Abstract
Cell therapy remains an experimental treatment for neurological disorders. A major obstacle in pursuing the clinical application of this therapy is finding the optimal cell type that will allow benefit to a large patient population with minimal complications. A cell type that is a complete match of the transplant recipient appears as an optimal scenario. Here, we report that menstrual blood may be an important source of autologous stem cells. Immunocytochemical assays of cultured menstrual blood reveal that they express embryonic-like stem cell phenotypic markers (Oct4, SSEA, Nanog), and when grown in appropriate conditioned media, express neuronal phenotypic markers (Nestin, MAP2). In order to test the therapeutic potential of these cells, we used the in vitro stroke model of oxygen glucose deprivation (OGD) and found that OGD-exposed primary rat neurons that were co-cultured with menstrual blood-derived stem cells or exposed to the media collected from cultured menstrual blood exhibited significantly reduced cell death. Trophic factors, such as VEGF, BDNF, and NT-3, were up-regulated in the media of OGD-exposed cultured menstrual blood-derived stem cells. Transplantation of menstrual blood-derived stem cells, either intracerebrally or intravenously and without immunosuppression, after experimentally induced ischemic stroke in adult rats also significantly reduced behavioral and histological impairments compared to vehicle-infused rats. Menstrual blood-derived cells exemplify a source of “individually tailored” donor cells that completely match the transplant recipient, at least in women. The present neurostructural and behavioral benefits afforded by transplanted menstrual blood-derived cells support their use as a stem cell source for cell therapy in stroke.
Introduction
S
Embryonic stem (ES) cells are pluripotent cells that can differentiate to all specialized cell types of the organism [7,8]. Unfortunately, numerous ethical and logistical considerations limit the utility of these cells, which can only be isolated from the inner cell mass of early embryos. Moreover, the tumorigenicity of ES cells remains a major obstacle for clinical application [9,10]. The advent of adult stem cells may circumvent the inherent problems of ES cells. Although the multipotent property of adult stem cells has been debated, accumulating evidence indicates that these cells possess ES cell-like features including their ability to differentiate into tissues of an entirely different germ layer [11 –17].
The bone marrow and umbilical cord blood are the 2 most studied adult stem cells, and have been proposed for autologous transplantation [11,17]. The availability of a transplant donor cell type that completely matches the transplant recipient appears as an optimal scenario for cell therapy in view of graft-versus-host complications, in the event of a mismatch between donor and recipient, largely resulting in transplant failure in hematopoietic stem cell transplantation [18,19], Of interest, immature donor cell sources, such as umbilical cord blood, seem to be relatively tolerated by the transplant recipient despite a HLA mismatch [20]. Accordingly, strategies designed to amplify autologous transplantation should benefit a large patient population. The derivation of adult stem cells from the bone marrow may be painful, whereas harvesting umbilical cord blood is, of course, limited during the baby delivery.
That menstrual blood that represents a novel source of stem cells [21] is recognized in the remarkable capacity of the lining of the uterus for regeneration after each menstrual cycle [22]. Extraction of this rich source of stromal cells is efficient and noncontroversial. In studying the cells released from the uterine lining as part of the menstrual blood, multipotent cells capable of differentiating into chrondrogenic, adipogenic, osteogenic, neurogenic, endothelial, pulmonary epithelial, hepatic/pancreatic, and cardiogenic cell lineages have been identified and characterized [21,23]. The cells maintain potency to differentiate and display highly proliferative capabilities that may be linked to ES cell surface markers retained on the cells (ie, SSEA-4, Oct4). Menstrual blood-derived stem cells thus pose as a novel cell population that may be routinely and safely isolated and provide a renewable source of stem cells from child-bearing women. In this study, we further characterized their neural stem cell features and evaluated their potential as graft source for stroke therapy.
Materials and Methods
Collecting and transporting menstrual blood cells
Detailed methods for the procurement and processing of cells obtained from a female and how the cells were isolated, collected, and preserved from the menstrual blood are described previously [21]. The menstrual blood cells were collected via a procurement kit prepared by the processing facility approved under an IRB study for the collection, processing, cryopreservation, and post-thaw viability of the harvested cells. Collections took place over 4 h or less. Menstrual blood was collected on day 1, 2, or 3 during the heaviest flow of the cycle. On an average 8–10 mL was collected per sample. An average collection is ∼30 million with anywhere from 0.5% to 40% of adherent cells. Only 1 million cells are required for the first cell culture to select the adherent cells with generally about 10% of the cells demonstrating adherence and about 100,000 cells sub-cultured. The cells double approximately every 24 h. All samples collected were stored between at ∼4°C post-collection. The samples were shipped to the laboratory on frozen bricks to assure shipment of cells at a cool temperature.
