Abstract
The Editor-in-Chief of Stem Cells and Development officially retracts the article entitled, “Detection, Characterization, and Spontaneous Differentiation In Vitro of Very Small Embryonic-Like Putative Stem Cells in Adult Mammalian Ovary,” by Seema Parte, Deepa Bhartiya, Jyoti Telang, Vinita Daithankar, Vinita Salvi, Kusum Zaveri, and Indira Hinduja (Stem Cells Dev 2011;20(8):1451-1464; doi: 10.1089/scd.2010.0461).
A reported comment on the PubPeer platform suggested that the “images in Figure 6D and Figure 6F appear to overlap but are described differently. The images are oriented differently, have different contrast, and are at a different magnification (red boxes are not the same size). Difficult to understand how these transformations would occur merely as errors during figure assembly,” (PubPeer, 2022).
The Editor-in-Chief of the journal contacted the corresponding author of the paper, Dr. Deepa Bhartiya, requesting a “prompt response and intended course of action” to the questions raised on PubPeer. Dr. Bhartiya responded, indicating that the errors in both figures were “an unintentional human error.”
After a series of communications between Dr. Bhartiya and the Editor-in-Chief of Stem Cells and Development, Dr. Bhartiya claimed full responsibility for the contents of the article and requested that corrections to the paper by removing the incorrect images, as indicated in the email below:
“A discrepancy in two figures (Fig 6D and negative control in Fig 11) was picked up by an Artificial Intelligence based tool and pointed out on Pubpeer. A careful examination shows that indeed there is a mistake but it was inadvertent and simply an oversight and not a deliberate manipulation of any kind. It is the same figure of a neuron turned up side down and pasted at two places. This remained unnoticed by the authors and also was not picked up by the reviewers or the readers [for] over a decade. It is our humble request to all those who have cited our work to be rest assured that the work integrity is intact. We apologize for the inconvenience caused to those directly and indirectly affected by this process. My students worked with great dedication to acquire the data and obtained results to compile into a manuscript. There is no manipulation whatsoever and this unintentional error remained unnoticed by everyone involved with the manuscript preparation and publication. We will try to submit the revised version again with due permissions. We request the editor to allow us to publish an addendum/erratum to take care of the mistake otherwise it is his decision to retract the paper.” [sic]
The Editor of the journal rejected the contention that the manipulation and combining of the same images within the panels purported to represent different experiments could be unintentional, and hence has denied the request to publish an erratum and offered the authors the opportunity to self-retract the article. The authors declined to do so and continued to request a corrigendum. The Editor determined that an editorial retraction was warranted based on the discovery of the discrepancies in the images. Dr. Bhartiya was notified via email of the decision to editorially retract the paper.
Reference
Introduction
O
Recent reviews have elegantly discussed the available data on the presence of putative stem cells (PSCs) in the adult mammalian ovary that may result in oogenesis in postnatal life [3 –7], similar to lower species like flies, birds, and fish [8 –11]. There is piling evidence hinting toward the existence of PSCs within the mice ovary [12,13], which may have an origin from bone marrow [14]; 6 months' culture of adult female mice germ-line stem cells and later transplantation in busulphan treated mice ovaries result in live offspring [15]; label-retaining stem cell population in mice OSE [16]; presence of multipotent stem cells with germ line potential in postnatal mouse ovary [17], and recent derivation of 2 colony-forming cell cultures with markers similar to embryonic stem (ES) cells, embryoid bodies, and teratomas from stem cells in the ovarian stroma [18,19]. Similarly, Bukovsky and his group [20] reported the presence of bipotent progenitor cells, and Virant-Klun and group [6,21,22] reported stem cells in the adult human ovaries that develop into oocyte-like, embryoid body-like, and parthenote-like structures. These data provide hope that in near future the potential of PSCs in OSE may be exploited to procreate life to help infertile couples. However, lot of skepticism exists regarding postnatal oogenesis since it contradicts 5-decade-old paradigm of fixed germ-cell pool in females, which has been aptly discussed by Tilly's group [23].
Bukovsky and co-workers have suggested that OSE cells, which differentiate from underlying mesenchymal cells in tunica albuginea, are the bipotent source of germ and granulosa cells [5,24,25], but if the views of Bukovsky's group are valid, one would not expect generation of chimeric follicles reported by others [12,15]. Ovarian grafting with EGFP-tagged germline stem cells result in chimeric follicles with EGFP-positive oocytes enclosed with EGFP negative granulosa cells in the wild-type ovarian tissue [12]. Also GFP-tagged germline stem cells on transplantation in the busulfan-treated mice ovary, result in similar chimeric newly assembled follicles [15].
There is a dearth of available information on the presence of stem cells in OSE, and in our understanding of germ cell renewal, follicular assembly and further studies are required to better understand these processes during postnatal life in higher mammals. Barring few studies on humans and mice, no information is available about postnatal oogenesis in other higher mammalian species so far. Hence, present study was conducted with the aim to investigate stem cells in OSE and in vitro oogenesis in higher mammals like rabbit, sheep, monkey, and menopausal women by using in vitro culture of OSE as the study model.
Materials and Methods
Ovarian biopsies were collected from menopausal women (n=6) with a mean age range of 46 years (40–55 years) undergoing total abdominal hysterectomy or ovariectomy due to various gynecological pathologies other than ovarian pathology, infection, or malignancy, after obtaining required ethical permissions. The tissue was collected during surgeries carried out at King Edwards Memorial Hospital and Jaslok Hospital and Research Centre, Mumbai, and brought to National Institute for Research in Reproductive Health (NIRRH) for further studies. Intact ovaries were also collected from normal adult rabbits (n=3), sheep (n=30), and marmoset monkeys (n=3) after approval from NIRRH Animal Ethics Committee. The samples were collected in 0.9% normal saline-containing antibiotics (penicillin 100 U/mL, streptomycin 100 μg/mL; Invitrogen) at ambient temperature for transport to the laboratory as soon as possible. A small ovarian cortical piece was fixed in 10% neutral buffered formalin at 4°C for preparing hematoxylin and eosin (H&E)-stained sections by standard methods. Various experiments carried out on the ovarian tissue obtained from different species based on ready availability and reagents are mentioned in Supplementary Table S1.
Scraping of OSE
The ovaries were gently rinsed several times in calcium- and magnesium-free Dulbecco's phosphate-buffered saline (DPBS; Invitrogen) containing antibiotics and kept in plain high-glucose DMEM/F12 and antibiotics before scraping of OSE. The ovarian surface was easily identified and was gently scraped to release cells with the help of a sterile blunt cell scraper into plain DMEM/F12 in a 60 mm dish under sterile conditions at 37°C on a preheated stage of IVF workstation (K Systems; Kivex Biotech Ltd), without damaging the underlying cortical tissue and unnecessary manipulations during harvesting were avoided. OSE is easily detached as it is loosely bound to its basement membrane [2]. The scraped OSE cells were examined under Hoffman optics on an inverted microscope (Eclipse TE 2000-S NIKON) for the presence of stem cells. H&E stained sections of ovarian tissue and scraped OSE cells were examined under bright field microscope (90i, NIKON). Images of representative areas were captured.
Culture of OSE
The scraped OSE cells with the medium were transferred to a 15 mL centrifuge tube and spun at 1,000 g for 10 min at 25°C. The pellet was later suspended in fresh medium and cultured in DMEM/F12 supplemented with 20% fetal bovine serum and antibiotics in 5% CO2 incubator at 37°C for all cultures except the sheep OSE cells which were maintained at 38.5°C for 3 weeks. Partial medium change was done every alternate day and cultures were carefully monitored under inverted microscope (Eclipse TE 2000-S; NIKON) with Hoffman optics on a warm-stage maintained at 37°C (38.5°C in case of sheep cultures). The cultures were dynamic in nature and within a few days one could see the epithelial cells attached to the bottom of the culture dish, whereas the round cells were slightly attached and became bigger in size and differentiated over time. All this was carefully (NUNC) observed and recorded regularly. Disposable culture-ware was used throughout the study. Cultures were terminated at the end of a 3-week period, and processed appropriately for reverse transcriptase–polymerase chain reaction (RT-PCR) and immunolocalization studies.
Characterization studies
Cells collected after scraping ovarian surface and those after 3 weeks of culture were fixed on ice in fresh 4% paraformaldehyde (pH 7.4; Sigma) for 10 min. The OSE smears were air-dried, washed twice with DPBS, air-dried again, and stored at 4°C till further use, whereas cultured cells were washed with DPBS postfixation, air-dried subsequently, and stored at 4°C till further use. Besides, the OSE cells were also stored in TRIZOL (Invitrogen) at −80°C for RNA extraction.
Both sheep and human OSE stem cells and oocyte-like structures and the phenomenon of epithelial-mesenchymal transition (EMT) were characterized using specific markers by immuno-localization and RT-PCR studies. In addition, alkaline phosphatase activity was assessed on ES cell-like colonies.
All the characterization studies were carried out on a minimum of 3 samples each and repeated at least 3 times.
Immunolocalization studies
Stem cell markers
Oct-4 and SSEA-4 staining was carried out on both human and sheep OSE. The details of various antibodies and detection system for immuno-localization of stem cell markers are listed in Supplementary Table S2. For immuno-localization of Oct-4, the cells were permeabilized with 0.3% Triton X-100 for 10 min at room temperature, whereas this step was avoided while staining cells for cell surface marker SSEA-4. Nonspecific epitopes were blocked by incubation with blocking buffer (DPBS containing 3% bovine serum albumin and 0.1 mM EDTA) for half an hour at room temperature. Briefly, after overnight incubation with respective primary antibodies at 4°C, cell smears were washed with washing buffer (DPBS containing 0.5% bovine serum albumin and 0.1 mM EDTA) and later incubated with Alexafluor 488 (Molecular Probes, Invitrogen) labeled anti- rabbit IgG or anti-mouse IgG (1:1,000) in wash buffer for 4 h. After several washes with washing buffer, the cells were counterstained for 20 s with 4’,6-diamidino-2-phenylindole (DAPI), mounted in Vecta Mount medium (Vector Laboratories Inc), and examined under confocal fluorescent microscope (LSM510-META; ZEISS). Respective negative controls with omission of primary antibody were also done. Experiments were repeated thrice and images of representative areas were captured. All images were captured by laser scanning confocal microscope at 882× magnification with 5× optical zoom using argon laser at λ=488 nm and blue diode laser at λ=405 nm for FITC and DAPI staining channels, respectively.
