Abstract
Observational studies and clinical trials indicate a link between arterial stiffness (AS) and sarcopenia (SAR), yet the causal relationship between these remains unclear. The study aims to investigate the causal connection from AS to SAR by Mendelian randomization (MR). We analyzed Genome-Wide Association Studies data for AS indicators: pulse wave arterial stiffness index (PWASI) and pulse wave peak-to-peak time (PPT), and SAR indicators: low hand grip strength (LHGS), usual walking pace (UWP), moderate-to-vigorous physical activity levels (MVPA), and walk or cycle unassisted for 10 minutes. The inverse variance-weighted, MR-Egger, weighted mode, and weighted median were applied to MR. There is a bidirectional causal relationship between the AS and SAR. The PWASI has a causation with UWP (odds ratio [OR] = 0.97, 95% confidence interval [CI] = 0.94–0.99). The PPT has a causal association with MVPA (OR = 1.08, 95% CI = 1.002–1.144) and UWP (OR = 1.05, 95% CI = 1.017–1.096). The LHGS is causally associated with PPT (OR = 0.95, 95% CI = 0.91–0.98) and UWP has a causal association with PWASI (OR = 0.77, 95% CI = 0.65–0.90) and PPT (OR = 1.37, 95% CI = 1.17–1.60). The increased AS could reduce the motor ability slightly and the lower upper and lower limb strength could lead to the higher AS. This bidirectional causal relationship of the two may offer novel perspectives for advancing the understanding of the underlying mechanisms related to AS and muscle pathophysiology.
Introduction
Arterial stiffness (AS), a pivotal determinant in cardiovascular pathology, is considered a critical catalyst for the emergence of cardiovascular diseases 1,2 Aging, obesity, amputation, and sedentary behavior are the risk factors of AS. 3 –6 AS may serve as an indicator of coronary heart diseases, hypertension, and atrial fibrillation. 7 –9 The inflammation and pro-ferroptotic signaling may initiate factors of AS. 2,10
Sarcopenia (SAR) is tied with an augmented risk of geriatric syndromes, which is characterized by the progressive and general loss of skeletal muscle function and mass. 11 Depending on different criteria, the prevalence of SAR ranges from 5% to 22% globally, which creates a huge medical burden due to the fall and disability. 12 It is also associated with the higher all-cause mortality. 13 Cardiovascular diseases such as hypertension could be the vital risk factors of SAR by lower vascular perfusion and malnutrition, 14 suggesting a possible connection between AS and SAR.