Menstrual blood cell processing and cryopreservation
A buffered saline media (DPBS) is used throughout the cell isolation process with heparin (heparin sodium 1,000 USP Units/mL; American Pharmaceutical Partners, Schaumburg, IL). The menstrual cells collected in a buffered saline conical collection tube are subjected to centrifugation at 2,000 rpm for 7 min at ∼4°C. The supernatant was used for microbiological testing. Pelleted cells are resuspended for a cell count and viability. The cells were prepared for cryopreservation. Bacteriological analysis of the supernatant was performed using the BacT/ALERT system (Biomerieux, Durham, NC). For the present study, the cells were grown to either passage 6 or 9 and cultured for additional 3 passages before testing without observing signs of contamination. Most products arrived to the laboratory with some level of contamination but after a treatment of an antibiotic cocktail, when the cells were thawed post-processing, the culture was found negative of contaminants. Two samples were used in this study, with cell samples tested at different passages (ie, passage 6 or 9 and cultured for additional 3 passages before testing). One milliliter of cellular suspension was tested for the total cell count, cell viability, and flow cytometric analysis for specific markers. The entire sample is initially filtered with a 100-µm filter prior to cryopreserving the cells. The cells are cryopreserved in a total volume of 10 mL comprising of 5 mL of cells, 3 mL of the buffered saline (DPBS), 1 mL of the protein HSA (Telacris Bio, Clayton, NC), and 1 mL of the preservative DMSO (99% Stemsol). Cells were cryopreserved in a controlled rate freezer (Controlled Rate Freezer 7454; Thermo Electron, Corp., Marietta, OH) until it reached −90°C, then the cryovials were transferred to a cryogenic storage unit and stored in the vapor phase of liquid nitrogen at a temperature at or below −150°C (LN2 Freezer MVE 1830; Chart Industries, Garfield Heights, OH).
Concentrated menstrual cell thaw, cell culture, and cell selection
Cells were thawed to expand in culture and selected for CD117 as previously described [21]. The freeze–thaw process demonstrated a high level of viability after adherent cells had been selected for CD117. Most passages revealed close to 100% viability as determined by 7-AAD. CD117 selection was performed via Miltenyi system at post-thaw, with positive selected cells subsequently expanded in culture. CD117 has been previously identified in endometrial cells, and shown to be closely associated with a highly proliferative cell type and appears to promote cell survival and migration [24]. In brief, to thaw cells they were agitated in a 37°C water bath, transferred to chilled Chang’s complete media [25,26] with DNase, centrifuged at 120g for 5 min, and resuspended in Chang’s complete media without DNase by gentle inversion. Cells were seeded in T-25 nontreated tissue culture flask with ∼1 million cells/flask. The leukocytes were not removed from the cell preparation, but only the adherent cells remained in culture and the adherent layer was sub-cultured to expand the cells. Once cells were sub-cultured, they were seeded at 2,000 cells/cm2. Cells were incubated in a 5% CO2 incubator at 36°C–38°C until they are confluent to 70%–80%. When cells were sub-cultured, the flask was rinsed with 5 mL DPBS and treated with 1.5 mL of prewarmed TrypLE (Invitrogen #12605-010; location Carlsbad, CA) incubated at 37°C. After incubation, cells were seeded to expand and when there was a minimum of 2.5 million total nucleated cells they were selected for CD117. The CD117 stem cells were separated from a cellular suspension in working buffer using a MS column and a MiniMACS kit (Miltenyi Biotec, Bergisch Gladbach, Germany).
In vitro study
Cell culture. Primary cultures of neurons were derived from the rat (Sprague-Dawley) striatum and maintained in culture following the supplier’s protocol (Cambrex, Walkersville, MD). In brief, immediately after thawing, cells (4 × 104 cells/well) were seeded and grown in 96-well plate coated by poly-
Oxygen glucose deprivation (OGD). Cultured CD117+ at passages 6 and 9 were cultured for additional 3 passages, and exposed to the OGD injury model as described previously [27] with few modifications (see Fig. 1). In brief, culture medium was replaced by a glucose-free Earle’s balanced salt solution (BSS) with the following composition: 116 mM NaCl, 5.4 mM KCl, 0.8 mM MgSO4, 1 mM NaH2PO4, 26.2 mM NaHCO3, 0.01 mM glycine, 1.8 mM CaCl2, and pH adjusted to 7.4 with or without minocycline. Cultured cells were placed in humidified chamber and equilibrated with continuous flow of 92% N2 and 8% O2 gas for 15 min. After this equilibrium, the chamber was sealed and placed into the incubator at 37°C for 48 h for MTT assay and Trypan blue stain. The MTT assay is a chemiluminescence-based assay, with spectrometric absorbance measured at 595 nm (for formazan dye) and with the absorbance at >650 nm for reference.

Schematic diagrams of in vitro and in vivo experimental procedures.
Cell viability. Cell viability was evaluated by ATP activity following the supplier’s protocol (Promega, Madison, WI) and by Trypan blue (Sigma, St. Louis, MO). In brief, MTT assay was carried out by adding MTT assay solution immediately after OGD. The intensities of chemiluminescence of ATP activity were measured and calculated by Image station 2000R system (Kodak, NY). In addition, Trypan blue exclusion method was conducted and mean viable cell counts were calculated in 3 randomly selected areas (0.2 mm2) in each well (n = 5 per treatment condition) to reveal the cell viability for each treatment condition.
Immunocytochemistry. Each 1 × 105 cells were plated on 8-well Permanox® slides (Nalge Nunc Int, Naperville, IL) at 2 days before fixation. Cultured cells were treated with 4% paraformaldehyde (PFA) for 10 min at room temperature after rinsing with phosphate-buffered saline (PBS). After blocking reaction with 10% normal goat serum (Vector, Burlingame, CA), cells were incubated overnight at 4°C with specific antibodies against Oct4 (1:1,000; Abcam, Cambridge, MA), SSEA (1:200; Abcam), Nanog (1:500; Abcam), CXCR4 (1:100; Abcam), Nestin (1:200; Abcam), MAP2 (1:1,000; Abcam), GFAP (1:100; Abcam), and NeuN (1:1,000; Abcam) with 10% normal goat serum. After several rinses in PBS, cells were incubated for 45 min at room temperature in FITC-conjugated anti-mouse IgG (1:1,000; Molecular Probes, San Jose, CA), or rhodamine-conjugated anti-rabbit IgG (1:2,000; Molecular Probes) for visualization. Cells were processed for DAPI staining then subsequently embedded with mounting medium. Immunofluorescent images were visualized using Zeiss Axiophot 2. In addition, control studies included exclusion of primary antibody and substituted with 10% normal goat serum in PBS. No immunoreactivity was observed in these controls. All studies were conducted in triplicates. Assessment was performed blindly by an independent investigator.