Germ cell markers
Germ cell markers c-Kit, DAZL, GDF-9, VASA, and ZP4 were localized in both human and sheep oocyte-like structures by using similar method as mentioned for Ck-18 (Supplementary Data for details; Supplementary Data are available online at
RT-PCR studies
Total RNA was extracted using TRIZOL (Invitrogen) from scraped OSE cells and cells postculture for studying stem and germ cell-specific gene transcripts by semi-quantitative RT-PCR method. Appropriate positive control, i.e., in-house-derived human ES cells [26] and adult human testicular tissue (collected from prostate cancer patients as part of their clinical management) were also processed similarly.
After incubating the extracted RNA samples with 1500U of ribonuclease-free deoxyribonuclease (DNase 1; Qiagen) at 37°C for 30 min, RT-PCR studies were carried out. First-strand cDNA was synthesized using Sensiscript RT Kit (Qiagen) according to the manufacturer's instructions. Briefly, 10 μM oligo dT primers, 0.5 mM dNTPs, 5 U of Moloney murine leukemia virus RT, and 10 U of RNase inhibitor in 20 μL reaction mixture were used to synthesize the first-strand cDNA by incubating at 70°C for 2 min and then at 37°C for 1h. Placental RNA (1 μg/μl, provided with the kit) was reverse transcribed simultaneously with sample RNA for use as positive control. Genomic DNA contamination was ruled out by omission of Reverse Transcriptase enzyme during reverse transcription reaction and use of this product as one of the control samples during PCR. Similarly RT kit component contamination was ruled out by omission of sample RNA in one sample used as negative control. cDNA mix (2.0 μL) was amplified using 10 pmol of each primer, 1U Taq DNA polymerase, 1.5 mM MgCl2, and 10 mM dNTPs in a 25 μL reaction volume in a G STORM thermal cycler (Gene Technologies). The products along with 100 bp DNA ladder were electrophoresed on a 2% agarose gel, stained with 0.5 μg/mL ethidium bromide, and observed under UV trans-illumination. The negative control did not include cDNA in the PCR reaction mixture. Respective RT and PCR negative controls were employed during all experiments conducted in triplicates.
Detection of alkaline phosphatase activity in ES cell-like colonies
ES cell-like colonies that appeared spontaneously in the OSE culture were fixed with 4% paraformaldehyde for 10 min, washed with DPBS, and then stained for alkaline phosphatase activity (Chemicon) as per the manufacturer's protocols. The stained structures were viewed and photographed under inverted microscope (CKX 41, Olympus Europa, GmbH).
Results
The H&E-stained sections of menopausal human ovarian samples, included in the present study, were devoid of follicles in the cortex and had a well-defined OSE comprising of continuous layer of columnar to cuboidal epithelial cells. The sections of adult sheep ovaries were rich in healthy follicles situated in the cortex covered with well-defined OSE comprising of flat to cuboidal epithelial cells (Fig. 1A, B).
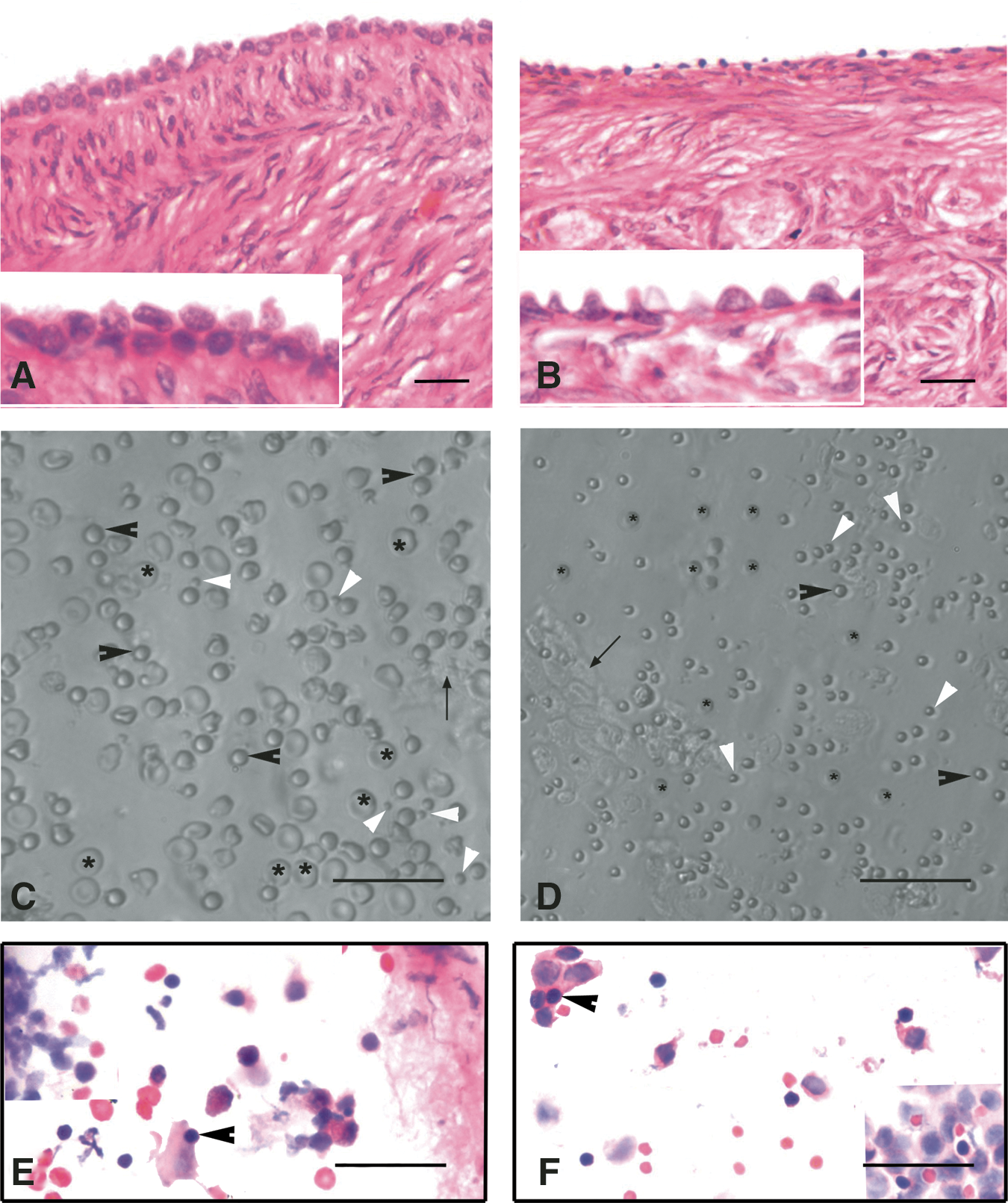
H&E-stained sections of menopausal human and adult sheep ovarian cortex
Stem cells in OSE
Small round PSCs were detected interspersed with few red blood cells (RBCs) or trapped in epithelial cell clusters in all the species studied (Figs. 1 and 2). They appeared dark as compared to the rest of the cell types, had a characteristic bubbly, shiny appearance, and could easily be distinguished from the surrounding (RBCs), which had typical biconcave disc-like appearance with an irregular shape.