Clinically, the correlation between AS and SAR has been highlighted as a critical factor in geriatric health assessments. A meta review suggested that AS may be a mediation phenotype of SAR and cardiovascular diseases. 15 The earlier cross-sectional studies indicated that AS is positively related to skeletal muscle loss and weak muscle strength 16,17 Additionally, the correlation between elevated AS and diminished hand grip strength (HGS) underscores the interaction between vascular and muscular health in the aging process. 18 Cohort studies showed that the older adults with SAR or SAR obesity had the highest pulse wave velocity. 19 Research in Gambian found that the higher negative correlation between pulse pressure and hand power in men than in women. 20 Moreover, the combination of liver fibrosis and SAR may result the vascular damage and AS in patients with diabetes. 21 However confounders like aging and obesity pose challenges in delineating the causality between AS and SAR. 15
Mendelian randomization (MR) analysis is an efficient way to explore potential causality between phenotype and diseases. This approach integrates genetic associations from separate Genome-Wide Association Studies (GWAS), allowing for the examination of causal effects of a particular exposure on a given outcome without confounding influences. 22
In our investigation, we explored the causal nexus between arterial rigidity and muscular atrophy through an MR analysis. AS was gauged via pulse wave velocity (PWV) analysis, employing the pulse wave arterial stiffness index (PWASI) and pulse wave peak-to-peak time (PPT) as indices of exposure. 23 For the outcome, SAR was represented by the low hand grip strength (LHGS), usual walking pace (UWP), moderate-to-vigorous physical activity levels (MVPA), and walk or cycle unassisted for 10 minutes (WU10). These measures assessed the extent of SAR and its potential effects on physical function. 24 –26 The key assumptions of MR are satisfied (Fig. 1). 22
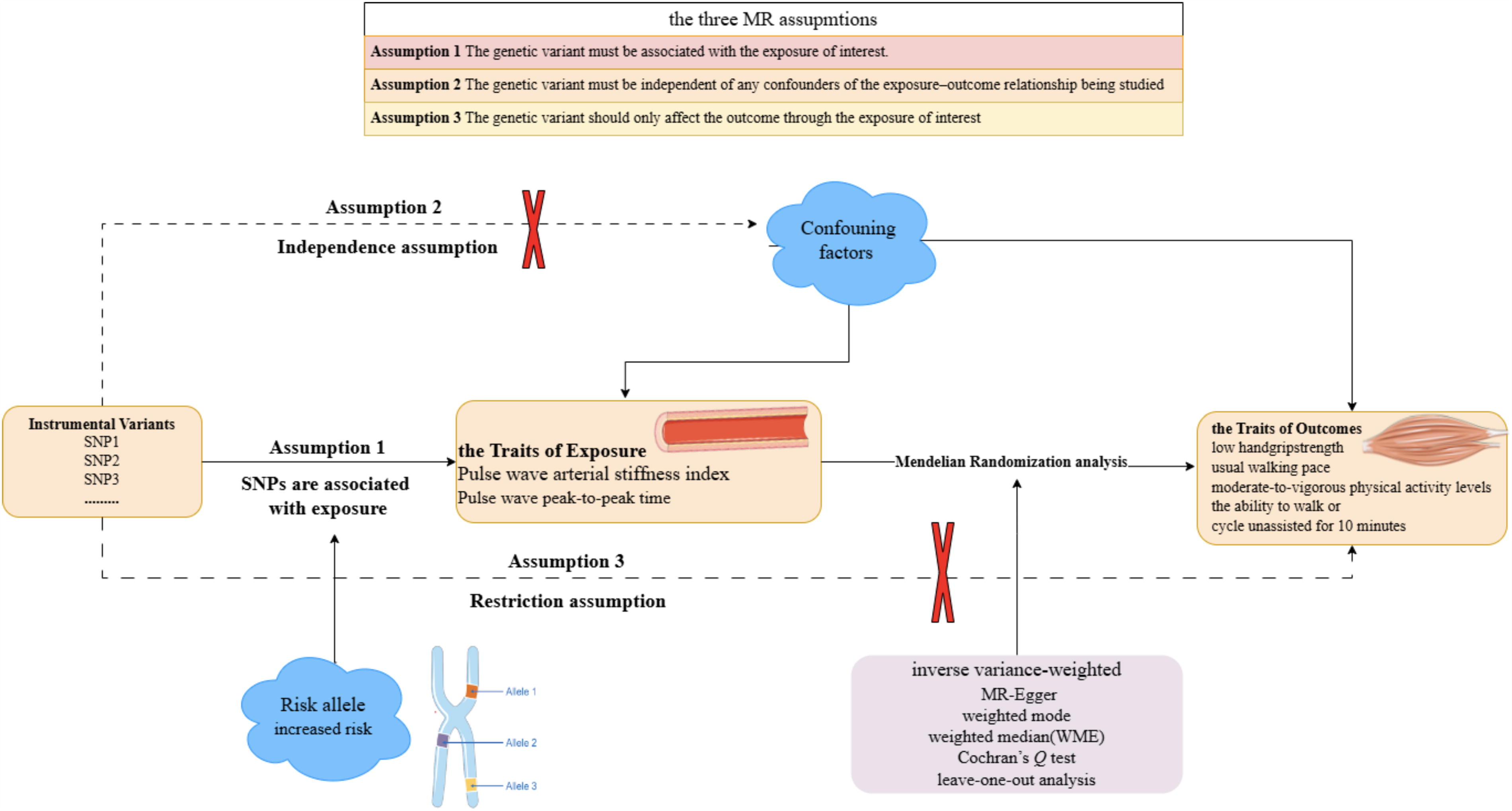
The directed acyclic graphs that key three assumptions for valid analysis are met.
