Abstract
Background:
In pediatric patients, the intracuff pressure of endotracheal tubes should be as low as possible to prevent injury to the tracheal mucosal wall. The conventional stethoscope-guided technique relies solely on the operator’s sensitivity of audible detection, which may lead to increased intracuff pressure. This study was conducted to compare the flow-volume loop guided technique for endotracheal tube cuff inflation with the stethoscope-guided technique and to determine whether the flow-volume loop guided technique results in lower and more consistent intracuff pressure.
Methods:
The participants were randomized to undergo either the flow-volume loop guided or the stethoscope-guided cuff inflation technique. In the flow-volume loop guided group, the cuff was inflated until the flow-volume loop was completely sealed. In the stethoscope-guided group, the cuff was inflated until the leakage was not audible. Cuff inflation was performed twice with incremental volumes of 0.5 mL and 0.2 mL to determine the consistency of the methods. The primary outcome was the intracuff pressure, and the secondary outcome was the incidence of postextubation complications.
Results:
Eighty participants (4 to 12 years old) were included in this study. The use of the flow-volume loop guided technique was associated with a lower cuff pressure versus use of the stethoscope-guided technique when inflated at the 0.5-mL increment [14 (6–18) cm H2O vs 19 (9–24) cm H2O; P < .001] and at the 0.2-mL increment [14 (6–18) cm H2O vs 18 (9–24) cm H2O; P < .001], with better consistency between the measured cuff pressures (z = –2.299; P = .02). The presence of postextubation complications (6/80) was not significantly different between the 2 groups but was associated with the American Society of Anesthesiologists physical status (P < .001).
Conclusions:
The flow-volume loop guided technique for endotracheal tube cuff inflation is a more objective technique that effectively seals the airway with the lower cuff pressure to allow for mechanical ventilation in pediatric subjects during anesthesia.
Introduction
The use of cuffed endotracheal tubes for general anesthesia in pediatric patients <8 years of age has been increasingly accepted for several reasons, including better anatomic studies of the larynx, 1 more suitable materials and improved designs of cuffed endotracheal tubes, 2 and the lack of differences in postextubation cuff-related complications between subjects in whom cuffed and those in whom uncuffed endotracheal tubes were used. 3 However, ensuring a safe cuff pressure is important when using cuffed endotracheal tubes, especially in young children. According to a study of adult subjects, the intracuff pressure should be monitored and maintained at <25 cm H2O because tracheal mucosal capillary pressure has been found to range between 25 and 30 cm H2O. 4 However, the tracheal mucosal capillary pressure has not been studied in pediatric subjects; therefore, the adult tracheal mucosal capillary pressure is typically used.
Logically, the tracheal mucosal capillary pressure in children should be lower than that in adults. Practically, the cuff pressure must be low enough to prevent injury to the tracheal mucosal wall and high enough to seal the respiratory circuit. Because the best practice for determining the intracuff pressure for pediatric patients has not yet been established, measuring intracuff pressure should be a routine practice in pediatric anesthesia despite the fact that a manometer may not be available in the resource-limited operating room. However, measurement of the intracuff pressure is not universally implemented due to many inconveniences, including the direct measurement of cuff pressure with a manometer, which may interrupt the surgical field during general anesthesia, and the necessity of multitasking. Therefore, a simple and reliable method for achieving a cuff seal without measuring the intracuff pressure every time to prevent overinflation is needed.
Currently, the conventional methods for inflating the balloon of an endotracheal tube cuff to achieve the sealing are manual palpation and the stethoscope-guided technique. These 2 methods rely on the sensitivity of tactile and audible sensations of the operators, in which the intracuff pressure in some patients may reach a harmful level, especially when the operator does not have precise tactile and audible sensations. A pressure-volume loop guided method, named the objective cuff inflation method, has recently been proposed as an alternative. Lower intracuff pressure with efficient sealing has been obtained when using this technique in adult subjects. 5 However, this method has not been tested in pediatric patients.
