Abstract
Background:
Passive humidifiers, also known as heat-and-moisture exchangers (HMEs) are used to warm and humidify inspired gases delivered during mechanical ventilation. Recent data demonstrate that the evaluation and selection of these devices is not optimal, leading to severe complications, such as endotracheal tube occlusions. The aim of the study was to assess the humidification performances of a large number of commercially available devices, with the psychrometric method, and to compare the results with manufacturer’s data.
Methods:
We assessed 113 devices using a bench test that simulated physiological ventilation conditions. Seventy-one devices were described by the manufacturers as HME without or with filtration properties (HMEF) and 42 as antimicrobial filters. Among the 71 HME/HMEF, 60% were foam-based, 32% were paper-based, 6% were fiber-based, and 1 had carbon media (HMEF with inhaled sedation function). Three hygrometric measurements using the psychrometric method for each device were performed after reaching a steady state and compared with manufacturers’ data (based on the International Organization for Standardization [ISO] method).
Results:
Among the 71 HME/HMEF tested, only 24 (34%) delivered absolute humidity above 28 mg H2O/L. Nineteen percent of the foam-based and 57% of the paper-based HME/HMEF delivered desired levels of humidity. Humidification data provided by manufacturers were available for 59 HME/HMEFs. In all but one device, humidity output was overestimated using the ISO method. The median (interquartile) difference between our measurements and the manufacturers’ data were 6.0 (3.8–8.7) mg H2O/L for devices described as HMEs (P < .001). Poor performing devices were detected only with the psychrometric method.
Conclusions:
Several HME/HMEFs performed poorly and should not be used for prolonged mechanical ventilation. The values determined by independent assessments were lower than values reported by manufacturers. Evaluation of a passive humidifier using the current ISO method does not guarantee provision of adequate humidification.
Keywords
Introduction
Invasive ventilation using tracheal intubation bypasses the normal mechanisms for heating and humidifying inspired gases. Medical gases are nearly anhydrous dictating that inspired gases must be heated and humidified. This is the standard of care. 1 The importance of humidification of inspired gases was noted at the inception of mechanical ventilation nearly 70 years ago. 2 Proper gas humidification avoids complications related to under-humidification: mucociliary dysfunction, 3 endotracheal sub-occlusions,4,5 or complete endotracheal tube occlusion (ETO), a life threatening complication, frequent during the recent COVID-19 pandemic. 6 Providing proper gas humidification is mandatory for invasively ventilated patients, with either heat-and-moisture exchangers (HMEs, without or with filtration properties [HMEF]) or heated humidifiers (HH). 1 While recent studies have demonstrated improvements in hygrometric performance with new generation of HH, 7 recent clinical reports have demonstrated high rates of ETO during the COVID-19 pandemic,8–10 as well as in other populations. 11 As a required adjunct to mechanical ventilation, humidification is often overlooked. Abrupt ETO is easy to recognize while other complications, sub-occlusions and airway mucosal dysfunction, are more frequent and difficult to diagnose. In addition to the difficulty in identifying these problems, the current ISO standard proposed in 1988 to evaluate HME humidification performance is based on the gravimetric method and appears to mischaracterize underperforming devices. 8 Several studies showed significant differences between independent measurements of the humidity delivered by the HME and manufacturers' data.8,12,13 Given the recent concerns associated with HME use during the pandemic, and the discussions on the gas humidity measurement methods, we conducted an independent, large-scale evaluation of these devices with a validated method of hygrometric measurement.
The objectives of the study were to evaluate hygrometric performances of HME, HMEF, and filters with the psychrometric method previously described8,12,13 and to compare these results with manufacturers’ data (based on the gravimetric method—ISO 9360).
QUICK LOOK
Current knowledge
Under-humidified gases may lead to severe complications including endotracheal tube occlusion (ETO), frequently reported in recent studies. There is a discrepancy between methods to evaluate passive humidifiers. Manufacturers’ evaluation of their devices are based on ISO standards and are intended to help users select efficient and safe devices. However, this measurement method may not detect poorly performing devices while independent measurements with the psychrometric method seem more selective.
What this paper contributes to our knowledge
This study demonstrated large differences between tested devices. Among devices with humidifying properties, only 34% provided safe humidity output with independent measurements while manufacturers evaluations found that 95% of the passive humidifiers to be efficient. Comparison of these data with the clinical studies reporting ETO found that only independent measurements could detect devices associated with this complication.
Methods
We tested the hygrometric performance of 71 HME/HMEFs (43 foam-based, 23 corrugated paper-based, 4 fiber-based, and 1 carbon-based) and 42 antimicrobial filters. The full list of the tested devices is provided in Supplementary Data S1. Institutional Review Board approval was not necessary because the primary data came from a bench study and the patient data came from previously published studies.
