Abstract
Purpose
: To evaluate myopic shift after the administration of omidenepag isopropyl (OMDI) in patients with normal-tension glaucoma (NTG).
Methods
: In this retrospective observational study, we enrolled 105 patients (105 eyes) with NTG who had received initial doses of OMDI between December 2018 and October 2022. We recorded the data on subjective refraction (diopters, D) at baseline and at the first visit after administration. We investigated and followed-up patients with myopic shift ≥0.75 D from the baseline value. The main outcome measures were refractive changes from baseline to the first visit after administration.
Results
: This study included 105 patients (52 men and 53 women). The mean refractions at baseline and at the first visit after administration were –3.87 ± 3.77 and –3.99 ± 3.79 D, respectively (p < 0.01). Three patients (2.9%) developed myopia ≥–0.75 D. Among these three patients, OMDI administration was continued in two, and their refraction value returned to <±0.75 D of the baseline value; the medication of the other patient was changed because of blurred vision, and the refraction returned to <±0.75 D of baseline after the change in medication.
Conclusions
: After administration of OMDI, 2.9% of the patients developed myopic shift ≥0.75 D. This myopic shift was transient, as refraction returned to <±0.75 D of baseline after follow-up or medication changes.
Introduction
Omidenepag isopropyl (OMDI) is a prostaglandin EP2 receptor agonist that has been available since 2018 in Japan. Its action in lowering intraocular pressure (IOP) affects the uveoscleral and trabecular pathways, facilitating aqueous humor outflow.1,2 Several reports of its sufficient IOP-lowering effects and high safety in clinical trials or studies have been published.3–12 The existing prostaglandin-analog drugs, including prostaglandin F receptor agonists, are known to cause occasional prostaglandin-associated periorbitopathy (PAP), including eyelid or iris pigmentation, elongated or darkened eyelashes, and deepening of the upper eyelid sulcus.13,14 However, it has been reported that PAP does not commonly occur during OMDI administration.3–12 The most frequent adverse reaction of OMDI is conjunctival hyperemia.3–11
In our hospital, we encountered a patient with normal-tension glaucoma (NTG) who complained of blurred vision after the administration of OMDI. Upon examination, we found that the patient had developed a myopic shift of –2.00 D. In the postmarketing surveillance of OMDI, Nakazawa etal. noted that the overall incidence of myopia in patients with glaucoma or ocular hypertension was 1.1%.3 Nevertheless, the progress after the myopic shift is unclear in these studies. Therefore, in the present study, we aimed to retrospectively investigate the myopic shift in patients with NTG who received initial doses of OMDI and to report the progress of the patients who developed myopia.
Methods
This study included 105 patients (105 eyes) at Inouye Eye Hospital (Tokyo, Japan) or Sapporo Inouye Eye Clinic (Sapporo, Japan). The participants were untreated, diagnosed with NTG, and administered OMDI for the first time between December 2018 and October 2022. This study was conducted in accordance with the tenets of the Declaration of Helsinki and was approved by the Inouye Eye Hospital Ethics Committee (approval number: 202208-4). The requirement for informed consent was waived because of the retrospective study. A notice was posted in the hospital specifying the subject of this study, and that the study would be conducted retrospectively. If patients refused to be part of the study, they had the prerogative to be excluded.
Using medical records, refraction values at baseline and at the first visit after administration were investigated and compared (before and after administration). Based on previously reported criteria,3 a patient with a change ≥0.75 D from baseline was defined as having myopic shift. In addition, the refractions at subsequent visits were also investigated in patients with refractive changes. Patients with myopic shift because of the progression of cataracts were excluded from this study.
The myopic progress was investigated using medical records. In addition, multiple regression analysis was performed with refractive changes as the dependent variable and age, sex, baseline IOP, baseline mean deviation (MD), and baseline refraction as independent variables. If only one eye met the inclusion criteria, that eye was considered in the study. When OMDI was administered to both eyes, the eye with the higher baseline IOP was considered. If the IOP was the same in both eyes, the right eye was considered.
Statistical analysis
A paired t-test was used to compare the refractions before and after administration. Statistical significance was set at p < 0.05. All statistical analyses were conducted using IBM SPSS Statistics for Windows, version 22 (IBM Corp., Armonk, NY, USA).
Results
Patients
Among the 105 participants, 52 were men and 53 were women. The mean age was 57.2 ± 13.4 years (mean ± standard deviation; range, 29–81 years). At baseline, the mean IOP was 15.9 ± 2.5 mmHg (range, 11–21 mmHg). The MD value at baseline, measured using the Humphrey visual field test (program: 30-2 SITA standard), was –4.49 ± 4.25 dB (range, –19.26 to 0.75 dB). The period from baseline to the first follow-up visit after administration was 1.15 ± 0.43 months (range, 1–3 months).