ELISA. The timing of ELISA paralleled the hypoxic condition (ie, OGD) using passage 6 or 9 and additionally cultured for 3 passages before testing, with the conditioned media collected at 2 days after OGD. VEGF, BDNF, NT-3, but not GDNF trophic factors such as vascular endothelial growth factor (VEGF), brain-derived neurotrophic factor (BDNF), glial cell line-derived neurotrophic factor (GDNF), and neurotrophin-3 (NT-3) have been detected as critical secretory factors in stem cells. Thus, we measured these molecules as possible neurotrophic factors secreted by menstrual blood-derived stem cells. The media collected as described above served as samples for evaluation of trophic factor secretion. The levels of VEGF, BDNF, GDNF, and NT-3 released from 1 million adherent cells were determined using ELISA kits according to the protocols of the manufacturer (BDNF and GDNF from Promega; VEGF and NT-3 from R&D Systems). The conditioned media were analyzed by interpolation from the standard curves assayed on individual plates.
In vivo study
Stroke surgery. Transient unilateral focal ischemia was produced using a well-established middle cerebral artery occlusion (MCAo) using the intraluminal suture model as previously described [28,29] (see Fig. 8). Male Sprague-Dawley rats weighing about 250 g served as subjects in this study. Animals were anesthetized with gas inhalation composed of 30% oxygen (0.3 L/min) and 70% nitrous oxide (0.7 L/min) mixture. The gas was passed through an isoflurane vaporizer set to deliver 3% to 4% isoflurane during initial induction and 1.5% to 2% during surgery. Physiological parameters, via blood gases assays, and ischemia and reperfusion levels determined by laser Doppler measurements, did not differ among all MCAo stroke groups. The body temperature of animals was maintained at 37°C during the surgery until they recovered from anesthesia. Based on our pilot studies, a 60-min MCAo produces a well-defined ischemic core and penumbra.
Grafting procedures. All surgical procedures were conducted under aseptic conditions. For intracerebral (IC) transplantation in MCAo stroke animals, anesthetized animals (equithesin 3 mL/kg i.p.) were implanted with menstrual blood-derived stem cells directly into the striatum (0.5 mm anterior to bregma, 2.8 mm lateral to midline, and 5.0 mm below the dural surface), using a 28-gauge implantation cannula [30]. For intravenous (IV) delivery, animals were anesthetized with 2% isoflurane in a jar for pre-anesthetic, and spontaneously respired with 1.5% isoflurane in 2:1 N2O:O2 mixture using a facemask connected and regulated with a modified vaporizer. Accurate placement of a 27-gauge needle within the jugular vein was confirmed by aspirate of blood into the syringe. Cell volume was preset at 400k in 3 µL solution (PS) and 4 million cells in 1 mL solution (PBS) for IC and IV transplantation, respectively. The rationale for the current IC and IV doses is based on our experience with similar stem/progenitor cell therapeutic doses, including bone marrow, umbilical cord blood, and fetal tissue-derived cells [28,29,31,32]. Delivery was via manual infusion with infusion rate approximately at 1 µL or 1 mL per minute, respectively. Cryopreserved cells were obtained from Cryo-Cell International Inc. and thawed just prior to transplantation surgery. Viability cell counts, using Trypan blue exclusion method, were conducted prior to transplantation and immediately after the transplantation on the last animal recipient. The cell viability criterion of at least 80% viable cells was used to proceed with the transplantation surgery. The predetermined cell dosages refer to number of viable cells. Transplantation surgery was carried out within 2 h post-stroke, and timed with thawing of the cells. A heating pad and a rectal thermometer allowed maintenance of body temperature at about 37°C throughout surgery and following recovery from anesthesia. In each treatment group, 4–5 animals were randomly assigned to receive either cells from passage 6 or 9, with both cells further cultured for 3 passages before testing. The original sample size was 10 rats per group, but 3 animals died during and/or immediately after stroke surgery, thus the study included the following subjects in each treatment condition: IV (n = 9), IC (n = 10), and vehicle (n = 8). Four animals received IC vehicle and another 4 animals received IV vehicle. Behavioral and histological analyses between these 2 control groups revealed tight behavioral scores and cell counts, without any significant statistical differences, thus data from both groups were combined and treated as one group.
