Abstract
In pluripotent stem cells (PSCs), there are 2 types: naive and primed. Only the naive type has the capacity for producing chimeric offspring. Mouse PSCs are naive, but human PSCs are in the primed state. Previously reported porcine PSCs appear in the primed state. In this study, putative naive porcine-induced pluripotent stem cells (iPSCs) were generated. Porcine embryonic fibroblasts were transduced with retroviral vectors expressing Yamanaka's 4 genes. Emergent colonies were propagated in the presence of porcine leukemia inhibitory factor (pLIF) and forskolin. The cells expressed pluripotency markers and formed embryoid bodies, which gave rise to cell types from all 3 embryonic germ layers. The naive state of the cells was demonstrated by pLIF dependency, 2 active X chromosomes (when female), absent MHC class I expression, and characteristic gene expression profiles. The porcine iPSCs contributed to the in vitro embryonic development (11/24, 45.8%) as assessed by fluorescent markers. They also contributed to the in utero fetal development (11/71, 15.5% at day 23; 1/13, 7.7% at day 65). This is the first demonstration of macroscopic fluorescent chimeras derived from naive-like porcine PSCs, although adult chimeras remain to be produced.
Introduction
P
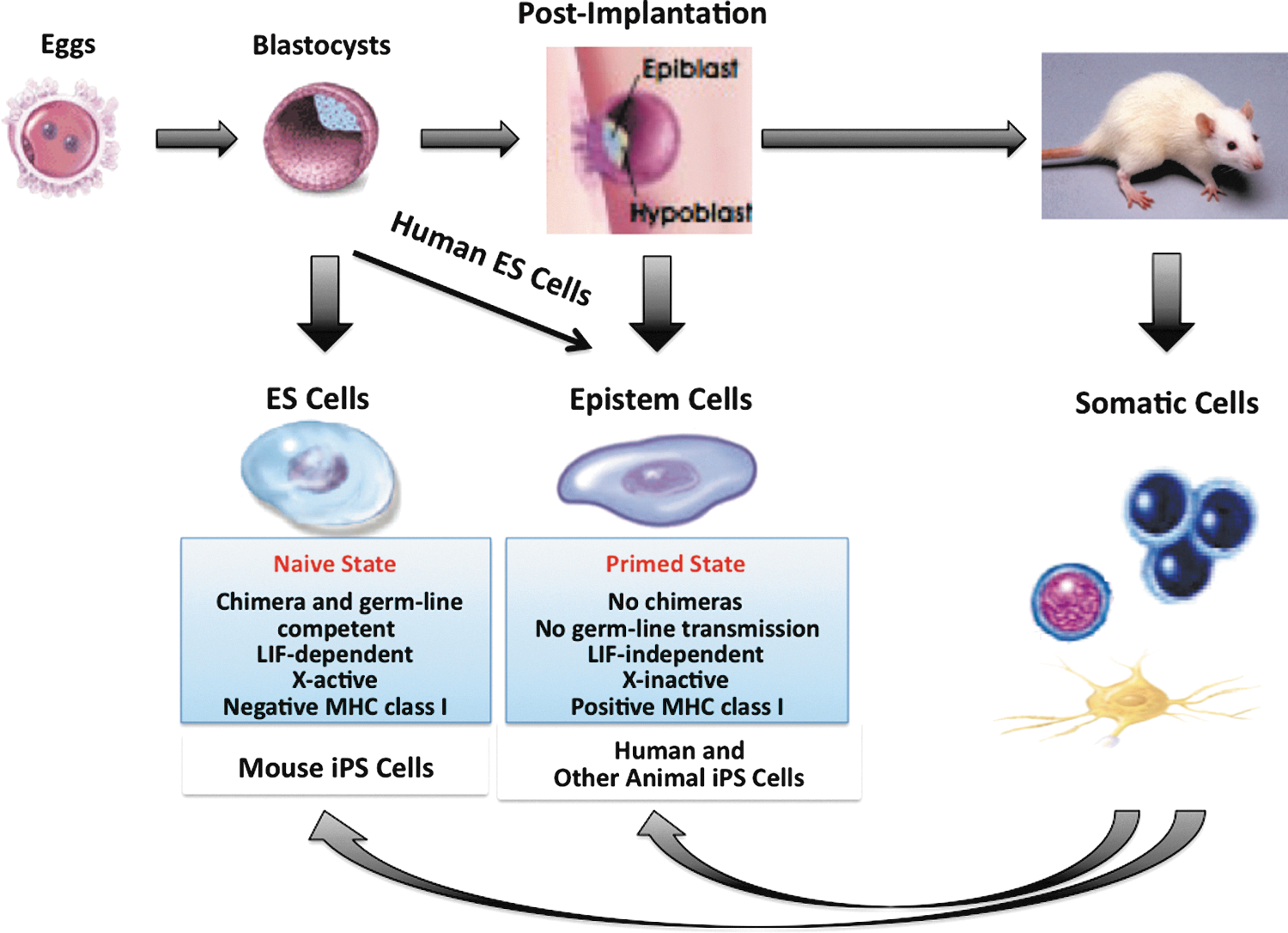
Naive and primed pluripotent stem cells. In mice, embryonic stem cells (ESCs) are derived from the preimplantation of inner cell mass (ICM) of blastocysts and can produce chimeras with germline transmission (naive state). Epistem cells are derived from the postimplantation of epiblasts. Epistem cells are also pluripotent, but they fail to respond to leukemia inhibitory factor (LIF) or to contribute to chimeric offspring (primed state). In humans, although ESCs are derived from the preimplantation of blastocysts, they are similar to epistem cells in their characteristics. Regarding induced pluripotent stem cells (iPSCs), mouse ones are naive, but human and other animal ones are primed. However, in pigs, non-naive or even nonpluripotent cells can also contribute to the fetal and adult development for some reason [18 –20].
