Abstract
Very small embryonic-like stem cells (VSELs) are possibly lost during cord blood banking and bone marrow (BM) processing for autologus stem cell therapy mainly because of their small size. The present study was conducted on human umbilical cord blood (UCB, n=6) and discarded red blood cells (RBC) fraction obtained after separation of mononuclear cells from human BM (n=6), to test this hypothesis. The results show that VSELs, which are pluripotent stem cells with maximum regenerative potential, settle along with the RBCs during Ficoll-Hypaque density separation. These cells are very small in size (3–5 μm), have high nucleo-cytoplasmic ratio, and express nuclear Oct-4, cell surface protein SSEA-4, and other pluripotent markers such as Nanog, Sox-2, Rex-1, and Tert as indicated by immunolocalization and quantitative polymerase chain reaction (Q-PCR) studies. Interestingly, a distinct population of slightly larger, round hematopoietic stem cells (HSCs) with cytoplasmic Oct-4 were detected in the “buffy” coat, which usually gets banked or used during autologus stem cell therapy. Immunohistochemical studies on the umbilical cord tissue (UCT) sections (n=3) showed the presence of nuclear Oct-4–positive VSELs and many fibroblast-like mesenchymal stem cells (MSCs) with cytoplasmic Oct-4. These VSELs with nuclear Oct-4, detected in UCB, UCT, and discarded RBC fraction obtained after BM processing, may persist throughout life, maintain tissue homeostasis, and undergo asymmetric cell division to self-renew as well as produce larger progenitor stem cells, viz. HSCs or MSCs, which follow differentiation trajectories depending on the somatic niche. Hence, it can be concluded that the true stem cells in adult body tissues are the VSELs, whereas the HSCs and MSCs are actually progenitor stem cells that arise by asymmetric cell division of VSELs. The results of the present study may help explain low efficacy reported during adult autologous stem cell trials, wherein unknowingly progenitor stem cells are injected rather than the pluripotent stem cells with maximum regenerative potential.
Introduction
B
Growing evidence suggests that besides the HSCs and MSCs, a small population of pluripotent stem cells, termed very small embryonic-like stem cells (VSELs), are also present in the UCB, which could potentially contribute to organ and tissue regeneration [1]. Recently, VSELs were isolated from various adult mice organs, blood, and BM as well as from human cord blood and BM by fluorescent activated cell sorting [2]. Our group has reported the presence of VSELs in situ in adult human gonads [3,4]. On processing the VSELs, isolated by scraping the sheep ovary surface epithelium, we observed that these cells with high nucleo-cytoplasmic ratio were invariably lost during processing by centrifugation. We hypothesized that, similarly, during processing of cord blood and BM on Ficoll-Hypaque gradient, VSELs may be lost and get discarded during processing. Thus, the present study was undertaken and our results may have serious implications, because VSELs being pluripotent are expected to have more regenerative potential than the HSCs and MSCs.
Materials and Methods
Sample collection and processing
For the study, UCB (n=6) from full-term deliveries scheduled for caesarean section, umbilical cord tissue (UCT; n=3), and discarded fraction of red blood cell pellet obtained after processing BM for autologus use (BM, n=6) were used. The UCB was collected in heparinized tubes from deliveries performed by Dr. Hinduja and Dr. Zaveri, whereas discarded fraction of red blood cell pellet from BM samples were transported from Dr. S.G.A. Rao's clinic in Mumbai suburbs to Stem Cell Biology Department at NIRRH for further studies. Small piece of UCT was fixed in normal buffered formalin and slides were prepared for immunohistochemical studies using standard protocols, whereas the UCB and discarded fractions of BM were processed to isolate cells by Ficoll-Hypaque centrifugation method.
