Abstract
The combination of allogeneic bone marrow transplantation (allo-BMT) and donor lymphocyte infusion (DLI) is a useful method for establishing donor chimerism and preventing a relapse of leukemia/lymphoma. However, there is a risk of inducing uncontrollable fatal graft-versus-host disease (GVHD). In fact, allo-BMT plus intravenous (IV)-DLI using donor splenocytes induces fatal GVHD in recipient mice. In this study, we examined the effects of the combination of intra-bone marrow (IBM)-BMT and the subcutaneous injection of donor splenocytes (SC-DLI) on the allo-BMT system. Recipient BALB/c mice were conditioned by sublethal irradiation (5 Gy), followed by IBM-BMT plus IV-DLI or SC-DLI in C57BL/6 mice. The IV-DLI group showed better engraftment of donor hemopoietic cells than the control group (without DLI) but showed fatal GVHD. The SC-DLI group, however, showed good reconstitution and mild GVHD. These results suggest that the combination of SC-DLI and IBM-BMT promotes the reconstitution of hemopoiesis and helps reduce the risk of GVHD.
Introduction
B
Donor lymphocyte infusion (DLI) is often used after allo-BMT to prevent disease relapse in the setting of T-cell–depleted BMT or nonmyeloablative conditioning regimens. It is also a combined method to convert mixed chimerism to full donor chimerism [17,18]. Donor T cells injected intravenously during DLI are activated in the host's lymphoid tissues, which then migrate to the target tissues of graft-versus-host disease (GVHD) and then mediate the GVHD. DLI, which is used as the combined conditioning therapy for BMT, helps to reduce relapse rates. However, DLI-induced GVHD is always associated with an increase in therapy-related morbidity because of its uncontrollable and fatal characteristics [19 –26]. A key challenge for DLI is to balance the positive and negative effects of donor T cells in order to optimize the outcome.
In mice, allo-BMT plus IV-DLI using donor splenocytes can induce fatal GVHD due to the donor T-cell infiltration and proliferation in the GVHD target tissues such as the liver, spleen, intestine, and skin [27]. In this study, we examined the localizable effects of donor T cells (splenocytes) by subcutaneous injection (SC) of donor splenocytes in the allo-BMT system using a preconditioning regimen (sublethal irradiation). Compared with IV-DLI plus IBM-BMT, the SC-DLI plus IBM-BMT group showed good reconstitution and only mild GVHD. The survival rate in the SC-DLI group was much higher than in the IV-DLI group. These results suggest that the combination of SC-DLI and IBM-BMT promotes reconstitution of hemopoiesis and helps to reduce the risk of fatal GVHD.
Materials and Methods
Mice
C57BL/6 mice (B6) and BALB/c were purchased from Shimizu Laboratory Supplies (Shizuoka, Japan). All the mice were maintained in a pathogen-free room, and 8–10-week-old male mice were used in the present studies. The university's committee for animal research approved all experiments.
Reagents
The antibodies used in this study were as follows: fluorescein isothiocyanate (FITC)-labeled anti-mouse H-2b Ab, phycoerythrin (PE)-labeled anti-mouse H-2d Ab, peridinin chlorophyll protein (PerCP)-Cy5.5-labeled anti-mouse CD45 Ab, and anti-mouse CD3 Ab (BD Pharmingen, San Diego, CA). Lysing buffer (BD Pharmingen) was used for the lysis of erythrocytes. Collagenase type IV, used for hepatocyte isolation, was purchased from Sigma (Sigma-Aldrich, St. Louis, MO).
Whole-body irradiation of recipient mice
Gamma-irradiation was delivered by a Gammacell 40 Exactor (MDS Nordion, Kanata, ON, Canada) with two 137Cs sources. Recipient mice were irradiated with 6, 5, or 4 Gy, the day before BMT.
IBM-BMT
BMCs were flushed from the medullary cavities of the femurs and tibias of donor mice with phosphate-buffered saline (PBS). After gentle dissociation, the BMC suspension was filtered through a 70-μm nylon mesh (Becton Dickinson Labware, Franklin Lakes, NJ). The BMC suspension was then centrifuged and the supernatant was aspirated. The BMCs were adjusted to 3 × 109 per mL. The thus-prepared BMCs (3 × 107) were injected directly into the tibial cavity of the recipient mice via the intra-bone marrow route (IBM-BMT) the day after irradiation, as previously described [13]. Briefly, the mice were anesthetized and the area from the inguinal region to the knee joint was shaved. The tibia was gently drilled with a 26-G needle through the patellar tendon into the BM cavity. The BMCs (3 × 107/10 μL) were then injected into the BM cavity using a microsyringe (50 μL; Ito, Fuji, Shizuoka, Japan).
Donor lymphocyte infusion
Spleens were removed from donor B6 mice and then minced with scissors. Single cells were prepared by milling in steel mesh, followed by filtering through a 70-μm nylon mesh in PBS containing 2% fetal calf serum. After centrifugation, the pellets were suspended in 1 × lysis buffer and kept for 15 min at room temperature for the lysis of erythrocytes. The erythrocyte-depleted splenocytes were adjusted to 5 × 107/0.2 mL (2.5 × 108 per mL) in PBS and were then injected intravenously into the tail vein in the IV-DLI group or subcutaneously in the back in the SC-DLI group. Figure 1 shows the experiment protocol, including the days for treatment.