Method
Study design
We selected the GWAS datasets from the MRC Integrative Epidemiology Unit (IEU) database. 27,28 We chose the PWASI (ukb-b-11971) and PPT (ukb-b-8778) as exposures, and LHGS (ebi-a-GCST90007526), UWP (ukb-b-4711), WU10 (ukb-b-14149), and MVPA (ebi-a-GCST006097) as outcomes. The information of GWAS datasets is provided in Supplementary Table S1. We used the selected single nucleotide polymorphisms (SNPs) for MR analysis. Then the sensitive analysis checked the robustness of the results. Finally, we reversed the roles of exposure variables and outcome measures and performed the analogous procedures. This study’s reporting follows the Strengthening the Reporting of Observational Studies in Epidemiology - Mendelian Randomization (STROBE-MR) guidelines (Supplementary Table S2). The flow is presented in Figure 2.
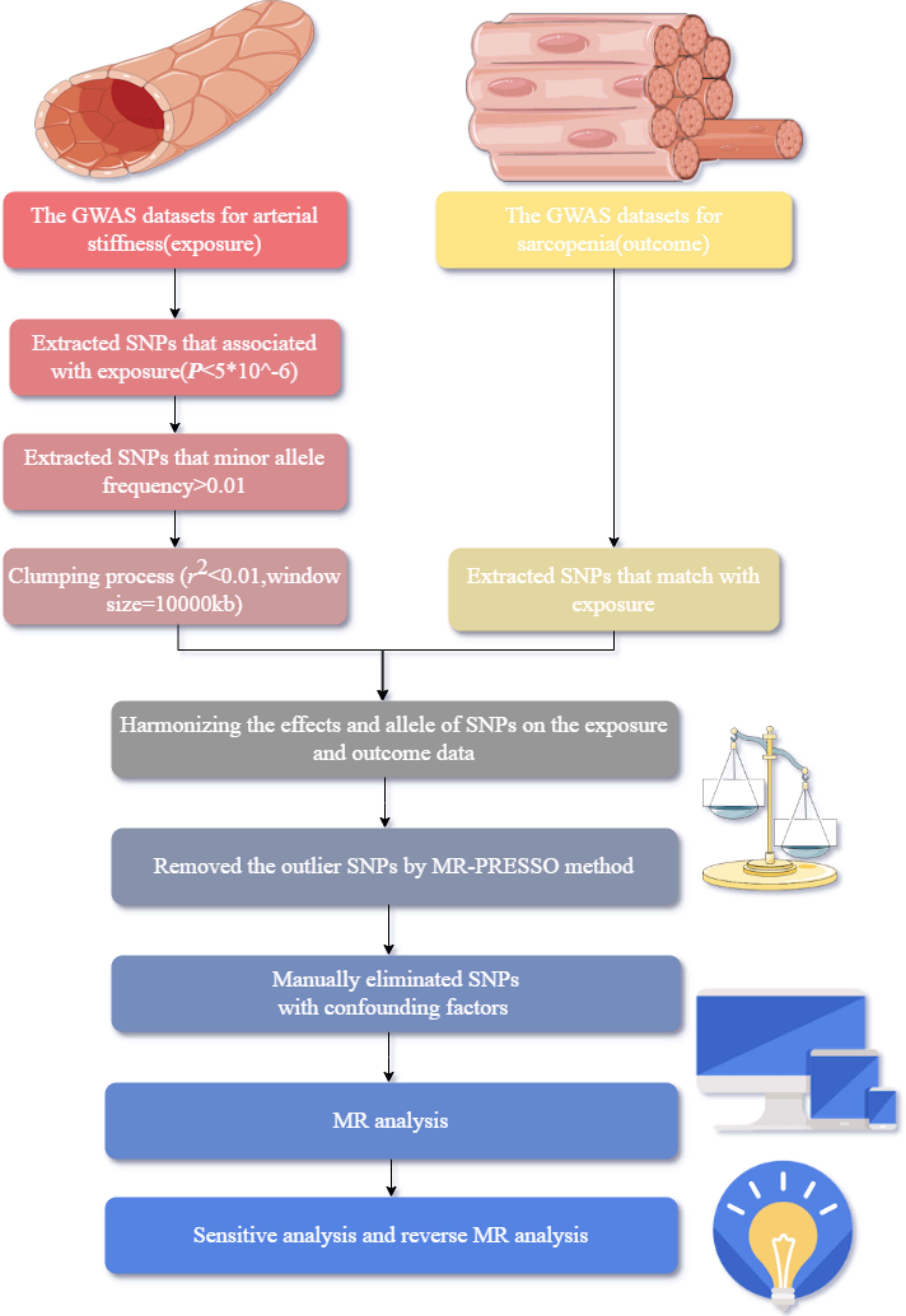
The study flow of Mendelian randomization (MR) analysis.
Exposures
Data associated with the PWASI were selected from a dataset of 151,053 participants of European descent. Likewise, genetic variants linked to PPT were identified from 151,466 individuals of European descent. They both are from the UK Biobank. The characteristics of arterial health were evaluated based on pulse wave morphology data derived from the finger, captured using the infra-red sensor device, the Pulse Trace PCA 2TM. A total of 26,232 participants were excluded from the analysis because of the data loss. 29 The PWASI is defined as the ratio of the PPT to the individual’s height. Conversely, PPT is characterized by the time interval separating the maximal amplitudes of the incident and reflected pressure waves within the pulse wave. According to the pathophysiology of AS, the higher PWASI value indicates greater AS, and lower PPT is associated with higher AS. Ethical approvals were obtained, ensuring compliance with ethical standards for GWAS. 29,30
Outcomes