The bench test apparatus was a stable, reproducible model and used the same ventilation parameters and physiological conditions (conditioned expiratory gases, dry inspired gases, and constant ambient temperature) for assessing the HMEs and antibacterial filters. This setup was very similar to the bench test used in previous studies to evaluate HME hygrometric performance.8,12,14 The bench apparatus is shown in Supplementary Data S1. The relative humidity of expiratory gases was 100% while the temperature at the Y-piece was 32–33°C, based on previous patient data,15–18 which corresponds to an absolute humidity (AH) of 35 mg H2O/L. We generated expiratory gases with an AH of 35 ± 0.5 mg H2O/L using an HH (MR 730, Fisher & Paykel, Auckland, New Zealand, with a heated wire circuit) connected to the ventilator to simulate expired gas parameters. The humidity of simulated expiratory gases was measured with the psychrometric method (see below) until the target value was stable for two consecutive measures separated by 30 min. A Dräger V500 ventilator (Dräger Medical, Lübeck, Germany) was used in assist control mode with a rate of 20 breaths/min, a tidal volume of 500 mL, a positive expiratory pressure at 5 cm H2O, and an FIO2 of 0.21. The room temperature was kept constant between 24.5°C and 25.5°C.
The parameters of the bench test were stabilized and each device placed in the circuit exposed to the expiratory gas with absolute humidity of 35 mg H2O/L for at least 90 min. Then the hygrometry of the inspiratory gases was measured using the psychrometric method. This method has been used in previous studies conducted on a bench test or in patients.8,12,14,16,19 Three measurements for each device were recorded on 3 separate days in random sequence. We also calculated the humidity efficiency of the devices using the following formula: humidity efficiency = (inspired AH/expired AH) × 100. This provided information on the device performance. A detailed description of the bench and the measurement method is provided in Supplementary Data S2.
Hygrometric data reported by manufacturers, based on gravimetric measurements when available for HME and HMEF, were compared with the psychrometric measurements. When humidity output was reported for several tidal volumes, we report those tested for tidal volume closest to 500 mL (used on the bench study). The difference between the gravimetric measurements reported by the manufacturer and those obtained using psychrometric methods was determined.
We defined the success of a test as follows: with the results of the psychrometric method, we considered that a device passed the test when the humidity output was above 28 mg H2O/L.12,20 With the gravimetric method when the humidity output was above 30 mg H2O/L, we considered that the device passed the test. 21
In an effort to assess the risk of ETO related to the level of humidification, we conducted an extensive literature review of studies that reported the rate of ETO with HMEs evaluated with the psychrometric method in the present study.5,9,10,22–38 For the devices for which we had data, we related the humidity levels measured in this study to the ETO rates described in the literature. For the majority of the devices, hygrometric data from the manufacturers were also available. We compared the humidity provided by the devices associated with high rates of ETO and those with low rates of ETO (below 1%). We did not consider devices for which clinical evaluation was conducted in fewer than 20 subjects.39,40 In addition, studies evaluating devices that were not available for the current bench evaluation were not reported.28,41–44 We did not consider the rate of ETO with HH given the variability of the gas humidity related to external conditions with these devices, as previously described. 19 Resistances, dead space values (device internal volume), as well as bacterial and viral filtration efficiency provided by manufacturers were also recorded.
Statistical analyses
Continuous variables are represented as mean ± SD. Relationship between hygrometric performances (absolute humidity measured by psychrometry) with independent measurement and differences between independent measurement and manufacturers’ data were expressed using a univariable linear regression model. The relationship between internal volume and material of the tested HME or HMEF on humidity output suggested that a linear regression was not appropriate and that a modification of the model adding a quadratic term improved the fit. To support the linear regression assumptions, fit diagnostics were investigated. The normality assumption was verified with the Shapiro–Wilk tests using studentized residuals from the statistical model. The comparison between foam- and paper-based HMEs to deliver humidity above 25 and 28 mg H2O/L was performed using the Fisher exact test. McNemar’s test was used for the comparison between proportion of the tested devices that pass the gravimetric tests in comparison with psychrometric tests. The comparison between HMEs made of paper and HMEs made of foam was performed using a one-way ANOVA. The normality assumption was verified with the Shapiro–Wilk tests using studentized residuals from the statistical model. The Brown and Forsythe’s variation of Levene’s test statistic was used to verify the homogeneity of variances. The same statistical analysis was used to the comparison among humidity output according to the devices’ performances based on psychrometric and gravimetric measurements. Posteriori comparisons were performed using the Tukey’s technique. For the comparison between psychrometric and gravimetric measurements, a linear mixed model with one fixed factor was performed. Variability among subjects was analyzed as a random effect. The dependence among residuals of repeated measurements from the same experimental unit were estimated with an unstructured covariance association. The normality hypothesis was verified using the Shapiro–Wilk test using residuals from the statistical model and transformed by the Cholesky’s metric. The Brown and Forsythe’s variation of Levene’s test statistic was used to verify the homogeneity of variances. Statistical significance was present with a two-tailed P value < .05. Analyses were performed using SAS version 9.4 (SAS Institute Inc, Cary, NC, USA).