Pediatric patients require a more sensitive monitoring method to achieve precise inflation of the cuff. The flow responds faster to triggers than to the pressure; thus, the flow is a more sensitive parameter that should be monitored. Therefore, this study was conducted to compare the flow-volume loop guided endotracheal tube cuff inflation technique and the stethoscope-guided technique in pediatric subjects. The flow-volume loop guided technique was developed based on the idea that, after endotracheal intubation, the only leakage in the breathing circuit should be within the area around the epiglottis, which can be sealed via cuff pressure. Therefore, the flow-volume loop guided method may be useful only in patients who are unlikely to have any respiratory leakage in the breathing circuit other than around the cuff. This study was performed to compare the endotracheal cuff pressure achieved in pediatric subjects when using the flow-volume loop guided technique to detect respiratory circuit closure to that achieved when using the stethoscope-guided technique to detect an audible air leakage. It was hypothesized that the less subjective flow-volume loop guided technique should result in a lower and more consistent intracuff pressure.
QUICK LOOK
Current knowledge
Evidence supports the safety of using cuffed endotracheal tubes in pediatric patients. The manual palpation method and the stethoscope-guided technique have commonly been used to inflate the cuff to achieve sealing pressure at 20 cm H2O. These methods depend solely on the audible and tactile sensitivity of the operators. An alternative objective cuff inflation method in pediatrics thus remains to be investigated.
What this paper contributes to our knowledge
In children, it is safer to keep the lowest possible cuff pressure because there is no evidence with regard to the most appropriate capillary perfusion pressure in children’s tracheas. The present study shows that the flow-volume loop guided cuff inflation technique provided lower and more consistent cuff pressure with proper sealing compared with the stethoscope-guided method.
Methods
Study design and participants
This prospective randomized controlled trial was conducted in the Department of Anesthesiology, Faculty of Medicine Ramathibodi Hospital, Mahidol University, Thailand, from September 1, 2018, to March 1, 2019, approved by the institutional review board of Mahidol University and registered at http://www.clinicaltrials.gov (NCT03902756). Written informed consent was provided by the parents of participants only <7 years old, and, for those ≥7 years old, consent was obtained from both the participants and their parents. School-age participants who were scheduled to undergo surgery under general anesthesia with an endotracheal tube were recruited to participate in this study. Patients who were predicted to have a difficult intubation, airway abnormalities, underlying respiratory diseases, a risk of pulmonary aspiration, or a recent respiratory tract infection (within 2 weeks of the surgery) were excluded from this study to prevent possible complications and to ensure participant safety. These patients are eligible for inclusion in future studies after the proposed method is verified as safe and appropriate.
Enrollment and randomization
The enrolled participants were randomized to one of 2 groups at a 1:1 ratio by using the sealed-envelope randomization method (the envelopes were prepared by statisticians). The flow-volume loop guided technique was used to inflate the endotracheal tube cuff in the participants in the flow-volume loop guided group. The participants in the stethoscope-guided group underwent a stethoscope-guided technique to check for audible leaks while the endotracheal tube cuff was being inflated (Fig. 1).
Flow chart.
Anesthesia
All the participants underwent anesthetic induction with thiopental (6 mg/kg), fentanyl (1 μg/kg), and atracurium (0.6 mg/kg). The appropriate size of the cuffed endotracheal tube (Shiley Oral/Nasal Tracheal Tube Cuff; Covidien, Dublin, Ireland) was determined by using the following formula: cuffed endotracheal tube size = (age in years/4) + 3.5. Anesthesia was maintained by using sevoflurane at a minimum alveolar concentration of one in an oxygen/air mixture. No nitrous oxide was used during the procedure. At the end of the procedure, after reversal of the effect of residual muscle relaxants with intravenous neostigmine (0.05 mg/kg) and atropine (0.02 mg/kg), the participants were extubated while fully awake.
Study intervention
Cuff inflation was performed after confirming endotracheal intubation via the standard monitoring of the end-tidal carbon dioxide in both groups. The presence of continuous waveform capnography observed on the anesthesia machine (Avance CS2; GE Healthcare, Milwaukee, Wisconsin) was used to confirm the end-tidal carbon dioxide pressure and proper endotracheal tube placement.