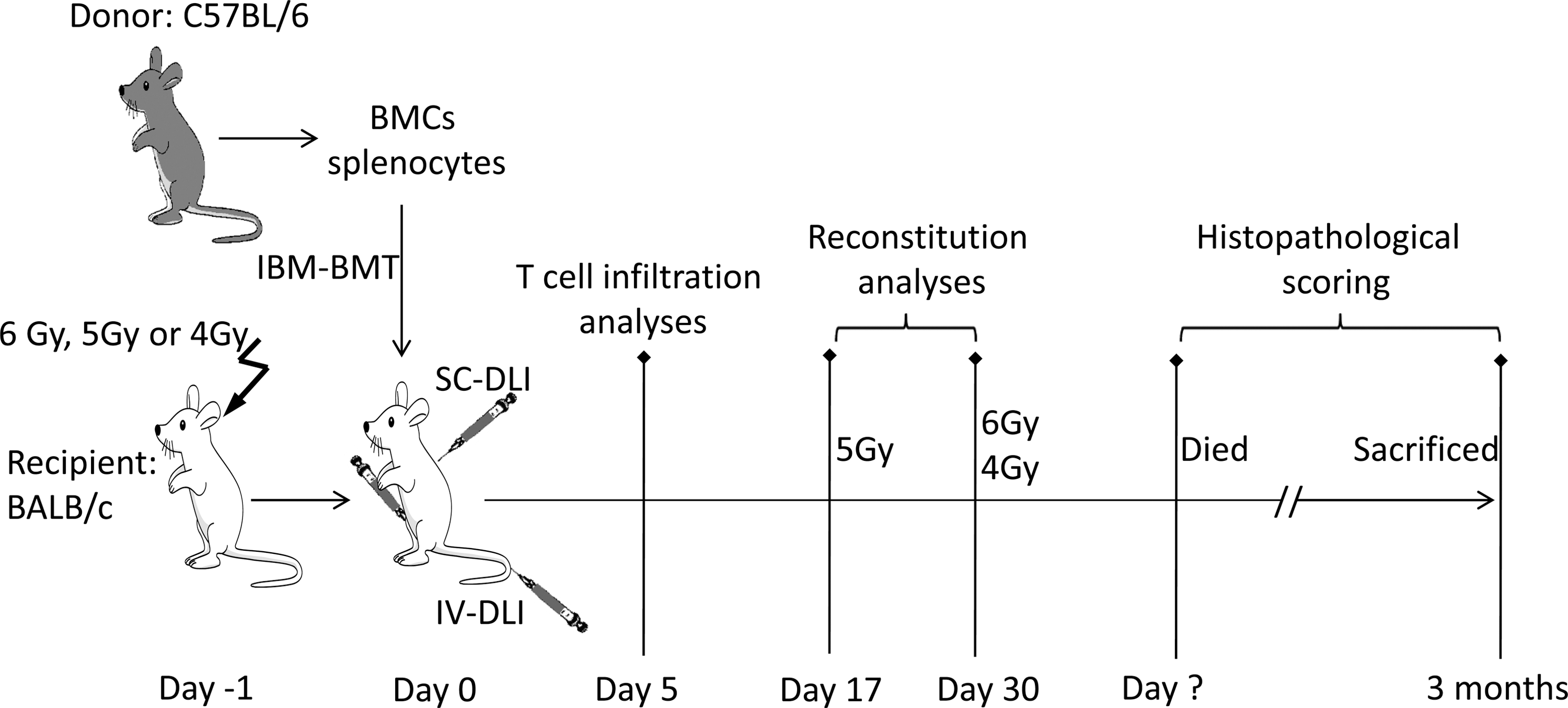
Experiment protocol. The days of irradiation, BMT, and DLI are shown. The observed days of T-cell infiltration, reconstitution, and the analyses of histopathological scoring are also shown. BMT, bone marrow transplantation; DLI, donor lymphocyte infusion.
GVHD analysis and scoring
The recipients were monitored daily for survival, and every 5 days for body weight changes and clinical signs of GVHD after BMT. The clinical scoring was based on 6 parameters: weight loss, posture, activity, fur texture, skin integrity, and diarrhea. A severity scale of 0 to 2 was used for each parameter, with a maximum score of 12 (Table 1). Clinical signs early after transplantation due to radiation toxicity were not considered as the appearance of GVHD [28,29].
The clinical signs of GVHD (based on 6 parameters: weight loss, posture, activity, fur texture, skin integrity, and diarrhea) were scored every 5 days after bone marrow transplantation. A severity scale of 0 to 2 was used for each parameter, with a maximum score of 12.
The carcasses of the recipients that had died or had been sacrificed at 3 months after BMT were kept in 10% formalin. Tissues from GVHD target organs (liver, intestine, and skin) were embedded in paraffin, sectioned, and stained with hematoxylin and eosin. Slides were observed using an Olympus BX41 light microscope (Olympus, Tokyo, Japan) with a UPlanFL N 20 × /0.50 objective. An Olympus DP-25 color camera using DP2-BSW software was used to acquire the images.
Histopathological analysis was performed “single blind” by scoring the changes in the skin (dermal/epidermal lymphocyte infiltration, dyskeratotic epidermal keratinocytes, and epidermal thickening), the intestine (crypt regeneration, apoptosis in crypt epithelial cells, crypt loss, surface colonocyte attenuation, inflammatory cell infiltration in lamina propria, mucosal ulceration, and thickening of mucosa), and the liver (bile duct injury manifested by nuclear hyperchromasia, nuclear crowding, infiltrating lymphocytes, and cytoplasmic vacuolation and liver inflammation due to the infiltration of lymphocytes, neutrophils, and eosinophils). A severity scale from 0 to 4 was used, with a maximum score of 12 (Table 2) [29,30].