Results
Seventy-one devices with HME function and 42 filters were evaluated. Among the devices with an HME function, 26 were only HME, 40 were HMEF, 4 were HMEs with bypass function (to deliver aerosols), and 1 was an HME with inhaled sedation function. Among the 71 HME devices, 43 were foam-based, 23 were paper-based, 4 were fiber-based (acrylic/polypropylene), and 1 was with carbon media (Fig. 1). The mean absolute humidity measured with psychrometry in the group of devices with heat and moisture function was 25.9 ± 3.2 and 14.1 ± 4.6 mg H2O/L for the filtering devices (P < .001). Sixty-six percent of the tested HME/HMEF delivered less than 28 mg H2O/L, a threshold considered safe for patients requiring prolonged mechanical ventilation.12,20,24 As expected, none of the tested filters attained this threshold. Twenty-one devices (30%) commercialized as HMEs or HMEF had humidity output below 25 mg H2O/L, considered at high risk of complications for patients with prolonged mechanical ventilation.12,35
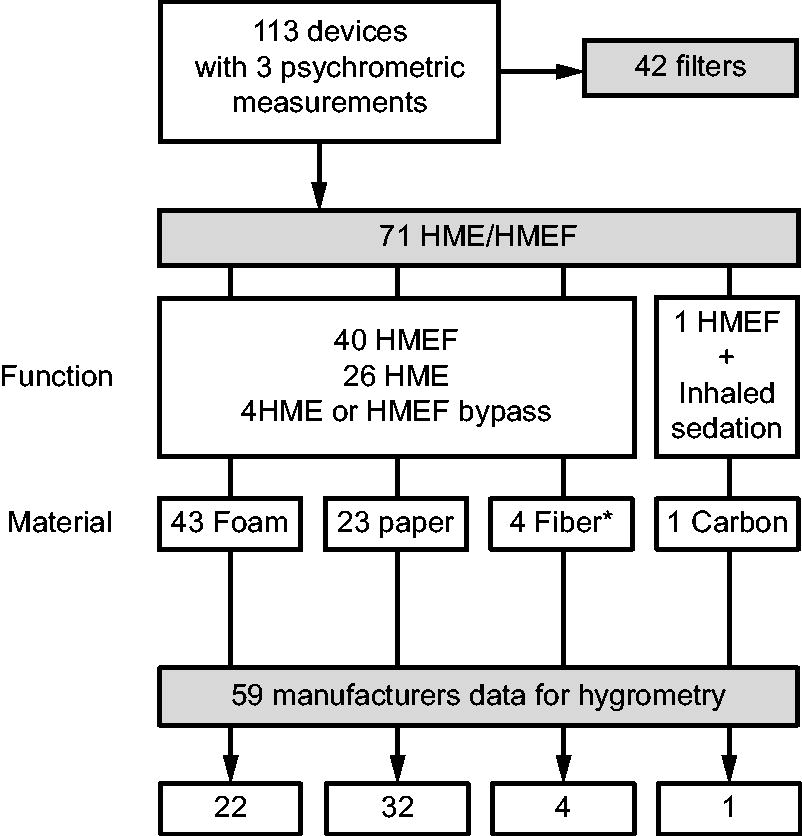
Categorization of the tested devices based on their functions and materials used for humidification properties: filters only, heat-and-moisture exchangers (HMEs), only and HMEs with filtration properties (HMEF) with different functions and material were tested. * polypropylene/acrylic fibers.
Among the 24 HME/HMEF delivering more than 28 mg H2O/L in the evaluation, 8 were foam-based, 14 were paper-based, 1 was fiber-based, and 1 was carbon-based. The proportion of foam-, paper-, fiber-, and carbon-based HME/HMEF considered performing (humidity output above 28 mg H2O/L) was 19%, 57%, 25%, and 100% (1/1) respectively. Four HME bypass (3 foam- and 1 paper-based) were evaluated, but none provided gas humidity above 28 mg H2O/L. Fifty-three devices provided more than 25 mg H2O/L, including 3 filters. The main results are presented in Figure 2 and Table 1.
