Abstract
In cellular transplantation strategies for repairing the injured central nervous system, interactions between transplanted neural precursor cells (NPCs) and host tissue remain incompletely understood. Although trophins may contribute to the benefits observed, little research has explored this possibility. Candidate trophic factors were identified, and primers were designed for these genes. Template RNA was isolated from 3 NPC sources, and also from bone marrow stromal cells (BMSCs) and embryonic fibroblasts as comparative controls. Quantitative polymerase chain reaction was performed to determine the effect of cell source, passaging, cellular differentiation, and environmental changes on trophin factor expression in NPCs. Results were analyzed with multivariate statistical analyses. NPCs, BMSCs, and fibroblasts each expressed trophic factors in unique patterns. Trophic factor expression was similar among NPCs whether harvested from rat or mouse, brain or spinal cord, or their time in culture. The expression of neurotrophin NT-3, NT-4/5, glial-derived neurotrophic factor, and insulin-like growth factor-1 decreased with time in culture. Induced differentiation of NPCs led to a marked and statistically significant increase in the expression of trophic factors. Culture conditions and environmental changes were also associated with significant changes in trophin expression. These results suggest that trophins could contribute to the benefits associated with transplantation of NPCs as well as BMSCs. Trophic factor expression changes with NPC differentiation and environmental conditions, which could have important implications with regard to their behavior after in vivo transplantation.
Introduction
C
Trophic factors could indeed underlie benefits associated with transplantation of both BMSCs and NPCs [10 –12]. Trophins have been ascribed wide-ranging and repair-promoting effects within the CNS. They recruit and stimulate proliferation and differentiation of NPCs. They also have anti-apoptotic effects [13] and enhance axonal regrowth [14], remyelination [15,16], and neuronal plasticity [17,18]. Such effects have been noted after the transplantation of both cell types [10,19 –21]. Unfortunately, the few studies published to date exploring trophin production by these cells have predominantly investigated undifferentiated, multipotent cells in vitro [9]. These findings have been extrapolated to the post-transplant environment [9,20,22 –24], despite the fact that trophin production is likely influenced by the host environment [25] and cellular differentiation in the host tissue [5]. Similarly, trophin expression by NPCs is likely influenced by the in vitro environment in which these cells are grown and passaged. To our knowledge, this possibility has not yet been explored. Due to limitations inherent to in vivo studies, in vitro experimentation can provide insightful information about factors that may influence the expression of trophins by NPCs. The present study thus examined the extent to which cell source, passaging, differentiation, and environment influence the expression of candidate trophic factors in vitro.
Materials and Methods
Animal care and surgical procedures
All experiments were conducted with institutional ethics approval and in accordance with the Canadian Council on Animal Care guidelines.
Isolation of cellular substrates
Adult mouse brain NPCs were isolated from enhanced yellow fluorescent protein (eYFP)-expressing transgenic mice [129-Tg (ACTB-eYFP) 2Nagy/J; The Jackson Laboratory]. The isolation and generation of neurospheres were performed based on methods previously described [26]). Briefly, mice were killed by cervical dislocation, and the brains were excised under sterile conditions and transferred to artificial cerebrospinal fluid (aCSF) solution containing 2 M NaCl, 1 M KCl, 1 M MgCl2, 155 mM NaHCO3, 108 mM CaCl2, 1 M glucose, and 1% penicillin/streptomycin (Sigma-Aldrich). After removing the overlying meninges and blood vessels, the subventricular zone of the forebrain was dissected and transferred to a low calcium aCSF solution (10 mL) containing 40 mg of trypsin, 20 mg of hyaluronidase, and 4 mg of kynurenic acid for 1 h at incubator. Then, the tissue was mechanically dissociated into a cell suspension with a fire-polished Pasteur pipette, and, after cell viability had been assessed by trypan blue staining, the dissociated cells were plated on uncoated tissue culture flasks (10 cells/μL) in a serum-free medium (200 mL) containing 20 mL of Dulbecco's modified Eagle's medium (DMEM)/F-12, 4 mL of 30% glucose, 3 mL of 7.5% NaHCO3, 1 mL of 1 M HEPES, 200 mg of transferrin, 50 mg of insulin, 19.25 mg of putrescine, 20 μL of selenium, 20 μL of progesterone and 1 μg of fibroblast growth factor 2 (FGF2), 2 μg of epidermal growth factor (EGF), and 1% penicillin/streptomycin. The neurospheres were passaged weekly by mechanical dissociation in the same medium (as just described).
Spinal cord NPCs were isolated from the spinal cords of transgenic adult or fetal embryonic day 16 (E16) Wistar rats expressing enhanced green fluorescent protein (eGFP) [Wistar-TgN(CAG-GFP)184ys] (YS Institute, Inc.) [27]. The isolation of NPCs derived from the adult periventricular spinal cord was performed as previously described [28]. For adult rats, cervical and thoracic spinal cord was excised under sterile conditions and washed in Dulbecco's phosphate buffered saline supplemented with 30% glucose (Sigma-Aldrich) and 1% penicillin/streptomycin (Sigma-Aldrich). The overlying meninges, blood vessels, and white matter were removed. Tissue from the periventricular region was then harvested. For the isolation of fetal NPCs, whole spinal cords from E16 rats were excised and similarly processed. Dissected spinal cord tissue was minced, enzymatically dissociated with 0.01% papain and 0.01% DNase I for 1 h at 37°C, and then mechanically dissociated into a cell suspension that was centrifuged through a discontinuous density gradient consisting of a 10 mg/mL albumin-ovomucoid protein inhibitor (Worthington Biochemicals) to remove cell membrane fragments [27]. Cells were resuspended in Neurobasal-A medium (Gibco-Invitrogen) supplemented with B27 (Gibco-Invitrogen), L-glutamine (Gibco-Invitrogen), penicillin/streptomycin (Gibco-Invitrogen), 20 ng/mL each of EGF and bFGF (Sigma-Aldrich), 2 μg/mL heparin (Sigma-Aldrich), and hormone mix consisting of 1:1 DMEM/F-12, 0.6% glucose, 25 μg/mL insulin, 100 μg/mL transferrin, 5 mM HEPES, 3 mM sodium bicarbonate, 30 nM sodium seleniate, 10 μM putrescine, and 20 nM progesterone (all from Sigma-Aldrich). Cells were plated in uncoated Nunc T25 culture flasks (VWR International), and the neurospheres generated were passaged weekly by mechanical dissociation in a serum-free medium as just described.
BMSCs were cultured as previously described [29,30]. Briefly, bone marrow was collected from the femurs and the tibia of eGFP transgenic rats (approximately 40–50×107 cells per rat) and resuspended in long-term bone marrow culture medium. The cells were incubated at 37°C in 5% CO2. Cells were passaged after 2 weeks, and then every 5–7 days. Cells were characterized according to the methods recommended by Dominici et al. [31]. Using this protocol, we have previously shown that BMSCs show no evidence of neural differentiation in vitro [29].
