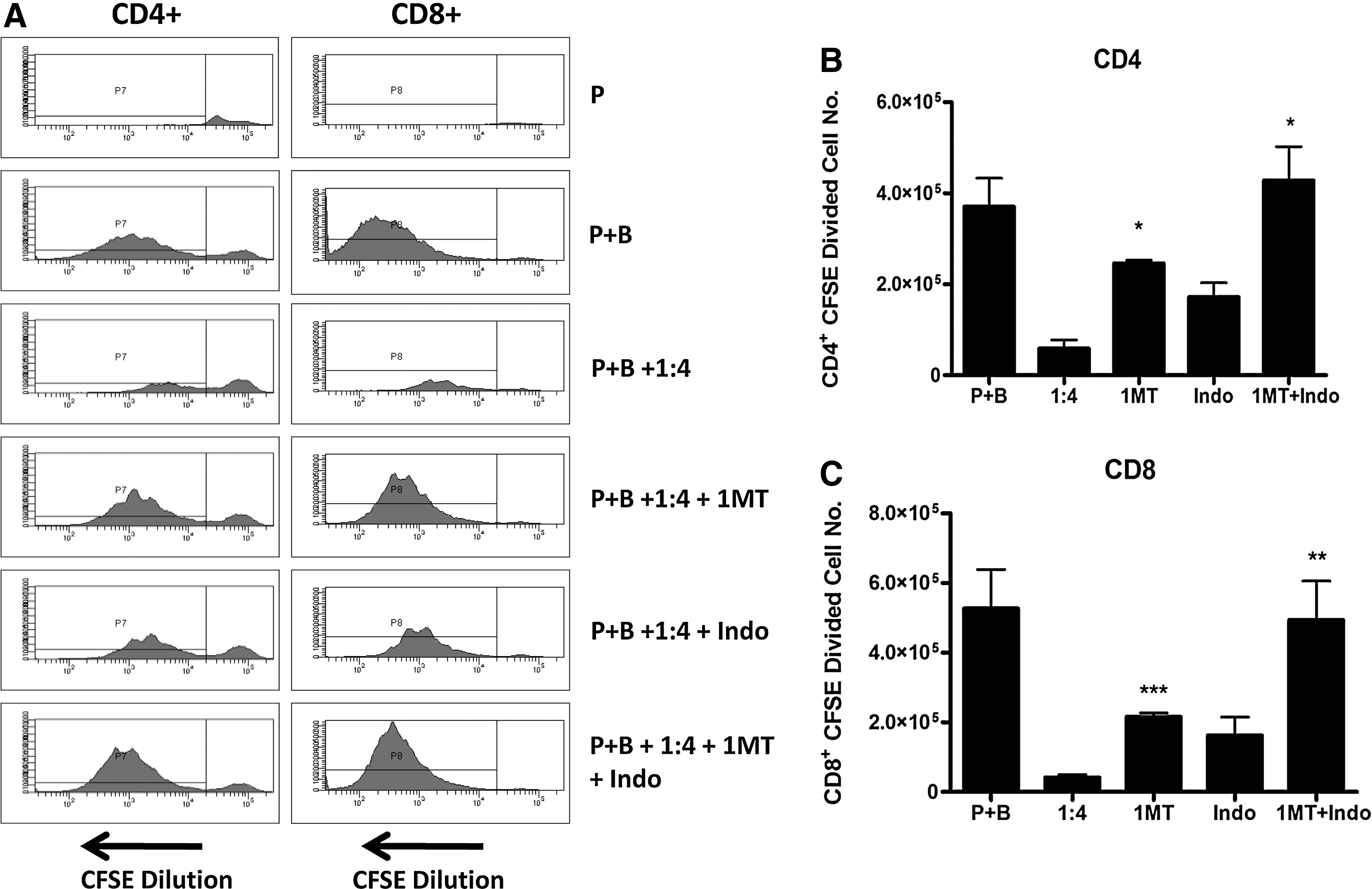
IDO and PGE-2 are involved in mesoangioblast suppression of T-cell proliferation. CFSE-labeled PBMCs were stimulated with anti-CD3/CD28 beads (P+B) in the presence of mesoangioblasts (1:4 ratio) with or without inhibitors of IDO and Cox-2 [1-Methyl-L-trypyophan (1MT) (0.5 mM), indomethacin (Indo) (10 μM)] or both. On day 6, cells were harvested and stained with anti-CD3, anti-CD4, anti-CD8, and 7AAD. Cells were gated on live CD4+ or CD8+ populations and analyzed for CFSE dilution
Discussion
Understanding the immunogenicity of allogeneic mesoangioblasts is critically important if this cell therapy strategy is to be successfully used to treat patients with DMD. Previous data have suggested that allogeneic mesoangioblasts may be immune privileged, although, after transplantation in alpha-sarcoglycan null mice, allogeneic mesoangioblasts or differentiated muscle fibers derived from mesoangioblasts were found to be more immunogenic [38]. The immunogenicity of mesoangioblasts and the interactions between these cells and immune cells under inflammatory conditions require careful investigation. Here, we have characterized the immunogenicity of human mesoangioblasts and have demonstrated that these cells do not evoke T-cell proliferation in vitro even after prestimulation with IFN-γ, a cytokine likely to be present at the site of implantation, which results in the increased expression of HLA-ABC, HLA-DR, and a low level expression of the costimulatory molecule CD40. Interestingly, IFN-γ but not TNF-α or IL-1β stimulation also induced the expression of PD-L1, an inhibitory molecule. Thus, the data suggest that while mesoangioblasts theoretically have the machinery required for nonself antigen presentation, they fail to induce allogeneic T-cell proliferation in vitro, and the expression of PD-L1 may play a role in facilitating their lack of immunogeneicity. Next, we investigated the effect of mesoangioblasts on T-cell proliferation in vitro. For the first time, we show that mesoangioblasts potently suppress T-cell proliferation driven by anti-CD3/CD28 stimulation in a dose- and time-dependent manner. Specifically, mesoangioblasts significantly inhibited T-cell proliferation at ratios of 1:1 to 1:4 but not 1:8. In addition, mesoangioblasts added to stimulated PBMC cultures on day 0 and day 2 inhibited proliferation by ∼80% and 50%, respectively; whereas the addition of mesoangioblasts on day 3 resulted in ∼10%–20% suppression of T-cell proliferation.