In a comprehensive analysis of 22 distinct cohorts, SNPs linked to LHGS were discovered, encompassing 254,894 participants of Caucasian heritage, all aged 60 years or older. 26 SNPs related to physical activity traits, including UWP, MVPA, and WU10, were derived from a total of 459,915, 377,234, and 68,537 European individuals from the UK Biobank and Atherosclerosis Risk in Communities study. The measurements were obtained by self-report and Axivity AX3 wrist-worn accelerometer. 24 LHGS is characterized by an HGS threshold (≤30 kg for males, ≤20 kg for females). The UWP is the usual walking distance in an hour. MVPA is quantified by aggregating the cumulative weekly duration of moderate and vigorous physical activities. WU10 criterion is defined by an individual’s capacity to ambulate or pedal unassisted for a continuous period of 10 minutes. Higher levels of LHGS are related to enhanced SAR while lower UWP, MVPA, and inability to WU10 are all associated with a higher likelihood of SAR.
Variant selection
We used a similar process of a performed cardiovascular MR study 31 to select the applicable SNPs. The three key assumptions of MR were met 22 : first, the significance threshold for instrumental variables (IVs) of the exposure was initially set to p < 5 × 10−8; however, this resulted in a limited number of SNPs. Consequently, we established the threshold at p < 5 × 10−6. 29,32 Second, the minor allele frequency cutoff was established to exceed 0.01, ensuring that only variants with a notable population prevalence were considered. Third, during the clumping process, we used a linkage disequilibrium r 2 < 0.01 and alongside a clumping criterion distance 10,000 kilobases, to ensure precise identification of independent genetic associations. Subsequently, SNPs with palindromic sequences were excluded through a process of standardization that effectively identified and removed these variants, ensuring the accuracy and reliability of our findings. Then, we employed Mendelian Randomization Pleiotropy RESidual Sum and Outlier (MR-PRESSO) method to identify and remove any outlier SNPs from the analysis. Finally, we manually eliminated SNPs with confounding factors SNPs with missing values are removed.