The mouse embryonic fibroblast (MEF) feeder cell line was a generous gift provided by the van der Kooy laboratory [32,33]. About 3×106 cells were provided frozen in a solution containing 500 μL DMEM (Gibco-Invitrogen), 300 μL fetal bovine serum (FBS), and 200 μL of dimethyl sulfoxide. Cells were thawed at 37°C and transferred to gelatin-coated T75 flasks containing 15 mL of medium [13.2 mL DMEM (Gibco-Invitrogen), 1.5 mL FBS, 0.15 mL Glutamax (Gibco-Invitrogen), and 0.15 mL Penicillin/Streptomycin (Gibco-Invitrogen)]. The medium was changed the next day and then twice weekly. Cells were passaged when growth became confluent.
Inducing differentiation
Mouse brain and rat spinal cord NPCs were induced to differentiate in vitro as previously described [5,28]. Briefly, NPCs were plated on Matrigel™ (BD Biosciences) coated culture plates in growth-factor-free media containing 1% FBS for 7 days.
Assessing the effect of environment on trophin expression in mouse NPCs
To determine how trophin production by NPCs is influenced by plating, dissociation, initial proliferating versus initial differentiating conditions, the addition of serum and the addition of EGF/bFGF, passage 5 (P5) mouse brain NPCs were subject to various experimental conditions as outlined in Fig. 1. These experiments were performed at least twice. Three flask environments were compared with 6 plated NPC environments. In all cases, cells were grown in an initial culture condition for 7 days before the media was changed. Cells were then dissociated where indicated, and then cultured for an additional 24 h before RNA was harvested or before cells were fixed for immunocytochemistry.
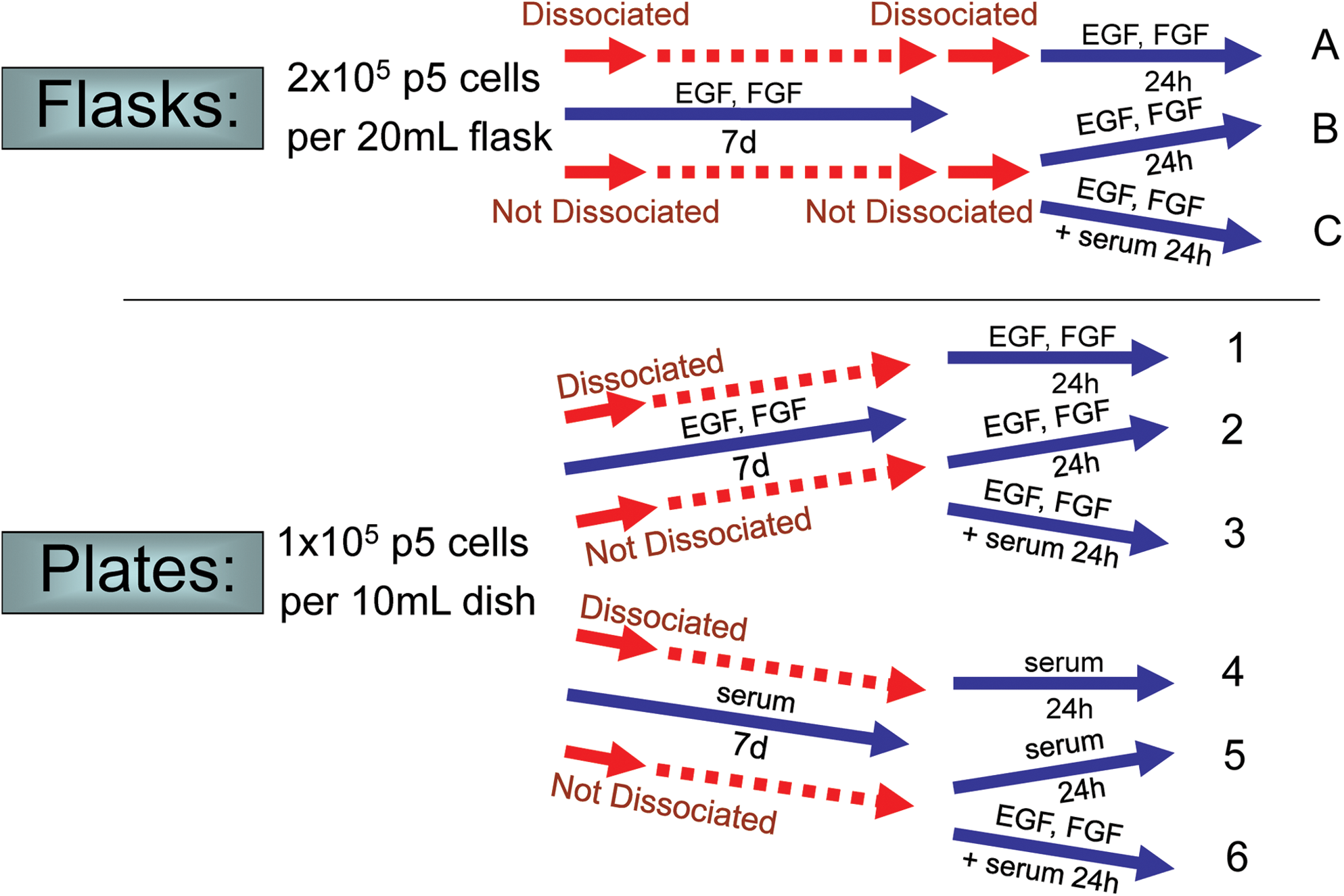
Experimental protocol exploring the effect of culture conditions. Culture conditions employed to explore environmental influences on trophin expression are shown. Color images available online at
For the flasks, 2×105 P5 mouse brain-derived NPCs were initially cultured in serum-free medium as just described containing EGF and bFGF. Cells in Flask A were dissociated by gentle mechanical dissociation with a Pasteur pipette initially and when the media was changed at 7 days. This dissociation was not performed for NPCs in Flasks B or C. At the time the medium was changed in all flasks, EGF and bFGF were maintained in the media; however, 1% FBS was also added to the media in Flask C only.
Plated NPCs were initially grown at a concentration of 1×105 adult mouse brain-derived NPCs per 10 mL matrigel-coated dish. Plates 1–3 contained initial proliferating conditions (EGF, bFGF), while Plates 4–6 were grown in initial differentiating conditions (no EGF/bFGF, addition of 1% FBS). At 7 days, the media was changed. For Plates 1, 2, and 3, the replaced media uniformly contained EGF and bFGF, while Plate 3 additionally contained 1% serum. For Plates 4, 5, and 6, all media contained 1% FBS; media for Plate 6 was additionally supplemented with EGF and bFGF.