Behavioral tests. Behavioral assessment was performed by using semiquantitative analyses of motor asymmetry (elevated body swing test, EBST) [30,33,34], motor coordination (cylinder test) [29], and neurological function (Bederson test) [30,33,34] at 14 days after stroke transplantation surgery. The EBST provided a motor asymmetry parameter. The test apparatus consisted of a clear Plexiglas box (40 × 40 × 35.5 cm). The animal was gently picked up at the base of the tail, and elevated by the tail until the animal’s nose is at a height of 2 in. (5 cm) above the surface. The direction of the swing, either left or right, was counted once the animal’s head moved sideways ∼10° from the midline position of the body. After a single swing, the animal was placed back in the Plexiglas box and allowed to move freely for 30 s prior to retesting. These steps were repeated 20 times for each animal, with performance expressed in percentage to show the biased swing activity. The cylinder test was used to assess the degree of forepaw asymmetry. Rats were placed in a transparent cylinder (diameter: 20 cm, height: 30 cm) for 3 min with the number of forepaw contacts to the cylinder wall counted. The score of cylinder test in this study was calculated as a contralateral bias, that is [(the number of contacts with the contralateral limb) – (the number of contacts with the ipsilateral limb)/(the number of total contacts) × 100]. The Bederson test is conducted by assigning a neurologic score for each rat obtained from a battery of 4 tests that which included: (1) observation of spontaneous ipsilateral circling, graded from 0 (no circling) to 3 (continuous circling); (2) contralateral hind limb retraction, which measured the ability of the animal to replace the hind limb after it was displaced laterally by 2 to 3 cm, graded from 0 (immediate replacement) to 3 (replacement after minutes or no replacement); (3) beam walking ability, graded 0 for a rat that readily traversed a 2.4-cm -wide, 80-cm -long beam to 3 for a rat unable to stay on the beam for 10 s; and (4) bilateral forepaw grasp, which measured the ability to hold onto a 2-mm -diameter steel rod, graded 0 for a rat with normal forepaw grasping behavior to 3 for a rat unable to grasp with the forepaws. The scores from all 4 tests, which were done over a period of about 15 min, were added to give a neurologic deficit score (maximum possible score of 12 points from the 4 component tests). To prevent any examiner’s bias, all behavioral evaluations were performed by an investigator blinded to the treatment conditions.
Immunohistochemistry. Under deep anesthesia, rats were sacrificed at 14 days after reperfusion, and perfused through the ascending aorta with 200 mL of cold PBS, followed by 100 mL of 4% PFA in PBS. Brains were removed and postfixed in the same fixative for 3 days followed by 30% sucrose in phosphate buffer (PB) for 1 week. Six series of coronal sections were cut at a thickness of 30 µm by cryostat and stored at −20°C. Free-floating sections for immunohistochemistry were incubated overnight at 4°C with anti-MAP2 polyclonal antibody (1:500; Chemicon), Anti-HuNu (1:50; Chemicon), and anti-Oct4 (1:100; Abcam) with 10% normal goat serum. After several rinses in PBS, the sections were visualized following the method described above with modification to accelerate FITC with biotin-conjugated anti-mouse IgG antibody and FITC-conjugated streptoavidin (1:500; Sigma, St. Louis, MO). Control studies included exclusion of primary antibody substituted with 10% normal goat serum in PBS. No immunoreactivity was observed in these controls. Finally, brain sections were counterstained with DAPI. Immunofluorescent and light microscopic examinations were carried out using Zeiss Axiophot 2. Sections were blind-coded and Abercrombie’s formula was used to calculate the total number of immunopositive cells (Abercrombie correction = C × (T/T+N), where C is crude cell counts, T is the section thickness, and N is mean nucleus size) [35].
Morphological analysis. The density of endogenous cells in the ischemic penumbra (determined by DAPI stain using consecutive sections) was estimated and analyzed as described previously [36,37]. In brief, the level of +0.2 mm anterior to the bregma based on the atlas of Paxinos and Watson [38] was selected for semiquantitative analysis. Based on our pilot studies, this brain area consistently exhibits the ischemic striatal penumbra following a 60-min MCAo. For estimation of DAPI-positive cells, 2 striatal areas (each area: 0.05 mm2) in the ischemic penumbra and 2 symmetrical areas in the contralateral side were analyzed using Scion Image software (Scion Corp., Frederick, MD). The areas were captured; binary images were created with a distinct threshold; and the positive areas were calculated and summed up. The ratio of lesion to intact side was used for statistical analyses. For estimation of graft survival, HuNu-positive cells were counted in 6 consecutive 0.05-mm2 regions of the inner and outer boundary zone of striatal penumbra. The total number of HuNu-positive cells in the 6 areas was counted and expressed as cells/mm2 for statistical analyses. Similarly, for determining whether the HuNu-positive cells remained as stem cells or differentiated into a neuronal lineage, double-labeling with Oct4 and MAP2 was examined in the same striatal ischemic penumbra as above.
Statistics
Significant differences in behavioral deficits and ischemic cell loss were analyzed using ANOVA followed by post hoc Fisher’s test. Statistical significance was preset at P < 0.05.
Results
Aliquots derived from the 2 samples of cells, passaged either 6 or 9, and grown for additional 3 passages in culture, were found to behave similarly, with cell yields, number of adherent cells and those expressing neuronal phenotypes, as well as graft survival and functional effects showing near complete resemblance of the 2 cell populations. Thus, the data from these 2 cell populations were collapsed into a single treatment condition. A flow chart of experimental procedures is provided (Fig. 1).
Cultured menstrual blood cells display embryonic stem cell-like features
Immunocytochemical assays of cultured menstrual blood reveal that they express embryonic-like stem cell phenotypic markers (Oct4, SSEA, Nanog) (Fig. 2). Greater than 90% of the cells were positive for these pluripotent markers. They maintained these stemness properties at least up to 9 passages plus the additional 3 culture passages (ie, longest time point the cells were cultured in this study). In addition, their growth rate or proliferative capacity did not change over time. Of note, human ES cells showed the typical specific nuclear staining, but we detected a few cells positive for Oct4 show cytoplasmic labeling, which appears to be the pattern of staining displayed by majority of menstrual blood-derived stem cells. While we do not have a solid explanation for such cytoplasmic labeling, this differential pattern of Oct4 labeling may distinguish ES cells from menstrual blood-derived stem cells. Furthermore, menstrual blood-derived stem cells (75%) were CXCR4-positive, a stem cell chemotaxis marker, also expressed by human ES cells. The cells were plated on a coated 10-cm dish in DMEM/F12 supplemented with ITS, and the medium was changed twice a week throughout the study.