Cuff inflation in the flow-volume loop guided group
After insertion of the endotracheal tube into the trachea, the tube was connected to the breathing circuit. Pressure-controlled volume-guaranteed ventilation was then initiated at a tidal volume of 8 mL/kg, and the breathing frequency was adjusted to attain an end-tidal carbon dioxide pressure of 30–35 mm Hg. The flow-volume loop was observed on the screen monitor of the closed-circuit anesthesia machine (Avance CS2) as illustrated in Figure 2. The endotracheal tube cuff was inflated in 0.5-mL increments. If the flow-volume loop was not closed (Fig. 2A), then incremental inflation was continued until complete closure was achieved (Fig. 2B); air was injected into the endotracheal tube cuff until the expiratory limb reached a volume of zero and met the starting point of the inspiratory limb. The first endotracheal tube cuff pressure (P1) was then measured. Subsequently, the air was aspirated in 0.2-mL increments until the loop opened again (a leak developed), after which the cuff was reinflated in 0.2-mL increments until the flow-volume loop was closed again. The second cuff pressure (P2) was then measured.
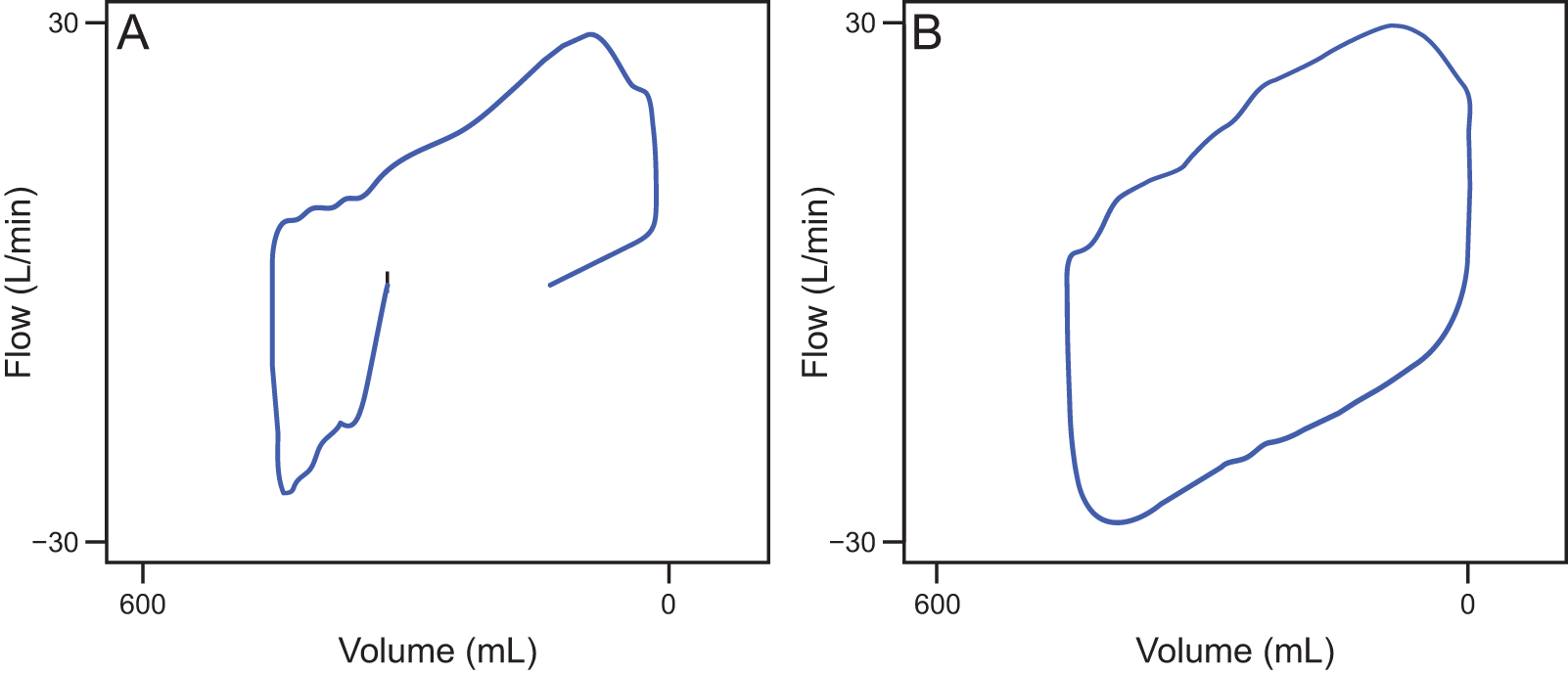
Flow-volume loop.