Immunocytochemistry
To assess cellular differentiation, mouse brain-derived NPCs were also plated on 6 Matrigel-coated 8-chamber glass slides (3000 cells per chamber). Media was administered to the six 8-chamber slides in exactly the same fashion as the larger plates. After 8 days, the cells were fixed with 4% paraformaldehyde and then washed thrice with phosphate-buffered saline (PBS). The slides were blocked with 1% bovine serum albumin and 0.3% Triton X-100 in PBS for 1 h at room temperature and exposed to the following primary antibodies: mouse anti-GFAP (1:1000; Chemicon) for astrocytes, mouse anti-CNPase (1:100; Chemicon) for mature oligodendrocytes, and mouse anti-Tuj1 (1:500; Covance) for neurons. The cultures were incubated with the primary antibody in blocking solution overnight at 4°C. Cultures were washed thrice with PBS and then incubated with fluorescent Alexa 568 anti-mouse or rabbit secondary antibody (1:300; Molecular Probes) for 1 h, washed thrice with PBS, and coverslipped with Mowiol containing the fluorescent nuclear stain 4′,6-diamidino-2-phenylindole. The determination of positive staining was based on a comparison with 2 negative controls that omitted either the primary or secondary antibody.
RNA isolation and cDNA synthesis
For each undifferentiated precursor cell, different passages were used in order to maximize the amount of information collected, while allowing comparison of trophin expression profiles for different generations of each cell type. Thus, RNA was collected from P2 and P3 passages of undifferentiated mouse brain-derived NPCs, as well as P1, P7, and P9 passages of undifferentiated embryonic rat spinal cord and P3, P6, and P12 passages of undifferentiated adult rat spinal cord. RNA was also collected from differentiated P3 mouse brain-derived NPCs, P9 embryonic rat spinal cord-derived NPCs, and P3 adult rat spinal cord-derived NPCs. In addition, RNA was collected from the cultured MEFs and rat BMSCs previously described.
For RNA isolation, RNaseZAP® (Gibco-Invitrogen) was used to treat all tools, gloves, and working areas employed. Cultured cells were collected and homogenized in Trizol® (Gibco-Invitrogen). After extraction with chloroform, removal of the aqueous layer, precipitation with isopropyl alcohol, and a wash with 75% ethanol, the RNA pellets were resuspended in RNase free water. Spectroscopy was then performed using the NanoDrop 1000 Spectrophotometer (Thermo Fisher Scientific) to ensure successful RNA harvest and determine its concentration. cDNA was subsequently synthesized using Superscript II reverse transcriptase (Gibco-Invitrogen) and Oligo(dT)12–18 primers (Gibco-Invitrogen).
Quantitative polymerase chain reaction analysis
Candidate trophic factors were identified in a literature review. We sought factors consistently found to be produced by neural cells, upregulated at the epicenter of a CNS injury, or of demonstrated benefit in CNS repair [12,25,34
–41]. Primers were then designed for these candidate factors, which included nerve growth factor (NGF), brain-derived neurotrophic factor (BDNF), neurotrophin-3 (NT-3), neurotrophin-4/5 (NT-4/5), glial-derived neurotrophic factor (GDNF), ciliary neurotrophic factor (CNTF), platelet-derived growth factor (PDGF-A), EGF, basic FGF (bFGF), leukemia inhibitory factor (LIF), insulin-like growth factor-1 (IGF-1), glial growth factor 2 (GGF2), transforming growth factor-β1 (TGF-β1), and vascular endothelial growth factor-A (VEGF-A) (Table 1). Primers were also designed for the housekeeping gene glyceraldehyde 3-phosphate dehydrogenase (GAPDH)—the housekeeping gene employed in trophin studies by a number of other groups [42]. Primers were designed with careful consideration of the structure of each gene ensuring the sequence was common to all active isoforms or unique to specific isoforms where indicated. The most highly conserved sequence in these regions was entered into PrimerQuest software (
cDNA template was diluted to 3 ng/μL, and quantitative polymerase chain reaction (qPCR) reactions were performed using SYBR Green chemistry and an ABI 7900HT fast realtime PCR thermal cycler (Applied Biosystems). Absolute quantitation was performed, and all assays were run in at least triplicate. Analysis was performed using SDS 2.3 software.
Statistical analysis
The experiments involving differentiation of mouse brain NPCs (Table 2A–F) were replicated thrice, and those assessing the effect of the environment on trophin expression in mouse brain-derived NPCs, BMSCs, and fibroblasts were replicated twice. These provided highly concordant findings. The results of single experiments were statistically analyzed and presented here. SAS 9.2 software was used for statistical analysis. A univariate analysis of variance (ANOVA) was employed to compare undifferentiated NPCs with BMSCs and MEFs (Fig. 2). Univariate ANOVA was also employed to assess the expression of markers of cellular differentiation). When the overall ANOVA had a P value <0.05, post-hoc testing between groups was performed with Bonferroni correction for multiple comparisons. For variables potentially affecting trophin expression by NPCs (Figs. 3 –5), univariate and multivariate ANOVA were performed. Results were very similar for both analyses, and we have chosen to report the more conservative multivariate statistics (Table 2). NPCs analyzed in distinct experiments were analyzed in separate multivariate models. The first model included species/CNS site, source maturity, passage, and state of differentiation (Table 2A–D). The second model included plate versus flask, cellular dissociation, initial proliferating versus differentiating conditions, additional of serum in the last 24 h, and the addition of EGF/bFGF in the last 24 h (Table 2G–K).