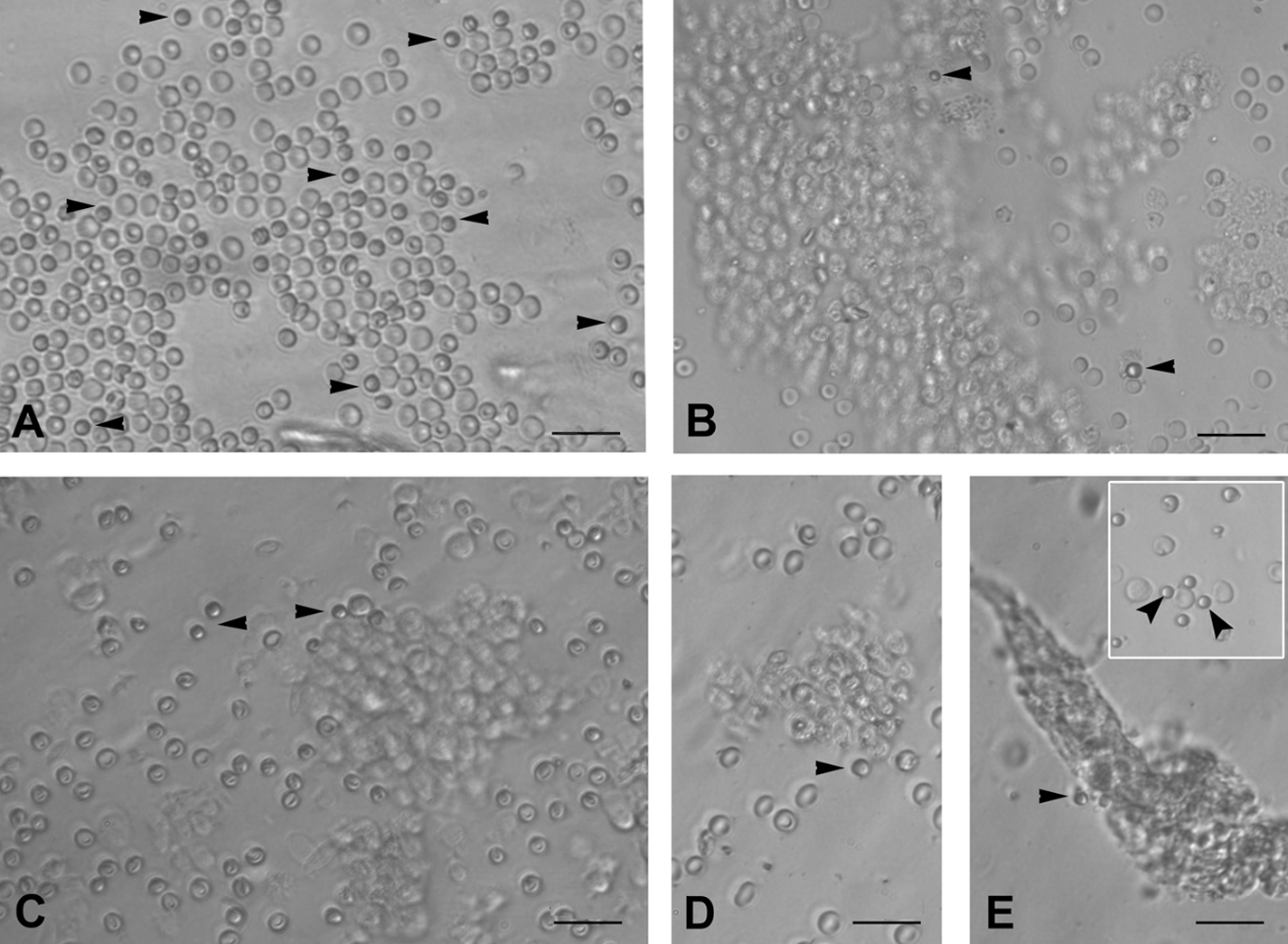
PSCs interspersed with RBC and clusters of epithelial cells just after scraping ovarian surface in different mammalian species:
A careful examination of the OSE cell smears demonstrated the presence of 2 distinct populations of round PSCs of different size (Fig. 1C, D). The stem cells were observed with greater clarity after H&E (Fig. 1E, F) and DAPI staining (Supplementary Fig. S1). H&E staining of both human and sheep OSE cells revealed the presence of RBCs, epithelial cells with abundant cytoplasm, and round PSCs (arrowhead) with large, darkly stained nuclei surrounded by a thin rim of cytoplasm. The PSCs nuclei stained dark as compared to the surrounding epithelial cells (Fig. 1E, F).
Culture of scraped OSE cells
Spontaneous differentiation of PSCs into oocyte-like structures
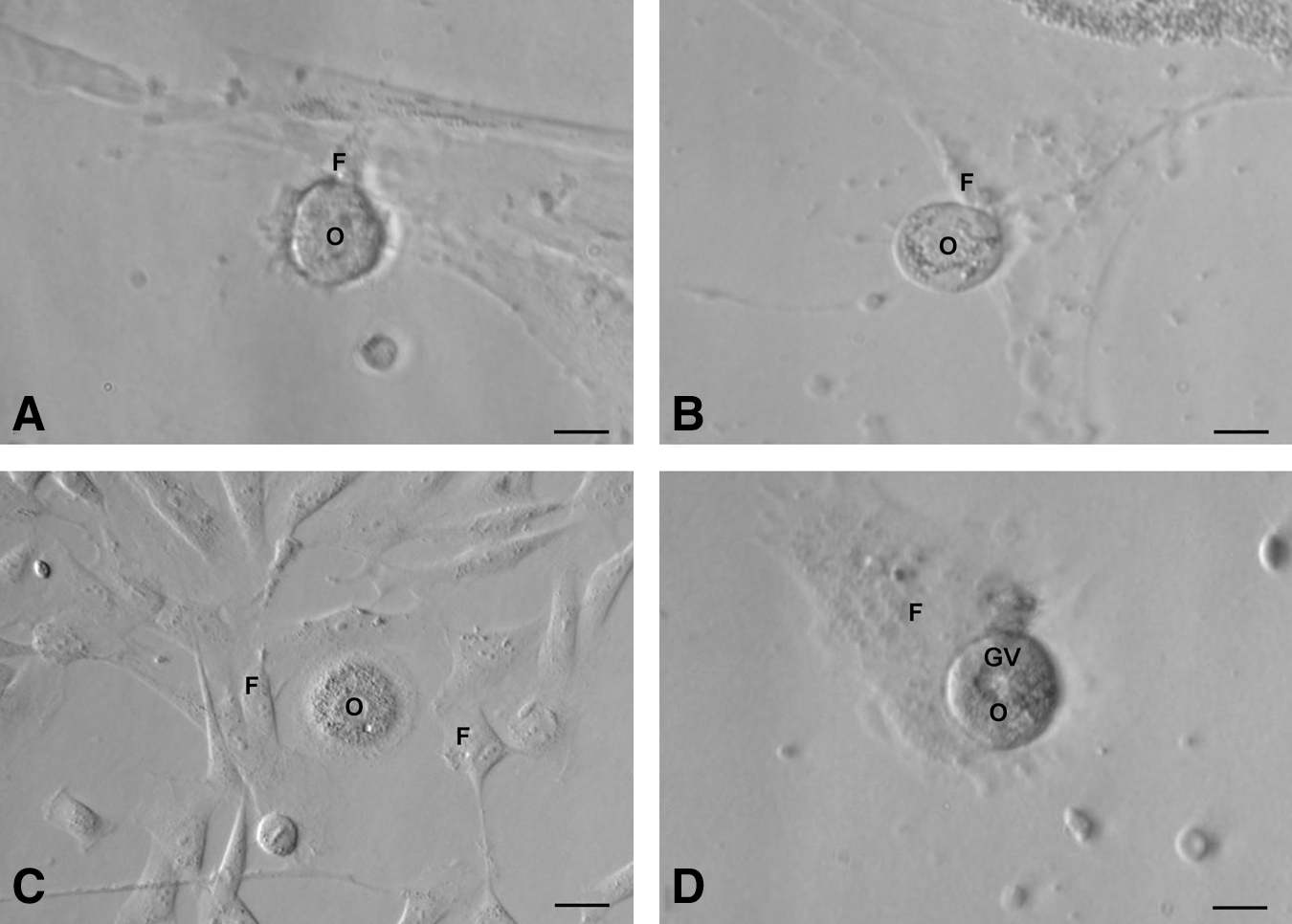
The stem cells appeared to increase in size and differentiate spontaneously into small oocyte-like structures (Fig. 3) by 3 weeks. Prominent polar body-like protrusions from the oocyte-like structures were occasionally evident in all species studied (Fig. 4A–D) and few of these oocyte-like structures appeared to be surrounded by distinct zona pellucida-like structure. Oocyte-like structures with an average diameter of 130 μm, comparable to naturally occurring oocytes, prominent nucleus and peri-nuclear accumulation of organelles were also observed (Fig. 4E–H).
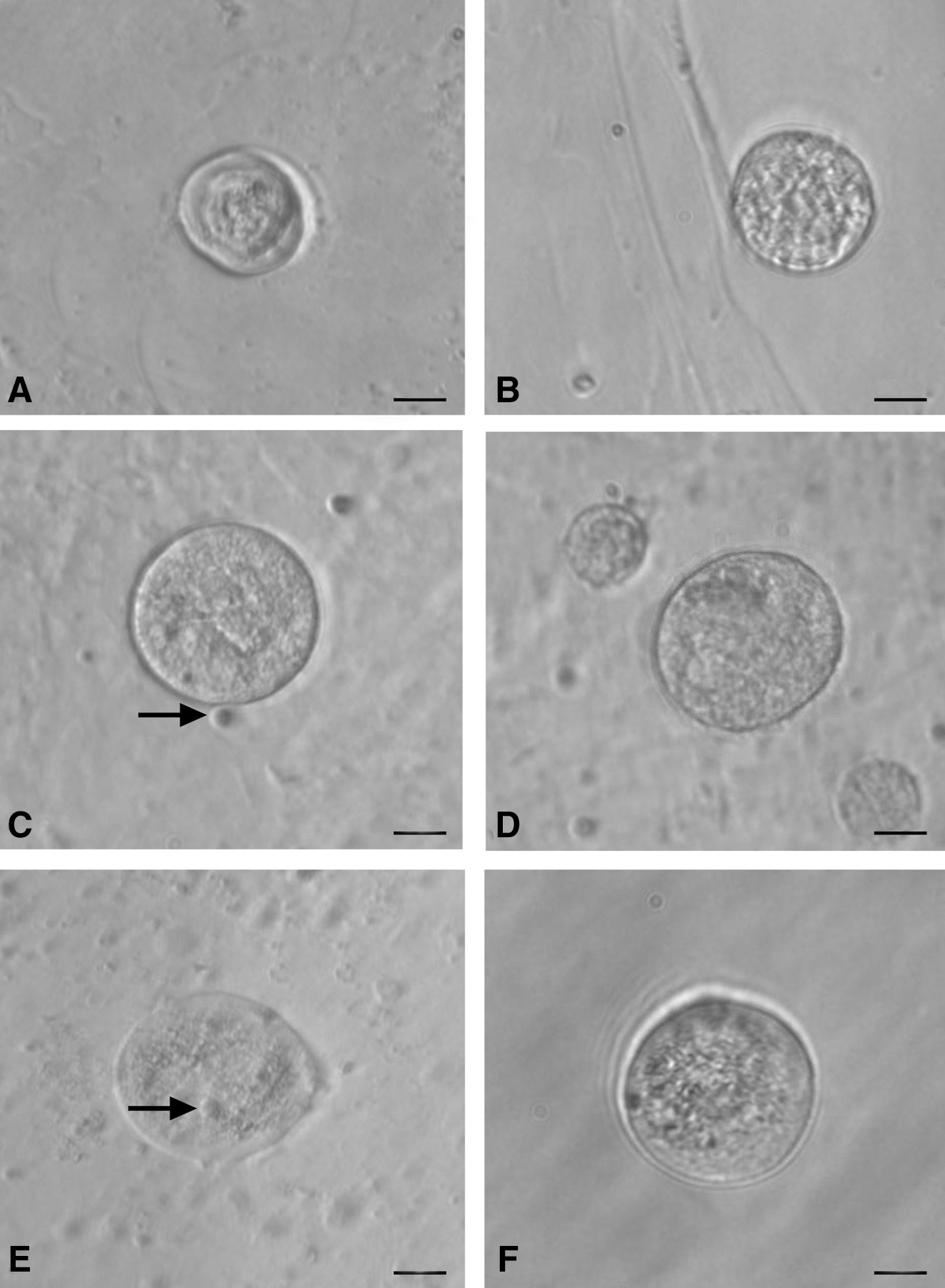
Smaller oocyte-like structures developed postculture from OSE isolated from