Ficoll-Hypaque density centrifugation
Collected UCB was diluted 1:1 in phosphate-buffered saline (PBS) and carefully overlaid in 1:2 ratio onto research-grade Ficoll-Hypaque solution (HISTOPAQUE-1077; Sigma; d: 1.077 g/cm3), followed by centrifugation at 1,500 rpm for 30 min at room temperature (RT). After centrifugation, all the 4 layers (1–4) (Fig. 1A) were easily visualized and collected in separate tubes.
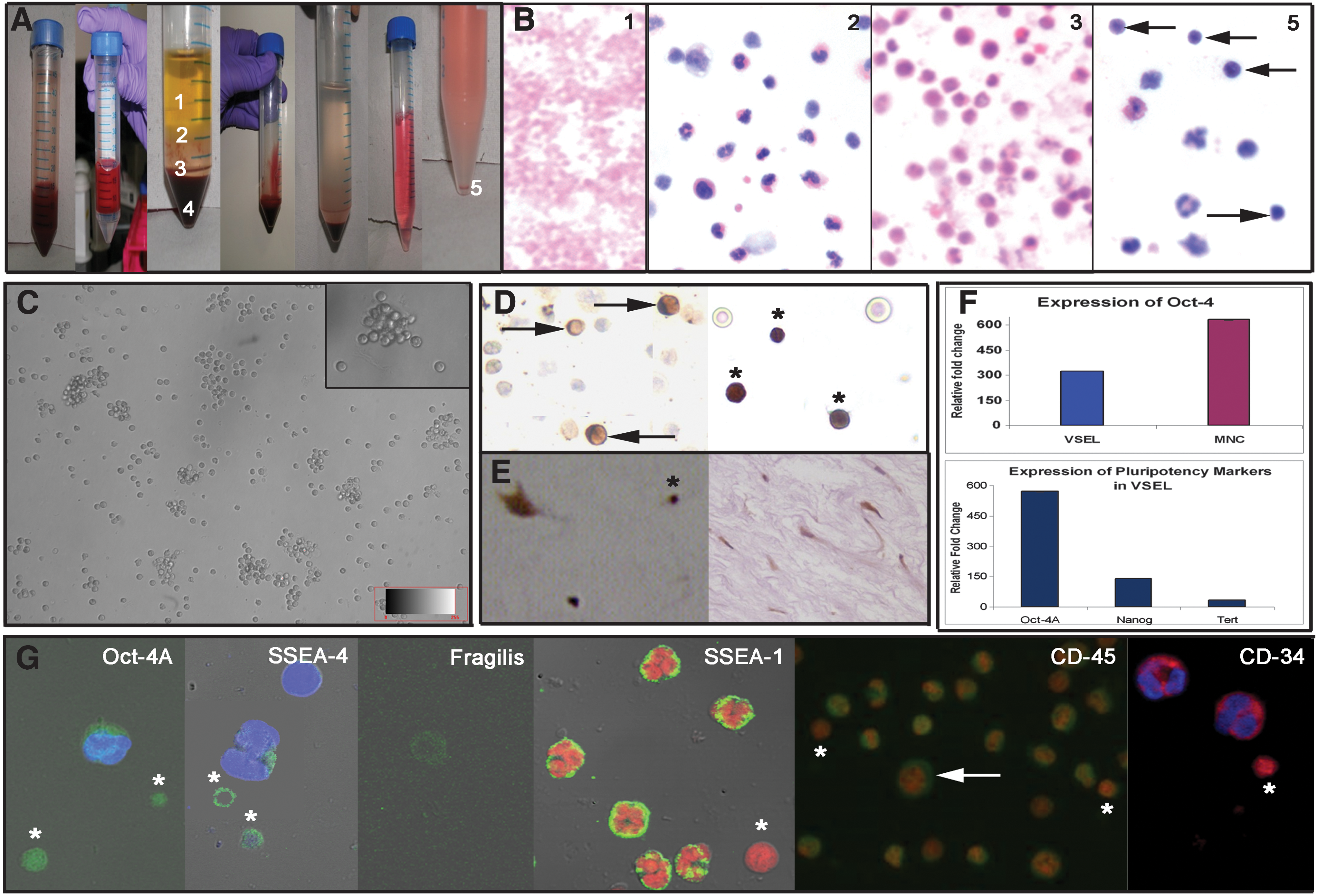
Processing of mononuclear cells
The layer 2 (buffy coat) was diluted 3 times its volume with DMEM F12 (Invitrogen) containing 5% FBS (Invitrogen) and centrifuged for 10 min at RT. The pellet was collected and resuspended in PBS to make a single-cell suspension. The platelet (layer 1) and Ficoll (layer 3) were also processed similarly.