In contrast, primed PSCs have very limited capacity to contribute to chimeric offspring. They form flat colonies. They have a proliferative response to basic fibroblast growth factor (bFGF) and activin instead of LIF, and have an inactivated X chromosome (XaXi). They are positive for MHC class I antigen expression. Human PSCs, both ESCs and iPSCs [3 –5], share defining features with mouse epistem cells derived from the postimplantation of epiblasts, and they appear in the primed state [1,2,6]. Previously reported PSCs, either ESCs or iPSCs, derived from other animals such as monkeys [7 –11], pigs [12 –15], and rabbits [16,17] also appear in the primed state, because these animal PSCs usually have a very limited capacity to contribute to chimeric offspring. However, it has been reported that non-naive or even nonpluripotent cells can also contribute to the fetal and adult development in pigs [18 –20].
In rats and nonpermissive mice, naive PSCs can be generated with LIF and 2 signal transduction inhibitors (2i, mitogen-activated protein kinase/extracellular signal-regulated kinase inhibitor PD0325901 and glycogen synthase kinase 3β inhibitor CHIR99021) [21 –24]. Recent studies have shown that human PSCs (ESCs and iPSCs) in the primed state can be converted to the naive-like state by cultivation with 2i, LIF, and forskolin [25,26]. The sustained expression of the exogenous Oct3/4, Sox2, Klf4, c-Myc, and Nanog genes would also induce the naive-like state in human PSCs [27]. Because it is impossible to test the ability of human PSCs to develop into chimeras, testing in other animal models is required. Here, the isolation of putative naive porcine iPSCs, which have characteristics very similar to mouse naive PSCs, are shown. In addition, they have the capacity to contribute to fetal development.
Materials and Methods
Pigs
The humanized Kusabira-orange (huKO) transgenic pigs (Porco Rosso) were bred at the Meiji University (Kanagawa, Japan) [28]. The inbred Clawn miniature pigs (Porco Clawn) were purchased from Japan Farm (Kagoshima, Japan). Crossbred (Large White/Landrace×Duroc) prepubertal gilts were used as surrogate mother pigs for allogeneic embryos. All studies involving pigs and mice were approved by the institutional animal care and use committees at Jichi Medical University and Meiji University.
Porcine LIF
Total RNA was isolated from porcine embryonic fibroblasts (PEFs) and was reverse-transcribed by using Super-Script III reverse transcriptase (Invitrogen, Rockville, MD) and oligo (dT) primers (Takara Bio, Shiga, Japan) to generate cDNA. Polymerase chain reaction (PCR) was then performed to amplify the porcine LIF (pLIF) cDNA using the Ex Taq kit (Takara Bio) and specific primers; the sense (5′-GCC CTC TGG AGT TCA GCC CAT AAT G-3′) and reverse primers (5′-GGT GCT AGG GGA CCT TCC ATC TAG-3′) were designed on the basis of pLIF cDNA sequence (GenBank accession no. AY585336.1). The amplified cDNA fragment (609 bp) was inserted into the TA cloning vector. After digestion with EcoRI and SacI (Takara Bio), the cDNA was inserted downstream of the chicken β-actin-based promoter (CAG) in pPyCAG to create the pLIF expression vector. This vector was transfected into 293FT cells by using lipofectamine 2000 (Invitrogen) and conditioned medium was collected, filtrated, and stored at −70°C until use. The conditioned medium was used at 1:200, corresponding to 5000 U/mL LIF (Supplementary Fig. S1; Supplementary Data are available online at
Porcine iPSCs
Retroviral constructs expressing Yamanaka's four genes were provided by S. Yamanaka. The retrovirus was produced as described previously [4,29]. PEFs were transduced with the retrovirus and cultured either in primate ESC medium (ReproCELL, Kanagawa, Japan) supplemented with human bFGF (4 ng/mL) (Wako, Osaka, Japan), mouse ESC medium with mouse LIF (1000 U/mL) (ESGRO, Chemicon, Temecula, CA), or PiPSM. PiPSM is the knockout Dulbecco's modified Eagle's medium (DMEM) (Invitrogen) containing 15% fetal bovine serum (FBS) (Invitrogen), 1% glutamax-L (Invitrogen), 0.1 mM 2-mercaptoethanol (Invitrogen), 1% nonessential amino acids (NEAAs) (Invitrogen), 50 U/mL penicillin (Invitrogen), 50 μg/mL streptomycin (Invitrogen), 10 μM forskolin (Biomol, Farmingdale, NY), and pLIF (1:200 of conditioned medium).
For serum- and feeder-free propagation, porcine iPSCs were cultured on collagen I-coated dishes (Iwaki, Tokyo, Japan) in serum-free medium (SFM); knockout DMEM containing 20% knockout serum replacement (Invitrogen), 1% glutamax-L, 0.1 mM 2-mercaptoethanol, 1% NEAAs, 50 U/mL penicillin, 50 μg/mL streptomycin, 10 μM forskolin, and 0.5% pLIF supernatants.
To establish iPSCs stably expressing enhanced green fluorescence protein (EGFP) (Porco Clawn Verde iPSCs), Porco Clawn iPSCs were transfected with the plasmid expressing EGFP (pPyCAG-EGFP-IP) by using the Lipofectamine 2000 reagent (Invitrogen) and subjected to selection with puromycin (2 μg/mL) (Sigma, St. Louis, MO). Resistant colonies were isolated and cultured in PiPSM.
To develop embryoid bodies, colonies of iPSCs were detached with trypsin and dispersed on noncoated flask in PiPSM. After 7 days of cultivation, aggregated cells were placed onto gelatin-coated tissue-culture dishes in DMEM containing 10% FBS and cultured for another 7 days.
Porcine embryos and fetuses
Detailed procedures are described in the section Supplementary Procedures. Briefly, parthenogenetic and in vitro-fertilized porcine morulae were prepared. Small clumps (10 to 20 cells) of iPSCs were injected into the morulae. The morulae developed in vitro into the blastocyst stage and were examined for fluorescence. Hatching blastocysts were transferred into the uterus of prepubertal gilts. Pregnant gilts were killed and the fetuses were isolated. The isolated fetuses were observed under a fluorescence microscope.