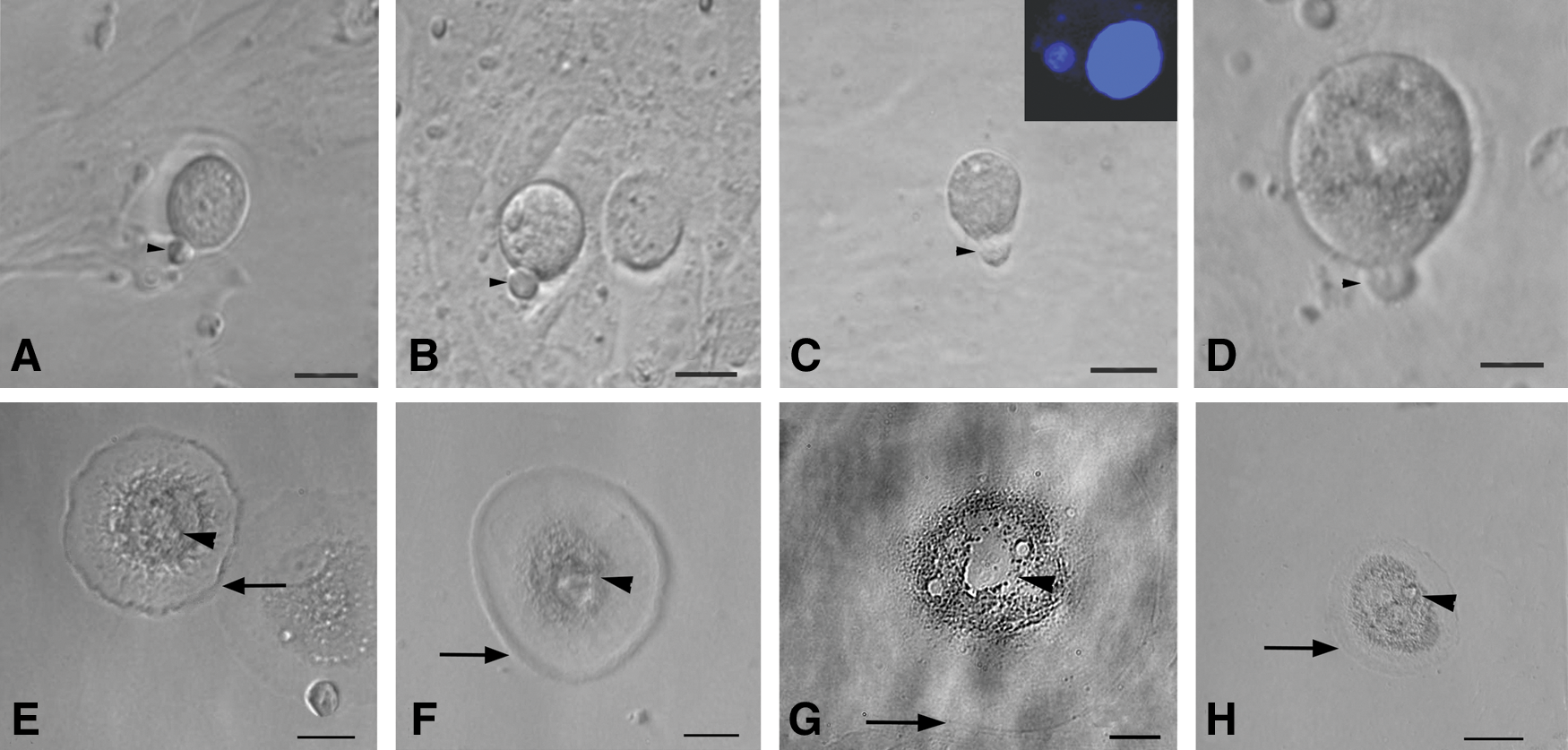
Polar body-like structures and larger oocyte-like structures in post-OSE cultures from all 4 mammalian species. Extruded polar body-like structures (arrow head) were observed in
Development of parthenote embryo-like structures
Structures resembling early embryos and blastocyst-like 3-dimensional structures with maximum diameter of 100–150 μm were prominently observed in sheep and human cultures (Fig. 5). Sheep embryo-like structure, stained with Hoechst 33342 to evaluate its viability as well as cell number, demonstrated that it was a 8–10 cells stage embryo-like structure with prominent nuclei (Fig. 5A, B). Well-defined trophoectoderm (TE) and fluid-filled blastocoel-like structures were also evident (Fig. 5C, F). Few of them revealed rolling movement (Supplementary Video 1).
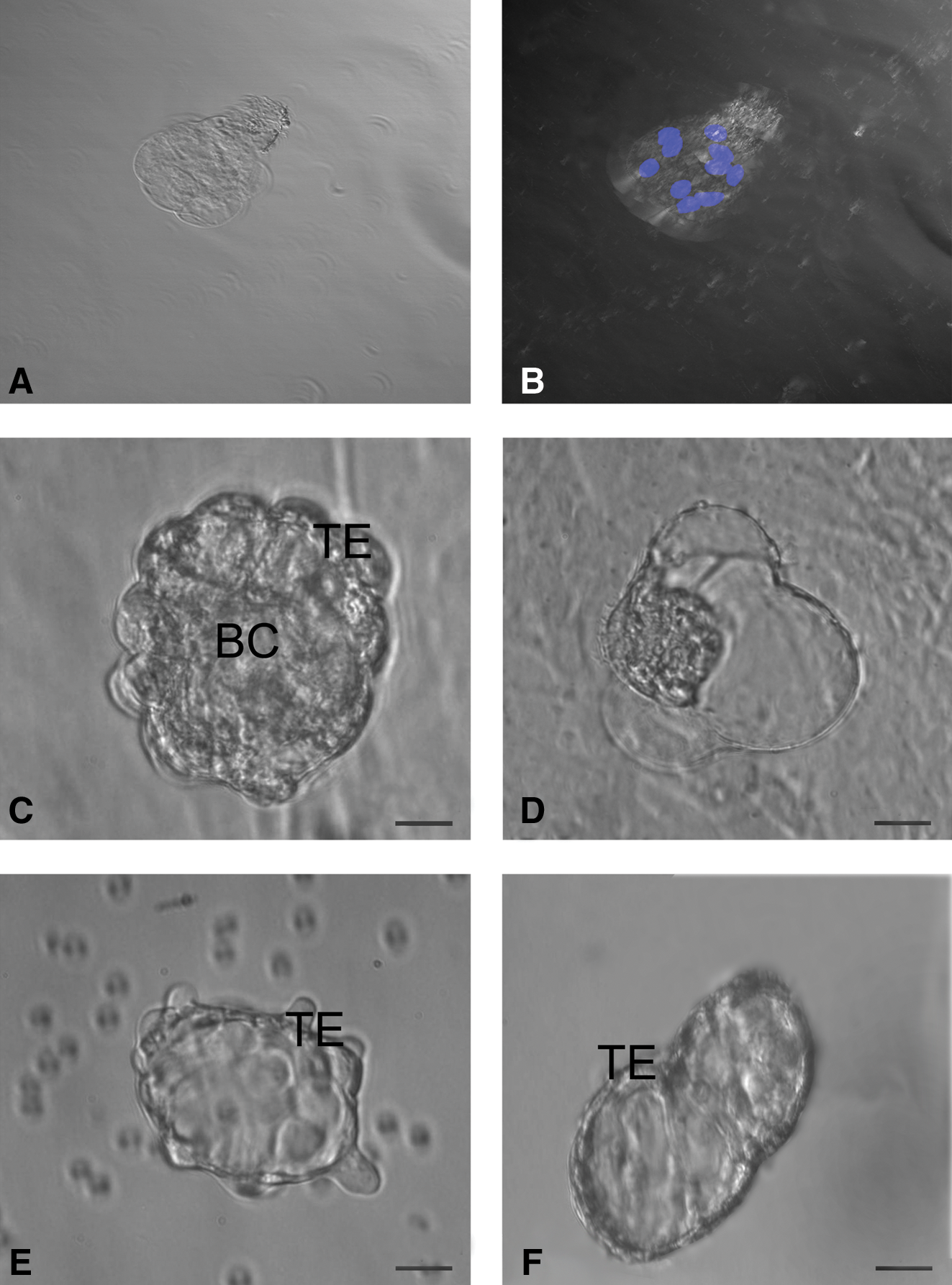
Parthenogenetic embryo-like structures observed post-OSE culture.
Appearance of ES cell-like colonies, embryoid body-like structures, and neuronal phenotype cells
Besides oocyte-like structures, a few ES cell-like colonies (Fig. 6A) and embryoid body-like structures (Fig. 6B) also appeared in human OSE cultures by day 10. The ES cell-like colonies were flat with a well-defined margin and were positive for alkaline phosphatase activity (Fig. 6C), whereas the embryoid bodies were 3-dimensional dense floating bodies (Fig. 6B). Besides cells with neuronal-like phenotype were also observed (Fig. 6D–F). Similar such structures were observed in all the other species studied.