Histopathological GVHD scoring was performed based on the changes in 3 GVHD target organs: the skin, intestine, and liver. A severity scale of 0 to 4 was used for each organ, with a maximum score of 12.
Analyses of donor cells in recipient peripheral blood, spleen, or liver by FACS
To detect the reconstitution of the recipients, the peripheral blood (PB) of the recipient mice was collected 17 days or 1 month after BMT. The PB was stained with FITC-labeled anti-H-2b Ab, PE-labeled anti-mouse H-2d Ab, and PerCP-Cy5.5-labeled anti-CD45 Ab. The erythrocytes were then lysed using lysing buffer. The stained cells were analyzed using FACScan (BD Biosciences, San Jose, CA). Leukocytes were first gated by CD45+ cells, which were estimated as nuclear cells. The percentage of donor leukocytes was estimated as H-2b+/CD45+ cells.
To detect donor-derived T cells infiltrating the GVHD target tissues at 5 days after BMT, mononuclear cells (MNCs) from the recipient's spleen and liver were collected as follows: 0.5 mg/mL collagenase type IV solution was prepared by PBS dilution, and then after euthanasia, 2 mL collagenase solution was injected intraportally in the recipient mice. The spleen and liver were surgically excised and a single-cell suspension was prepared. The spleen and liver MNCs were then isolated by Lymphoprep (AXIS-SHIELD PoC AS, Oslo, Norway). The MNCs were stained with FITC-labeled anti-H-2b Ab, PE-labeled anti-mouse H-2d Ab, and PerCP-Cy5.5-labeled anti-CD3 Ab. The percentage of donor T cells was analyzed by FACScan estimated as H-2b+/CD3+ cells.
Statistical analysis
Survival data were analyzed using the Kaplan–Meier method in the Stat Mate software. Other results are represented as means ± standard deviation (SD). The Student's t-test was used to determine any statistical significance. A P value of <0.05 was considered to be a significant difference.
Results
No GVHD occurs in the BMT-only group
Previous studies have shown that, in contrast to humans and other primates, no or only mild GVHD is observed in the case of allo-BMT without DLI in the murine setting [31 –33]. To confirm this, BMCs from B6 mice were transplanted into sublethally irradiated (6, 5, or 4 Gy) BALB/c recipients by IBM-BMT (control group). All the control groups (6, 5, or 4 Gy) survived until 90 days after BMT (Fig. 2). No severe complications, such as fatal GVHD, occurred in the BMT-only control group according to clinical observations (Fig. 4) and histopathological evaluation (Fig. 5).