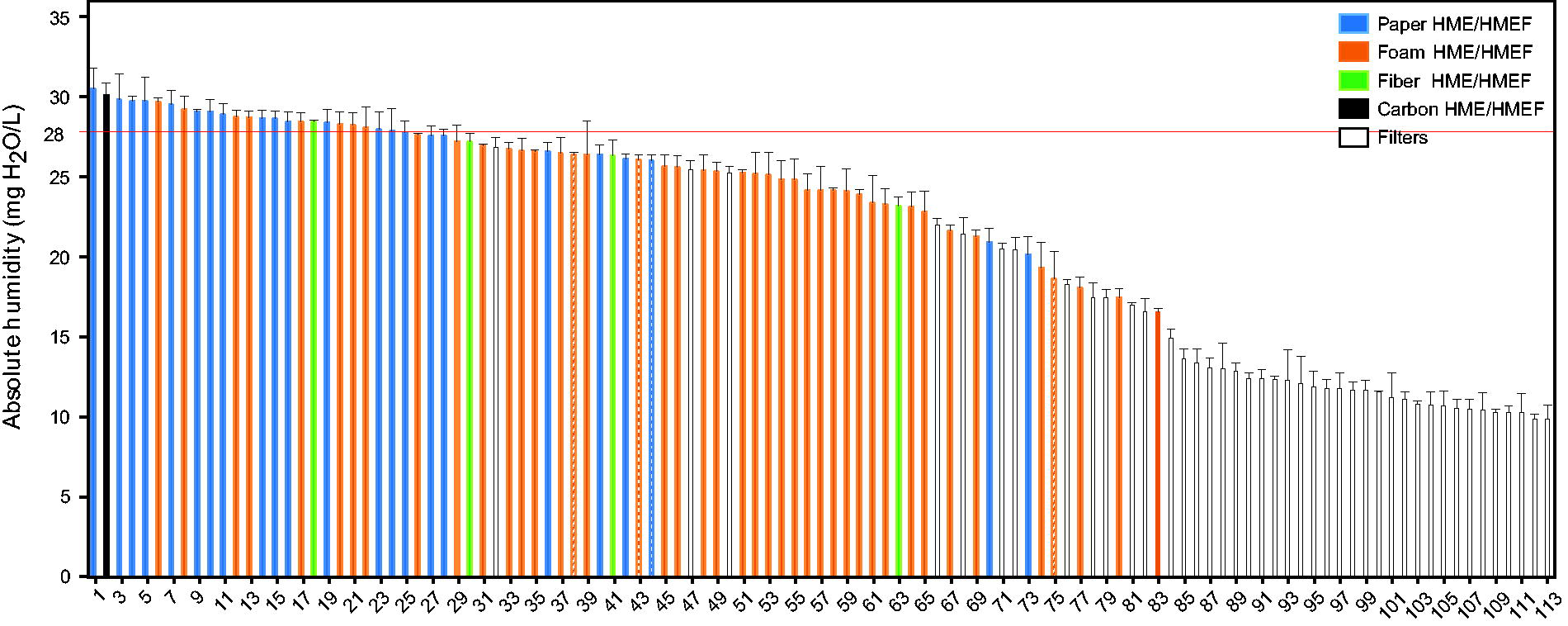
Absolute humidity (mean ± SD, in mg H2O/L) of the 113 tested devices based on the measurement with the psychrometric method from the highest to the lowest humidity delivered. Bypass HMEs are represented with hatched bars. The minimum absolute humidity considered as safe for utilization in patients with prolonged mechanical ventilation; 28 mg H2O/L is indicated with a red line.12,20
Absolute humidity delivered with tested devices that provide more than 25 mg H2O/L with independent measurements (psychrometry) (mean ± SD)
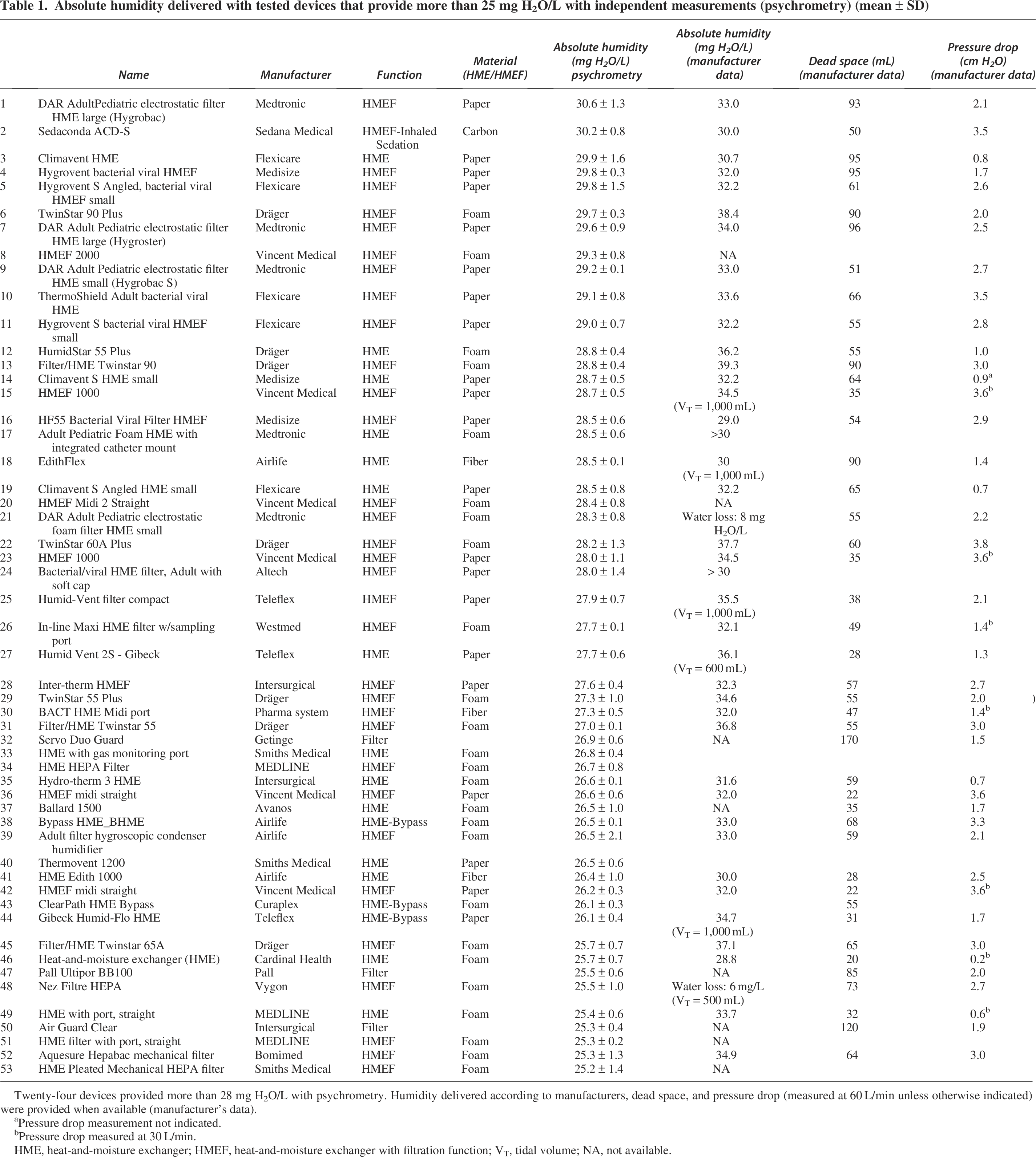
Twenty-four devices provided more than 28 mg H2O/L with psychrometry. Humidity delivered according to manufacturers, dead space, and pressure drop (measured at 60 L/min unless otherwise indicated) were provided when available (manufacturer’s data).
Pressure drop measurement not indicated.
Pressure drop measured at 30 L/min.
HME, heat-and-moisture exchanger; HMEF, heat-and-moisture exchanger with filtration function; VT, tidal volume; NA, not available.
Another way to report the hygrometric results is the humidification efficiency (humidity output/expiratory humidity). The most efficient device had an 87% efficiency and the least efficient, a filter, had an humidification efficiency of 28% (Supplementary Data S1).
Manufacturers’ data for humidity output were available for 59/71 (83%) of the HME/HMEF included in the present study. In this group of 59 HME/HMEF, the mean absolute humidity measured with psychrometry was 26.1 ± 3.3 mg H2O/L, while the