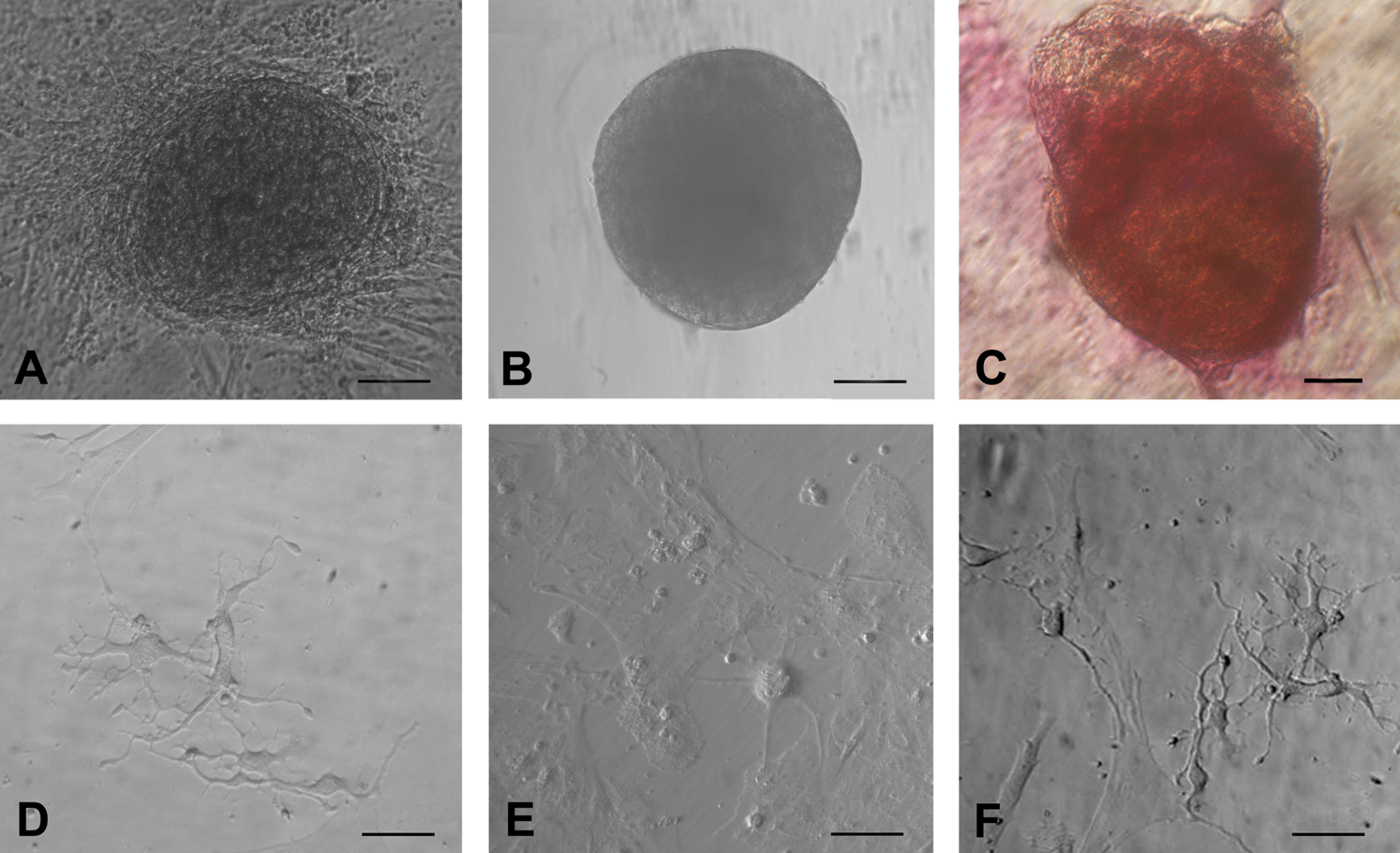
Other cell types observed post-OSE culture.
Epithelial–mesenchymal transition (EMT)
At the time of scraping the ovarian surface, round or cuboidal epithelial cells were found either dispersed singly or in small clusters. They attached to the surface of culture dish and assumed fibroblast-like appearance over time and thus exhibited a distinct phenomenon of EMT by 2 weeks of culture (Fig. 7A–D) in all the 4 species studied. In sheep cultures, a phenomenon of possible assembly of follicle-like structures was also observed (Fig. 7E, F). Initially, the spindle-shaped fibroblasts organized themselves (Fig. 7E) and assembled together to form a round structure (Fig. 7F), which grew in size and appeared like a follicle-like structure. Further characterization of these structures is required. Interestingly, a close proximity of developing oocyte-like structures with mesenchymal fibroblasts (Fig. 8) was also observed in all the species studied.
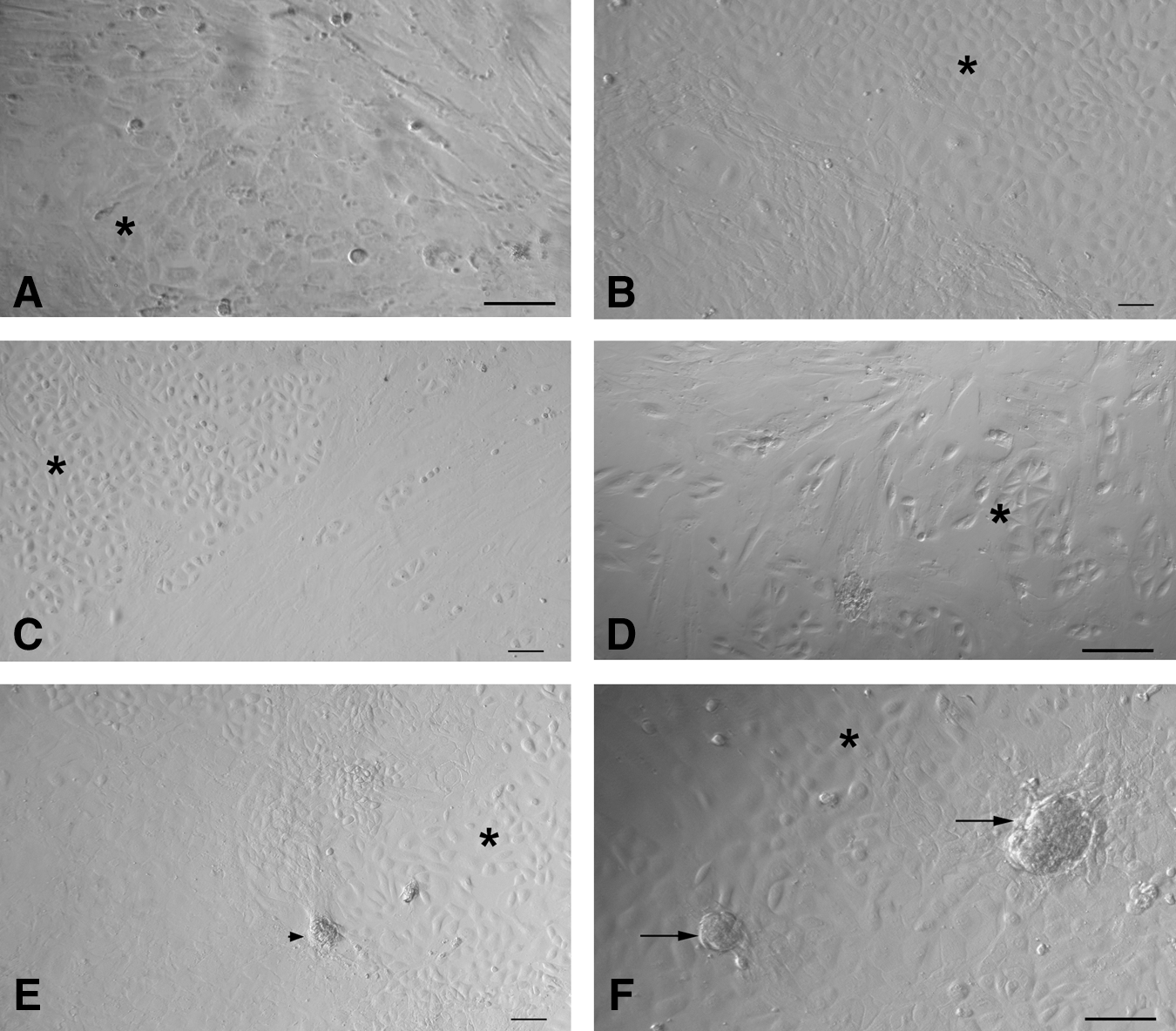
Epithelial to mesenchymal transition
Close association of oocyte-like structures with surrounding mesenchymal fibroblasts produced by epithelial–mesenchymal transition (refer to Fig. 7) postculture of
Characterization studies
Immunolocalization studies
Stem cell markers in human and sheep OSE: The use of polyclonal Oct-4 antibody revealed staining in the nuclei of small stem cells and also to the cytoplasm of relatively larger DAPI positive cells (Fig. 9). The DAPI-negative, small stem cells also stained positively for cell surface marker SSEA-4 (Fig. 10) in both sheep and human ovarian tissue.