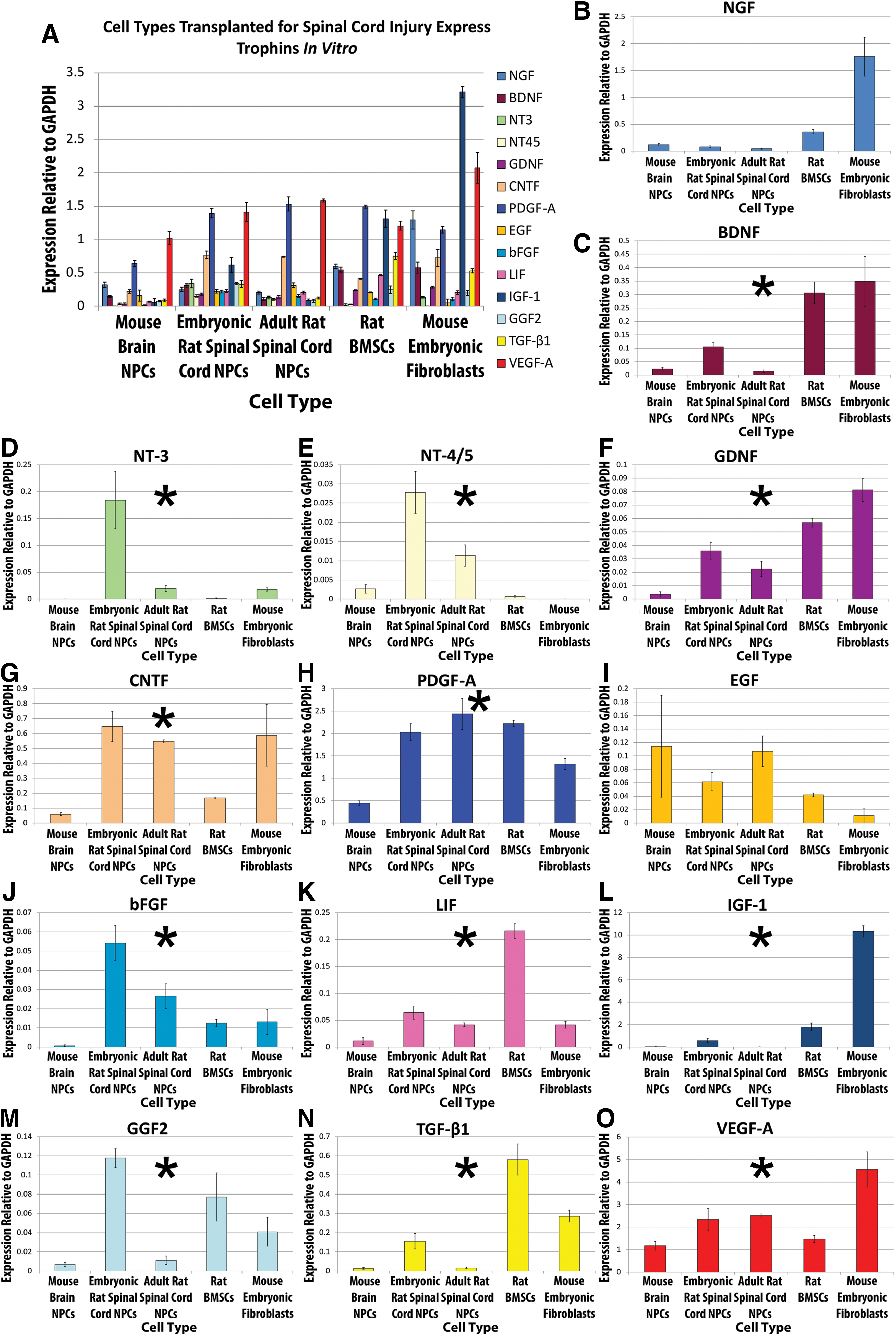
Trophin expression in cultured NPCs, bone marrow stromal cells, and mouse embryonic fibroblasts.
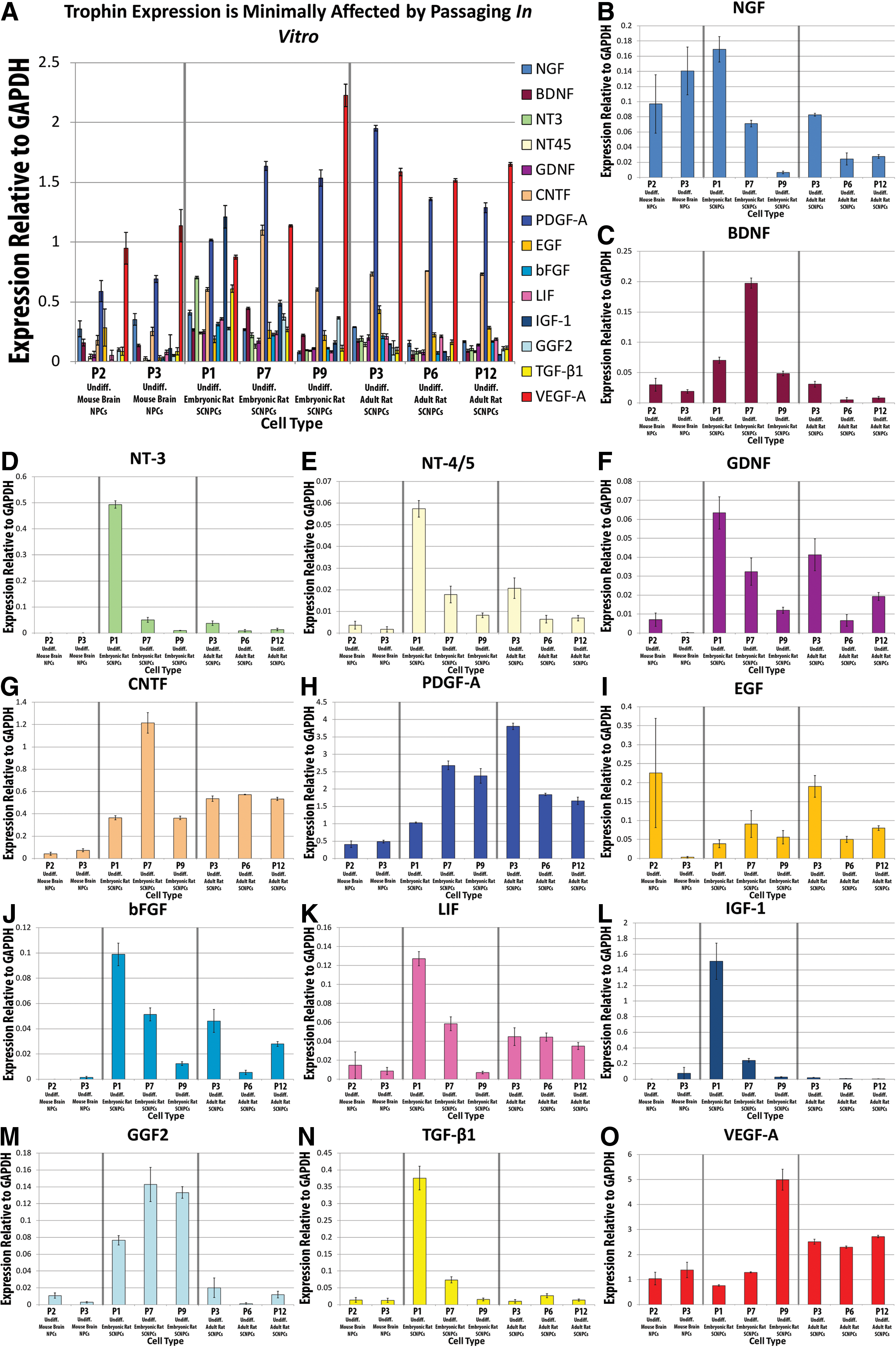
Passaging influences trophin expression in undifferentiated NPCs.