Processing of red blood cells (4) to further enrich VSELs
The red blood cells (RBC) pellet (layer 4) collected from both UCB and BM was diluted 3 times its volume with DMEM F12 and centrifuged at 800 g for 15 min at RT to remove most of RBCs. The supernatant was collected in a fresh centrifuge tube and spun at 1,000 g for 10 min at RT. The collected pellet was resuspended in PBS to make a single-cell suspension (layer 5).
The cell suspensions obtained from layers 1, 2, 3, and 5 were used to make cell smears for immunolocalization studies, whereas part of the cell pellet (2 and 5) was stored in TRIZOL (Invitrogen) for RNA extraction. Table 1 is a comprehensive representation of various immunolocalization studies undertaken.
HSC, hematopoietic stem cell; PSC, pluripotent stem cell.
The air-dried cell smears were fixed with 4% paraformaldehyde for 10 min, washed twice with PBS, air dried, and stored at 4°C till further use. Smears were stained with hematoxylin and eosin (H&E) using a standard method and viewed under a bright-field microscope. Representative fields were photographed under a Nikon 90i microscope (Nikon). The smears prepared from layers 2 and 5 were used for immunolocalization and confocal studies.
Immunolocalization studies
Immunolocalization studies were performed on paraffin-embedded sections of cord tissue and on cell smears from layers 2 and 5.
Immunocytochemistry
The cell smears were processed for immunolocalization of Oct-4 using polyclonal Oct-4 antibody (Abcam) and Vectastain Elite ABC kit (Vector Laboratories) as detection system.
In brief, the endogenous peroxidase was blocked using 0.3% hydrogen peroxide for 45 min in dark at RT. Antigen retrieval was done by treating the smears with sodium citrate buffer (10 mM sodium citrate, pH 6.0) at high power for 5 min in a microwave oven. This was followed by a permeabilization step with 0.3% Triton X-100 for 10 min. Blocking was done with 10% normal goat serum for 1 hr. The cells were then incubated with the primary polyclonal Oct-4 antibody (1:100) at 4°C overnight. Primary antibody was omitted and smears were incubated with the blocking solution for negative control. After washing with PBS, the cells were incubated with the biotinylated secondary antibody (anti-rabbit IgG) for 30 min followed by avidin–biotin complex formation step for 30 min; the cells were then extensively washed with PBS and detected using 3,3′ diaminobenzidine (DAB) (Sigma-Aldrich) and later counterstained with hematoxylin.
Immunohistochemistry on UCT sections
The paraffin sections were deparaffinzed and rehydrated through a graded methanol series. Endogenous peroxidase was blocked using 0.3% hydrogen peroxide for 90 min in dark at RT and then the sections were processed in a similar manner to the cell smears.
Representative areas of smears and sections were photographed under Nikon 90i microscope and results were recorded.