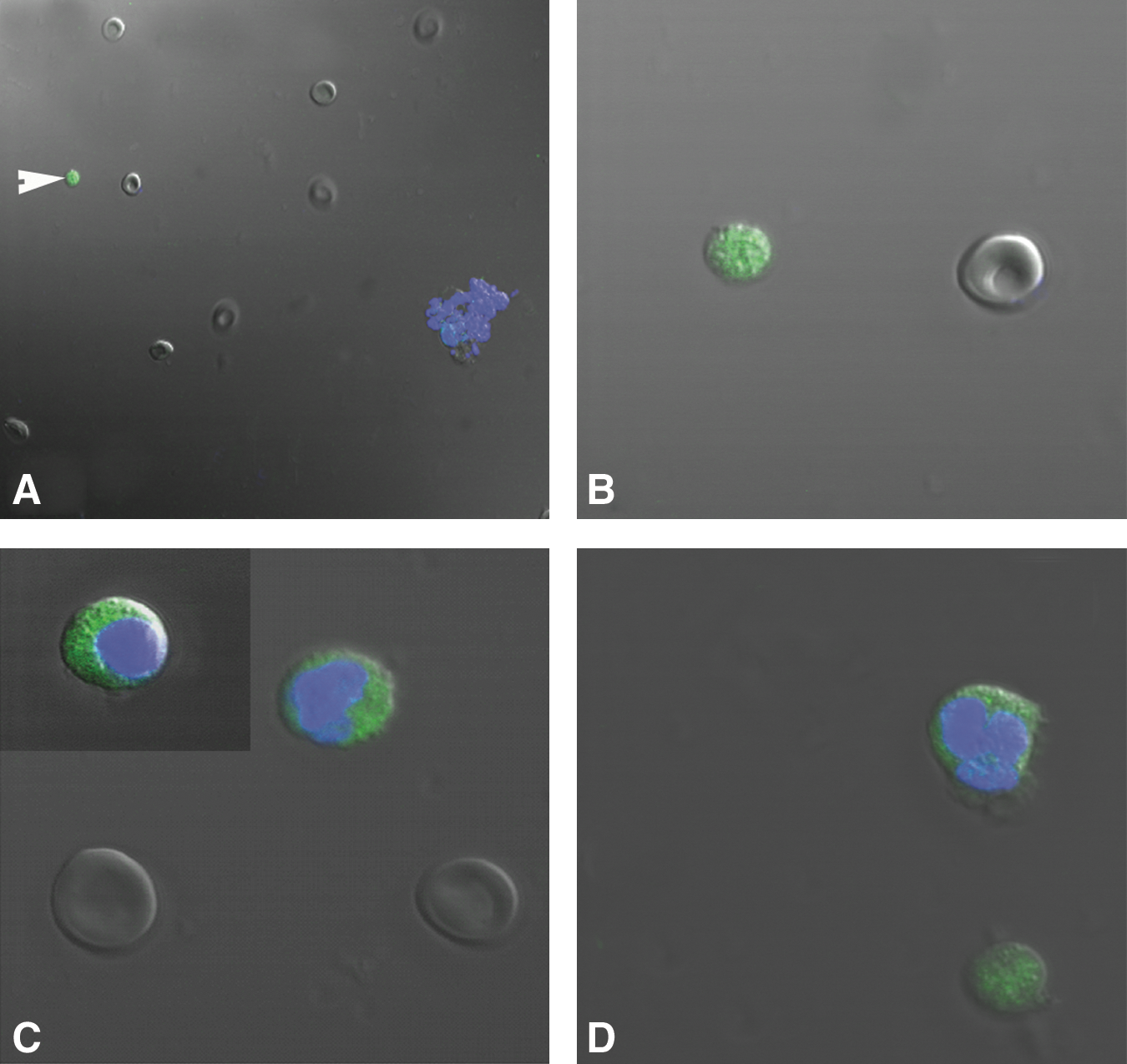
Differential immunofluorescent localization of pluripotent marker Oct-4 in stem cells isolated from human OSE, using polyclonal antibody by confocal microscopy. Note the presence of nuclear staining in very small stem cells (white arrowhead) stained negatively for DAPI
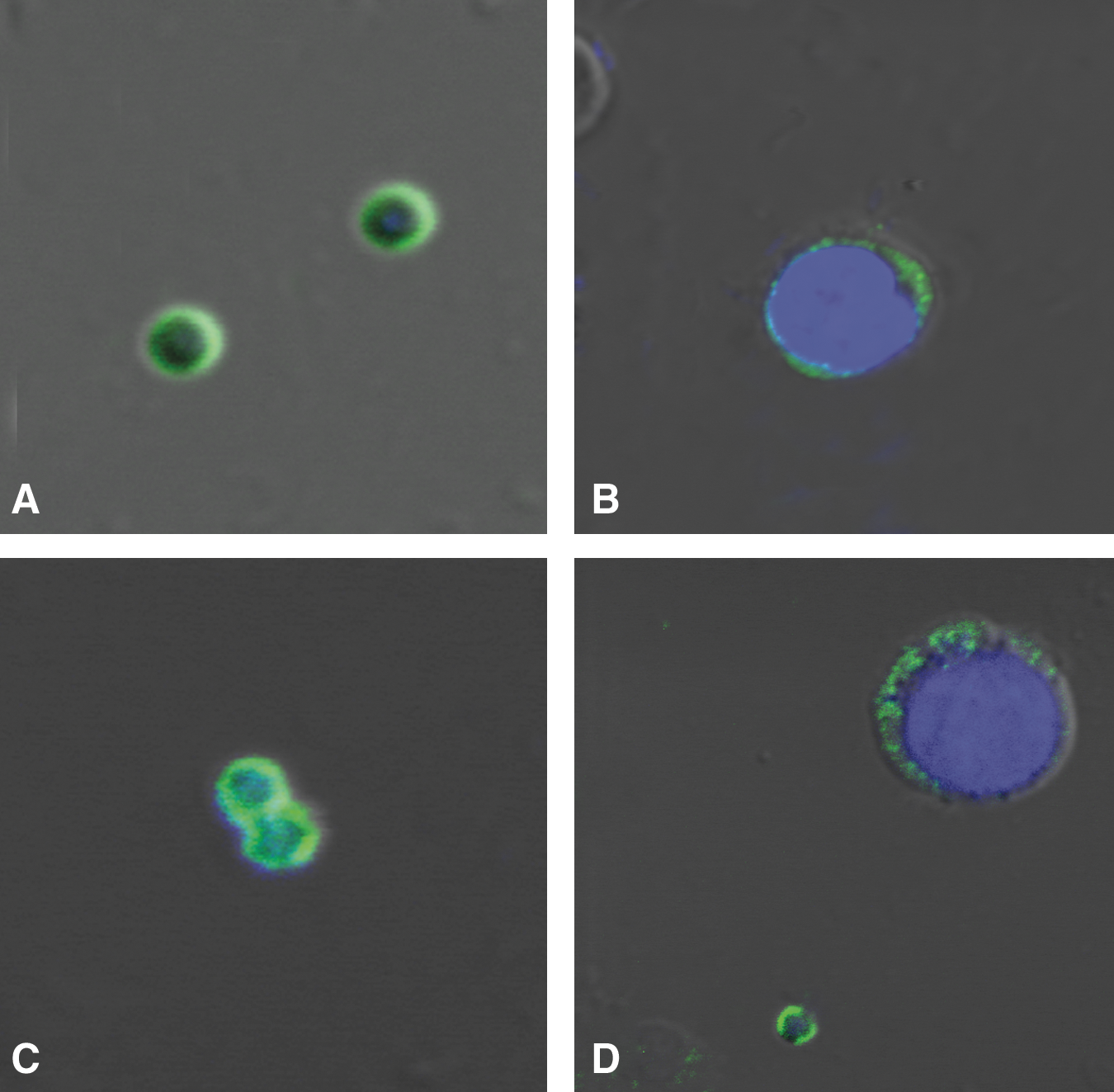
Differential immunofluorescent localization of a pluripotent stem cell surface marker SSEA-4 in stem cells isolated from human OSE by confocal microscopy. Note the presence of cell surface staining in very small stem cells in human
Germ cell-specific markers in human and sheep OSE: The oocyte-like structures obtained postculture exhibited positive cytoplasmic staining for c-Kit, DAZL, GDF-9, VASA, and ZP4 (Fig. 11). The adjacent somatic fibroblasts were negative, indicating specific localization in the germ cells. Respective negative controls with omission of primary antibody were also employed which showed absent staining (Fig. 11). All the staining procedures were repeated in triplicate.
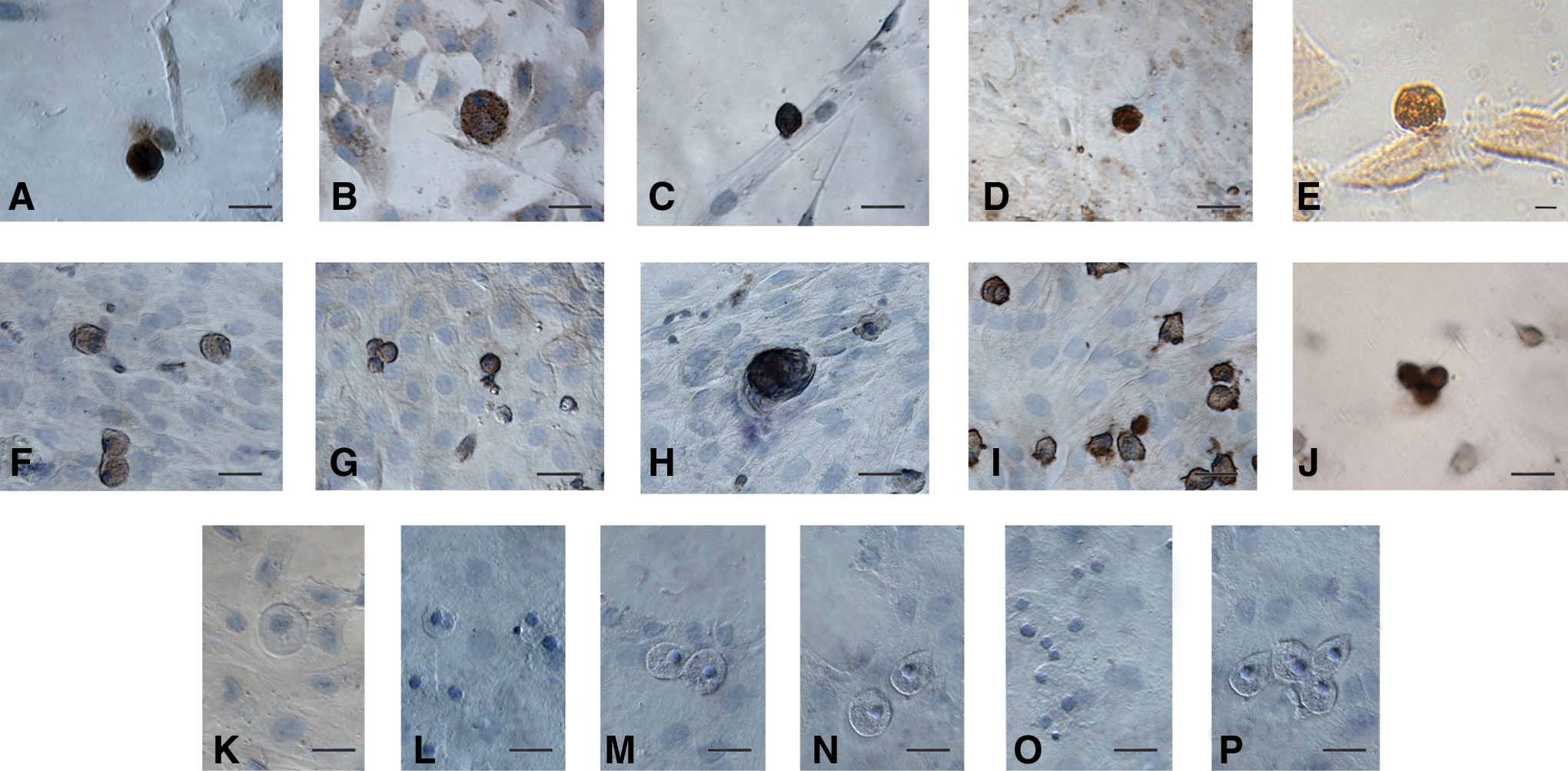
Characterization of oocyte-like structures observed postculture of human and sheep OSE by immuno-localization of germ cell markers. The oocyte-like structures stained positive in human and sheep cultures for
RT-PCR studies
Pluripotent markers: The gene expression profile of ovarian cortex with intact OSE and scraped OSE cells showed the presence of pluripotent transcripts viz. Oct-4, Oct-4 A, Nanog, Sox-2, and TERT in human and Oct-4, Nanog, Sox-2, and Stat-3 in sheep samples, respectively (Fig. 12).