MR analysis
We used four methods to assess the causal connection: the inverse variance-weighted (IVW), weighted median (WME), the MR-Egger regression, and the mode-based estimate. 31 The IVW method exhibits enhanced statistical strength compared with other methodologies when employed under the appropriate conditions, making it a preferred choice for certain meta-analytic scenarios. 33 Thus, the principal findings stem predominantly from the IVW approach, complemented by corroborative insights from alternative methodologies. Furthermore, a randomized-effects IVW analysis was employed to counteract possible biases.
Sensitive analysis
The strength of the association between each IV and the exposure was evaluated using the F-statistic measure. 34 The formula is: F = r2 × (n − 2)/(1 − r2 ). 35 Cochran’s Q test checked the heterogeneity, ensuring a nuanced understanding of their interrelationships with the exposure variable. The pleiotropy was evaluated by the MR Egger. Leave-one-out analysis verified whether any single SNP disproportionately influenced the outcome.
Reverse MR analysis
We reversed the roles of the traits, using SAR-related traits as the exposure and AS-related traits as the outcome, and repeated the analysis. The statistical significance threshold was set at p < 5 × 10−8. A stringent criterion is designed to safeguard against the influence of spurious correlations in our genetic association analysis. Utilizing the MR Steiger directionality test, an inquiry was undertaken to ascertain the existence of a unidirectional causal influence. 34 The R packages of twosampleMR were used. 28 We didn’t register. We employed the Kimi AI to verify the errors and enhance fluency.
Results
SNP selection
For the PWASI, we excluded 9,851,829 SNPs out of 9,851,867 samples resulting in the selection of 22 SNPs (Supplementary Table S3). For the PPT, we identified 20 SNPs (Supplementary Table S3).
Causal effects of AS traits and SAR traits
Through the MR analysis, we found that the PWASI has a causality with UWP (odds ratio [OR] = 0.97, 95% confidence interval [CI] = 0.94–0.99, p = 0.025, IVW). In addition, the PPT has a causal connection with MVPA (OR = 1.08, 95% CI = 1.002–1.144, p = 0.01, IVW) and UWP (OR = 1.05, 95% CI = 1.017–1.096, p = 0.009, IVW) (Table 1, Fig. 3). Details are in Supplementary Table S4 and Supplementary Figure S1. F-statistics for all analyses are >10 (Supplementary Table S3). The direction is confirmed to be true (Table 1). Although some analyses exhibit heterogeneity (Supplementary Table S5, Supplementary Fig. S2), we selected the random-effects model to reduce the bias and none exhibited pleiotropy (Supplementary Table S6). Leave-one-out analysis revealed no SNPs with significant effects on the outcomes, implying a reliable and uniform association among the evaluated genetic variants (Supplementary Fig. S3).