Sodium bisulfite genomic sequencing
Genomic DNA was purified using the DNeasy Blood & Tissue Kit (Qiagen, Hilden, Germany) and digested with HindIII (Takara Bio). Digested genomic DNA was purified using the QIAquick Gel Extraction Kit (Qiagen), and the bisulfite reaction was performed with EZ DNA Methylation-Direct Kit (Zymo Research, Irvine, CA). Bisulfite-treated DNA was amplified using the BioTaq HS DNA polymerase (Bioline, London, United Kingdom) and Oct3/4 primers (Oct3/4-Bis-F, 5′-GGG GGT TTA GTA AAA TTA GGG TTT T-3′; Oct3/4-Bis-R, 5′-AAA ACT TAA CAC AAA CAC CAA ACC T-3′). Primer sequences were designed based on the pig genome assembly of version Sscrofa9.2 (November 2009 build) from the UCSC genome database. PCR reactions were performed under the following conditions: 95°C for 10 min, 40 cycles at 95°C for 30 s, 60°C for 30 s, and 72°C for 1 min, followed by a final extension step at 72°C for 2 min. Amplified PCR products were cloned into the pGEM T-Easy vector (Promega, Madison, WI), and 10 to 16 clones were sequenced to determine the DNA methylation status.
Statistical analysis
Data are expressed as means±SD. The significance was tested by the Student's t-test.
Results
Novel porcine iPSCs
LIF is indispensable for the growth of naive PSCs, although it is dispensable for primed PSCs [1,2]. However, mouse LIF failed to generate naive-like porcine iPSCs. In addition, human LIF has weak effects on porcine cells [30]. Since it would be better to use porcine LIF (pLIF) to generate naive porcine iPSCs, recombinant pLIF was produced (Supplementary Fig. S1).
Porcine iPSCs were generated from PEFs of Porco Rosso, the transgenic pig expressing the huKO fluorescence gene, with retroviral vectors expressing 4 reprogramming genes (human Oct3/4, Sox2, Klf4, and c-Myc) [4]. After 2 weeks of cultivation with pLIF and forskolin, compact, dome-shaped colonies, similar to mouse ESC colonies, appeared at an efficiency of 0.02%. Three of them were plucked and propagated for further analyses. The cells were stably propagated over 40 passages. In this study, 2i was dispensable for the maintenance of these cells. They expressed huKO fluorescence (Fig. 2A). The cells were positive for alkaline phosphatase activity. They had a normal karyotype (Supplementary Fig. S2A). The cells differentiated into all 3 germ-layer cells in vitro in embryoid bodies as assessed by immunocytochemistry (Supplementary Fig. S2B). The putative naive porcine iPSCs, however, developed only immature teratomas in immunodeficient (NOD/SCID) mice. It is not porcine-specific, but it is also the case with de novo naive-like human iPSCs (unpublished data). Other investigators have also claimed the difficulty of naive-like human iPSCs to develop mature teratomas [27,31,32].
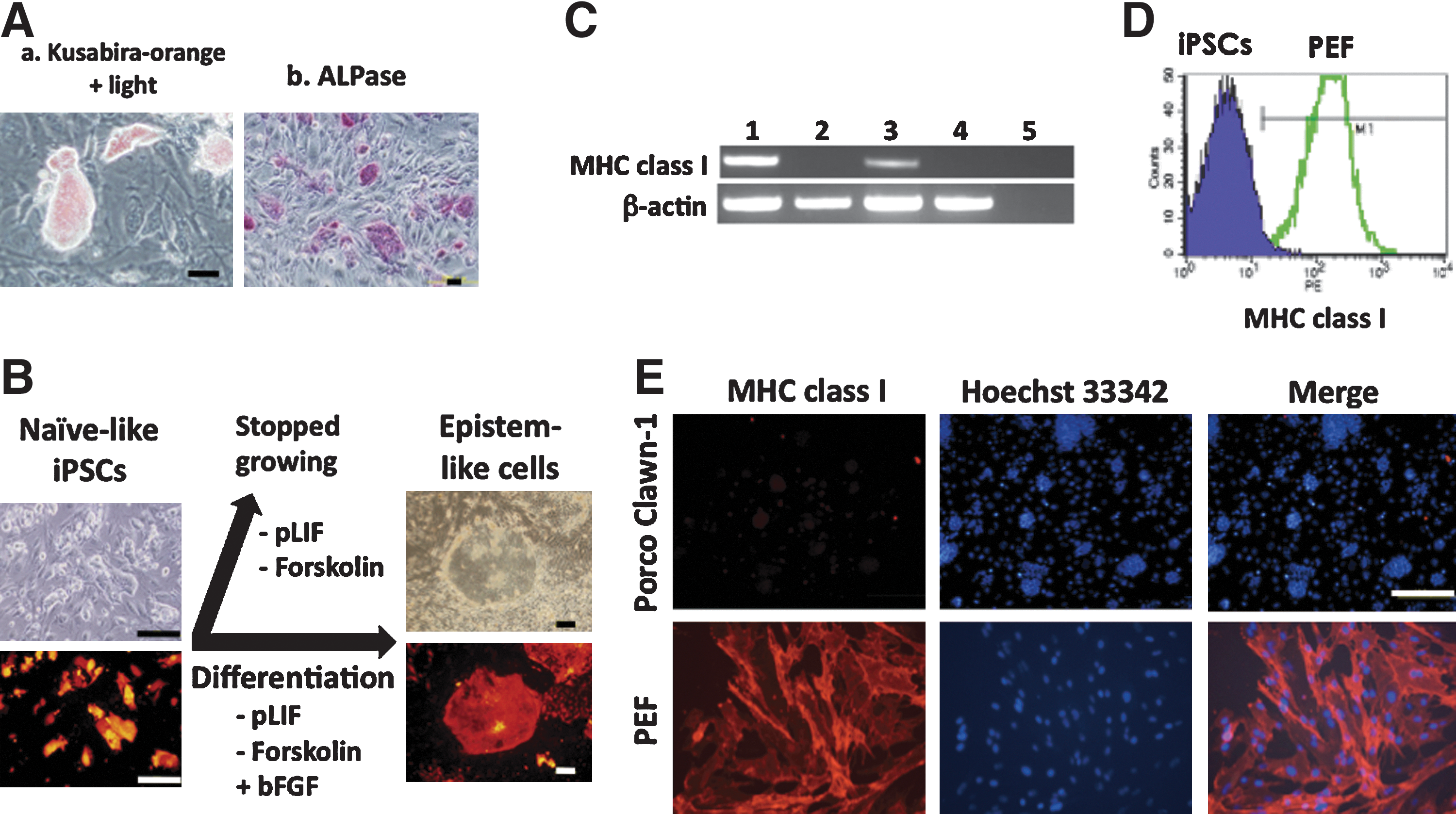
Putative naive porcine iPSCs are LIF-dependent and MHC class I-negative.