Immunofluorescent staining
The expression of nuclear antigen Oct-4A, cell surface antigen SSEA-4, CD45-FITC, CD34-PE, and Fragilis were studied on cell smears from layers 2 and 5 only. Immunofluorescent staining was performed using specific antibodies listed in Table 1. Briefly, fixed cell smears were washed with wash buffer (PBS containing 0.3% bovine serum albumin and 0.1 mM EDTA), permeabilized by 0.3% Triton X-100 for 10 min, washed in PBS, preblocked with 15% normal goat serum, and subsequently incubated with antibodies to Oct-4, CD45 FITC, CD34 PE, Fragilis, and SSEA-4. For detection, cell smears were incubated with secondary antibody indicated in Table 1; however, for CD34 and CD45 this step was omitted as the primary antibodies were directly labeled with the flurochrome. Negative controls were incubated overnight in the blocking solution with the omission of primary antibody. Counterstaining was done using (1 mg/mL) 4′,6-diamidino-2-phenylindole (DAPI; Molecular Probes). All images were captured by laser scanning confocal microscope (Carl Ziess) at 63×magnification with 5×optical zoom using argon laser at λ=488 nm and blue diode laser at λ=405 nm for FITC and DAPI staining, respectively. For staining of cell surface markers, permeabilization with Triton X-100 was omitted.
RNA extraction and cDNA synthesis
Total RNA was isolated from cell pellets (2 and 5) obtained after processing UCB and discarded fractions of BM, using TRIZOL according to the manufacturer's instructions. The extracted RNA was treated with ribonuclease-free deoxyribonuclease (DNase 1; Qiagen) to remove any contaminating genomic DNA. Two micrograms of total RNA was reverse-transcribed using the iScript RT Kit according to the manufacturer's instructions (Biorad) to synthesize the first-strand cDNA in a GSTORM thermocycler (Gene Technologies).
Quantitative polymerase chain reaction
Expression of various pluripotent markers, viz. Oct-4A, Oct-4, Nanog, Rex-1, Tert, and Sox-2, was studied by Q-PCR using a CFX96 real-time PCR system (Bio-Rad Laboratories) and SYBR Green chemistry (Bio-Rad Laboratories) (Table 2).
The amplification conditions for Oct-4 were as follows: initial denaturation at 95°C for 5 min, followed by 40 cycles comprising denaturation at 95°C for 10 s, primer annealing at 57°C for 30 s, and extension at 72°C for 30 s. For the rest of gene transcripts, viz. Oct-4A, Nanog, Rex-1, Sox-2, Tert, and 18S rRNA, all the amplification conditions were the same except that the annealing temperatures were optimized at 62°C.
The threshold cycle (Ct) was determined subsequently using the CFX Manager software (Bio-Rad Laboratories) and normalized to the housekeeping gene (18S RNA). Relative fold change of Oct-4 and Nanog mRNA over the calibrator was expressed as 2−ΔΔCt, where ΔCt=Ct of target genes (Oct-4, Nanog)−Ct of endogenous control gene (18s), and ΔΔCt=ΔCt of samples for target gene−ΔCt of calibrator. Melt curve analysis was performed at the end of every run to confirm the homogeneity of the PCR products, which was also confirmed by running the same on 2% agarose gel.
Results
Enrichment of VSELs from UCB and BM
Careful processing of different fractions obtained after Ficoll-Hypaque separation of UCB showed that the VSELs separated along with the RBCs in layer 4. Layers 1, 2, and 3 comprised of platelets, mononuclear cells (MNCs), and mostly granulocytes, respectively (Fig. 1A), as evident from the H&E staining (Fig. 1B). Further centrifugation of the RBC pellet (layer 4) at different speeds enabled separation of RBCs from the VSELs, as RBCs settled down at 800 g and later the VSELs were pelleted at 1,000 g. H&E staining of the smears prepared from this layer comprised of very small round cells with high nucleo-cytoplasmic ratio, which ranged in size from 3 to 5 μm (Fig. 1B) along with few multilobed nucleated cells. Similarly, it was possible to separate the BM VSELs from the RBC pellet collected after separation of the MNCs. The VSELs enriched in layer 5 were present in large numbers, were round, and had a characteristic shiny appearance as seen under Hoffman optics (Fig. 1C). They had striking similarity in their appearance to the VSELs isolated by scraping the ovarian surface epithelium, reported earlier by our group [4].