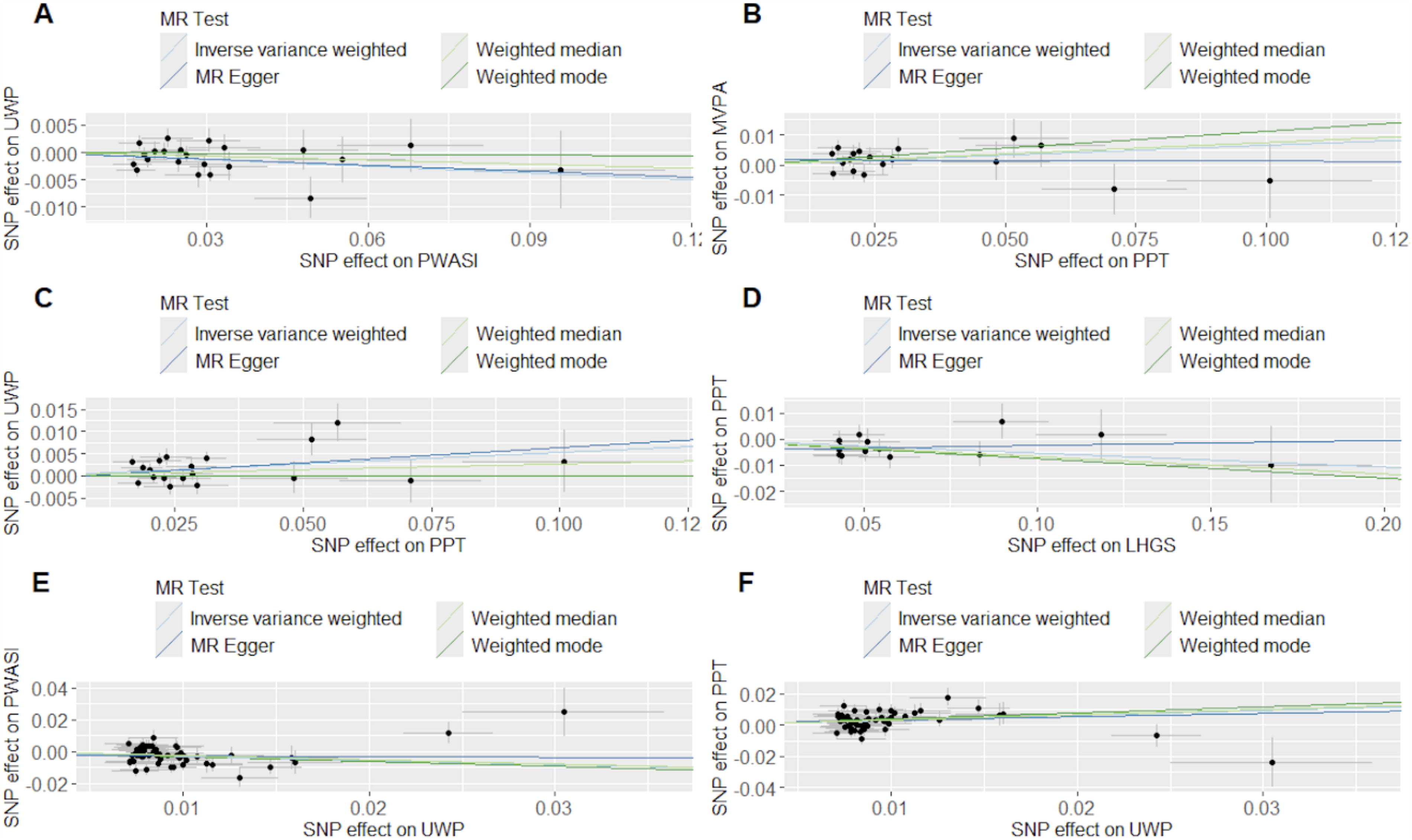
The Mendelian randomization results between traits of arterial stiffness and traits of sarcopenia;
The Mendelian Randomization Results Between Traits of Arterial Stiffness and Traits of Sarcopenia
p < 0.05.
IVW, inverse variance-weighted method; LHGS, low hand grip strength; MVPA, moderate-to-vigorous physical activity levels; OR, odds ratio; PPT, pulse wave peak-to-peak time; PWASI, pulse wave arterial stiffness index; SE, standard error; SNP, single nucleotide polymorphism; UWP, usual walking pace; WME, weighted median estimator.