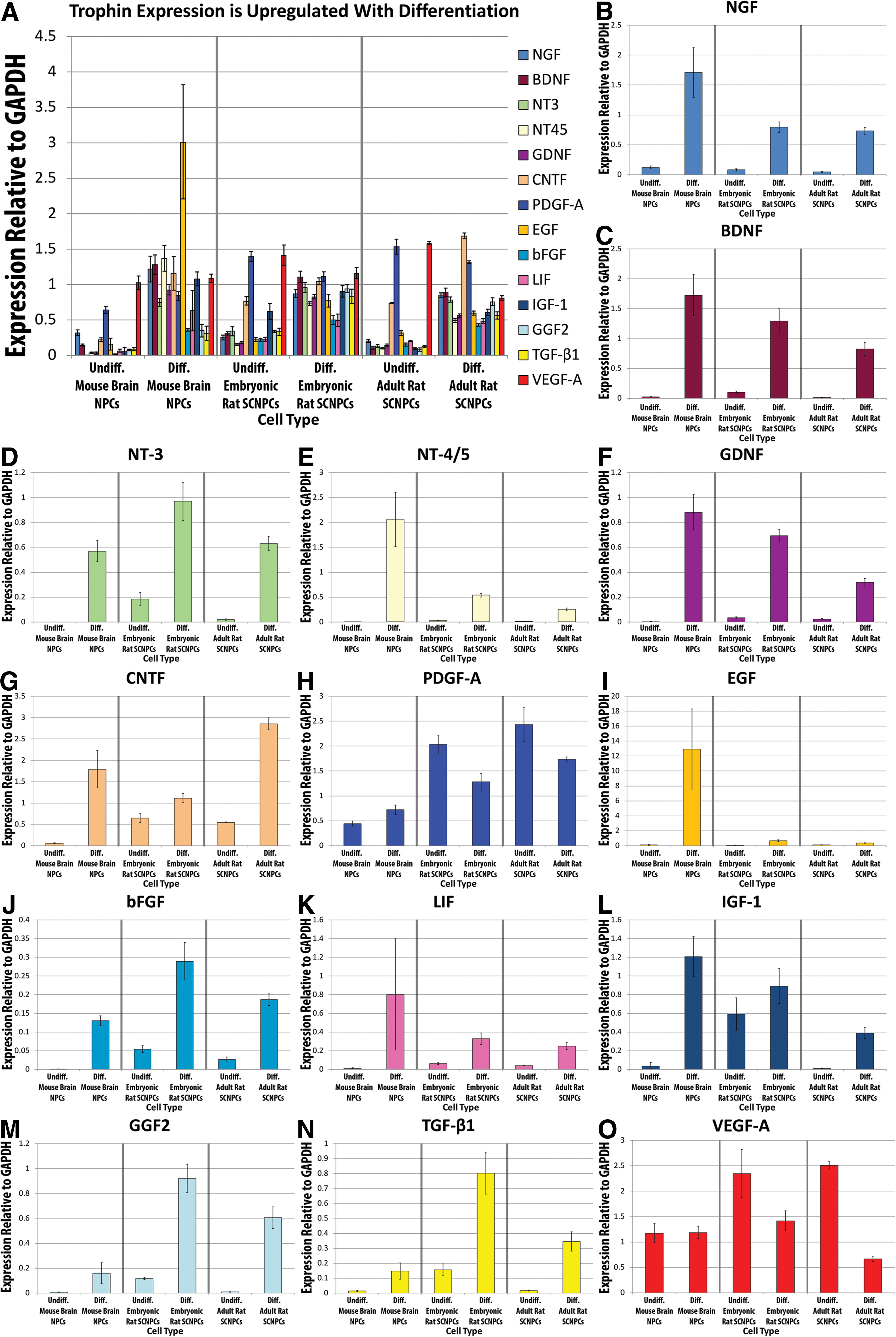
Induced differentiation upregulates trophin expression in cultured NPCs.
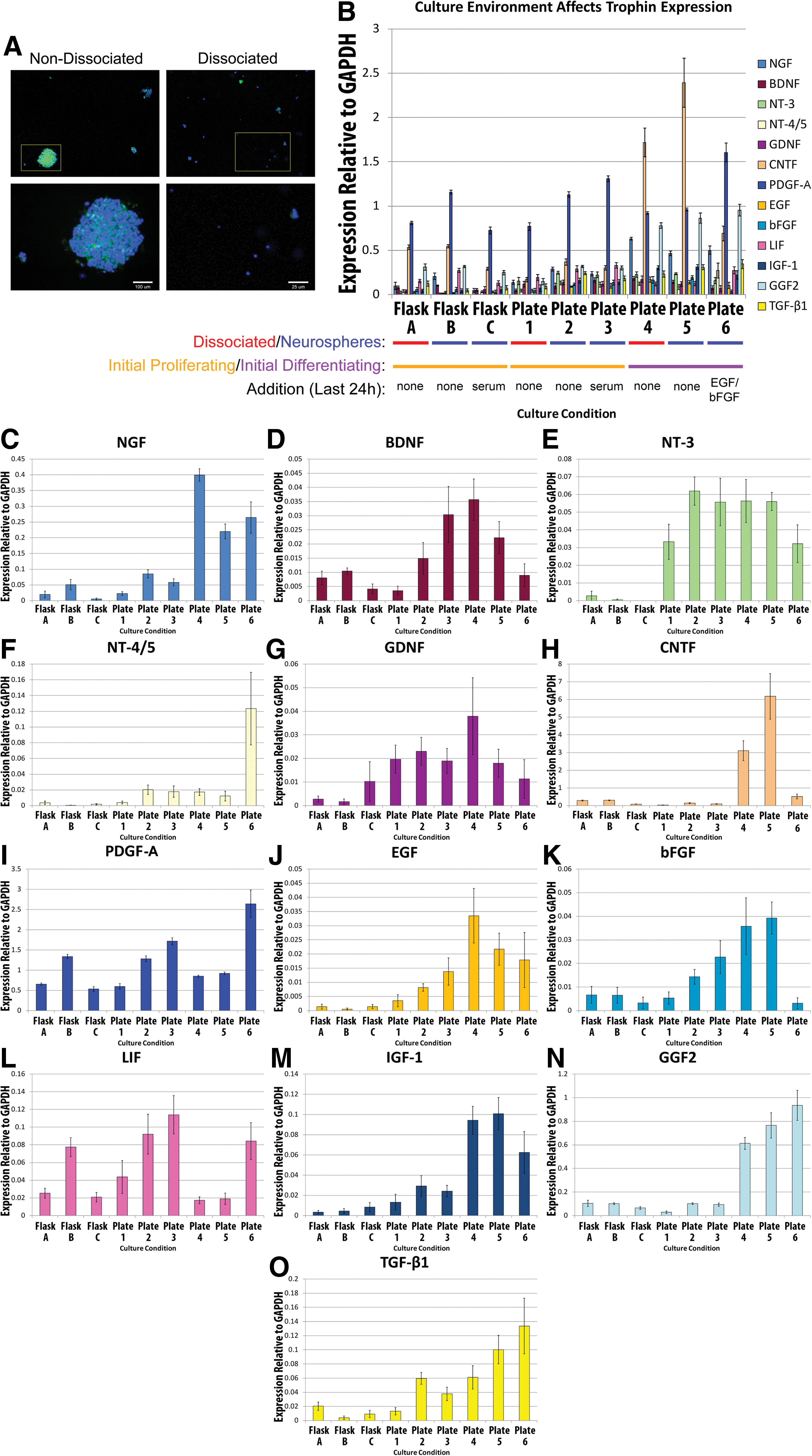
Culture conditions influence trophin expression in NPCs.
A–D and G–K were separately analyzed with multivariate analysis of variance and Bonferroni's post-hoc test. Values for species/CNS site (A) and source maturity (B) are controlled for the effect of passaging and differentiation, while passage (C) has been statistically controlled for NPC type and differentiation. Differentation (D) has been statistically controlled for NPC type and passage. For G–K, each analyzed variable has been controlled for the others.