Porco Rosso iPSCs express pluripotency-related genes, including endogenous Oct3/4, Nanog, ERas, Sox2, Lin28, and Stella, as assessed by immunocytochemistry or reverse-transcription PCR (RT-PCR) (Supplementary Fig. S3). However, the four transgenes were still detectable by RT-PCR (Supplementary Fig. S3A). The expression levels of the transgenes and endogenous genes were compared by quantitative RT-PCR, but any consistent, convincing result was not obtained. The transgenes were human but the endogenous genes were porcine. The primers for RT-PCR were designed to distinguish the human and porcine homologues, and they were different (Supplementary Tables S1 and S2). Under such different primer conditions, the two exogenous human versus endogenous porcine homologues were not quantitatively comparable. On the other hand, if the same primer pairs were used, the two were not distinguishable. Therefore, it is only possible to conclude that they were both expressed in the porcine iPSCs, and that the transgenes were expressed at higher levels in the porcine iPSCs than in the human iPSCs that Yamanaka has generated [4].
The iPSCs expressed SSEA-1, -3, and -4 (1=3>4). RT-PCR showed that the expression of endogenous Klf4 and c-Myc genes was only slightly enhanced, which is in agreement with the finding of Okita et al. [33]. E-Cadherin, which is abundantly expressed in mouse ESCs [34], was also expressed at high levels. On the other hand, N-cadherin was hardly expressed, as is noted in mouse ESCs [34]. The cells expressed Tra-1–60 and −81, although at low levels (data not shown). Quantitative RT-PCR revealed that these iPSCs expressed the endogenous Oct3/4, Nanog, and Sox2 genes at high levels (Supplementary Fig. S4).
Evidence of the naive-like state
Next, the novel Porco Rosso iPSCs were examined to determine whether they are in the naive state. LIF-dependency is a hallmark of the naive state. The dependency of porcine iPSCs on pLIF was clearly shown by culturing the cells in the absence of pLIF; that is, the cells stopped growing. In addition, in response to pLIF, STAT3 was tyrosine-phosphorylated and transferred into the nuclei (Supplementary Fig. S1C), and the expression of the pLIF receptor was upregulated in the cells (Supplementary Fig. S3A). Notably, cultivation of the cells with bFGF in the absence of pLIF and forskolin allowed them to convert to epistem-like cells (Fig. 2B).
No or low levels of MHC class I expression is another hallmark of the naive state [2,25]. Porcine iPSCs were negative for MHC class I as assessed by RT-PCR, immunocytochemistry and flow cytometry (Fig. 2C–E), implying that the cells were in the naive state. On the other hand, epistem-like conventional porcine iPSCs turned positive for MHC class I (Fig. 2C).
In female iPSCs, the reactivation of X chromosome (XaXa) should be a hallmark of the naive state [2,35,36]. The activated X chromosome is associated with the depression of the Xist gene [37]. According to RT-PCR analysis, Xist mRNA was little expressed in porcine iPSCs, although it was expressed in epistem-like conventional porcine iPSCs and, of course, in female PEFs (Fig. 3A). In addition, the histone H3K27 trimethylation spots associated with the inactivation of X chromosomes were observed in primed iPSCs, but not in naive-like iPSCs (Fig. 3B).
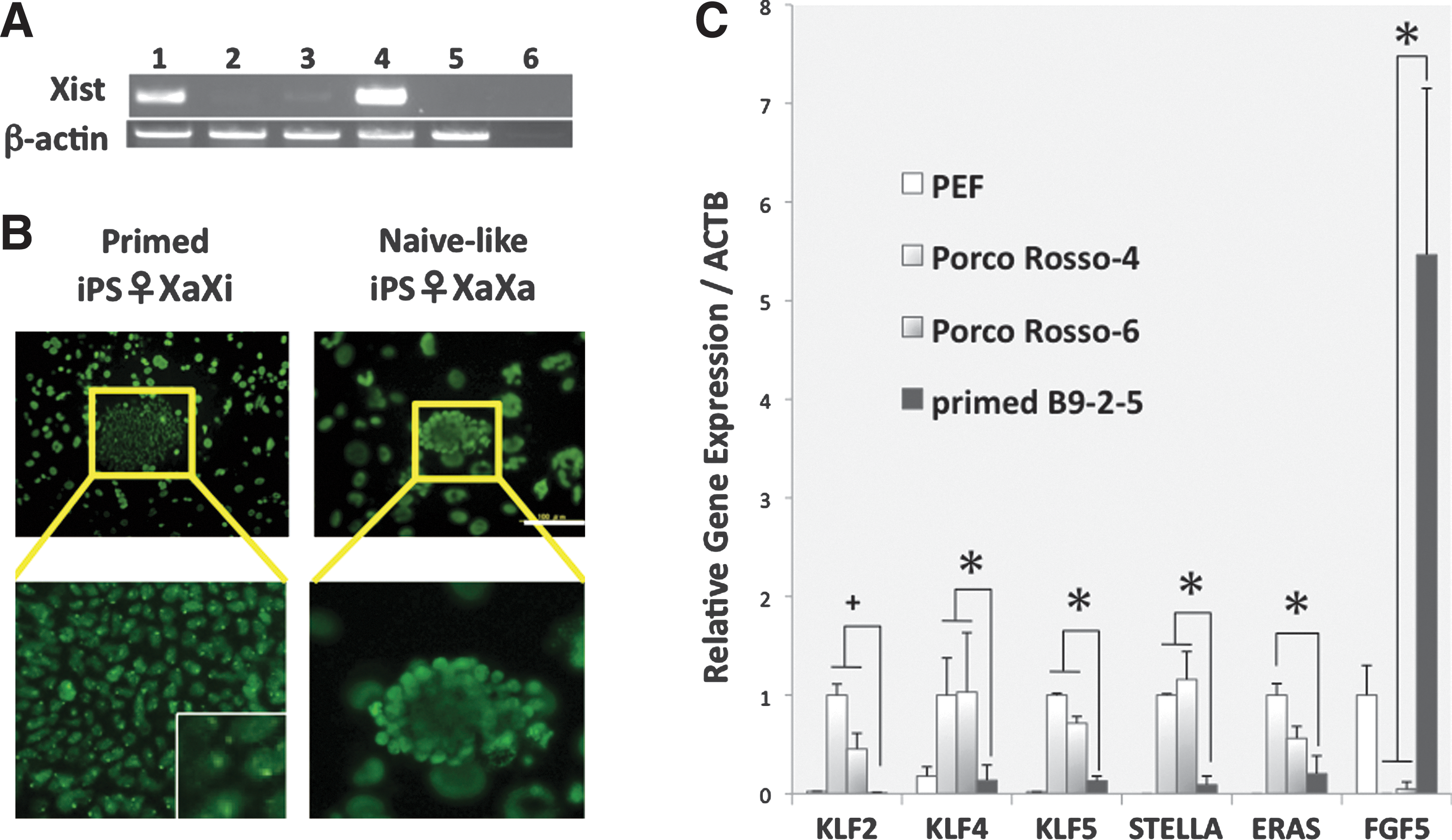
Putative naive porcine iPSCs have XaXa and distinct profiles of gene expression.