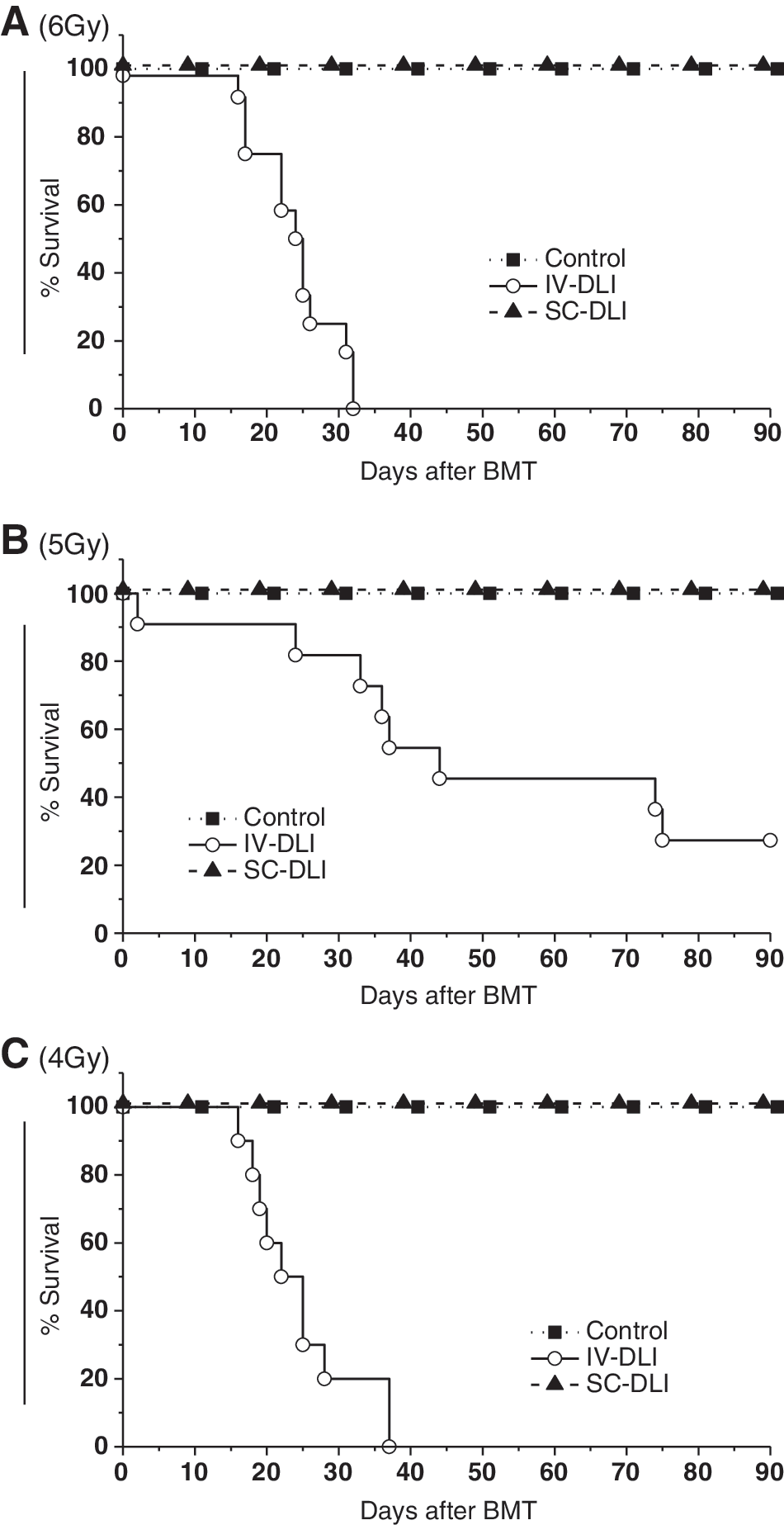
IV-DLI shortens the survival but SC-DLI does not. Recipient (BALB/c) mice were irradiated at 6 Gy
The SC-DLI group shows lower mortality than the IV-DLI group
The PB and spleen are commonly used as the source of lymphocytes for DLI. In the present study, we carried out DLI using the erythrocyte-depleted splenocytes (5 × 107 per mouse). After BMT, the recipients were monitored daily for survival. For the 6 Gy irradiated mice, all the mice in the IV-DLI group died within 32 days after BMT (Fig. 2), whereas all the mice in the SC-DLI group survived until 90 days after BMT. Similarly, high mortality rates were also observed in the IV-DLI group for 5 or 4 Gy irradiated recipients. To our surprise, the 5 Gy IV-DLI group showed significantly improved survival compared with the 4 Gy IV-DLI group. In contrast to the high mortality caused by IV-DLI, the SC-DLI groups (6, 5, or 4 Gy) showed a 100% survival rate, as seen in the control groups (Fig. 2).
Better reconstitution of donor hemopoietic cells is observed in both the IV-DLI and SC-DLI groups than the control group (without DLI)
Next, we examined the reconstitution degree of the recipients with donor-type cells in the PB of the recipient mice after BMT. In the experiment with preconditioning with 6 Gy irradiation, the PB was collected from the surviving recipient mice to analyze the chimerism at 1 month after BMT. All the recipients of IBM-BMT, either combined with DLI or not, showed nearly complete donor-type (H-2d) hematopoietic cells (Fig. 3A). Moreover, there was no significant difference between the control group and the SC-DLI group: the mean and SDs of the percentages of donor hematopoietic cells were 92.4% ± 3.7% and 95.3% ± 3.1%, respectively (Fig. 3D). We therefore reduced the radiation dose to 5 Gy. With 5 Gy, reconstitution was examined earlier (on day 17). Donor-type hematopoietic cells were detected in 4 of 10 control group recipients, 10 of 10 IV-DLI group recipients, and 9 of 11 SC-DLI group recipients (Fig. 3B). Statistically significantly higher percentages of reconstitution with donor cells were observed in the SC-DLI group than in the control group, and there was no significant difference between