absolute humidity measured by the manufacturers (based on the ISO method) was 32.6 ± 2.8 mg H2O/L (P < .001). Absolute humidity measured by the manufacturers was above the humidity measured with the psychrometric method for 58/59 (98%) tested devices. The mean differences between measured humidity and data provided by the manufacturers for HME/HMEF were 6.5 ± 3.8 mg H2O/L, with 43/59 (73%) of devices having absolute humidity differences of 4 mg H2O/L or above, 34% with differences of 8 mg H2O/L or above. The maximum difference was 17.2 mg H2O/L. The Δ between gravimetry and psychrometry was influenced by the material (Supplementary Data S1). Mean differences between psychrometric measurements and manufacturers claim for foam-, paper-, fiber-, and carbon-based HME/HMEF were 8.1 ± 3.5, 4.9 ± 3.2, 4.2 ± 2.2, and −0.17 mg H2O/L, respectively (P < .001). The difference between what is measured by the manufacturer and the performances measured by the psychrometric method was inversely proportional to the HME/HMEF absolute humidity measured by the psychrometric method (Fig. 3). Most devices passed the ISO test, while the psychrometric test was more restrictive (Supplementary Data S1). Mean psychrometric results of the HME/HMEF for each manufacturer and differences between psychrometric and gravimetric data are displayed in Supplementary Data S1. Dead space and resistances of the devices as provided by the manufacturers are displayed in the Table 1. Few devices combine proper humidification performances and low dead space (Supplementary Data S1).