Cuff inflation in the stethoscope-guided group
Cuff inflation in the stethoscope-guided group was performed similarly to the technique commonly used by pediatric anesthesiologists. Pressure-controlled volume-guaranteed ventilation was then initiated at a tidal volume of 8 mL/kg, and the breathing frequency was adjusted to attain an end-tidal carbon dioxide pressure of 30–35 mm Hg. The endotracheal tube cuff was inflated in 0.5-mL increments until the disappearance of any audible air leak via the auscultation with a Littmann Classic II Pediatric Stethoscope (3M, Maplewood, Minnesota) placed at the right lamina of the thyroid cartilage. The first P1 was then measured. Subsequently, the air was aspirated in 0.2-mL increments until an audible air leak was heard, after which the cuff was reinflated at 0.2-mL increments until the air leak was no longer audible via auscultation. Then, the P2 was measured.
Endotracheal tube cuff pressure measurement
Endotracheal tube cuff pressure was assessed by a second investigator (SP) who was blinded to the method of cuff inflation by using a Portex (Smiths Medical, Minneapolis, Minnesota) endotracheal tube cuff pressure manometer. The measurements were labeled P1 and P2 in both groups. If the participant’s head and neck were repositioned during the operation, then cuff inflation was repeated by using air increments of 0.2 mL according to the assigned method for each participant, and the last value was used in the analysis rather than the P2.
Outcomes
The primary outcome in this study was the intracuff pressure. The secondary outcome was the presence of postoperative respiratory complications related to excessive cuff pressure, including sore throat, hoarseness, pulmonary aspiration, and postextubation laryngeal edema.
Statistical analysis
Based on a previous similar study 5 conducted in adults, in which the mean ± SD difference in the intracuff pressure between the 2 groups was 14.75 ± 7 cm H2O, a sample size of 40 participants per group was estimated by using a type-1 error of 0.05 and a power of 0.8. All statistical analyses were performed by using SPSS version 29.0 (SPSS, Chicago, Illinois). Normally distributed data (determined by using Shapiro-Wilk test) are presented as mean ± SD. Data without normal distribution are presented as median (minimum-maximum range). The Mann-Whitney U test was used to compare the independent samples, and the Wilcoxon signed-rank test was used to compare the repeated-measures samples. Categorical data are presented as number and percentages. The Pearson chi-square test was used to compare categorical data and to assess the associations. Statistical significance was set at P < .05.
Results
Subject characteristics
Eighty participants (ages 4 to 12 years) were included in the study (Fig. 1). There were no differences in participant sex, age, weight, height, body mass index, American Society of Anesthesiologists physical status, or operation time between the study groups. The number of ear, nose and throat procedures was different between the groups (flow-volume loop guided group [0/40] vs stethoscope-guided group [4/40]; P = .041) (Table 1). No participant developed tube leakage during the operation. The head or neck was not repositioned during the operation.
Subject characteristics
Data are presented as median (interquartile range) unless otherwise noted.
ASA, American Society of Anesthesiologists.
Primary outcome
The flow-volume loop guided group had significantly lower intracuff pressures than those in the stethoscope-guided group, (P1: U = 1,321, P < .001; P2: U = 1,300, P < .001) (Fig. 3). The P1 was 14 (6–18) cm H2O in the flow-volume loop guided group and 19 (9–24) cm H2O in the stethoscope-guided group. The P2 was 14 (6–18) cm H2O in the flow-volume loop guided group and 18 (9–24) cm H2O in the stethoscope-guided group. In the flow-volume loop guided group, the P1 and P2 were not significantly different (z = –1.724, P = .09), although they were significantly different in the stethoscope-guided group (z = –2.299, P = .02). This result indicated the consistency of inflating the cuff pressure in the flow-volume loop guided group was better than in the stethoscope-guided group (Fig. 4).