Immunocytochemical localization of Oct-4
Polyclonal Oct-4 antibody was used to study cell smears from layers 2 and 5 only. As evident in layer 2, few large cells with cytoplasmic Oct-4 were observed (possibly the HSCs; Fig. 1D), whereas majority of cells remained unstained. In layer 5, the VSELs showed distinct nuclear staining for Oct-4.
Immunohistochemical localization of Oct-4 in UCT sections
The cell population in the mucous connective tissue also termed as the Wharton's jelly of the umbilical cord comprised mainly of spindle-shaped fibroblasts after H&E staining. However, 2 distinct cell populations were easily identified after immunolocalization studies using polyclonal Oct-4 antibody, viz. very small cells with nuclear Oct-4 and the fibroblasts (possibly the MSCs) with cytoplasmic Oct-4. Similar results were obtained using VSELs isolated from BM samples.
Immunofluorescence staining and confocal microscopy
Various pluripotent, germ cell, and hematopoietic markers were studied by confocal microscopy.
(i) Pluripotent markers Oct-4 and SSEA-4 revealed characteristic nuclear and cell surface staining, respectively, on VSELs in layer 5 only (Fig. 1G). A distinct larger cell with abundant cytoplasm revealed minimal cytoplasmic staining for both Oct-4 and SSEA-4. All other cell types in layer 2 remained unstained for Oct-4 and SSEA-4.
(ii) Primordial germ cell marker Fragilis showed cell surface staining only on the VSELs (Fig. 1G).
(iii) Hematopoietic marker CD45 stained all the blood cells in the MNC fraction (layer 2), whereas the VSELs remained distinctly negative.
(iv) CD34, a more primitive marker for hematopoietic progenitor cells, stained all the cells including the small cells, which were easily identified because of their size and because they stained negative for DAPI. Interestingly, SSEA-1 also stained the granulocytes but VSELs were distinctly negative for SSEA-1, confirming their primitive status.
Similar results were obtained using VSELs isolated from BM samples.
Q-PCR results
As evident, gene transcripts for various pluripotent markers including Oct-4, Nanog, and TERT were present and exhibited several fold higher expression in the VSELs (layer 5) when compared with the MNC fraction (layer 2) isolated from the UCB. Similar results were obtained using VSELs isolated from BM samples.
Discussion
The present study describes a simple method based on differential centrifugation to isolate pluripotent stem cells, which are termed VSELs (because of their size), from both UCB and BM. This method is relatively cheaper, is practical, and can be easily carried out in a laboratory setting when compared with available sophisticated approach using specific markers by FACS [2]. It is an Indian Council of Medical Research (ICMR), Government of India, New Delhi, patent-protected technology (1257/DEL/2011). Multiparameteric analysis was employed to characterize and confirm the pluripotent nature of the VSELs by visualizing morphology under Hoffman optics, H&E staining, immunolocalization for pluripotent, germ cell, and hematopoietic markers, and Q-PCR. The VSELs exhibit a characteristic round shape and high nucleo-cytoplasmic ratio and showed expression of pluripotent markers, such as Oct-4A, Nanog, Rex, and TERT, at the mRNA level and immunolocalization for nuclear Oct-4 and cell surface SSEA-4. The VSELs present in layer 5 were strikingly similar to the VSELs obtained by scraping ovary surface epithelium, recently reported by our group [4].
Results clearly show that UCB and BM VSELs settle down along with RBCs in the lowermost layer (Fig. 1A, layer 4) during Ficoll-Hypaque processing. RBCs settle down, because they possess hemoglobin, whereas the VSELs possibly settle down as per laws of physics. VSELs being smaller in size have lower mass and so require higher centrifugation speeds to pellet when compared with larger HSCs, which get separated in the upper buffy coat (layer 2). This is similar to the situation wherein ship (HSCs) floats despite being bigger in size, whereas small bullets (VSELs) sink in water.