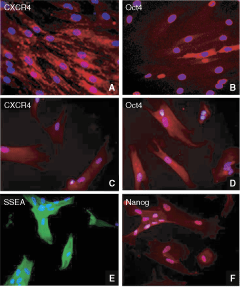
Cultured menstrual blood cells display embryonic stem cell-like features. Panels
Cultured menstrual blood cells can be steered toward neural lineage
After passage 6 or 9, the cells were transferred to coated dishes in neural induction medium (DMEM/F12 supplemented with N2 and FGF-2) for a week, and retinoic acid was added to the medium over the next 3 weeks. Cells were Nestin-positive, indicative of an early neural lineage commitment, and readily differentiated into intermediate neuronal (30% MAP2-positive, but the mature neuronal marker, NeuN, labeling not detected) and astrocytic phenotype (40% GFAP-positive) upon withdrawal of FGF-2 (Fig. 3). Thus, when grown in appropriate conditioned media, cultured menstrual blood stem cells express neural phenotypic markers (Nestin, MAP2).
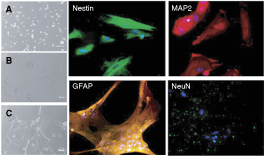
Cultured menstrual blood cells can be steered toward neural lineage. Morphological changes in cultured menstrual blood cells immediately following thawing (
Co-cultured menstrual blood-derived stem cells protects against in vitro stroke insult
In order to test the therapeutic potential of these cells, we used the in vitro OGD stroke model and found that OGD-exposed primary rat neurons that were co-cultured with menstrual blood-derived stem cells or exposed to the media collected from cultured menstrual blood-derived stem cells exhibited significantly protected against ischemic cell death (Fig. 4). ANOVA revealed significant treatment effects in both Trypan blue exclusion method (F 2,6 = 58.78, P < 0.0001) and MTT assay (F 2,6 = 45.60, P < 0.001) for detecting cell death and cell survival, respectively. Post hoc tests revealed that menstrual blood-derived stem cells (Trypan blue exclusion method and MTT assay, P < 0.0001 vs. controls) or the media collected from cultured menstrual blood-derived stem cells (Trypan blue exclusion method, P < 0.0001 vs. controls; MTT assay, P < 0.001 vs. controls) significantly reduced cell death and improved cell survival of OGD-exposed primary neurons. There were no significant differences in the protective effects afforded by co-culturing with menstrual blood-derived stem cells and exposure to the media collected from cultured menstrual blood-derived stem cells (P > 0.1).
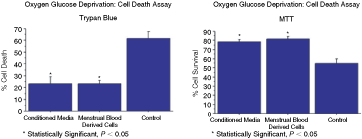
Co-cultured menstrual blood-derived stem cells protects against in vitro stroke insult. Cell viability tests using Trypan blue exclusion method and MTT assay revealed that oxygen glucose deprivation (OGD)-exposed primary rat neurons that were co-cultured with menstrual blood-derived stem cells or exposed to the media collected from cultured menstrual blood-derived stem cells exhibited significantly protected against ischemic cell death. There were no significant differences in the protective effects afforded by co-culturing with menstrual blood-derived stem cells and exposure to the media collected from cultured menstrual blood-derived stem cells. Error bars represent standard deviations. Data were generated from 2 triplicates of 2 different menstrual blood-derived stem cell samples. Asterisk corresponds to statistically significant difference between conditioned media or menstrual blood-derived stem cells and control.
Cultured menstrual blood-derived stem cells secrete growth factors
As an approach to reveal a mechanism of action underlying the therapeutic benefits of cultured menstrual blood-derived stem cells, we assayed for growth factors implicated as neuroprotective in stroke models. ELISA data showed elevated levels of trophic factors, such as VEGF, BDNF, and NT-3, in the media of OGD-exposed cultured menstrual blood-derived stem cells (Table 1).
ELISA R
Trophic factors, such as vascular endothelial growth factor (VEGF), brain-derived neurotrophic factor (BDNF), and neurotrophin-3 (NT-3), were up-regulated in the media of oxygen glucose deprivation (OGD)-exposed cultured menstrual blood-derived stem cells.
Transplantation of menstrual blood-derived stem cells attenuates behavioral and histological deficits in stroke animals
In order to further evaluate the therapeutic potential of menstrual blood-derived stem cells in stroke, we next examined the efficacy of transplanting these cells in an established in vivo rat stroke model. Transplantation of menstrual blood-derived stem cells, either intracerebrally (IC) or intravenously (IV), after experimentally induced ischemic stroke in adult rats significantly reduced behavioral abnormalities compared to vehicle-infused rats (Fig. 5). ANOVA revealed significant treatment effects in all 3 behavioral tests (EBST, F 2,24 = 29.71, P < 0.0001; Cylinder test, F 2,24 = 103.87, P < 0.0001; and Bederson test, F 2,24 = 16.27, P < 0.0001), with post hoc t-tests showing that both IC- and IV-delivered menstrual blood-derived stem cells ameliorated these motor and neurological impairments. In addition, both IC and IV route produced the same degree of behavioral recovery in EBST and Bederson test (P > 0.1), but direct transplantation into the stroke brain promoted significantly better improvement of motor coordination in the cylinder test than peripheral administration (P < 0.05).