the IV-DLI and SC-DLI groups (Fig. 3E). Moreover, better reconstitution was confirmed by long-term (not transient) chimerism (data not shown). When the radiation dose was reduced to 4 Gy, neither the control group nor the SC-DLI group could reconstitute the recipients with donor BMCs. The two survivors in the IV-DLI group showed donor-type hematopoietic cells but these mice died soon (Fig. 3C, F).
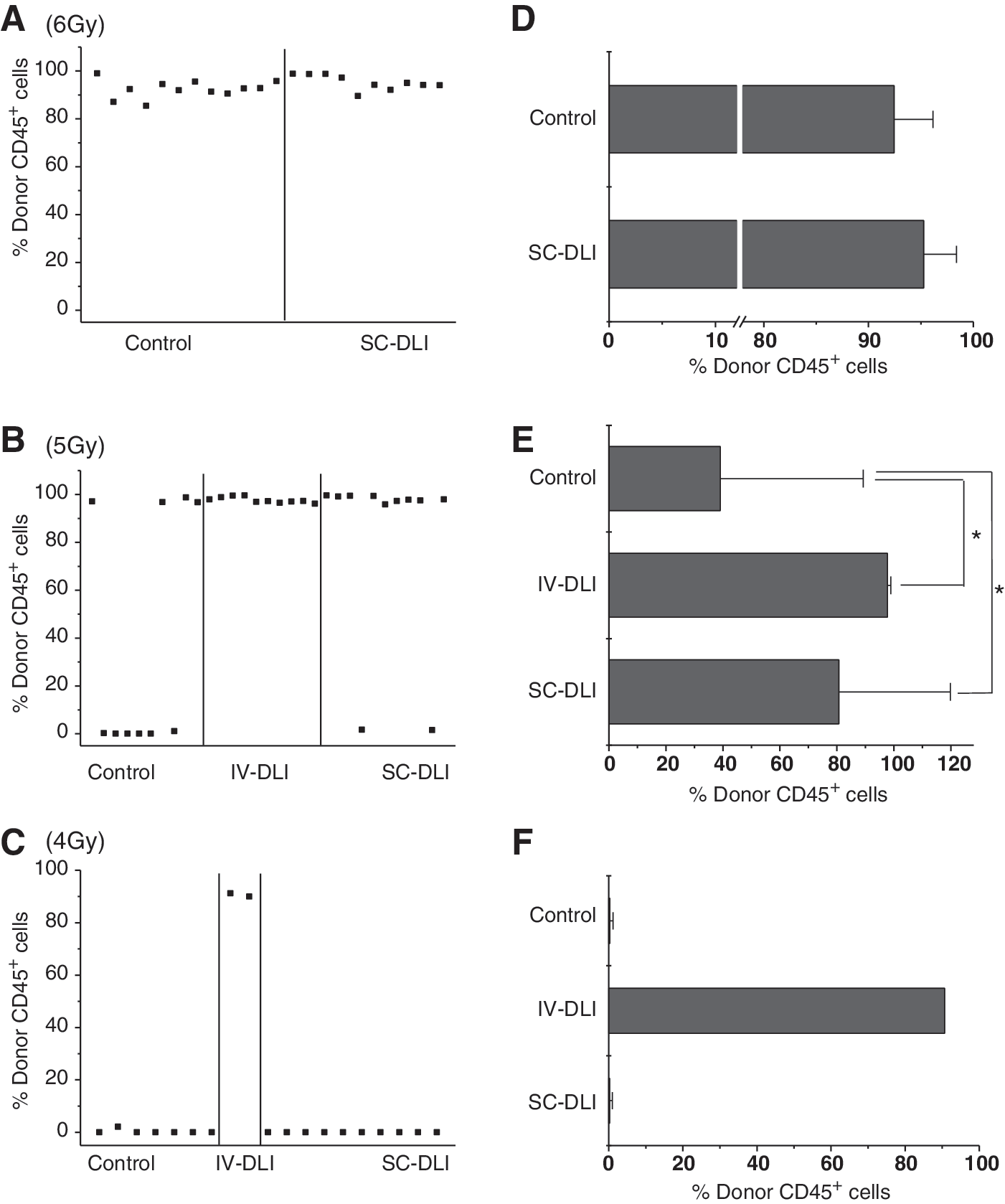
Reconstitution in different irradiation doses. Recipient mice were irradiated with 4, 5, or 6 Gy on day −1. BMT and DLI were carried out on day 0. Peripheral blood of 6 Gy
Serious GVHD is observed in the IV-DLI group
After BMT, clinical signs of GVHD in the recipients (including weight loss, posture, activity, fur texture, skin integrity, and diarrhea) were assessed every 5 days and the score was calculated. In the 6 Gy IV-DLI group, all the recipient mice showed hunchback; several showed loss of weight, inaction, ruffled fur texture, and slight diarrhea on day 15. The signs of GVHD became progressively more serious: angular, severe hunching, stationary unless stimulated, severe ruffling, obvious areas of denuded skin, and severe diarrhea. The clinical score reached a peak on day 30: 7.33 ± 0.58. No obvious signs of GVHD appeared in the other two groups: 0.25 ± 0.87 (control group) and 0.50 ± 0.71 (SC-DLI group) (Fig. 4A). In the 5 Gy experiment, the clinical scores in the control group, IV-DLI group, and SC-DLI group were 0.10 ± 0.31, 6.44 ± 2.60, and 0.81 ± 0.30, respectively, according to observations on day 30. The mice in the IV-DLI group retained the tendency to develop serious GVHD during the period from day 30 to day 70. The other two groups displayed mild GVHD (Fig. 4B).
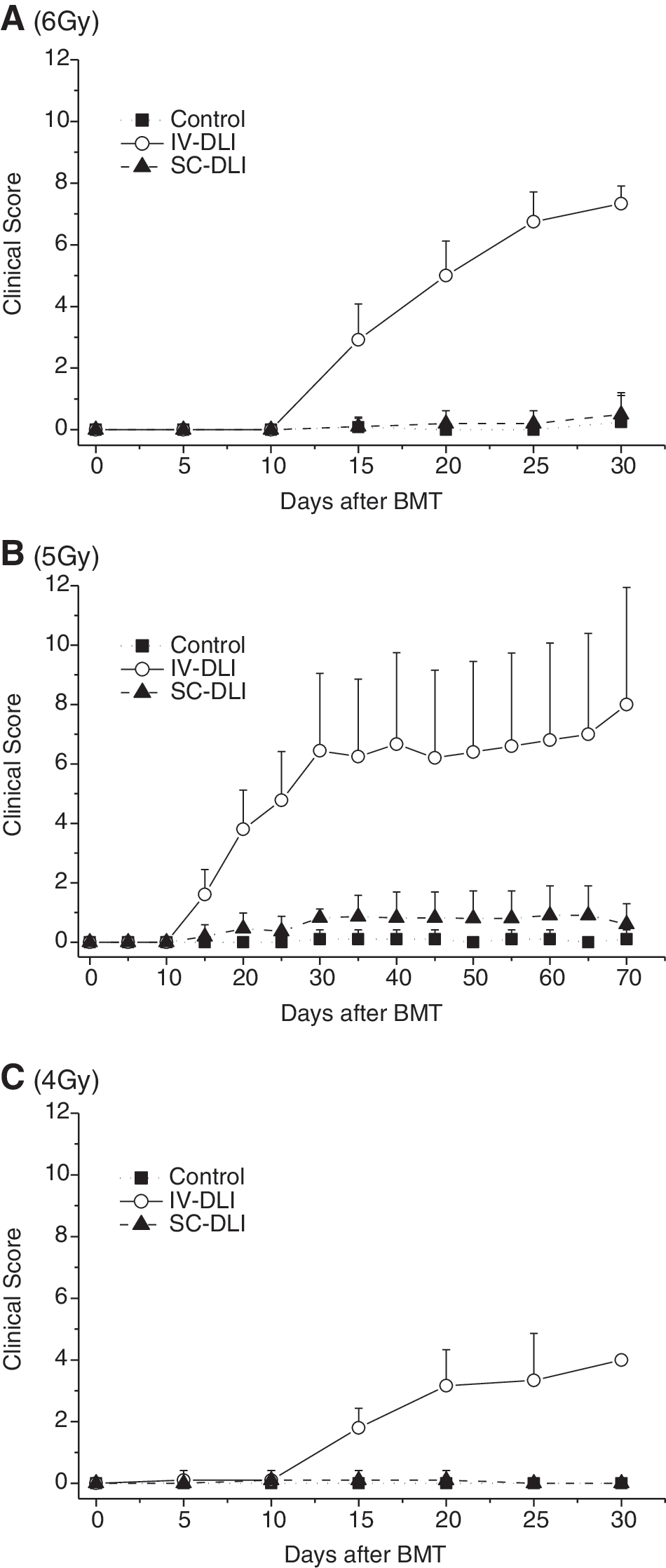
SC-DLI significantly ameliorates GVHD. Recipient mice were irradiated at 6 Gy
In the 4 Gy experiment, the IV-DLI group displayed moderate but not severe GVHD, [although the recipient mice survived short-term (they died between days 16 and 37)]. Pathological diagnosis after autopsy indicated that infection due to graft failure (not GVHD) was the cause of death (data not shown).The clinical score was 4.00 (only two mice survived) on day 30 (Fig. 4C). It is not surprising that no GVHD was observed in either the control group or the SC-DLI group due to a failure of reconstitution.