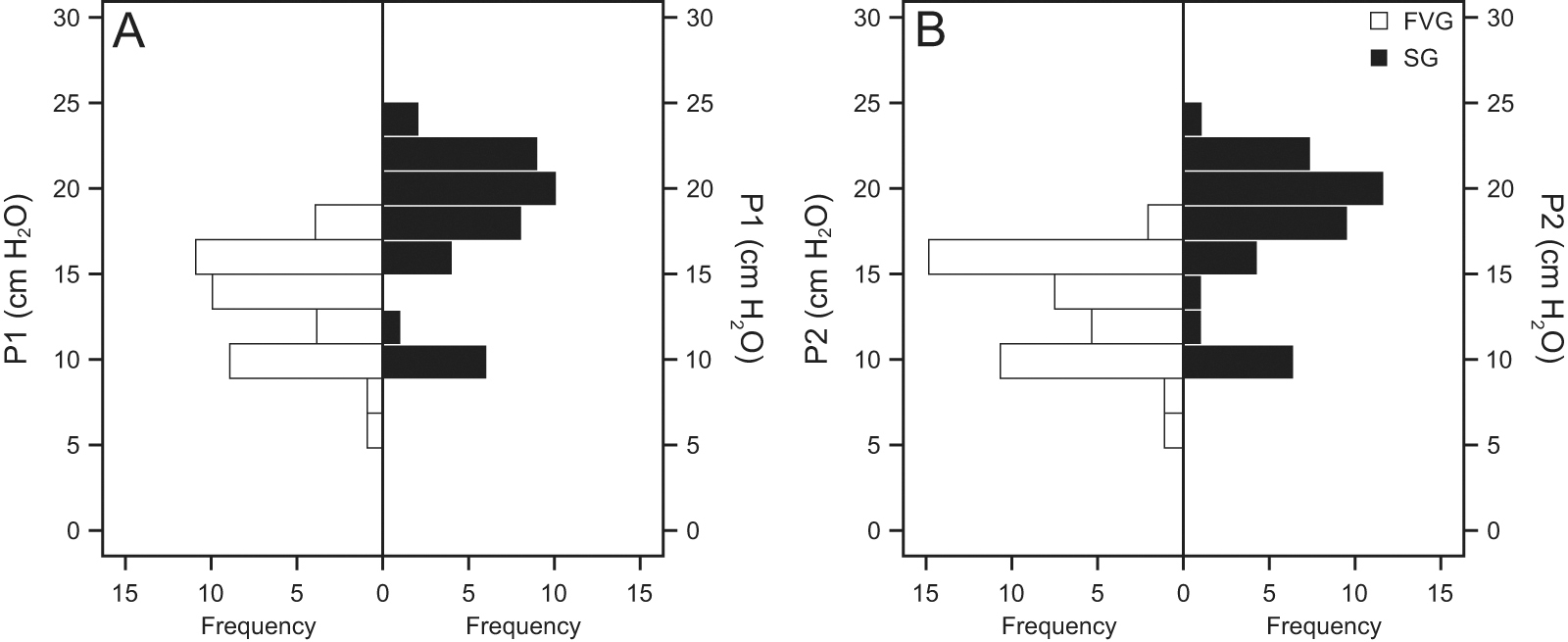
Intracuff pressures comparison. Intracuff pressures in the flow-volume loop guided (FVG) and stethoscope-guided (SG) groups for
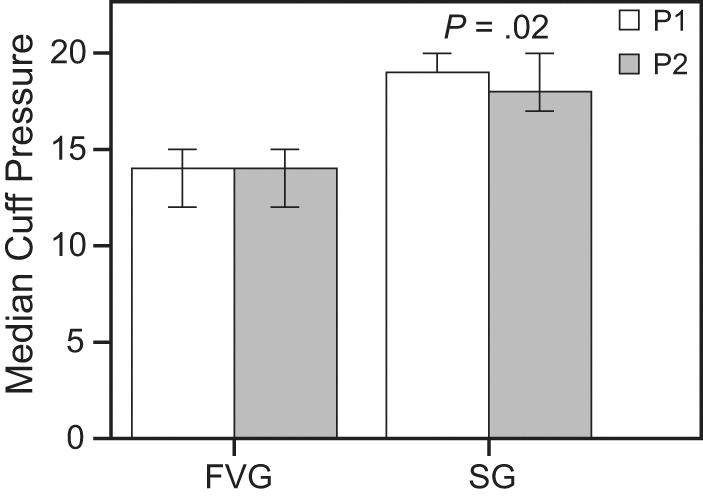
Consistency of the cuff pressure inflation. The consistency of adjusting the cuff pressure in the flow-volume loop guided (FVG) group is better than that in the stethoscope-guided (SG) group. No significant difference was detected between the first and second cuff pressures in the FVG group (z = –1.724; P = .09), although the cuff pressures are different in the SG group (z = –2.299; P = .02).