Reverse MR analysis
Using SAR traits as the exposure and AS traits as the outcomes (Supplementary Table S7), we found that LHGS is causally associated with PPT (OR = 0.95, 95% CI = 0.91–0.98, p = 0.01, IVW; OR = 0.94, 95% CI = 0.88–0.98, p = 0.012, WME) and UWP has a causal association with PWASI (OR = 0.77, 95% CI = 0.65–0.90, p = 0.001, IVW; OR = 0.76, 95% CI = 0.63–0.93, p = 0.006, WME) and PPT (OR = 1.37, 95% CI = 1.17–1.60, p < 0.001, IVW; OR = 1.40, 95% CI = 1.15–1.69, p < 0.001, WME) (Table 1, Fig. 3). The details are in Supplementary Table S4 and Supplementary Figure S1. The direction of all associations is confirmed to be true (Table 1). The F-statistics are >10 (Supplementary Table S7), manifesting the strength of the IVs. Although heterogeneity was observed (Supplementary Table S5, Supplementary Fig. S2), no pleiotropy was detected (Supplementary Table S6). Employing the leave-one-out approach, no particular SNPs were discovered to have a significant effect on the analysis results, maintaining the consistency of the observed genetic relationship (Supplementary Fig. S3).
Discussion
The study’s findings delineate a reciprocal causality between AS and SAR, where AS is implicated in the genesis of SAR, and conversely, SAR is suggested to aggravate AS. This bidirectional relationship may be underpinned by shared risk factors and analogous pathophysiological mechanisms fundamental to both conditions.
Our results show that AS has a certain effect on usual walking muscle ability. A community-based investigation has revealed an inverse correlation between aortic stiffness and gait speed among participants aged 70 years and older. 36 Several studies from all kinds of populations showed a negative relationship between AS and MVPA. 37 –40 Consequently, the conclusion suggests that AS exerts a causal negative impact on muscle output function, affecting both the routine and the strenuous aspects of physical performance.
In addition, the UWP may be causally associated with PWASI and PPT and higher LHGS is negative with PPT, indicating that the muscle function can affect the AS. A retrospective observational study demonstrated that a 6-minute walk distance is an independent determinant of the AS. 41 In conclusion, AS and muscle function have a partly bidirectional causal relationship, which may be specifically suitable for individuals of European ancestry.
However, there are conflicting conclusions in the literature. A longitudinal study involving 497 older adults found that HGS didn’t have a relationship with PWV. 42 An observational study indicated that the UWP of these individuals bore no significant association with PWV. 43 Given the age of the participants in these studies, confounders such as senile depression and aging may have influenced the results, and the number of participants is still small. Consequently, subsequent studies should account for these possible confounding factors and strive for a more precise comprehension of the correlation, ideally by enrolling a larger participant pool. The cross-sectional research indicated a significant association between an increased resting brachial artery size and superior grip strength, hinting at a probable vascular influence on muscle functionality. 44 A landmark study (n = 5352) employing a PWV measurement demonstrated that elevated PWV values were significantly linked to diminished physical functioning even after rigorous adjustment for confounding variables such as age, sex, and ethnic group. 45 The credibility of the two studies is bolstered by their large population samples, with the second study particularly enhancing its validity by adjusting for the effects of aging. Consequently, these robust findings incline us toward acknowledging a potential relationship between AS and SAR.
Given the established bidirectional causal relationship between AS and SAR, it is plausible that they share common underlying mechanisms that drive their progression. Aging has been associated with impaired mitochondrial function, potentially through modulation of peroxisome proliferator-activated receptor γ coactivator 1α signaling pathways, leading to inflammation and oxidative stress that contribute to muscle mass reduction and vascular elasticity deterioration. 46,47 Testosterone deficiency could decrease muscle fibers due to lack of insulin-like growth factor 1 levels and damage the arterial wall through low-grade inflammation and reduced vasodilatory effect. 48 –50 Insulin resistance may precipitate muscle catabolism by triggering mitogen-activated protein kinase and S6K1 pathways, also implicated in adipose tissue augmentation by diminishing nitric oxide production and exacerbating inflammatory responses. 51 –54 Activation of the renin-angiotensin-aldosterone system has been shown to escalate SAR and AS, influencing metabolic and cardiovascular homeostasis. 55,56 Fatty acids like pentadecanoic acid are crucial in the development of adipose tissue and sympathetic nervous system activity, leading to endothelial dysfunction and initiating inflammatory responses. 19,57,58 In conclusion, aging, insulin resistance, testosterone deficiency, increased fatty acids, and activation of the renin-angiotensin-aldosterone system are common factors, with inflammation, oxidative stress, mitochondrial dysfunction, and metabolic disorder as primary pathological reactions. However, the precise mechanisms underlying this interplay remain to be fully explored.