GVHD is main cause of death in the IV-DLI group
We next examined the histopathological changes in the liver, skin, and intestine from randomly selected recipient mice that had died in the IV-DLI group or were sacrificed at 3 months after BMT in the control group and the SC-DLI group (four mice per group). The severity of histopathological GVHD nearly paralleled the clinical signs. In the IV-DLI group, either 6- or 5-Gy-irradiated recipients showed lymphocyte infiltration in the bile duct, epidermal thickening, and dermal lymphocyte infiltration, occasional crypt apoptosis, and mild inflammation in the intestine (Fig. 5A, C). The pathological scores in the IV-DLI group in the 6 and 5 Gy experiments were 5.75 ± 0.43 and 5.67 ± 0.47, respectively (Fig. 5B, D). In the control group and the SC-DLI group, no or only mild lesions were found in the GVHD target tissues, even in the skin at the site of the injection of splenocytes. The pathological scores in these two groups were also significantly lower than in the IV-DLI group in either radiation dose experiment. These data indicated that GVHD was the main cause of death in the IV-DLI group and that DLI via the SC route could diminish the risk of fatal GVHD.
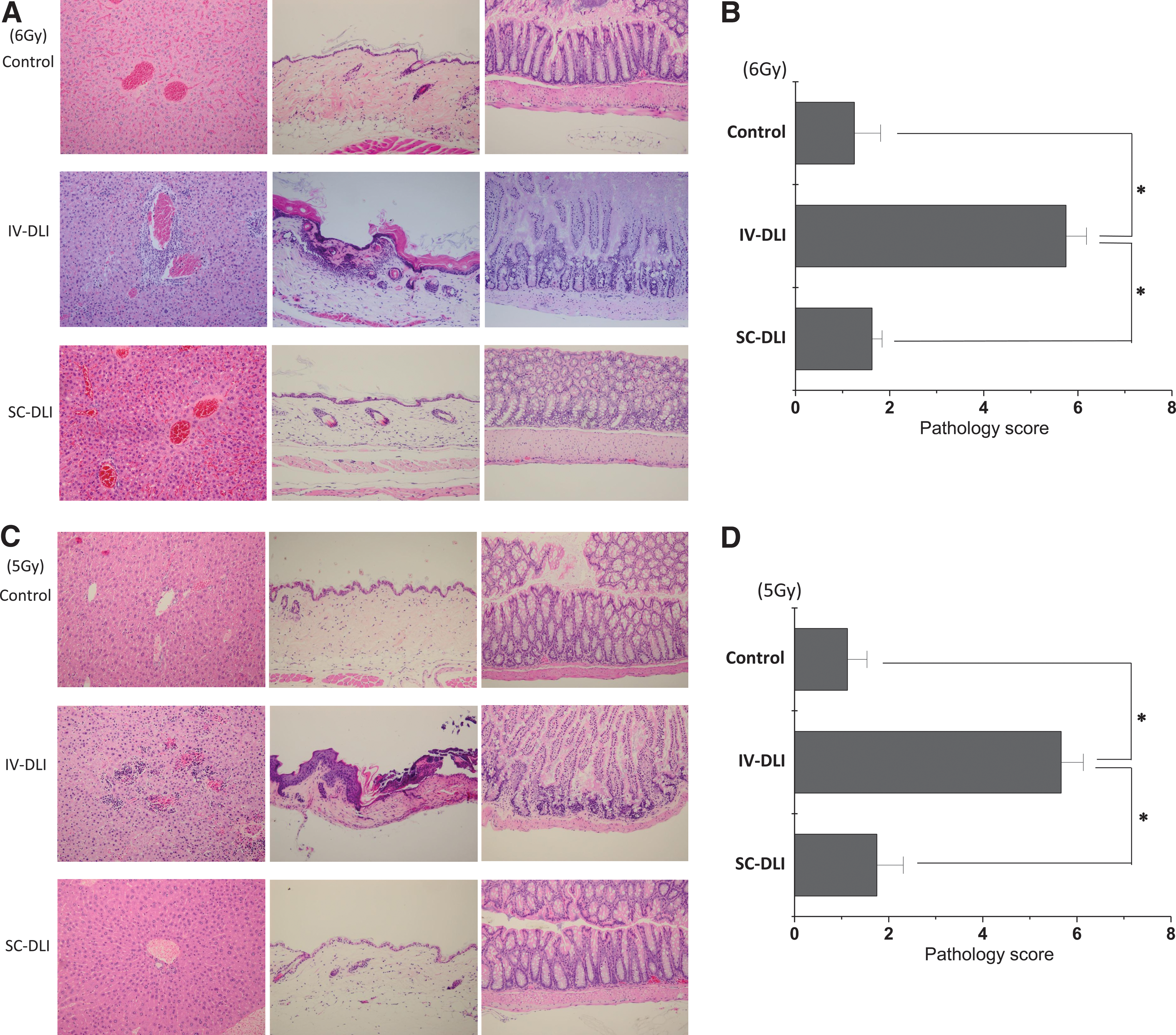
Histopathological score and representative photographs of GVHD target tissues: liver, skin, and intestine. Autopsies were performed on mice that had died (IV-DLI group) or had been sacrificed at 3 months after BMT (control group and SC-DLI group). Tissues from GVHD target organs (liver, skin, and intestine) were prepared for histopathological scoring. For the SC-DLI group, the skin from the site of the injection is shown. Representative photographs of 6 Gy