We have observed several folds higher expression of various pluripotent makers in layer 5 compared with layer 2 (Fig. 1F), which further confirms the pluripotent nature of these cells. Oct-4A mRNA was expressed several folds higher in layer 5 compared with layer 2. Interestingly, total Oct-4 was also detected in layer 2, suggesting presence of an alternative spliced variant Oct-4B in the HSCs rather than pluripotent Oct-4A. Immunolocalization studies also confirmed that MNC in layer 2 had HSCs with cytoplasmic Oct-4, whereas the VSELs in layer 5 had predominantly nuclear Oct-4–positive cells. Immunohistochemical studies on UCT sections also yielded intriguing results, because a heterogeneous population of both VSELs with nuclear Oct-4 and spindle-shaped, fibroblast-like MSCs with cytoplasmic Oct-4 were identified (Fig. 1E).
Current banking protocols cryopreserve only cells of the buffy coat for future autologus stem cell therapy and large sums of money in tune of $2,000 and yearly fees are charged for the same. However, our results imply that instead of true stem cells (VSELs), only the progenitors are being banked for therapeutic use. On a similar note, huge expenses are incurred for stem cell therapy using autologus BM, because the public is vulnerable and at any cost wish to save lives of their dear ones.
Oct-4 studies have confused stem cell researchers in the past, because it has several transcripts and pseudogenes. However, using specific primers and 3 different antibodies, we have earlier reported the presence of similar VSELs with nuclear Oct-4 and slightly bigger progenitor stem cells with cytoplasmic Oct-4 in the adult mammalian testis (Adark spermatogonial stem cell) [3] and also ovaries (ovarian germ stem cells [OGSCs]) [4]. Results are depicted in Table 3.
These results are indeed startling. It is apparent that there is nothing like adult stem cells in somatic body tissues and embryonic stem cells isolated from spare human embryos, which has divided the scientific community into 2 groups. Embryonic-like stem cells, that is, VSELs, are present in various adult body tissues. They are relatively quiescent, serve as a backup, and give rise to the progenitor stem cells (which are currently termed the adult stem cells) by asymmetric cell division.
The progenitor stem cells exhibit high proliferation activity and differentiate further to give rise to various differentiated progenies. The question that arises is if it is the same VSEL in different body tissues then why the progenitors differ from each other and give rise to different end points? We propose that this indeed highlights the importance of the somatic microenvironment, which differs from tissue to tissue and thus directs the progenitors to follow different differentiation pathways. Thus, in contradiction to earlier reports that Oct-4 is dispensable to somatic tissues, the present study results in contrast show that indeed Oct-4 pluripotency network is crucial for proper homeostasis of body tissues throughout life. Being pluripotent in nature, VSELs have the ability to differentiate into any cell lineage based on what genes get turned ON when the pluripotent stem cell chromatin gets reprogrammed during the transition of the VSELs into the progenitor. The present study results are in agreement with the huge body of literature published by Ratajczak and group and Li and Clevers [5], who have proposed that 2 distinct populations of stem cells exist in various body tissues. The results are also in agreement with Virchow and Connheim, who had proposed almost 150 years back that adult tissues contain dormant embryonic remnants.
It is concluded that adult stem cells are actually progenitor stem cells derived from VSELs in the adult body. It has always been the pluripotent embryonic-like stem cells, which bring about homeostasis in the adult body. The debates between the groups who work on adult versus embryonic stem cells and the pros and cons of embryonic versus adult stem cells research become irrelevant. Hopefully, VSELs will unify both the groups and new paradigms will emerge wherein more emphasis will be to better understand the somatic niche that controls the fate of VSELs.
Footnotes
Acknowledgments
This study (BT/PR14026/MED/31/100/2010) was financially supported by Department of Biotechnology and Indian Council of Medical Research, New Delhi, India.
Author Disclosure Statement
No competing financial interests exist.