Dietary interventions and regular physical exercise are recognized as crucial strategies for enhancing muscle strength. The glucose-lowering agents (GLAs), including glucagon-like peptide-1 receptor agonists (GLP-1RAs), are as therapeutic agents of AS. According to our results, improving the muscle ability could decrease the AS and vice-versa. Therefore, diet, exercise, and GLAs may be therapeutic methods for both AS and SAR. Recent research has demonstrated that a combination of resistance and aerobic exercises can significantly augment baPWV and muscular strength. 59 A randomized trial demonstrated that moderate-intensity walking can improve PWV, 60 and dietary nitrate supplementation can enhance AS and SAR by increasing nitric oxide bioavailability. 61 Additionally, dietary sodium restriction has been shown to delay AS and metabolic acidosis in the context of kidney failure. 62 Previous research showed that GLP-1RAs could improve the muscle proportion in diabetes. 63,64 Consequently, GLP-1RAs, exercise, and dietary interventions are considered effective methods for improving AS and SAR.
While contributing valuable findings, this study has inherent limitations: initially, the study’s two specific traits of AS, namely the PWASI and the PPT, may engender bias due to the limited scope of the examined traits. Second, the measurement based on a single position may be subject to greater influence from random factors. Third, the reliance on self-reported data for some of the SAR-related traits introduces the potential for subjective bias. Moreover, the research’s ethnic limitation to participants of European heritage could potentially hinder the broader applicability of its conclusions across various demographic groups. Additionally, the significance threshold used for selecting SNPs (p < 5 × 10−6) is relatively lenient, which could lead to a lower connection between the SNPs and the traits of AS. Despite providing valuable insights, there are clear areas for improvement.
Further research can focus on the specific pathways and mechanisms that link AS and SAR, as well as explore potential therapeutic strategies that can address both conditions simultaneously. Enhancing our knowledge of the aging process and associated health challenges can contribute to advancing preventive and therapeutic approaches for cardiovascular and musculoskeletal diseases.
Conclusion
To sum up, the study aims to dissect the etiological confluence between AS and SAR. The results reveal a substantial reciprocal causal linkage between AS and SAR, corroborating the widely observed phenomenon that cardiovascular conditions, including hypertension, and decrements in muscle strength frequently co-occur within the aging demographic.
Footnotes
Acknowledgments
The authors extend their sincere gratitude to the IEU for generously supplying the GWAS data, which was instrumental to the execution of this research endeavor. Furthermore, the authors are profoundly appreciative of the peerless mentorship and unwavering support received from colleagues and academic advisers alike, whose contributions were invaluable throughout the conception and progression of this scholarly work.
Data Availability Statement
Authors’ Contributions
H.L.: Review and editing (equal), methodology (lead), formal analysis (lead), investigation (lead), conceptualization (lead), writing—original draft (lead), formal analysis (lead), writing—review and editing (equal), resources (equal). T.M.: Software (lead), writing—review and editing (equal), validation (lead), visualization (lead). R.Q.: Funding acquisition (lead), supervision (lead).
Author Disclosure Statement
There are no competing interests or affiliations with any entities or collectives.
Funding Information
This work was supported by the Heilongjiang Province Traditional Chinese Medicine Research Project (grant numbers ZYW2023–066).
Supplementary Material
Supplementary Figure S1
Supplementary Figure S2
Supplementary Figure S3
Supplementary Table S1
Supplementary Table S2
Supplementary Table S3
Supplementary Table S4
Supplementary Table S5
Supplementary Table S6
Supplementary Table S7
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