↑ denotes that the variable of interest is associated with increased trophin expression, ↓ denotes that the variable of interest is associated with decreased trophin expression, ↔ denotes an uncertain trend despite statistical significance; where a group name is present, it indicates a significantly higher level trophin expression for that group.
CNS, central nervous system; EGF, endothelial growth factor; bFGF, basic fibroblast growth factor; B, brain; E, embryonic; R, rat; M, mouse; SC, spinal cord.
Results
Trophic factor expression of NPCs in vitro
Trophic factor expression was characterized in vitro from 3 sources of undifferentiated NPCs and compared with the expression of BMSCs and MEFs. Most trophins were expressed at detectable levels by each cell type as demonstrated in Fig. 2. Univariate statistical analysis revealed that BMSCs and MEFs expressed significantly higher levels of BDNF (P<0.0001), GDNF (P<0.0001), IGF-1 (P<0.0001), and TGF-β1 (P<0.0001) than NPCs. BMSCs expressed significantly higher levels of LIF (P<0.0001) and TGF-β1 (P<0.0001) than either NPCs or MEFs. MEFs expressed significantly higher levels of IGF-1 (P<0.0001) and VEGF-A (P<0.0001) than BMSCs or NPCs.
Qualitatively, undifferentiated NPCs expressed trophins at a similar level and in a similar pattern irrespective of source or species. In a multivariate statistical analysis comparing trophin expression among the NPC sources, statistical differences were seen (Table 2). Mouse brain-derived NPCs expressed a significantly higher level of NGF, NT-4/5, GDNF, and EGF than rat spinal cord NPCs. Murine brain-derived NPCs also expressed significantly lower mRNA levels of BDNF, CNTF, PDGF-A, IGF-1, GGF2, and VEGF-A than rat spinal cord NPCs. Based on multivariate statistical analyses, cell maturity only influenced PDGF-A expression with significantly higher levels seen in the embryonic spinal cord NPC source (Table 2).
Passaging NPCs decreases the expression of many trophins
Trophin expression was compared between numerous passages of NPCs from the adult mouse brain as well as the adult and embryonic rat spinal cord (Fig. 3). Visual inspection suggests that passaging led to a decreased expression for NGF, NT-3, NT-4/5, GDNF, bFGF, LIF, IGF-1, and TGF-β1 as well as an increase in VEGF-A. Statistical analysis summarized in Table 2 , however, demonstrated a significant effect of passaging only for NT-3, NT-4/5, GDNF, IGF-1, and VEGF-A. Significant differences with passaging were also noted for CNTF and PDGF-A however, clear increasing or decreasing trends were not apparent.
Induced differentiation of NPCs led to a significant increase in the expression of trophins
Figure 4 and Table 2 demonstrate that induced differentiation of NPCs in vitro is associated with a marked and statistically significant upregulation of the expression of all trophic factors assayed, except for VEGF-A—which was significantly decreased—after controlling for the effects of cell source, maturity, and the effect of passaging. In all but one instance, the relevant P values were <0.0001, demonstrating a high level of statistical significance.
Culture environment influences trophin expression in NPCs
Analysis of the 9 samples delineated in Fig. 1 demonstrated that the culture environment influences trophin expression in mouse brain-derived NPCs (Fig. 5, Table 2). A multivariate analysis of flasks and plates found that plating was associated with a significant increase in the expression of NT-3, GDNF, PDGF-A, LIF, IGF-1, and TGF-β1. Cellular dissociation was associated with a significant decrease in the expression of PDGF-A, LIF, GGF2, and TGF-β1 as well as a significant increase in NGF expression. Initial differentiating conditions (trophin withdrawal and FBS administration) were associated with a significant increase in the expression of NGF, NT-4/5, PDGF-A, IGF-1, GGF2, and TGF- β1 as compared with initial proliferating conditions. After the initial proliferating conditions, exposure to FBS for 24 h was associated with a significant decrease in CNTF. After the initial proliferating conditions, the addition of serum for 24 h did not influence the expression of any trophins. In contrast, after differentiating conditions, exposure to EGF/bFGF for 24 h was associated with a significant increase in NT-4/5, PDGF-A, and LIF as well as a significant decrease in BDNF, CNTF, EGF, bFGF, IGF-1, and GGF2.
Though cellular differentiation after EGF/bFGF withdrawal and FBS supplementation is well described, Fig. 6 provides evidence that the differentiating conditions in Plates 4–6 were indeed associated with cellular differentiation based on increased expression of the mature cellular markers glial fibrillary acidic protein (GFAP) (astrocytes), 2' 3'-cyclic nucleotide 3'-phosphodiesterase (CNPase) (mature oligodendrocytes), and β3 tubulin (neurons). EGF and bFGF administered for 24 h led to a significant reduction in GFAP and CNPase expression as well as a significant increase in β3 tubulin expression. High baseline expression of CNPase correlates with a high level of CNPase background immunocytochemical staining seen in all specimens. Differentiating conditions were associated with clear morphological changes in cellular appearance (Fig. 6D) including an increase in cell size and complexity as well as reduced eYFP intensity. Positive immunocytochemical labeling for mature astrocytes, oligodendrocytes, and neurons was observed in Plates 4, 5, and 6, which is consistent with the qPCR results shown in Fig. 6A–C.