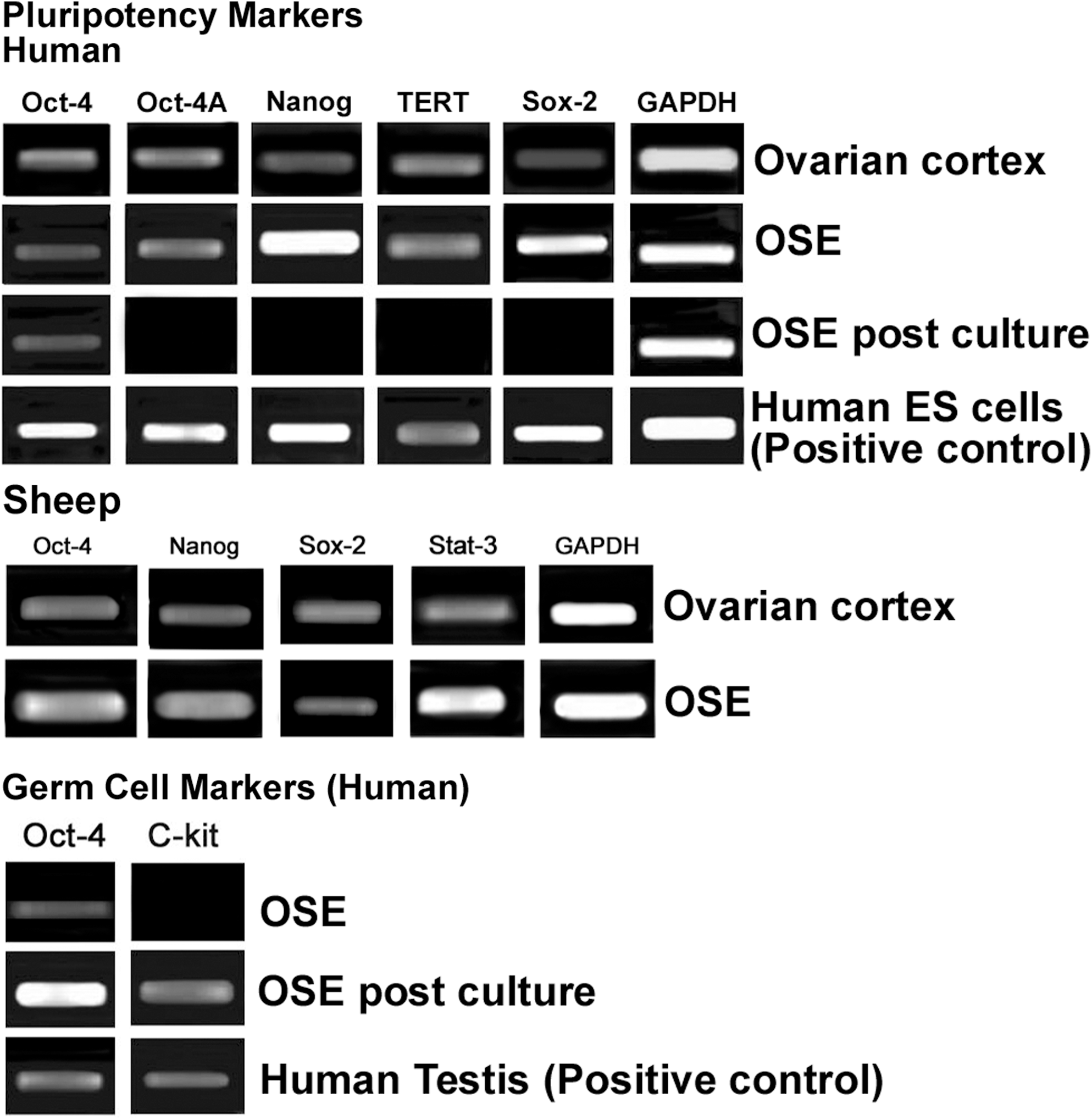
Reverse transcriptase–polymerase chain reaction analysis of human and sheep intact ovarian cortex, scraped OSE cells, and OSE cells postculture. Reverse transcriptase–polymerase chain reaction expression of transcripts for pluripotency markers showed positive bands of expected size for Oct-4, Oct-4A, Nanog, TERT, and Sox-2 in ovarian cortex and scraped OSE cells; in-house-derived human ES cells served as positive control. Sheep ovarian cortex and OSE also showed presence of transcripts for pluripotency markers viz. Oct-4, Nanog, Sox-2, and Stat-3. Transcripts for germ cell markers c-Kit and Oct-4 were observed postculture and human testicular tissue was used as positive control. Glyceraldehyde-3-phosphate dehydrogenase (GAPDH), a house-keeping control gene, was detected in all samples.
Germ cell markers: Pluripotent transcripts for Oct-4, Oct-4A, Nanog, Sox-2, and TERT, which were earlier detected in scraped OSE cells, were undetectable postculture except for Oct-4. Germ cell markers Oct-4 and c-Kit transcripts were observed in postcultured OSE cells of human, whereas scraped OSE was negative for c-Kit (Fig. 12).
Detection of alkaline phosphatase activity
ES cell-like colonies post-human OSE cultures showed positive alkaline phosphatase activity (Fig. 6).
Discussion
PSCs in OSE
Results of the present study provide evidence for the presence of PSCs with pluripotent characteristics in the OSE, of adult mammalian species like rabbit, monkey, sheep, and menopausal women. Besides epithelial cells with abundant cytoplasm and contaminating RBCs, we report for the first time, the presence of 2 distinct populations of spherical PSCs with high nucleo-cytoplasmic ratio in the scraped OSE. The unstained PSCs had a characteristic shiny appearance (Figs. 1C, D, and 2) and H&E staining revealed the presence of dark stained nuclei surrounded by a thin rim of cytoplasm and were easily distinguished from the surrounding epithelial cells with pale nuclear staining (Fig. 1E, F). The 2 populations of PSCs were distinguished based on their size (Fig. 1C, D), one population comprised of 1–3 μm cells (smaller than RBCs), whereas the second population was 4–7 μm in diameter (similar in size to the RBCs). The smaller PSCs stained negative with DAPI as compared to the bigger PSCs and epithelial cells (Supplementary Fig. S1) and this may possibly reflect presence of abundant euchromatin in them, since DAPI is understood to preferentially stain heterochromatin [27,28]. Similarly, the flow sorted very small embryonic-like stem cells (VSELs) isolated from various adult tissues, always stain much lighter with DAPI as compared to hematopoietic stem cells during the gating experiments reported by Ratajczak et al. [29 –31, personal communication with Dr Ratajczak's group]. Pluripotent markers also showed differential staining pattern in the 2 populations of PSCs. The smaller PSCs had nuclear Oct-4 and SSEA-4 localized on the cell surface, whereas the larger PSCs had cytoplasmic Oct-4 and minimal cytoplasmic SSEA-4 (Figs. 9 and 10). Additional pluripotent gene transcripts in both human (Oct-4, Oct-4A, Nanog, Sox-2, and TERT) and sheep (Oct-4, Nanog, Sox-2, and Stat-3) were detected by RT-PCR (Fig. 12).
Similar to earlier published data in adult human testis [32] it is hypothesized that in adult mammalian ovary, the small PSCs (abundant euchromatin, nuclear Oct-4, and cell surface SSEA-4; Figs. 9 and 10) are the pluripotent VSELs [30,31] and the larger cells (compacted heterochromatin, cytoplasmic Oct-4, and minimal SSEA-4; Figs. 9 and 10) are the immediate tissue committed progenitor stem cells derived from them. The VSELs possibly undergo asymmetric cell division to self-renew themselves and give rise to tissue committed progenitor stem cells in agreement with the recent concept published by Li and Clevers [33], indicating the co-existence of quiescent and active adult stem cells in several body tissues. Interestingly, the ovarian tissue committed progenitor stem cells had characteristic dark hematoxylin-stained nuclei (Fig. 1E, F) similar to the testicular Adark spermatogonial stem cells and may represent extensive nuclear reprogramming associated with dramatic changes in chromatin structure [34], when a pluripotent stem cell undergoes differentiation. Possibly the bigger PSCs (Figs. 1, 9, and 10) are a female counterpart to the Adark spermatogonial stem cells in the adult testis. It seems that intricate pluripotent stem cell biology is conserved in adult gonads—which need to be further explored.