Gene expression profiles are different between naive and primed PSCs. Notably, the Klf-family (Klf2, 4, and 5) and Stella genes are expressed at high levels in naive PSCs, but at low levels in primed PSCs [6,38,39]. Conversely, the FGF5 gene is expressed at high levels in primed PSCs, but at low levels in naive PSCs [40,41]. The expression profiles of these genes in porcine iPSCs were compatible with those of naive-type cells (Fig. 3C). ESC-expressed Ras (ERas), which is expressed in mouse ESCs/iPSCs but not in human ESCs [42 –44], is also expressed in porcine iPSCs (Fig. 3C). Taken together, the gene expression profiles of porcine iPSCs were more similar to those of naive PSCs than to those of primed PSCs.
Therefore, the Porco Rosso iPSCs were in the putative naive state. Naive-like porcine iPSCs can be easily passaged with trypsin and, unlike human PSCs, no longer require a Rho-kinase (ROCK) inhibitor for passaging or subcloning [45]. The doubling time and plating efficiency of naive-like porcine iPSCs were close to those of mouse PSCs (Supplementary Table S3). Naive-like porcine iPSCs can survive at high rates (more than 50%) after freezing and thawing. The cells can be cultured in the absence of serum and feeders on collagen I-coated dishes (Supplementary Fig. S3Bd). Porcine iPSCs from PEFs of inbred Clawn miniature pigs (Porco Clawn) were also generated. The Porco Clawn iPSCs showed characteristics very similar to those of Porco Rosso iPSCs (Supplementary Figs. S1 and S2).
Chimera formation
Porco Clawn iPSCs expressing EGFP (Porco Clawn Verde iPSCs) were injected into porcine parthenogenetic morulae to examine whether they are incorporated into the normal development of porcine embryos (Fig. 4A). One to 2 days after injection, successful incorporation of iPSCs into the ICM was observed. Some blastocysts were hatching, indicating their development (Fig. 4Ba). Similar results were obtained when Porco Rosso iPSCs (expressing the huKO fluorescence gene) were injected (Fig. 4Bb). Notably, fluorescent donor cells (especially Porco Rosso iPSCs) were also detected in the trophectoderm (TE) (Fig. 4C). Incorporation into the TE was also frequently observed (80%) after porcine ICM cells were injected into allogeneic morulae (Fig. 4D). Although the incorporation of ESCs or iPSCs into the TE has not been observed in mice, the observation is in agreement with a report showing that rapid TE differentiation occurs in mice compared to cattle [46]. The in vitro development of porcine iPSCs is summarized in Table 1.
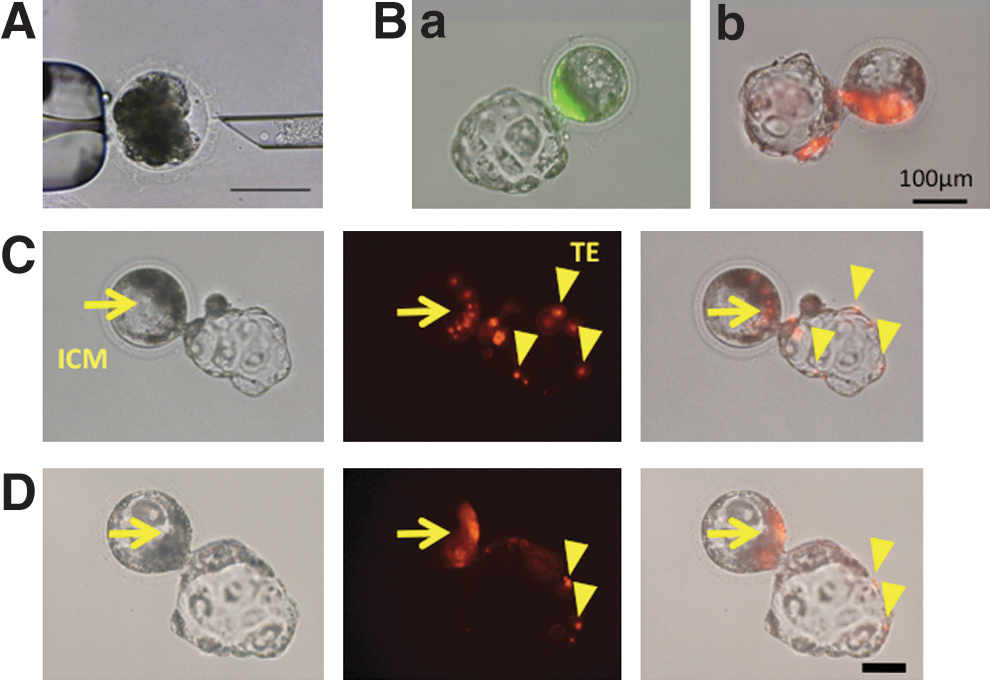
Injection of porcine iPSCs into morulae and their in vitro development.
iPSCs, induced pluripotent stem cells; huKO, humanized Kusabira-orange; EGFP, enhanced green fluorescence protein; ICM, inner cell mass; TE, trophectoderm.