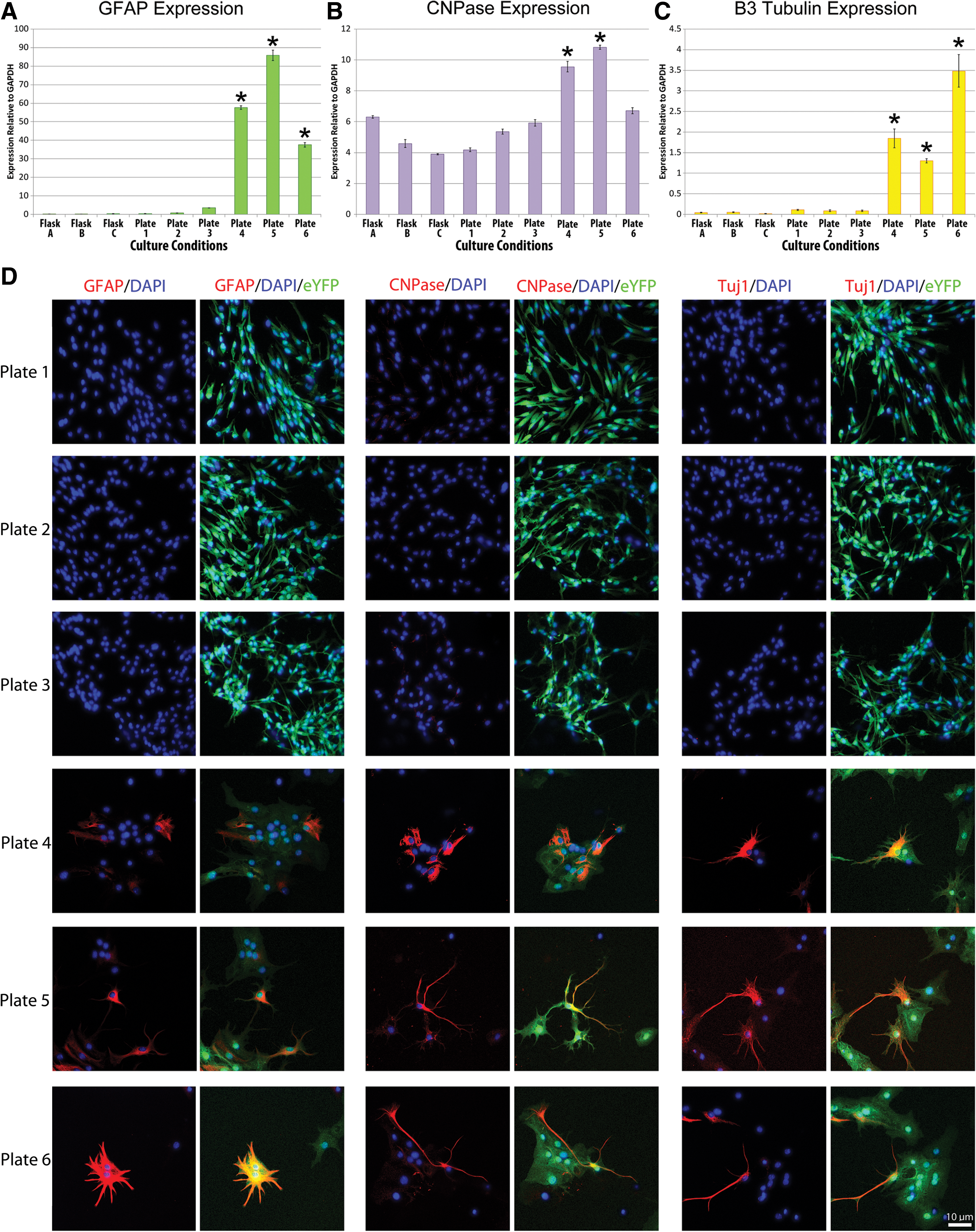
Culture conditions influence NPC differentiation. qPCR results for the expression of cellular differentiation markers are shown. Experiments have been performed in triplicate, and error bars represent standard error. Overall ANOVA for
Discussion
Our study adds to the published literature by presenting the most thorough characterization of trophin expression by NPCs to date with a comparison to BMSCs and MEFs. The data presented here also delineate factors that influence the level of expression by NPCs and provide important information on the relative expression of trophins by the different cell types.
Undifferentiated NPCs, BMSCs, and MEFs express trophins in vitro
NPCs and BMSCs (Fig. 2) have been extensively studied in pre-clinical studies in CNS repair [5,28,43 –46]. Similar cell types have been employed in human studies [46 –51]. Here, we present a direct comparison of trophin expression by these cell types with comparison to MEFs. Of importance, the trophin values presented in Fig. 2 likely reflect cellular production at the time of transplantation.
Our study demonstrates that undifferentiated NPCs express detectable levels of all trophins in culture. Trophin expression among the 3 NPC sources was qualitatively similar, although some significant differences were apparent (Fig. 2). We are unable to determine whether trophin expression differences between rat and mouse NPCs are related to species, brain versus spinal cord source, or differences in culture methods. Nonetheless, the degree of similarity observed is surprising, given that Neuhuber et al. found that cells isolated from the same site of different human donors can differ in their trophin level [47]. Smith et al. found that proliferation of NPCs was more dependent on site of origin than culture conditions [48]. The degree of similarity observed was also unexpected given that mouse and rat cells were cultured using different methods. The method of culture preparation has been shown to have an important influence on the biology and biological effects of cells transplanted for SCI [48,49]. Such differences may at least partly account for the differences observed in the present study.
Where comparison is possible, our characterization of trophin expression by NPCs is generally consistent with previous reports. Several other studies have provided more limited characterizations of trophin production by NPCs in vitro. In 2004, Lladó et al. found that conditioned medium from C17.2 neural stem cell cultures contained NGF and GDNF but not BDNF or NT-4/5 based on enzyme-linked immunosorbent assay (ELISA) assays. This medium supported axonal growth in vitro, which could be prevented by GDNF sequestration [50]. Kamei et al. reported similar findings in an organotypic slice culture model [51]. This group reported positive immunostaining of undifferentiated NPCs for BDNF, NT-3, NT-4/5, and CNTF; however, there is a possibility that this represents misinterpretation of background staining. Sequestration of BDNF, NT-3, and NT-4/5 restricted axon outgrowth in their brain and spinal cord co-culture model; however, there was no evidence that the sequestered factors were produced by “transplanted” NPCs. Zhang et al. used microarray and confirmatory ELISA to demonstrate that human embryonic stem cell-derived oligodendrocyte precursor cells secrete functional levels of midkine, hepatocyte growth factor, activin A, TGF-β2, and BDNF in vitro, and additionally demonstrated the stimulatory effect of these proteins on neurite outgrowth of adult rat sensory neurons in vitro [52]. Einstein et al. also showed that their NPCs in culture express IGF-1, NGF, BDNF, GDNF, NT-3, CNTF, LIF, and erythropoeitin [53].
Similar to NPCs, BMSCs express detectable levels of all assayed trophins, but significantly higher levels of LIF and TGF-β1 than any other cell type. Elevated LIF could be beneficial, as it enhances cell survival after insult [54 –59] as well as corticospinal tract growth [39], though it also increases astrogliosis [60 –62] and the inflammatory response [54]. TGF-β1 is, however, an anti-inflammatory cytokine [63,64] that has been shown to decrease neuronal loss after injury [65]. Relevant to the present work, several studies have investigated trophin production by BMSCs. Supporting our findings, Nakano et al. recently characterized BMSC conditioned media and using ELISA demonstrated the presence of IGF-1 and TGF-β1 [66]. Of note, Neuhuber et al. also found that BMSCs secrete VEGF, BDNF, IL-6, monocyte chemotactic protein-1, stem cell factor, and stromal cell-derived factor-1a into media in vitro [47]. Others have shown that BMSCs additionally express or secrete NGF, NT-3, GDNF, and hepatocyte growth factor in vitro [67,68].
Trophin expression by MEFs is of interest not only because they are a substrate for culturing human embryonic stem cells, but also because they have also been viewed as a potential cellular control for transplantation studies. While we are unaware of studies exploring trophin expression by these cells, conditioned media from MEFs has been subject to repeated proteomic analysis, which is not surprising given its scientific importance in supporting the growth of human embryonic stem cells and maintaining their pluripotency [69 –71]. In our hands, our MEF cells express very high levels of IGF-1. In accordance with this finding, high levels of IGF-1 have been detected in MEF conditioned media along with IGF binding proteins, and additional trophins pigment epithelium derived factor, follistatin-related protein 1, prostacyclin stimulating factor, and Gremlin 1 [69 –71]. Our results show that it may be appropriate to reconsider the use of fibroblasts as a cellular control for transplantation studies, as IGF-1 has been demonstrated to preserve neurons [72,73] and to increase the outgrowth of corticospinal axons [74]. Further studies are required in order to fully delineate the validity of using fibroblasts as a cellular control in transplantation studies.