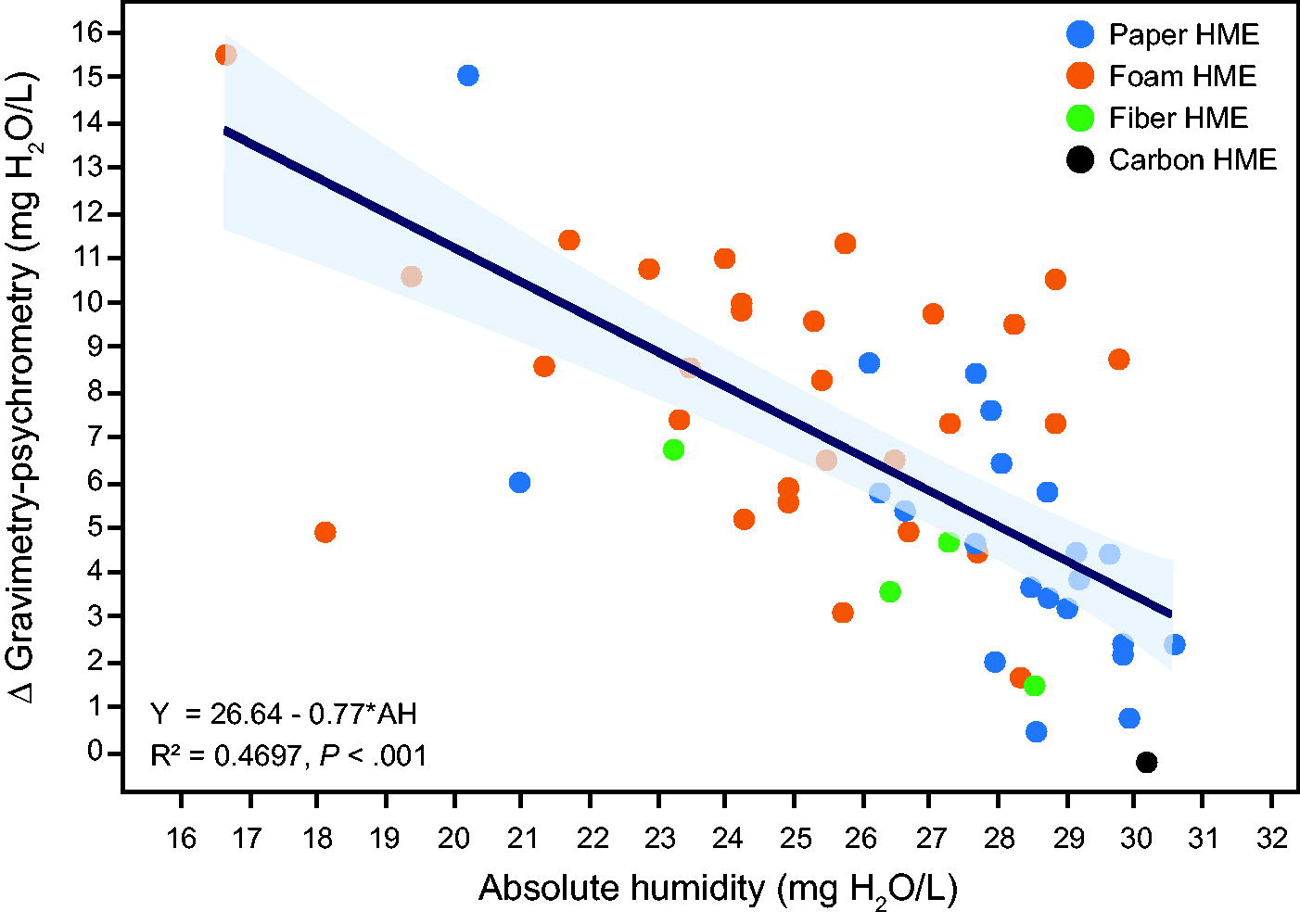
Representation of the relation between hygrometric performances (absolute humidity measured by psychrometry) with independent measurement and differences between independent measurement and performance claimed by manufacturers. The less efficient the passive humidifiers, based on the psychrometric method, the greater the difference with gravimetry. Coefficient of determination and P values are displayed.
The relation between humidity output measured in this study and the rate of ETO was available for 10 devices evaluated in 17 studies. Five passive humidifiers associated with low rates of ETO (below 1%) were considered: Hygrobac,5,22–26 Hygroster, 27 Edithflex, 28 Humid Vent Light,25,29–33 BB100.33,34 In addition, five devices associated with high rates of ETO were considered: BB2215,5,35–38 DYNJAAHME1B,9 FH603008,9 Ballard 1500,10 and Filtatherm. 10 Mean absolute humidity delivered by devices with low and high rates of ETO were 28.4 mg H2O/L and 23.0 mg H2O/L, respectively (P < .001) when measured with psychrometry. According to the manufacturer’s data, humidity delivered was 34.3 mg H2O/L and 30.9 mg H2O/L, respectively (Supplementary Data S1).
Full results including manufacturer’s data are available in a separate online Supplementary Data S2.