Secondary outcome
The incidences of sore throat was higher in the stethoscope-guided group versus the flow-volume loop guided group (12.5% vs 5%) but, although the difference was not significant (Pearson chi-square test = 0.721, P = .40). The incidence of hoarseness was equal between the 2 groups (2.5% in both groups; P > .99). Other postextubation cuff-related complications, including stridor, pulmonary aspiration, and postextubation laryngeal edema, were not observed (Table 2). No participants developed cuff leakage problems during general anesthesia, and no cases of pulmonary aspiration were observed. The presence of postextubation complications was not associated with an operation time >100 min (Pearson chi-square test = 0.430; P = .51) or a P2 of >20 cm H2O (Pearson chi-square test = 3.204; P = .07). However, postextubation complications were associated with preanesthesia medical comorbidities according to the American Society of Anesthesiologists physical status (Pearson chi-square test = 16.892; P < .001) (Fig. 5).
Postextubation complications
Data are presented as n (%).
NA, not applicable.
Discussion
The use of cuffed endotracheal tubes for pediatric patients who are receiving general anesthesia has recently increased. 6 –8 Several studies have shown no difference in postextubation cuff-related complications between subjects who were intubated either with cuffed or uncuffed endotracheal tubes. 3 , 9 –16 However, when using cuffed endotracheal tubes, the lowest possible cuff pressure must be used, especially in young children, to ensure that no tissue injury occurs in the endotracheal tract. A lower cuff pressure results in less pressure and less injury to the tracheal wall, which is beneficial during lengthy operations, in patients who tend to have a lower mean arterial pressure (such as neonatal patients), or in children with hemodynamic compromise. Although the evidence of tracheal mucosal capillary pressure is still not well established, maintaining a low cuff pressure while the respiratory circuit is sealed is necessary to ensure safety.
Currently, conventional methods such as palpation or stethoscope-guided inflation may not allow for the maintenance of a low cuff pressure. Studies in adult subjects have reported that the palpation technique results in an endotracheal tube cuff pressure >25–30 cm H2O, even when performed by experienced clinicians. 17 –20 A cuff pressure of 20 cm H2O is acceptable when the stethoscope-guided technique is used, as reported in another study. 21 However, the conventional stethoscope-guided inflation method may not be sensitive enough to prevent overinflation because the ability to detect air leakage depends on the operator’s hearing. Machine sensors are likely able to detect respiratory circuit closure better than humans.
The flow-volume loop guided technique uses a flow-volume loop from the anesthesia machine to indicate appropriate cuff inflation. In the current study, the median cuff pressure in the flow-volume loop guided group was significantly lower than that in the stethoscope-guided group at both air incremental volumes (0.5 mL in P1 and 0.2 mL in P2) with a maximum cuff pressure <20 cm H2O. Therefore, the proposed flow-volume loop guided technique may provide an effective method to seal the airway with a lower intracuff pressure. The intracuff pressure for surgical ear, nose, and throat patients may be higher in some cases. However, the significantly greater number of ear, nose, and throat surgical subjects in the stethoscope-guided group than in the flow-volume loop guided group did not affect the findings in the current study because the intracuff pressure of these 4 subjects in the stethoscope-guided group consisted of both low pressure ([P1,P2]; [10,10] and [10,10]) and high pressure ([P1,P2]; [20,19], [21,20]), which were not extreme values.