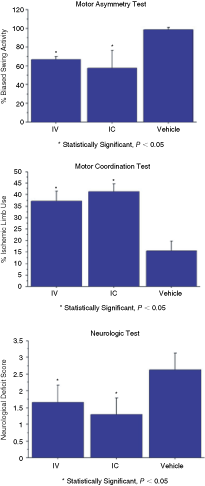
Transplanted menstrual blood-derived stem cells rescue against in vivo stroke injury. Transplantation of menstrual blood-derived stem cells, either intracerebrally (IC) or intravenously (IV), after experimentally induced ischemic stroke in adult rats significantly reduced behavioral abnormalities, including motor asymmetry, motor coordination, and neurological performance, compared to vehicle-infused rats. Error bars represent standard deviations. Asterisk corresponds to statistically significant difference between IC- or IV-delivered menstrual blood-derived stem cells and control; however, in the motor coordination test, IC shows significantly better recovery of ischemic limb compared to IV.
Immunohistochemical examination using DAPI staining revealed that there were more surviving host cells in the striatal ischemic penumbra of stroke animals that received either IC and IV transplantation of menstrual blood-derived stem cells compared to those that received vehicle infusion (ANOVA F2,24 = 55.91, P < 0.0001; post hoc t-tests: IV or IC vs. controls, P < 0.0001; no significant difference between IC and IV, P > 0.05) (Fig. 6).
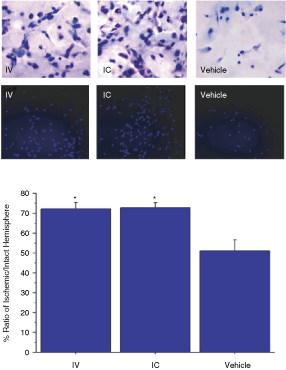
Transplanted menstrual blood-derived stem cells increase survival of host cells within the ischemic penumbra. Histological and immunohistochemical examination using H&E (top panel) and DAPI staining revealed that there were more surviving host cells in the striatal ischemic penumbra of stroke animals that received either IC and IV transplantation of menstrual blood-derived stem cells compared to those that received IC (shown) or IV vehicle infusion (*P < 0.0001). Error bars represent standard deviations. Asterisk corresponds to statistically significant difference between IC- or IV-delivered menstrual blood-derived stem cells and control.
Immunofluorescent microscopic evaluation of the status of grafted cells revealed that menstrual blood-derived stem cells either transplanted intracerebrally or intravenously survived in the stroke rat brain (ischemic penumbra). At 14 days after transplantation, IC-delivered menstrual blood-derived stem cells (HuNu-positive) were detected in the ischemic striatal penumbra within and a short distance away from the original transplant site (Fig. 7). Most of the cells (>80%) retained their stem cell marker (Oct4). Similarly, IV-delivered menstrual blood-derived stem cells (HuNu-positive) were detected in the inner and outer boundary of the ischemic striatal penumbra, with most of the cells (>90%) retaining their stem cell marker (Oct4). Approximate graft survival rates were 15% and <1% for IC- and IV-delivered cells, respectively.
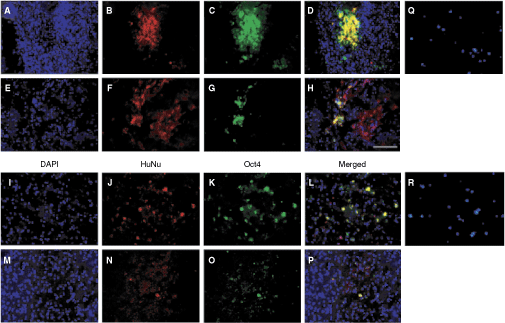
Transplanted menstrual blood-derived stem cells survive in the stroke brain. Immunofluorescent microscopic evaluation of the status of grafted cells revealed that menstrual blood-derived stem cells either transplanted intracerebrally (
Discussion
We demonstrate that cultured menstrual blood-derived cells express stem cell markers, and some exhibit neural markers. Menstrual blood-derived stem cells afford protection in co-cultured primary neurons subjected to experimental in vitro stroke. More importantly, transplantation of menstrual blood-derived stem cells, either directly into the brain or peripherally, ameliorated stroke-induced behavioral and histological deficits. These robust effects of menstrual blood-derived stem cells make them appealing for early cell therapy for stroke.
The existence of a population of stem cells in the intact endometrium that were believed to die and later shed during the menstrual cycle has been suggested 30 years ago [22]. However, not until recently did a study show that adherent cells derived from the endometrium are capable of differentiating into 9 lineages, namely, cardiomyocytic, respiratory epithelial, neurocytic, myocytic, endothelial, pancreatic, hepatic, adipocytic, and osteogenic cells [23]. In parallel, stromal cells harvested from menstrual blood display multipotent markers such as Oct4, SSEA-4, and c-kit at the molecular and cellular level [21]. In addition, menstrual blood-derived mesenchymal cells have been demonstrated to differentiate into cardiac precursor-like cells [39]. These adherent, stromal, and mesenchymal cells appear to exhibit characteristics similar to stem cells derived from the endometrium present in the menstrual blood. Indeed, it has been suggested that endometrial stem/progenitor cells exist in the endometrium and in menstrual blood. For example, menstrual blood could contain circulating bone marrow mesenchymal stem cells, in addition to the cells shed from the endometrium, which have been implicated to contribute to endometrial regeneration [40]. Not all the endometrium is shed; the basal layer remains, where endometrial stem/progenitor cells are thought to reside. However, equally compelling evidence from a recent study identifies partially purified endometrial mesenchymal stem cell-like cells incorporated within the functional layer that is shed [41]. This would perhaps explain why some endometrial mesenchymal stem cell-like cells are present in menstrual blood in addition to the stromal fibroblasts present in human endometrium. At this time, we cannot conclusively state whether the menstrual blood-derived stem cells are uniquely different from bone marrow mesenchymal stem cells without performing a vis-à-vis comparative study between the 2 cell populations. Based on the stem cell markers we employed here (Oct4, Nanog, SSEA, and CXCR4), both menstrual blood-derived stem cells and bone marrow stem cells appear to share similar phenotypic properties.