Discussion
The choice of an HME/HMEF should primarily be based on humidification properties (first criteria), internal volume, and resistance. The recent pandemic has been a reminder of the necessity to provide adequate humidity to avoid severe complications.8–10 Only 34% of the tested HME/HMEF in this study provided sufficient gas humidity output while almost all tested HME/HMEF passed the ISO standards as reported by the manufacturer. Most of foam-based HME/HMEF did not perform well under independent evaluations. Data provided by manufacturers obtained with the gravimetric method (ISO standard) systematically overestimated humidity output and did not detect passive humidifiers at risk for complications related to under-humidification. The difference between manufacturers’ data and our independent measurements has more than doubled compared with another study published 15 years ago. 12 The current ISO 9360 standard for measuring the performance of passive humidifiers gives overestimated and misleading results and does not guarantee patient safety.
The main finding of this study was a striking heterogeneity of the humidification performance of the devices proposed for passive humidification during mechanical ventilation. Only 1 of 3 HME/HMEF reached the minimum threshold of humidity of 28 mg H2O/L that was considered safe by Branson and Davis 20 and by another group of experts. 12 Almost 1 of 3 of the tested devices commercialized for passive humidification provide humidity below 25 mg H2O/L, which put patients at risk of ETO, especially in the case of prolonged mechanical ventilation.5,9,10,35–38 These data show a deterioration of the situation compared with an evaluation of 48 devices published in 2009. 12 In the 2009 study using the same methods of measurement, 42% of the tested devices provided humidity above 28 mg H2O/L.12
We found, in the present study, that the material used for moisture retention had an important impact on the performance, which has not been reported previously. Only 8/43 (19%) of the foam-based HME/HMEF, 1/4 (25%) of the fiber-based HME/HMEF, and 13/23 (57%) of the paper-based HME/HMEF provided humidity above 28 mg H2O/L (P < .001). The only carbon-based HME/HMEF tested in this study performed well in terms of humidity output. This is line with the absence of ETO described with this inhaled sedation and HMEF device in the literature.45,46 This is the first study, to our knowledge that demonstrates a clear impact of the material used for hygrometric properties of these devices. It was previously shown that hygroscopic material was superior to hydrophobic devices and several materials have already been tested.47–49 However, this study allows an evaluation of a large number of devices with different materials. While foam-based devices seem more frequently used, with lower production costs, only a minority of these devices provide sufficient humidification when utilized for prolonged mechanical ventilation. It is noteworthy that very high rates of ETO were described during the pandemic with foam-based HME/HMEF.9,10 However, the material alone is not enough to determine the HME/HMEF performance and other parameters (eg, membrane surface area, added hygroscopic salts, foam or paper quality, volume of humidification material) may have an impact but were not evaluated here. The humidity output for several comparable passive humidifiers is slightly lower (1–2 mg H2O/L less) in the present study compared with a study published in 2009. 12 These results may be explained by slightly lower expiratory humidity; however, the same range of expiratory humidity was targeted (35 ± 0.5 mg H2O/L), with the same method of measurement (psychrometry), same ambient temperature (25°C), and same ventilator settings with intensive care unit ventilators using dry gases in both studies. It is also possible that different qualities of the corrugated papers, foam, and different concentrations of hygroscopic salts have been used over the years to reduce the production costs. However, this cannot be evaluated as these data are not reported by the manufacturers.
Measurement of gas humidity is a difficult task and there is no agreement on the best method to use. 50 However, Chatburn and Primiano published “A rational basis for humidity therapy,” 51 mainly based on physiological measurements obtained with the psychrometric method52,53 or mass spectrometry. 17 While there are few data on expiratory humidity in intubated patients,15–17 none of these studies support the utilization of more than 35 mg H2O/L for simulated expiratory humidity. With the current ISO standard, the expiratory humidity is set at 44 mg H2O/L, which partly explains the differences between manufacturers and our data. Interestingly, 22% of the tested passive humidifiers provide more than 35 mg H2O/L according to manufacturers, above physiologic expiratory humidity, which is nonsensical. Setting simulated expiratory humidity too high with the ISO standard may explain these differences, given that inspiratory humidity delivered with passive humidifiers is directly correlated with expiratory humidity. 16 However, there are other measurement errors with gravimetry used by ISO standards, related to the gas volume and weight measurements that were described and may also explain overestimation of hygrometric measurements.20,47,54
The gravimetric method has been used as the standard method for ISO certification since 1988. This method was validated by independent university teams20,47 but sources of error were described. In previous studies, a discrepancy between the gravimetric and psychrometric measurements were reported.12,13 It must be noted that there has been a progressive drift of the differences between independent and manufacturers measurements. In the 2009 study evaluating 48 passive humidifiers, the mean difference between manufacturers and psychrometric measurements was 3.0 ± 2.7 mg H2O/L with a maximum difference of 8.9 mg H2O/L.12 In the present study, the mean difference has more than doubled (6.5 ± 3.8 mg H2O/L), and the maximum difference between manufacturers and psychrometric method has almost doubled (17.2 mg H2O/L). Large differences (4 mg H2O/L or above) are present in 43/59 (73%) of devices in the present study, which also doubled. 12 Almost all tested HME/HMEF devices that are proposed to humidify gases passed the ISO test (to provide at least 30 mg H2O/L of humidity output as recommended 21 ), including 83% of those considered with very low humidity output (below 25 mg H2O/L with the psychrometric method). In opposition, only 34% of the tested HME/HMEF passed the psychrometric test (to provide at least 28 mg H2O/L12). The difference between the techniques of measurements and the impossibility to detect poor performing devices with the ISO standard is concerning for patient safety and is in line with the high rates of ETO reported during the COVID-19 pandemic.8–10 This life-threatening complication, however, is only the visible tip of the iceberg, as endotracheal tube sub-occlusions responsible for increased tube resistance4,55,56 are much more frequent and the mucociliary dysfunction, associated with low humidity output, is difficult to quantify in patients. 3 These silent complications may increase the duration of mechanical ventilation and worsen outcomes of critically ill patients. It is time to face the problem head on and provide ourselves with the means to detect faulty devices.