SC-DLI reduces donor T-cell infiltration into GVHD target tissues
Previous studies reported that donor T cells reached a peak on day 5 after expansion and infiltration into target organs and tissues [27,31]. Li and colleagues claimed that the anti-CD3 treatment as precondition reduced donor T-cell infiltration of GVHD target tissues and prevented GVHD [32]. These results indicate that GVHD may be reduced by infiltration block. Moreover, the results of GVHD could be predicted by the analysis of donor T-cell infiltration to specific tissues. First, the spleens of the recipients were weighed on day 5. The means and SDs in the control group, IV-DLI group, and SC-DLI group were 50.25 ± 5.19, 93.00 ± 15.19, and 46.67 ± 5.69 mg, respectively. The spleens in the IV-DLI group were significantly heavier than in the other two groups (Fig. 6A). We also compared the percentage of donor T cells in the recipient spleen and liver at 5 days after BMT. The percentages of donor T cells in the spleen and liver of the recipients in the IV-DLI group were much higher than in the control group and the SC-DLI group (Fig. 6B, C). These results suggest that the splenocytes administered by SC inhibited donor T-cell migration into the GVHD target tissues more effectively than those injected by the conventional IV method.
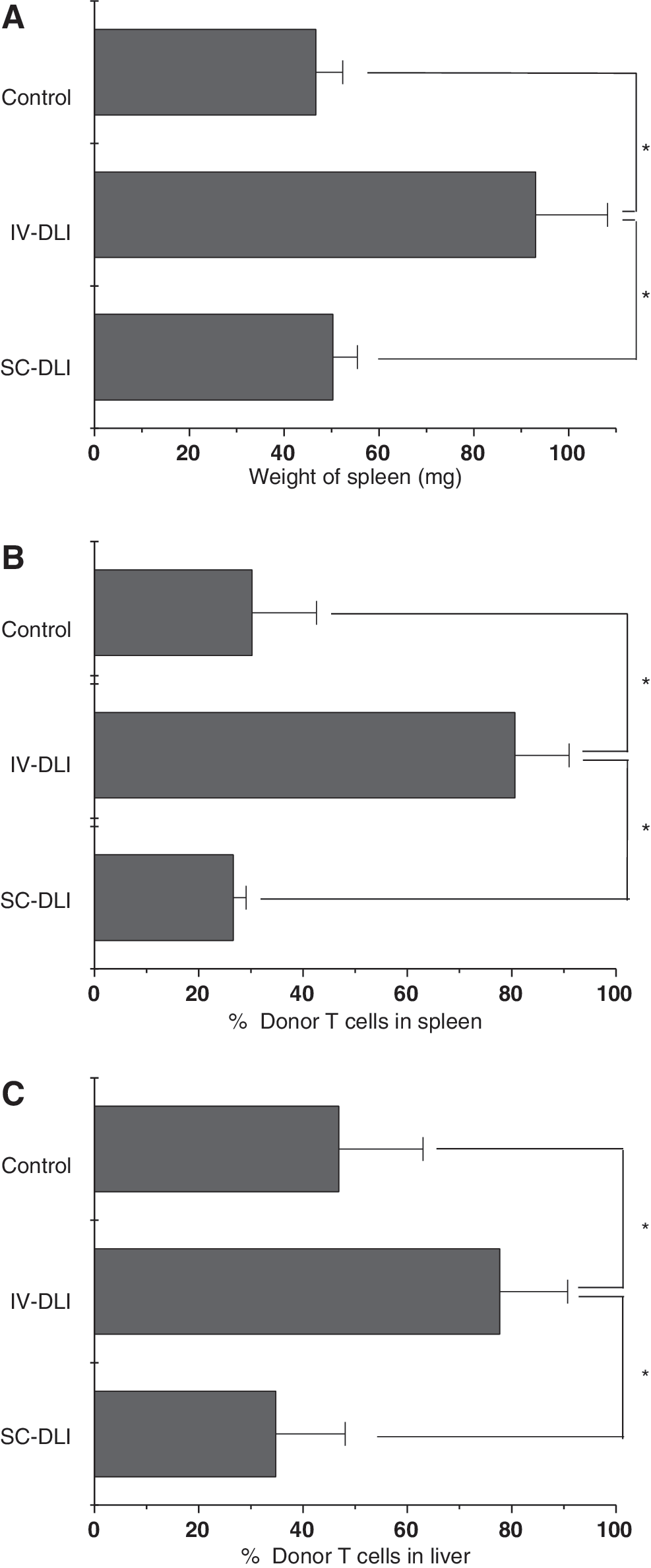
SC-DLI inhibited donor T-cell infiltration of the GVHD target tissues. Five days after BMT and DLI, the weight of the spleen
Discussion
In the present study, we have shown that IV-DLI enhances the dominance of donor hematopoiesis but induces uncontrollable GVHD, followed by death due to infection. On the other hand, SC-DLI enhances the dominance of donor hematopoiesis and induces mild GVHD, which is controllable.