The flow-volume loop guided technique also provided better consistency of cuff inflation at inflation increments of 0.5 mL and 0.2 mL. An incremental volume of 0.5 mL was used based on the syringe scale to accelerate the airtight seal at the beginning on inflation. A 0.2-mL incremental volume, selected based on a previous study, 22 was used to achieve precise inflation after fast sealing. This finding may be due to the consistency of the flow sensors in the anesthesia machine as opposed to human sensation. Such consistency could make the use of the flow-volume loop guided technique a practical method for intracuff inflation, especially for intra-operative anesthesia.
In the current study, postextubation complications were associated with the American Society of Anesthesiologists classification rather than with the inflation method, cuff pressure, or operation time. Few cases of postextubation complications were observed, which may be due to the proper selection of the tube that could lead to a few cuff-related postoperative complications because the cuff pressure also depends on the materials. In this study, single-tube material (Shiley) was used based on a study by Dullenkopf et al, 23 in which the cuff pressure range in the nonpolyurethane Shiley cuffed endotracheal tube was within a safe range.
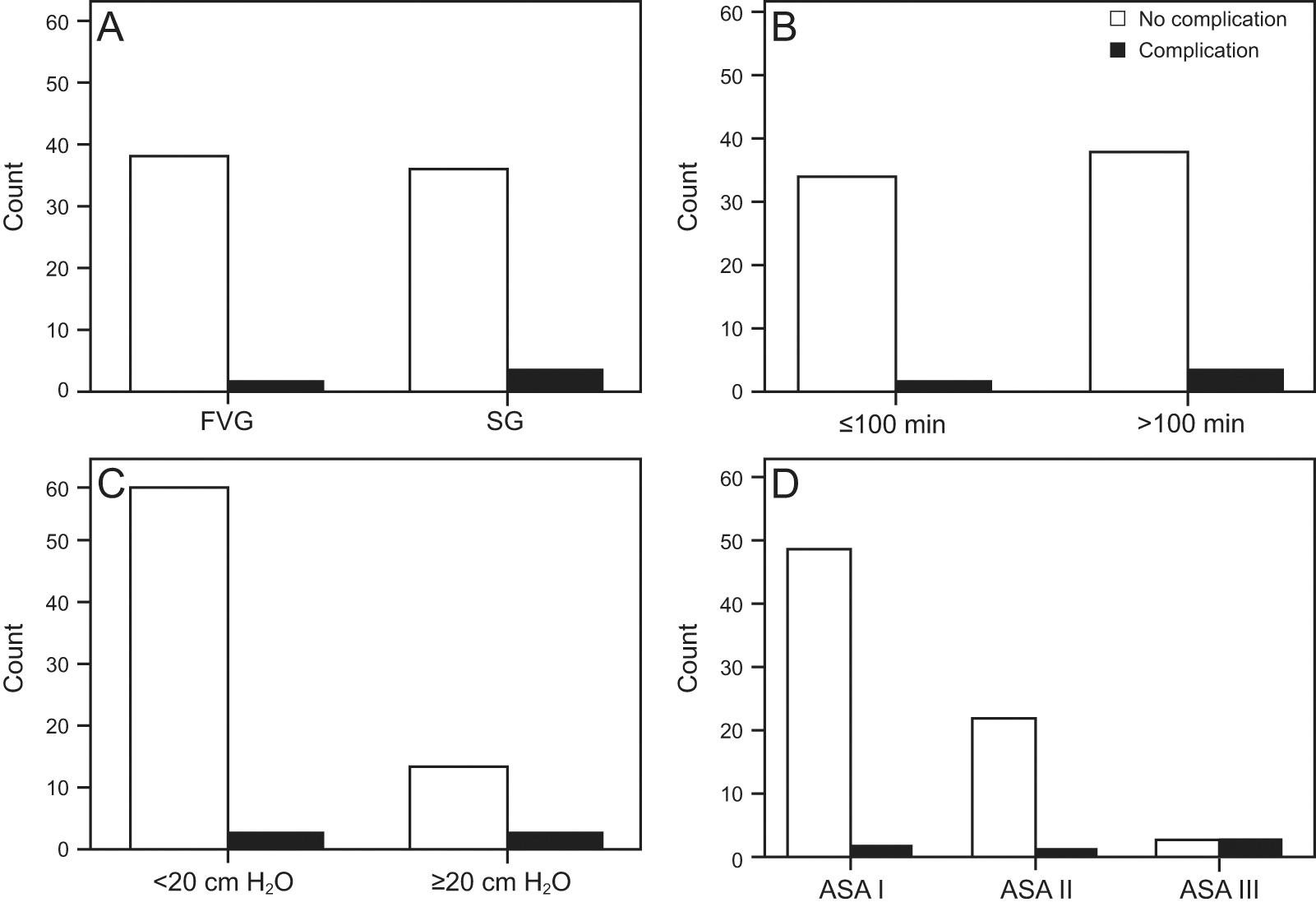
Association of the postextubation complications. The presence of postextubation cuff-related complications was not associated with
Therefore, the proposed flow-volume loop guided method is a practical technique for several reasons. Currently, the modern anesthesia machine includes a respiratory graphic monitor on the screen; therefore, it is easier to implement the flow-volume loop guided technique in routine practice. The flow-volume loop can also be used as a continuous monitor for earlier detection of air leakage in the breathing circuit, especially around the endotracheal tube after the patient’s position has been changed during an operation. Readjustment of the cuff seal after repositioning guided by the flow-volume loop can be performed to ensure the appropriate intracuff pressure. Krishna et al 24 and Ganigara and Ramavakoda 25 used the transducer of an invasive pressure monitoring device to measure the cuff pressure at the time of inflation and after inflation. The flow-volume loop guided technique can be combined with the method described by Krishna et al 24 to provide continuous monitoring of the intracuff pressure and air leakage around the cuff while the tracheal tube is retained.