ISO standards should therefore be more restrictive and rely on methods allowing the detection of poorly performing devices, the comparison of active humidification and passive humidification. ISO standards or regulatory agencies should also limit the authorizations to use poor performing passive humidifiers in patients requiring prolonged invasive mechanical ventilation. While there are more than 20 HMEs that provide humidity output > 28 mg H2O/L, which is deemed a safe level of humidity, devices with lower humidity output should not be authorized for utilization in these populations. However, devices with moderate humidity (between 25 to 28 mg H2O/L) may be used for short-term mechanical ventilation (eg, postoperative patients) and utilization of devices that deliver even lower humidity may be discussed in the operating room where the situation is really different (duration of invasive ventilation of a few hours, recycling of heat and humidity in anesthesia circuits). 18 In the operating room, the reaction of exhaled carbon dioxide with the carbon dioxide absorber (eg, soda lime) produces water vapor and contributes to humidification in the circuit. Kleemann proposed a minimum level of 20 mg H2O/L for gas humidity during anesthesia. 18
This study has limitations. First, this is a bench study and the simulated expiratory humidity refers to the physiological humidity of normothermic patients. Therefore, humidification performance does not reflect the humidity output in hypothermic patients, which is much lower, 16 and passive humidifiers are contraindicated in these patients. In addition, due to the large number of devices tested, it was not possible to evaluate several conditions that may have an impact on passive humidifiers performances, including different tidal volumes and minute ventilations, 57 as well as different durations of use. 58 Furthermore, due to the limited number of large studies evaluating the rate of ETO with different passive humidifiers, it was not possible to correlate humidification performance and this complication for all HME/HMEFs. It must be acknowledged that this complication is multifactorial, however, the humidity delivered and the duration to the exposure to the gas are the main factors of complications associated with under-humidification.3,8,12
Conclusions
This study demonstrated highly heterogeneous performance of passive humidifiers used to humidify inspiratory gases delivered to patients requiring invasive mechanical ventilation, with an obvious impact of the hygroscopic material used for humidification properties. Only a small proportion of these devices sold for this purpose were effective. Current ISO standards have important limitations, lead to systematic overestimation of the humidification performances, and do not make it possible to identify devices that deliver insufficient humidity. The overestimation of humidification performance with the ISO method does not constitute an incentive for manufacturers to improve this technology and does not protect patients. These ISO standards should be changed to protect the mechanically ventilated patients in hospitals. There is no mechanism that prevents the use of inefficient humidification systems, which seems inconsistent with regulatory goals. This situation exists because of the difficulty of recognizing this clinical problem, the vagueness in current recommendations, and the knowledge gaps in the protection of patients.
Footnotes
Acknowledgments
The authors thank Dr. Sahar Ahmad (Stony Brook University Hospital, Stony Brook, NY USA), Dr. Eduardo Mireles-Cabodevila, M. Scott Neuhofs, and Dr. Samuel Wiles (Cleveland Clinic, Cleveland, OH, USA) who provided the HMEs used at their centers to conduct bench tests. The authors thank Dr. Tsuneaki Kenzaka (Kobe, Japan) and Dr. Rohan K. Panchamia (New York, NY, USA) for their help to determine the reference of the HMEs used at their center. The authors thank Serge Simard, MSc, senior statistician, for the complete review of the analysis.
Author Disclosure Statement
F.L.: Research funding from different companies working in the field of humidification to evaluate HH/HME (Fisher & Paykel, Vincent Medical, Dräger, Sedana). Development of the VentilO Application (implementation and optimization of protective ventilation—dead space), partly financed by Fisher & Paykel. Consultancy Medtronic: Series of presentations on humidification during respiratory support (Medtronic medical education program). None of the companies whose devices were evaluated participated in the development, measurements, or analysis of the study. R.B. discloses a consulting relationship with Lung Pacer LLC and is the Editor-in-Chief of R
Funding Information
The study was not funded. All the devices were obtained from different hospitals in Canada and United States except for the devices from Dräger Medical, Vincent Medical, Sedana, and Pneumocare supplied by the companies.