The in vivo fates of Porco Clawn Verde and Porco Rosso iPSCs in parthenogenetic embryos were traced after transferring the embryos into surrogate mother uteruses. The fetuses were recovered at day 23 of gestation (full term, 114 days) and fluorescence (huKO or EGFP) was detected in 11 out of 71 fetuses (Fig. 5A and Supplementary Fig. S5A for high resolution). Porcine iPSCs actually differentiated into various tissues. In the fetuses in Fig. 5A, relatively strong fluorescence was observed in the head, brachial arch, atrium, ventricle, liver, and limb bud (indicated by arrowheads).
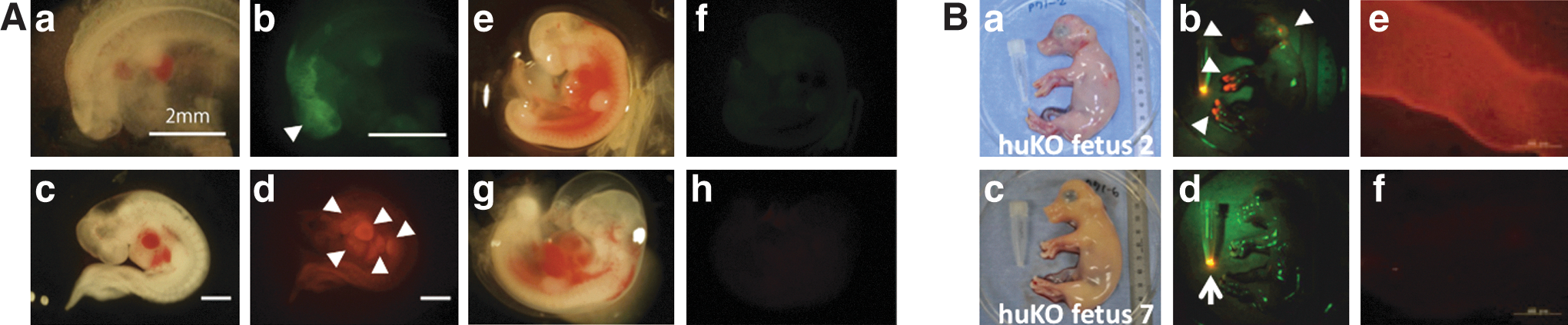
The in utero development of porcine iPSCs.
Because parthenogenetic embryos cannot develop at further time points, in vitro-fertilized morulae were then used as host embryos for long-term observation. The morulae injected with iPSCs were transferred into surrogate mother uteruses, and the fetuses were recovered at day 65 of gestation. Fluorescence was observed in 1 out of 13 fetuses (Fig. 5B and Supplementary Fig. S5B for high resolution). In the fetus, fluorescence was observed in the ectodermal skin around the head, neck, and hooves (arrowheads in Fig. 5B). Expression of the fluorescence protein (huKO) was confirmed by immunohistochemistry (Fig. 5B). Fluorescence was not detected in other parts of the fetus.
The contribution of porcine iPSCs in the in utero development is summarized in Table 2. The frequency of fluorescence-positive chimeras and the fluorescent area decreased over gestation days. Any contribution of injected iPSCs to the germline was not observable by 65 days of gestation. In our laboratory, iPSC-like candidate cells were stocked, which had been propagated in a process of establishing porcine iPSCs but had eventually failed to meet some criteria of iPSCs; that is, they were not iPSCs. When those cells were transferred to the 4-cell or 8-cell stage, they did not incorporate into blastocysts (0/13), thus not contributing to the embryonic development.
The reprogramming status
The methylation status of the Oct3/4 promoter in porcine iPSCs was examined by the bisulfite sequencing method. In 2 lines examined (Porco Rosso 4 and 6), the methylation status was heterogenous and only a part of clones were completely demethylated, that is, reprogrammed (Fig. 6). This may be a reason for the low efficiency to produce chimeras. There was always a huKO-negative fraction (4%–5%) in the Porco Rosso iPSCs (Fig. 7A). Considering that the huKO gene was driven by the retroviral long terminal repeats, it was likely that the silencing of the huKO gene was correlated with the retroviral transgene silencing, that is, completely reprogrammed status. The huKO-positive and negative cells were sorted, and the gene expression profiles were examined (Fig. 7B). Notably, the huKO-negative cells expressed pluripotency-related endogenous genes at high levels (Fig. 7C), but little expressed the transgenes (Fig. 7D). Transgene-silenced iPSCs are enriched in the huKO-negative fraction. It is possible that completely reprogrammed porcine iPSCs are enriched in this fraction.