Both CD4+ and CD8+ T cells are known to contribute to the destructive immune response that can be attributed to allogeneic cells. Significantly, mesoangioblasts were found to exert their immunosuppressive effect on both CD4+ and CD8+ T cells, indicating that they have the capacity to prevent an immune attack. Although mesoangioblasts significantly inhibited T-cell proliferation, they did not reduce the number of T cells expressing the early activation markers CD25 or CD69, a phenomenon also seen with MSC [34]. Nevertheless, a significant decrease in the number of IL-2+ and IFN-γ+ T cells after coculture with mesoangioblasts was observed at the peak of cytokine production. In contrast, mesoangioblasts did not reduce the total amount of IL-2 and IFN-γ produced over time on days 3, 4, 5, or 6, and it may be that the proportion of T cells that are not affected (in terms of proliferation) by mesoangioblasts may compensate and produce relatively more cytokines. Alternatively, cells other than T cells within the PBMC population may be responsible for the differences observed in cytokine production. Interestingly, the suppression of T-cell proliferation induced by mesoangioblasts was demonstrated to be reversible, as PBMCs that were restimulated with alloantigen could proliferate in the absence of IL-2. Collectively, mesoangioblasts exert their inhibitory effects predominantly at the level of T-cell proliferation and partially on T-cell effector function with direct effects on T-cell cytokine production, while T-cell activation is spared. Notably, these effects are reversible and do not involve the induction of T-cell anergy.
Mesoangioblast suppression of T-cell proliferation does not require cell contact, indicating that soluble factors are the likely mediators. IDO and PGE-2 have been demonstrated to play important roles in the immune modulation of MSC [32]. Mesoangioblasts were found to dramatically up-regulate IDO and Cox-2 and to produce high levels of PGE-2 after coculture with stimulated PBMCs. Similarly, IDO expression by mesoangioblasts was induced after stimulation with IFN-γ but not TNF-α or IL-1β. In contrast, Cox-2 expression and PGE-2 production was induced by TNF-α or IL-1β, but not IFN-γ. Partial but significant restoration of proliferation was observed in neutralizing studies using anti-IFN-γ, anti-TNF-α, or a combination of these with/without IL-1RA. These data suggest that IFN-γ and TNF-α are indeed key soluble factors which are involved in the suppression of T-cell proliferation by mesoangioblasts, but other factors may also play a role. Interestingly, prestimulation with these cytokines did not enhance mesoangioblast suppression of T-cell proliferation, and while there was no significant difference between un-stimulated and stimulated mesoangioblasts in their ability to suppress T-cell proliferation, the number of T cells proliferating was increased. It is unclear why these differences exist between mesoangioblasts and MSC with regard to prestimulation with IFN-γ in particular. Perhaps the key to the differences observed lies in the origin of these 2 cell types (muscle vs. bone marrow) and the types of environments that these cells are exposed to.