Despite robust differentiation of menstrual blood-derived stem cells into cardiac cells [39], they seem to have limited ability to commit into neuronal lineage as shown in the present study. Although pathophysiological symptoms of stroke and cardiac arrest significantly overlap (eg, tissue infarcts), a major difference between the 2 diseases is the underlying cell damage, that is, muscles versus neurons. This host environmental niche has been shown to participate in directing the lineage commitment of transplanted stem cells, thereby differentially regulating the eventual fate of menstrual blood-derived stem cells. The enhanced tendency of menstrual blood-derived stem cells to commit more preferentially to cardiomyocytes when transplanted to the ischemic heart over neuronal differentiation following transplantation into the stroke brain may be due to the relative tissue-specific differentiation signals rendered by the injured organ, which can be highly time-dependent post-injury. The original source of the stem cells may also likely dictate their lineage commitment. In particular, menstrual blood stem cells may be derived from the endometrial mucosa lined with a luminal epithelium from which glands extend to the myometrial layer through a supportive stromal tissue. The glands are lined by a pseudo-stratified columnar epithelium. Accordingly, the biased potency of menstrual blood stem cells to commit toward muscle cells, that is, cardiomyocytes over neural cells may have been a predestined process even prior to transplantation. In the end, host microenvironmental cues and tissue origins of the stem cells, among other factors (diseased state), may contribute to the differentiation of transplanted stem cells.
The exact mechanism underlying the observed functional benefits of menstrual blood-derived stem cells remains to be determined. However, the retention of stemness phenotypic characteristics by menstrual blood-derived stem cell grafts coinciding with immediate behavioral recovery at an early period after transplantation suggests that a bystander effect rather than a cell replacement via neuronal differentiation mechanism likely mediates the observed therapeutic benefits of this cell transplant regimen. Indeed, examination of trophic factor levels in the media of OGD-exposed cultured menstrual blood-derived stem cells reveals the up-regulation of VEGF, BDNF, NT-3, but not GDNF. Recent reports have implicated this set of trophic factors as mediating therapeutic benefits of transplanted stem cells in a variety of CNS disorders, including stroke [42 –47]. In view of clinical product development, there may be less safety and toxicity concerns for peptide delivery compared to cell therapy. Indeed, safety and efficacy of trophic factors have been demonstrated in stroke models [48,49]. Most of these compounds have been examined as a monotherapy, and clinical trials, at least for GDNF treatment in Parkinson’s disease, have been mixed [50 –52]. The ability of menstrual blood-derived cells to secrete a cocktail of growth factors and the cells’ potential to respond to host cues as well as to stimulate the microenvironment are deemed more advantageous than the exogenous application of these growth factors.
We recognize that OGD and normal culture conditions are 2 distinct culture conditions. Because the present disease indication targeted stroke, the choice to demonstrate trophic factor secretion in the in vitro stroke OGD model is deemed a logical approach. Our previous studies [34,53], as well as those by several others [54 –58], have similarly employed experimental in vitro paradigms in providing insights into mechanistic pathway of neural repair in stroke. The present in vitro data provide evidence lending support to the notion that menstrual blood-derived stem cells likely exerted neuroprotection against stroke via a trophic factor mechanism. Consideration was given to clarify trophic factor secretion of transplanted stem cells in the in vivo setting [28,31,53], but the practical application of infusing conditioned medium, as opposed to transplanting stem cells, is limited since product manufacturing likely poses as a challenge in obtaining therapeutic doses of cocktails of growth factors in ample supply to carry out large-scale clinical trials. Moreover, the rationale to pursue the present experiments on neural differentiation in an in vitro setting resonates the guidelines set forth by STEPS (Stem cell Therapeutics as an Emerging Paradigm for Stroke; 5,6), in that the initial step for advancing cell therapy for stroke (or any disease indication for that matter) is by providing a phenotypic characterization of the donor cells. Hence, we provide here the phenotype of menstrual blood-derived stem cells following cell culture differentiation.
The observation that IV transplanted animals displayed functional recovery equal to IC transplanted animals despite less donor cell graft survival bolsters our case that graft survival per se is not the primary mode of action of the cells. Indeed, we previously showed that the entry of grafted cells (ie, umbilical cord blood-derived cells) [28] is not a prerequisite for behavioral recovery of transplanted stroke animals. As our overarching theme here, we highlight that neurotrophic factor secretion by the graft, the host, or combination of both is likely the key neuroprotective mechanism underlying the therapeutic benefits of menstrual blood-derived stem cells. Moreover, it is logical to think that IV administered menstrual blood-derived stem cells possess similar safety and toxicity profile as IV-delivered CXCR4-positive stem cells derived from bone marrow and umbilical cord. To date, no deleterious side effects have been reported with any of these CXCR4-positive cells. Moreover, following stroke, the ligand of CXCR4, SDF-1, is elevated in the brain, allowing a chemotactic signaling pathway for these cells to hone preferentially toward the ischemic brain site from the periphery [59,60]. The deposition of these cells in peripheral organs may also be beneficial; for example, the anti-inflammatory function of the spleen implicated as an exacerbating factor in stroke progression can be blocked by grafted cells lodging into this organ resulting in reduction of stroke-induced inflammation and neurobehavioral deficits [61,62].