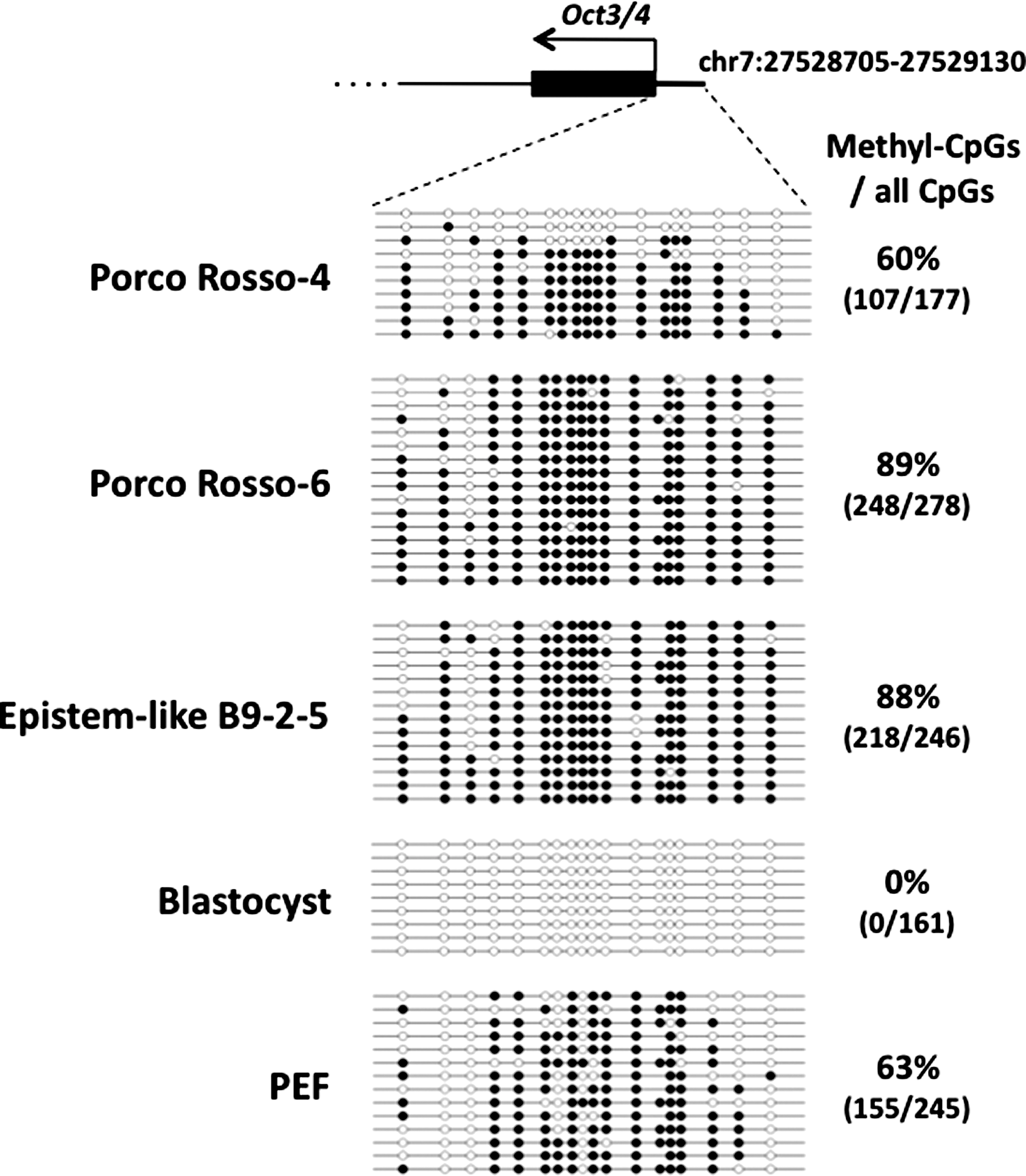
The DNA-methylation status of the endogenous Oct3/4 promoter region in porcine iPSCs. The methylation patterns of the Oct3/4 upstream region of individually sequenced clones were examined in porcine iPSCs (Porco Rosso-4, -6, and epistem-like B9-2–5), in vitro fertilization-derived blastocysts, and PEFs by using the bisulfite sequencing method. Open and closed circles indicate nonmethylated and methylated statuses, respectively. The numbers of methyl-CpGs out of all CpGs and the overall percentage of methylated CpGs are shown on the right side.
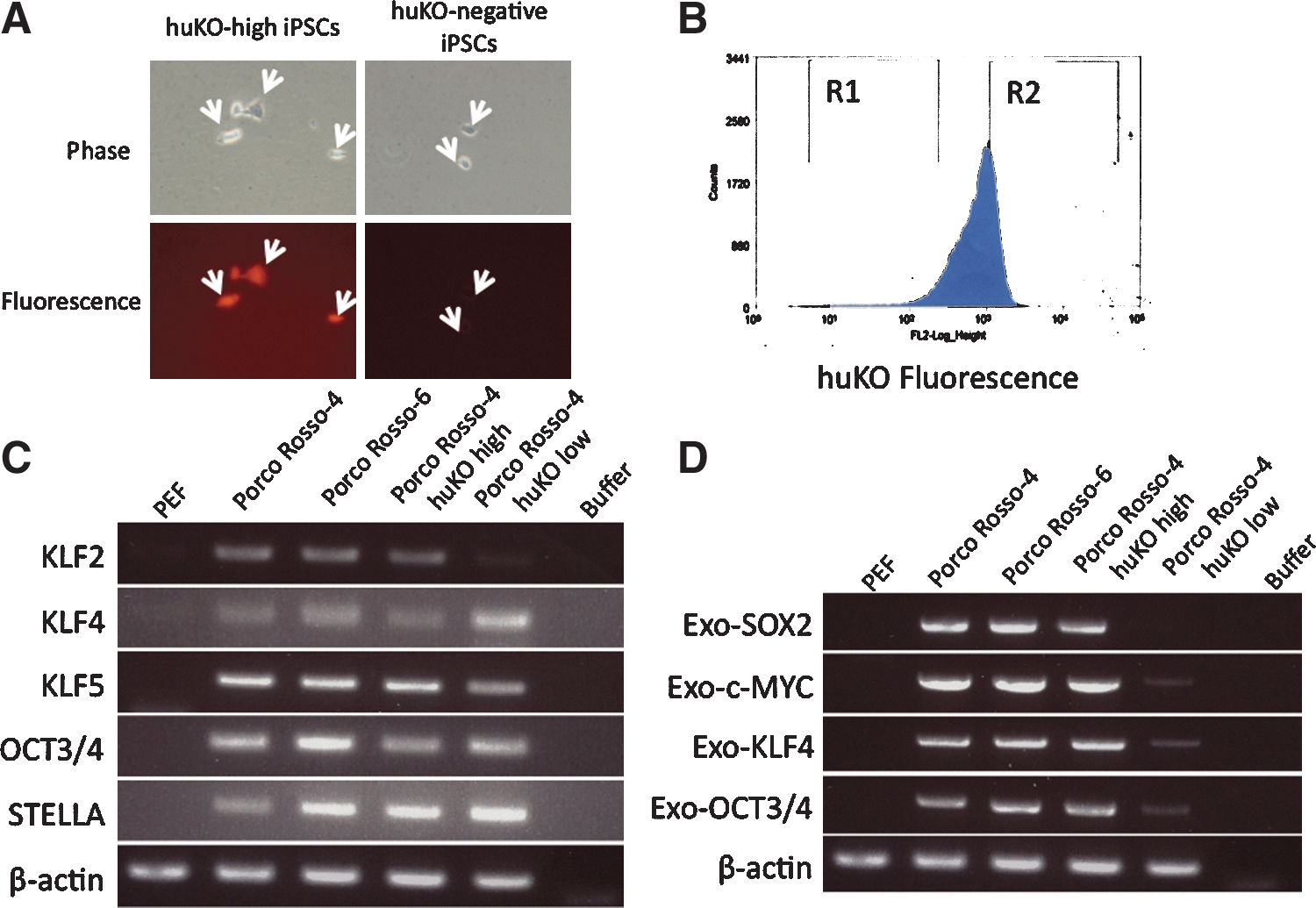
The silencing of the huKO-fluorescence gene implies completely reprogrammed status. There is a huKO-negative fraction in Porco Rosso-4 iPSCs indicated by arrows
Discussion
The putative naive porcine iPSCs were generated using pLIF and forskolin. pLIF and forskolin were indispensable to establish and maintain putative naive porcine iPSCs. In fact, the naive characters were lost without them (Fig. 2B). On the other hand, 2i was dispensable for maintaining the putative naive state in the culture. It is possible that the sustained expression of the transgenes would help to maintain the putative naive state without 2i [27]. The putative naive porcine iPSCs showed the ability to contribute to the normal embryonic and fetal development (Figs. 4 and 5). However, considering that porcine non-naive or even nonpluripotent cells such as neural stem cells can contribute to the embryonic and fetal development [18 –20], the ability to develop chimeras may not be good criterion for naivety in pigs. That used to be the case in mice indeed. It was reported that mouse neural stem cells can contribute to the formation of chimeric mouse embryos and give rise to cells of all germ layers [47], but it is considered unlikely at present. Presumably, they saw cell fusion [48]. On the other hand, the in vitro characteristics that have been shown in this study including LIF-dependent growth, negative MHC class I, active X chromosomes, and distinct gene-expression profiles (Figs. 2 and 3) might be definitive criteria for naivety. The present paper for the first time shows porcine iPSCs that meet all of such in vitro criteria and therefore the porcine iPSCs may be referred naive. To show that the cells are truly naive, germline competency of the cells should be demonstrated.
However, expression levels of the Nanog gene are actually higher in primed PSCs than in the putative naive ones (Fig. S4). Regarding the porcine Nanog gene, there is a confusing situation. First, in pigs, the Nanog gene is little expressed in ICM but is expressed at higher levels in epiblasts [49,50]. Second, the Nanog promoter is considerably demethylated even in the liver in pigs (unpublished data). This is also a reason why the data of the methylation status was presented only in the Oct3/4 promoter but not in the Nanog promoter in Fig. 6. Third, the so-called Nanog gene is located on the chromosome no. 1 in pigs, but it has no introns and lies between LINE sequences (