However, the flow-volume loop guided technique may be impractical in some cases. Because the leakage at any position in the breathing circuit can result in a flow-volume loop pattern similar to the cuff leakage pattern, the use of the flow-volume loop guided technique may not be appropriate for patients with leakage anywhere in the respiratory tract. Careful examination of other possible airway leakage must be performed. For other circumstances of endotracheal tube intubation, such as spontaneous breathing or while in the ICU, further studies are needed. The aim of this study was to determine a practical way to obtain the appropriate intracuff pressure in pediatric subjects under anesthesia. The assumptions of the proposed method were based on controlled ventilation.
Limitations
This study was not without limitations. First, the study was limited to children ages 4–12 years. Despite the favorable results shown in this population, infants and toddlers who are more susceptible to tracheal intubation-induced damage may have different results due to the smaller tracheal tubes needed in such patients; therefore, further research is necessary. In addition, patients with a full stomach were not included in this study; therefore, it is not possible to conclude whether the use of the flow-volume loop guided technique for cuff inflation can prevent pulmonary aspiration. Finally, only one type of cuffed endotracheal tube was used in this study. The outcomes must be verified by using other brands of endotracheal tubes constructed with different materials and with variations in cuff compliance.
Conclusions
The flow-volume loop guided technique was an effective sealing method for endotracheal tube cuffs in school-age children and a lower and more-consistent endotracheal tube intracuff pressure was achieved by using the flow-volume loop guided technique. The technique also uses the continuous monitoring of the anesthesia machine as an objective means to inflate the cuff, minimizing endotracheal injury while preventing endotracheal air leakage. However, because this study was performed only under controlled ventilation during anesthesia, the use of this technique under other circumstances requires further study.
Footnotes
Author Disclosure Statement
The authors have disclosed no conflicts of interest.