Adoptive immunotherapy with DLI has provided one of the most effective methods after allo-BMT as a treatment and prophylaxis of relapse in the setting of a nonmyeloablative conditioning regimen. DLI is also carried out as a combined method with BMT to convert mixed chimerism to full donor chimerism [17,18,26]. However, the donor T cells administered via the conventional IV route are associated with a major immune-mediated complication, namely GVHD. And the most severe form of GVHD has a high risk of transplant-related mortality [19,21 –23,25]. Here we have demonstrated that, when a large dose of donor splenocytes (5 × 107 per mouse) is used as IV-DLI with IBM-BMT, the dominance of the donor hematopoiesis is obtained, but high mortality is induced at different irradiation doses. On the other hand, SC-DLI could enhance the dominance of the donor hematopoiesis and induce mild GVHD, which is controllable.
In the SC-DLI group, we did not find severe infiltration of lymphocytes in the main GVHD target organs, such as the liver, skin, and intestine, even at the site of injection in the skin, but we did find the mild infiltration of cells in the organs. As we found very few T cells in the organs in the control group, the T cells infiltrating the organs in the SC-DLI or IV-DLI group should be the injected donor T cells. Thus, the difference in the severity of the GVHD depended on the injection route of donor spleen cells. It has been reported that the injected T cells go into the spleen, proliferate there, and then migrate into the target organs of GVHD [33]. Therefore, the severity of GVHD could be predicted by the number of donor T cells in the recipient spleen at several days after DLI. In our experiment, we found a much greater number of donor T cells in the spleen in the IV-DLI group than in the SC-DLI or control group. In the IV-DLI group, severe GVHD was observed clinically and pathologically. Namely, many lymphocytes infiltrated the target organs, such as the skin, liver, and intestine. However, there were only a few lymphocytes in the target organs in the control group and the SC-DLI group, even at the site of injection in the skin. As shown in previous reports [27,31], the mice in the control or SC-DLI group, which had few donor T cells in the spleen and liver, showed no or only mild GVHD. These results suggest that the subcutaneous injection of donor T cells may disturb the infiltration of the T cells.
In the conventional IV infusion method, all the splenocytes enter blood circulation within a short time. These donor splenocytes will then migrate to lymphoid tissues within hours after the injection. And the initial location of the donor splenocytes in the peripheral lymph nodes is not dependent on recipient conditioning or allogeneic disparity [34,35]. The donor allogeneic T cells expand in an explosive manner in lymphoid tissues within 2–3 days, followed by homing and reexpanding in the GVHD target organs [27,34,35]. As a result, the explosively expanded donor T cells in the GVHD target organs induce serious GVHD.
In the SC-DLI group, we also confirmed that donor T cells played an important role in the reconstitution (data not shown); after T-cell depletion, the donor splenocytes lost the ability to reconstitute hematopoiesis with donor-derived cells. In murine allo-BMT, it is well known that donor CD8+ and CD4+ T cells are activated by major histocompatibility complex (MHC) I and MHC II antigen-presenting cells (APCs) (both host- and donor-derived) separately and mediate GVHD in two different ways. The host-derived APCs play a more important role in initiating GVHD than donor APCs [36]. Langerhans cells, a subtype of dendritic cells found only in the skin-draining lymph nodes, show high surface levels of MHC II [37]. Therefore, in the SC-DLI group, only the CD4+ population of T cells can be effectively activated by the Langerhans cells, and the degree of the GVHD is reduced. On the other hand, as we know, subcutaneous injection is widely used as a long-acting depot injection method, and the skin, which is a physical obstacle, can localize the large number of donor splenocytes (5 × 107/0.2 mL) in a not-so-small area (about 7 cm2, measured after injection). The speed at which donor T cells migrate into the recipient's lymphoid tissues is limited by the physical obstacle. Therefore, the downstream of the GVHD process will be slowed down. However, donor T cells appear to migrate in a long-lasting way and the development of GVHD is thus at a low-enough level to be controllable. Moreover, the subpopulation of donor T cells and the mechanisms underlying the reduction of GVHD in the SC-DLI group should be further investigated.
In this experiment, we have shown that SC-DLI can accelerate the replacement of recipient cells and reduce the severity of GVHD. Therefore, SC-DLI is more controllable than IV-DLI in allo-BMT and should thus be an easy and safe method for performing allo-BMT.
Footnotes
Acknowledgments
The authors thank Ms. Y. Tokuyama and Ms. A. Kitajima for their expert technical assistance and also Mr. Hilary Eastwick-Field and Ms. K. Ando for the preparation of this manuscript. Grant support: a grant from The 21st Century COE Program of the Ministry of Education, Culture, Sports, Science, and Technology, the Department of Transplantation for Regeneration Therapy (Sponsored by Otsuka Pharmaceutical Company, Ltd.), Molecular Medical Science Institute, Otsuka Pharmaceutical Co., Ltd., Japan Immunoresearch Laboratories Co., Ltd (JIMRO), and Scientific research (C) 18590388.
Author Disclosure Statement
No competing financial interests exist.