It is important to identify surrogate markers for the naivety of PSCs. Although mouse PSCs and the naive-like porcine iPSCs express the stage-specific embryonic antigen (SSEA-1), conventionally primed porcine iPSCs or human PSCs do not express it. In mice and pigs, SSEA-1 is ontogenetically expressed in the ICM; however, in humans, it is not expressed in the ICM but in the morulae and trophoblasts [51,52]. Therefore, SSEA-1 is a candidate marker for naivety, at least in mice and pigs, but it remains to be elucidated whether this is also the case in humans. ERas [42] is another candidate because it is expressed in naive but not in primed PSCs [43,44]. In this context, it would be interesting to examine whether ERas is also expressed in human PSCs that have been turned naive-like as described by Hanna et al. [25].
The incorporation of the porcine iPSCs into chimeras is low, especially at day 65 of gestation. It is likely that this is because they are not completely reprogrammed, which may be associated with the sustained expression of the transgenes in the cells. The sustained expression would slow down the in vivo differentiation and development of iPSCs when they are injected into embryos. The usefulness of Tet-on/off lentiviral vectors and temperature-sensitive Sendai virus vectors for the conditional expression of transgenes is being explored [23,53].
The methylation status of the Oct3/4 promoter region can also explain the low incorporation of the porcine iPSCs into chimeras (Fig. 6). In the figure, it was shown that the region is not fully demethylated. Although the methylation status is not a criterion to evaluate whether the cells are naive or primed, it is rather a criterion to evaluate whether they are completely reprogrammed or not. In this context, it is possible to say that the porcine iPSCs are putative naive, although they are only partly demethylated. It is thus necessary to demethylate these promoter regions to achieve full reprogramming. In mouse PSCs that can produce adult chimeras, these regions are demethylated [33]. How are the porcine iPSCs converted to the completely reprogrammed state so that they can efficiently develop into adult chimeras? One approach would be the enrichment of huKO-negative fraction of Porco Rosso iPSCs, as suggested from the experiment of Fig. 7. However, this approach is not applicable to other porcine iPSC lines. Other approaches are needed, for instance, the additional transduction of porcine iPSCs with the Oct3/4 or Klf4 gene, which may demethylate the promoter region of the Oct3/4 gene, as shown by Buecker et al. [27] and Telugu et al. [54,55].
In conclusion, putative naive porcine iPSCs were generated with porcine LIF and forskolin. The putative naive porcine iPSCs share characteristics with mouse iPSCs but not with human iPSCs. The putative naive porcine iPSCs can contribute to embryonic and fetal development of pigs. They are also incorporated into TE as well as ICM. This is the first demonstration of macroscopic, fluorescent chimeras derived from naive-like porcine PSCs. The production of adult chimeras is a next step to be achieved.
Footnotes
Acknowledgments
We thank Shin-ichi Nishikawa (RIKEN CDB), Kazutoshi Takahashi, Keisuke Okita (Kyoto University), Masahito Sato (Kagoshima University), Jun Ohgane, and Masaki Nagaya (Meiji University) for helpful discussion. We acknowledge Jun-ichi Tottori (Japan Farm), Kasumi Honda (Meiji University), and Hiroshi Kadoi (Kadoi Ltd.) for technical help. This study was in part supported by Yamanaka iPSC special project of JST.
Author Disclosure Statement
S.Y. is a member without salary of the scientific advisory boards of iPierian, iPS Academia Japan, Megakaryon Corporation, and Retina Institute Japan.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