The actual source of these PSCs in the ovary needs to be further investigated. Possible origin of these pluripotent stem cells from peripheral blood is speculated in both ovary [14] and testis [35]. However, in agreement with earlier reports [15,20,22], our results suggest that these cells may be resident cells present in the OSE. This is strengthened by the the H&E (Fig. 1E, F) and cytokeratin staining (Supplementary Fig. S2) of OSE, where these cells are observed entrapped in the epithelial cells, and also by a recent report [18] wherein no colony-forming cells were detected after 13 days of culture of whole blood-derived mononuclear cells from adult female mice by heart puncture. Original reports from Tilly's group, including Niikura et al. [13], have reported the presence of stem cells in adult mouse ovary but are noncommittal on their specific localization. Bukovsky and group [24] hold the view that bipotential progenitors in tunica albuginea give rise to germ cells. Gong et al. [18,19] have suggested ovarian stromal origin of the stem cells, but the conclusions are based on immunolocalization studies on ovarian cell smears (where the epithelial cells cannot be distinguished from the stromal cells) and their in situ hybridization results lack negative controls, thus making interpretation of results difficult.
Although the present study is focused on adult gonadal tissue, the presence of Oct-4 in adult somatic tissue has been subject of controversy and is considered dispensable for tissue homeostasis and regeneration. It has been suggested that Oct-4 pluripotency network may not exist in adult somatic tissue [36,37] and may be a technical artifact resulting from presence of pseudogenes, etc., but these concerns were recently clarified [38]. Shin et al. [38] have reported that the Oct-4 promoter in VSELs has an open chromatin structure that is actively transcribed and the cells lack uncontrolled proliferation and teratoma formation similar to ES cells in vitro because of unique methylation pattern. The cells identified in the present study as VSELs, besides nuclear Oct-4 also express SSEA-4 and other pluripotent gene transcripts. Oct-4 detected in these cells cannot be an artifact due to the pseudogenes since care was taken to use specific primers that do not amplify pseudogenes [39] and DNase I treatment of extracted RNA ensured no genomic DNA contamination, but what is the developmental origin of these cells? They may be the primordial germ cells that survive into adulthood [40,41] and this needs to be further investigated since the migrating primordial germ cells (PGCs) are much larger, 15–20 μm in contrast to the 1–3 μm size of VSELs. It has been recently suggested that perhaps a small number of PGCs or undifferentiated cells derived from them with stem cell characteristics could remain in the postnatal ovary and under certain conditions may resume mitosis, enter meiosis, and give rise to oocytes [42]. During any kind of tissue injury, both tissue-specific VSELs and also those in bone marrow/peripheral blood get mobilized in both mice [31,43,44] as well as in humans [45 –47] for regeneration to occur. If this is truly the case and VSELs exist in the adult ovary, the putative germ cells detected by Tilly's group [13] in the bone marrow/peripheral blood in mice with compromised ovarian function due to chemotherapy, are easily explained based on VSELs biology. Also, the regeneration observed by Lengner and his group [36,37] could have occurred because of these circulating VSELs.
Besides embryonic (ES) and induced pluripotent (iPS) stem cells, potential of VSELs for regenerative medicine needs to be explored further. The main advantage being their quiescent nature, which protects them from accumulation of chromosomal aberrations associated with the process of aging. Success has already been achieved to derive, propagate, and long term culture ES cells-like colonies without any genetic manipulation, from adult testicular tissue [48,49]. Transplantation of expanded bone marrow VSELs improved symptoms of myocardial infarct in mice [43]. Similar initial success of derivation of ES-like colonies was recently reported using postnatal ovaries. Gong et al. [18,19] have reported ES cell-like cultures from adult mice ovary, which formed embryoid bodies and also teratomas, whereas the present study demonstrates the development of embryoid bodies and ES cell-like colonies in all the mammalian species studied after 3 weeks in culture.
Spontaneous differentiation of PSCs in vitro
Present study results of in vitro culture of scraped OSE from adult ovarian tissue of rabbits, monkey, sheep, and menopausal women, which comprised of epithelial cells, PSCs, and contaminating RBCs, resulted in the development of oocyte-like structures, parthenote-like structures, cells with neuronal phenotype and alkaline phosphatase positive ES cell-like colonies, and embryoid bodies (normally not present in ovary, Figs. 3 –6) in agreement with earlier human studies [21,22]. The results provide additional evidence suggesting the pluripotent to totipotent nature of OSE stem cells. During culture, the somatic epithelial cells attached to the bottom of the culture dish, similar to when testicular cells are cultured [50] and formed a bed; RBCs were lost over time and thus evidently the PSCs underwent spontaneous differentiation into various structures. The differentiating human and sheep oocyte-like structures were characterized with germ cell-specific markers like c-Kit, DAZL, GDF-9, VASA, ZP-4, and Oct-4 (Figs 11 and 12). They also exhibited various characteristic features like germinal vesicle formation, extrusion of polar body, and formation of distinct zona pellucida (Fig. 4). The presence of PSCs in the OSE and their ability to differentiate spontaneously into oocyte-like structures in vitro provides indirect evidence to support postnatal oogenesis in females. Further, as PSCs isolated from various species in the present study exhibited similar potential in vitro, possibly postnatal oogenesis may be an evolutionarily conserved phenomenon in higher mammalian species.
The phenomenon of epithelial–mesenchymal transition (Fig. 7) was evident in cultures of all the species studied at the cellular level in the present study and has been reported earlier to be a part of normal OSE physiology [2,24] and also during in vitro culture of OSE in collagen gels [51] and 3D matrices [52]. The epithelial cells that are initially cuboidal and tightly attached to each other lose their polarity, cell cytoskeleton gets remodeled, and OSE gets transformed into mesenchymal cells. The oocyte-like structures appeared to grow in close vicinity of mesenchymal fibroblasts (Fig. 8) and this close association has been earlier implied to demonstrate the nutritional support, aromatase expression which catalyzes the turnover of C [19] steroids into estrogens, and also as a source of energy by mitochondrial trafficking provided to the growing germ cells by the surrounding somatic cells [21,25,53 –56]. It is tempting to speculate that these fibroblast cells derived by EMT may act as the supporting somatic granulosa cells for the maturing oocyte-like structures derived from putative OSE stem cells. Auersperg and group [2] have also earlier suggested that granulosa cells may be embryologically derived from OSE, intra-ovarian rete, or both. Our results are in contradiction to the existing bipotent stem cell theory [5,25] and explain earlier reports of the formation of chimeric follicles with only GFP-positive oocyte [12,15] when GFP-tagged female germ stem cells are transplanted in busulphan-treated mice ovary.
The formation of sheep and human aggregates—“parthenote-like structures” in the present study (Fig. 5)—provides indirect evidence regarding the potential of the oocyte-like structures. The rolling over movement observed in the sheep parthenote-like structure was remarkable (Supplementary Video 1). It appeared to roll as it is conceptualized while passing through the oviduct and in the uterine lumen before implantation, unlike the nonmotile embryos created in vitro in IVF clinics.
An intriguing outcome of the present study in agreement with the earlier reports [21,22] is that the postmenopausal human ovary, devoid of any follicles in the ovarian cortex, is a rich source of stem cells which develop in vitro into oocyte-like structures. Then, why do women experience menopause? What suppresses follicular development in situ with advancing age? Menopause is best understood as sudden loss of follicles in the aged ovary. However, it is now becoming apparent that the surrounding somatic cellular niche that supports the stem cells in OSE has an important role in the onset of menopause, which possibly gets compromised with age and thus ability of stem cells to develop into germ cells and follicular assembly is reduced during menopause [3,4,57]. Besides, immune system has also been implicated to determine the ability of ovarian stem cells to differentiate into germ cells and its impairment may result in menopause [5]. Can we coax the PSCs to undergo folliculogenesis in situ by improving the somatic niche? This may be a novel approach to treat premature ovarian failure due to aggressive oncotherapy in future. The OSE cultures could also become a source of oocytes to reprogram somatic cells during SCNT—a field severely restricted at present, due to the nonavailability of oocytes. It is only a matter of time when one could use the parthenotes or embryos derived using the oocytes generated in vitro or expand the OSE pluripotent stem cells per se to derive pluripotent cell lines for regenerative medicine.
To conclude, present study reports the presence of pluripotent very small embryonic-like stem cells in adult OSE, which may undergo asymmetric cell division to maintain homeostasis and have the ability to differentiate into oocyte-like structures in vitro. Further studies are warranted at single-cell level, including complete gene expression profiling to further confirm whether postnatal oogenesis is a conserved phenomenon in adult mammals and to evaluate the functional potential of in vitro derived oocyte-like structures.
Footnotes
Acknowledgments
The authors would like to thank Dr. A. Bukovsky (Department of Obstetrics and Gynecology, The University of Tennessee Graduate School of Medicine, TN) for initial guidance with culture studies; Dr. S.K. Gupta (National Institute of Immunology, New Delhi, India) for ZP-4 antibody and Dr. S. Bapat (National Centre for Cell Sciences, Pune, India) for useful discussions related to EMT studies. The authors also thank Confocal Microscope Central Facility at NIRRH for help. Thanks to Ms. Harshada Modak and Mr. H. Karekar for typing assistance and the art work, respectively. This study was financially supported by Department of Science and Technology, New Delhi, Government of India (D.O.SR/SO/HS-41/2003).
Author Disclosure Statement
The author(s) declare that they have no competing financial interests.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