Equally an important finding in this study is the safety of transplanting human menstrual blood-derived stem cells in an animal model of stroke. There is no instance of detectable tumor or ectopic formation, as well as overt graft-versus-host complications in any of the transplanted animals, despite the absence of immunosuppression in this xenograft (ie, cross species) transplantation paradigm. Long-term monitoring of graft maturation will further reveal the safety profile of menstrual blood-derived stem cells in the stroke brain. Subsequent studies should also be designed to reveal the immune status of the host especially in view of the lack of immunosuppression in this xenogeneic transplant paradigm. Gross examination of the transplanted site demonstrated no overt inflammatory response (via Nissl and H&E stains), which might have been mediated by the CNS harboring a partially immunoprivileged property, but more importantly the potential for menstrual blood-derived stem cells to exert immunosuppressive effects, as seen with bone marrow- and umbilical cord blood-derived stem cells [28,31,53]. Interestingly, menstrual blood-derived stem cells also contain the same markers that have been identified on mesenchymal stem cells known for their immunosuppressive effect such as CD29, CD44, CD73, CD90, and CD105. Our pilot studies also showed that these menstrual blood-derived stem cells in a mixed lymphocyte reaction demonstrated a very weak stimulatory response. Given the fact that the uterus lining does not reject allogeneic fetuses, it is likely that menstrual blood-derived stem cells may afford such immunosuppressive effects. Finally, graft-versus-host complications normally accompany xenografts [30,32,63], thus the absence of any overt graft rejection in the present cross-species paradigm parallels similar observations seen with human bone marrow-, amnion-, and umbilical cord blood-derived stem/progenitor cells [28,29,31,64]; the latter cells’ ability to circumvent host immune response has been suggested to be due to the immature immune system of these grafted cells or their capacity to secrete immunosuppressant factors [28,29,31,64].
The lack of tumor formation in the transplanted menstrual blood-derived stem cells is interesting in view of the observed Oct4+ labeling of majority of these cells. Whereas Oct4 has been used as a marker of pluripotent ES cells, the ability of these cells to form tumors may be affected by the immune status of the transplant recipient. The routine functional assay for determining pluripotency of Oct4-positive stem cells is to demonstrate teratoma formation in immunodeficient mice. Here, our transplant recipients comprised of nonimmunosuppressed rats. Moreover, the present subacute post-transplant period (14 days) and the diseased (stroke) state of the host brain might have contributed to the Oct4-labeled menstrual blood-derived stem cells to not fully manifest their pluripotent features. Alternatively, these cells might be expressing the Oct4 marker, but not truly exhibiting pluripotent features, including teratoma formation, thereby distinguishing them from the ES cells with robust tumorgenic potential. Indeed, other non-ES cells such as amnion-derived cells express Oct4 but appear not to form tumors in vivo [65]. This topic of Oct4 labeling of menstrual blood stem cell grafts requires a series of studies to unravel their relatively dormant proliferative status.
Compared to other sources of adult stem cells, such as bone marrow and umbilical cord blood, the menstrual blood offers much relative ease and wider window of harvesting the cells (ie, cord blood only available during birth). Such ready availability and cryopreservable features of menstrual blood-derived stem cells should allow the entry of autologous transplantation for large-scale clinical application. The present data suggest that stem/progenitor cells shed during menstruation can be retrieved from menstrual blood and that they may provide a novel autologous source of adult stem cell for neural regeneration in women. Finding the male counterpart of these stem/progenitor cells (eg, testis-derived cells) will further advance the potential for autologous transplantation. Of interest, we have shown that testis-derived Sertoli cells stand as efficacious transplantable cells for CNS disorders [66,67], although Sertoli cells are likely not stem cells. Sex-specific stem cell sources may reveal an evolutionary gender difference. Notwithstanding, the use of menstrual blood-derived stem cells for allogeneic transplantation may be an equally potential approach to cell therapy, along the line of investigations on therapeutic indications rendered by stem cells harvested from umbilical cord or bone marrow. Of note, menstrual blood-derived stem cells, referred to as endometrial regenerative cells (ERC), have been shown to be safe when transplanted intravenously and intrathecally in 4 patients with multiple sclerosis, characterized by absence of immunological reactions or treatment-associated adverse effects [68].
A small number of cell passages is the logistically preferred approach for clinical application. The present menstrual blood-derived stem cells were transferred to the study at passage 6 or 9 and cultured in the laboratory prior to infusion for an additional 3 passages, so the cells were confirmed to be at passage 9 or 12 at the time of transplantation. Additional studies are warranted to determine cell phenotypes over multiple passages, although we have observed that menstrual blood-derived stem cells continued to maintain expression of ES cell-like phenotypes up to at least 20 passages (unpublished data).
In summary, menstrual blood-derived cells recapitulate pluripotency properties, thereby providing an alternative adult stem cell source that circumvents ethical and logistical limitations of ES cells. In vitro and in vivo assessments of transplanting menstrual blood-derived stem cells reveal their efficacy and safety in stroke, and potentially an equally therapeutic cell source for treating other CNS disorders.
Footnotes
Acknowledgments and Disclosures
The authors thank the technical assistance of Ms. Eunkyung Cate Bae in the final preparation of the manuscript. C.V.B. and P.R.S. serve as consultants, and P.R.S. is a co-founder of Saneron-CCEL Therapeutics, Inc, C.V.B., P.R.S., and J.G.A. have a patent application in this area, owned jointly by Cryo-Cell International, Inc. and Saneron-CCEL Therapeutics, Inc, and USF. Cryo-Cell International, Inc. provided the foundational menstrual stem cell technology in the patent applications of M.A. Walton and J.G.A. wholly owned by Cryo-Cell International, Inc.