Effect of cell source, passaging, and differentiation on trophin expression in NPCs
Although in our multivariate analysis only PDGF-A was expressed at significantly higher levels in embryonic than adult NPCs, there was a general suggestion that embryonic NPCs may be a richer source of trophins than their adult-derived counterparts. This is concordant with the work of Nakamura and Bregman, who characterized trophin expression in the neonatal and adult rat spinal cords after injury [75].
Passaging led to a decrease in the level of many trophins over time (Fig. 3, Table 2). This effect was most evident in our rat samples whose passages were widely spaced. These differences were generally modest and of uncertain biological significance, but may be of relevance to cell lines that are maintained in culture for prolonged periods of time. Indeed, Smith et al. provide evidence for differences in NPC biology with prolonged culture duration, noting a decline in proliferation and neuronal differentiation with time [48]. If trophins prove to be important to the mechanism of cells transplanted for SCI, cells from earlier passages may be better to transplant.
Induced differentiation of NPCs led to a marked and statistically significant upregulation of all the trophins examined except for VEGF-A (Fig. 4). These findings suggest that NPCs may upregulate trophin production once introduced into the injured spinal cord; however, this finding cannot be extrapolated to the post-transplant environment with any certainty. Trophin production by cells post-transplant should be measured directly although this presents significant methodological challenges, particularly if secretion of trophin proteins by transplanted cells is to be demonstrated.
Effect of culture environment on trophin expression in NPCs
BMSCs have been shown to alter trophin expression in response to environmental changes in vitro [76,77]. However, this has not been demonstrated for NPCs. To determine the extent to which changes in the environment influence trophin production by NPCs, we performed an experiment in which multiple aspects of the NPC culture environment were altered (Fig. 1). This experimentation explored many of the variables involved in cellular differentiation to determine whether the results shown in Fig. 4 may be confounded by effects of plating, cellular proximity, or differential presence of EGF/bFGF or FBS. Figs. 4 and 5 suggest that trophin upregulation associated with induced differentiation may relate at least in part to the plating process as well as exposure to old media. Plating alone led to an upregulation of many trophins, in particular NT-3, GDNF, PDGF-A, LIF, IGF-1, and TGF-β1. To our knowledge, these variables have not been previously examined for their influence on trophin expression.
Cellular dissociation was investigated, because cell-cell contact has demonstrated important biological effects in stem and progenitors cells. For instance, cellular dissociation promoted cellular differentiation in some in vitro culture conditions [78]. Preventing cellular dissociation during passaging has been shown to enhance neurosphere growth and, thus, could have an influence on trophin production [79]. In our analysis, cellular dissociation led to significant decreases in LIF and PDGF-A; given the well-known proliferative effects of these trophins on NPCs, it is possible that this may in part explain why cellular dissociation inhibits NPC proliferation [80 –82].
Of interest, Schwindt et al. studied the effect of EGF/bFGF withdrawal on NPCs. Their group noted that this leads to an upregulation of IGF-1 and that IGF-1 can replace the biological effects of EGF and bFGF. Erickson et al. also noted that IGF-1 could lead to prolonged NPC survival when substituted for EGF and bFGF [83]. EGF and bFGF withdrawal led to marked IGF-1 upregulation in our study (Fig. 5). Of interest, EGF and bFGF removal in plates 4, 5, and 6 led to upregulation of these factors as compared with plates 1, 2, and 3; in plate 6 where these factors were re-introduced, their expression levels decreased. These findings are suggestive of negative feedback.
Factors influencing induced differentiation of NPCs
The differentiating conditions employed in the present study are well known to induce the in vitro differentiation of NPCs into mature neural cell types [5]. Figure 6 confirms that these conditions promote cellular differentiation. Moreover, these results demonstrate that plating, serum administration, and cellular dissociation alone do not appear to influence cellular differentiation within the experimental conditions explored. As might be expected, at the 7-day time point, EGF and bFGF administration to differentiated NPCs reduced the expression levels of surrogates of cellular differentiation GFAP and CNPase consistent with their role in stimulating survival and proliferation but not differentiation of NPCs [84]. This is consistent with the results of Schwindt et al. who found that EGF and bFGF withdrawal increased the expression of these genes [84]. The finding that EGF and bFGF increased β3-tubulin expression is consistent with recent findings from Garcez et al., who found that EGF and bFGF together enhance neurogenesis in neural crest cells [85].
Relevance to cellular transplantation
Our findings have important implications for cellular transplantation strategies in regenerative medicine. The present study confirmed our hypothesis that differentiation and the environment influence the trophin expression profile of NPCs and suggests that one should not extrapolate trophin levels measured in one environment to another—in particular the post-transplant environment as many have done. Indeed, selecting a given cell type based on its in vitro expression profile (as shown in Fig. 2) is likely of little value, as this probably reflects trophin production at the time of transplant only. This study thus highlights the importance of studying trophin production by transplanted cells in vivo.
It should be noted that we cannot be certain that the trophin expression levels presented here reflect protein levels nor protein secretion. For instance, the proteomic analyses of MEF conditioned media identified only the most highly expressed trophin we measured (IGF-1) above detection threshold [69]. qPCR results may detect multiple isoforms that can have different biological activity. Also problematic is that qPCR can precisely define an expression level leading to statistically significant differences that may not be of biological significance. Analysis of trophin expression, thus, has not only inherent limitations but also important advantages in comparison with protein studies. qPCR is extremely sensitive, which is important given that many trophins have biological effects at picomolar concentrations. Also immunocytochemistry performed on cultured cells can be misinterpreted [51,67]. Moreover, Okragly and Haak-Frendscho demonstrated that trophin proteins are subject to poorly understood species and tissue-specific sequestration that can render protein assays inaccurate [86].
Conclusions
Our results have quantitated the expression of numerous trophic factors produced by NPCs and compared these results to those obtained from BMSCs and MEFs. We have also characterized numerous factors that influence the expression of trophins by NPCs and have shown that this expression is highly dependent on environmental influences. While these findings suggest that these cells could be poised to provide trophic support after transplantation into the CNS, our results also suggest that environmental cues could have a dramatic influence on the trophic factor expression by NPCs.
Footnotes
Acknowledgments
The authors gratefully recognize the gift of the embryonic feeder cells from the van der Kooy laboratory. They also wish to thank Xing-Hua Wang and Armand Keating for provision of BMSCs. They are grateful to Craig H. Neilsen Foundation, the Cervical Spine Research Society, the Canadian Institutes of Health Research and the Krembil Foundation who provided funding for these experiments. G. Hawryluk was supported by fellowships from Johnson and Johnson Medical Products, the McLaughlin Centre for Molecular Medicine, and the Congress of Neurological Surgeons/American Association of Neurological Surgeons.
Author Disclosure Statement
No competing financial interests exist.