While an essential role for cell contact was ruled out using transwell assays, the importance of PD-L1 expression in mesoangioblasts induced by IFN-γ in mesoangioblast suppression of T-cell proliferation was unclear. Neutralizing studies demonstrated that PD-L1 was not required for mesoangioblast suppression of T-cell proliferation. Nevertheless, the role of PD-L1 in activated (+IFN-γ) mesoangioblasts remains to be elucidated. Significantly, the soluble immunomodulatory factors IDO and PGE-2 were identified as fundamental players in mesoangioblast suppression of T-cell proliferation. A combination of both 1MT and indomethacin abolished mesoangioblast inhibitory effects on T-cell proliferation, and these data are consistent with studies involving MSC [32].
Conclusion
Overall, we have demonstrated that mesoangioblasts interfere with T-cell proliferation and effector function, a phenomenon that is reversible. The mechanisms involved require an initial activation of mesoangioblasts driven by IFN-γ, TNF-α, or IL-1β followed by downstream secretion of IDO and PGE-2, which mediate the effects observed.
Mesoangioblast cell therapy will encounter an inflammatory milieu in vivo. Evidence suggests that signals arising from local damage within inflammatory infiltrates attract mesoangioblasts. Specifically TNF-α and HMGB1 secreted by inflammatory macrophages have been shown to initiate mesoangiobalst recruitment [39,40]. Furthermore, it has been demonstrated that prestimulation of mesoangioblasts with TNF-α significantly enhances their migration to the site of muscle damage [41]. We have shown that exposure to pro-inflammatory cytokines does not significantly impair, but, in fact, plays important roles in mesoangioblast-mediated suppression of T-cell proliferation in vitro through the initial activation of mesoangioblasts and downstream induction of immunomodulatory soluble factors. Thus, should it prove necessary to prestimulate mesoangiobalsts with TNF-α to enhance migration to damaged muscle, the intrinsic immunosuppressive characteristics of mesoangioblasts would not be impaired. Our data suggest that once recruited to the sight of muscle damage/degeneration, mesoangioblasts will not only participate in muscle regeneration but may also enhance regeneration and muscle repair through paracrine effects on immune cells involving the modulation of T-cell proliferation and effector function. Although the results from this study are promising, our previous work suggests that allogeneic mesoangioblasts will be subject to allogeneic rejection mechanisms in the absence of immunosuppression [38]. Data from this previous study demonstrated that allogeneic mesoangioblasts engrafted and gave rise to α-Sarcoglycan expressing muscle fibers in dystrophic mice, and these fibres were observed for up to 3 months in the absence of immunosuppression. Significant immune reactivity to the allogeneic cells was present as measured through in vitro proliferation assays. However, the level of immune reactivity was not consistent with the significant engraftment and survival time of the allogeneic cells. It was hypothesized that while mesoangioblasts that differentiated and gave rise to muscle fibers were immunogenic, undifferentiated mesoangiobalsts exerted some level of immune suppression/modulation, leading to a prolonged survival time [38]. It should be noted that immune responses to allogeneic cells differ between rodents and primates [42], and, thus, results obtained in mice may not be fully predictive of the fate that human mesoangioblasts will undergo when transplanted in vivo. Although we have not examined the immune suppressive capacity of mesoangiobalsts in vivo, the data we have generated in vitro are consistent with the aforementioned study, and support the idea that mesoangioblasts may function in muscle repair on 2 fronts through direct regeneration and paracrine effects on the inflammatory environment. In addition, this study highlights novel potential uses for mesoangioblasts as an immunomodulatory cell therapy.
Footnotes
Acknowledgments
This research was supported by the European Community grant OptiStem. Karen English was supported in part by a Marie Curie Intra-European Fellowship for career development. The authors thank Kate Milward and Ou Li for technical assistance with flow cytometry assays.
Author Disclosure Statement
No competing financial interests exist.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