Refractive changes
The mean refractions were −3.87 ± 3.77 and −3.99 ± 3.79 D at baseline and at the first visit after the administration of OMDI, respectively; a significant myopic shift occurred after OMDI administration (p < 0.01; Fig.1). The mean change in refraction from baseline to the first visit was −0.10 ± 0.31 D. The refraction developed a myopic shift (from −0.25 to −2.00 D) and a hyperopic shift (from +0.25 D to +0.50 D) in 36 and 14 patients, respectively (Fig.2). There was no change in 55 patients. Three patients (2.9%) had a myopic shift ≥0.75 D from the baseline value. Among these three patients, OMDI administration was continued in two patients, and their refraction values returned to <±0.75 D of the baseline value at 6 months and 9 months after administration; the medication of the other patient was changed because of blurred vision at 1 month after administration, and the refraction returned to <±0.75 D of baseline at 3 weeks after the change in medication.

Mean refraction before and after the administration of omidenepag isopropyl.
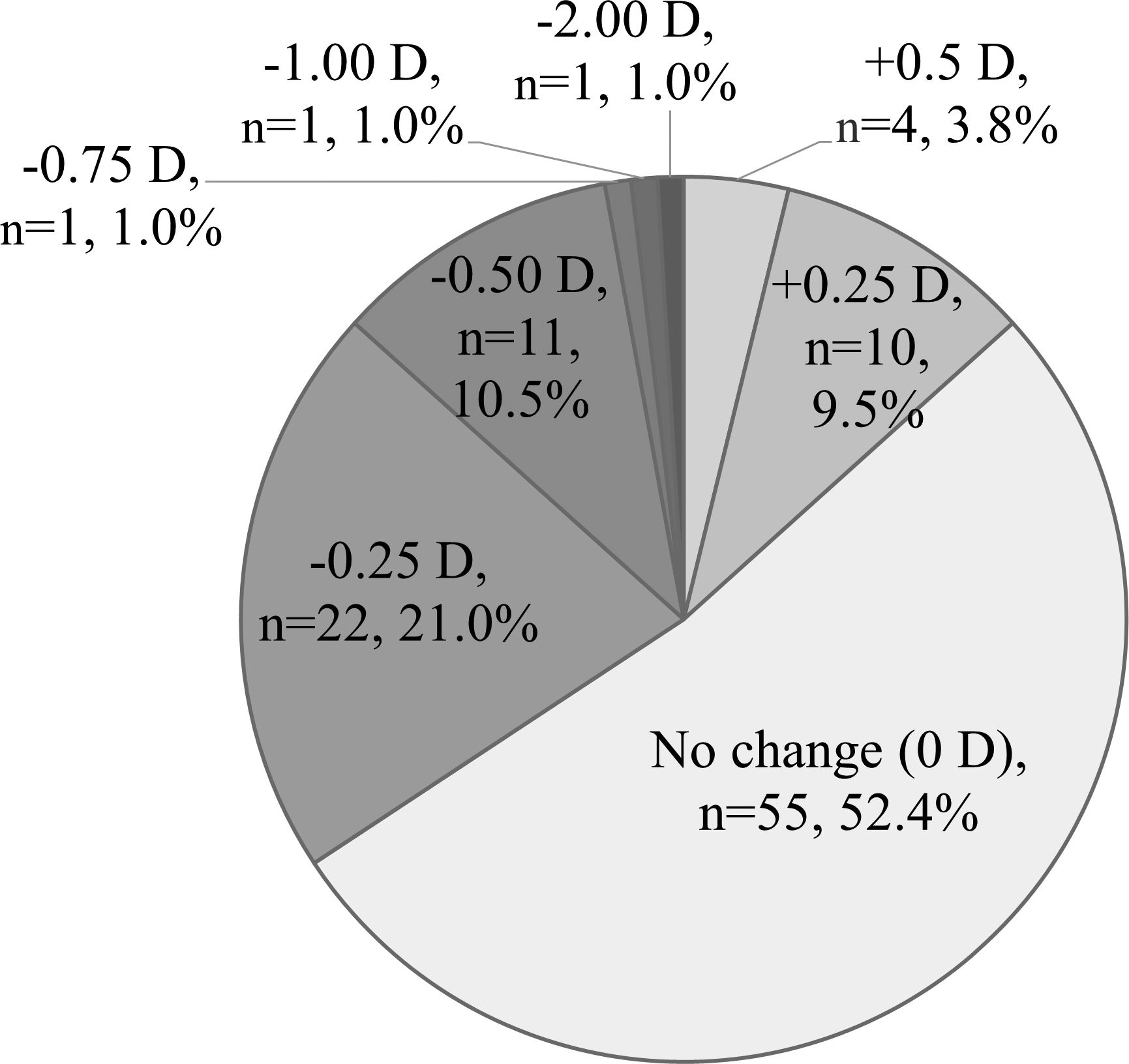
Refractive change between baseline and after the administration of omidenepag isopropyl.
Multiple regression analysis
In the multiple regression analysis, no factors correlated with the refractive changes (Table1).
Factors affecting refractive changes at the first visit after the administration of omidenepag isopropyl
IOP, intraocular pressure; MD, mean deviation; HF, Humphrey visual field test.
Discussion
In this study, we investigated refractive changes after the administration of OMDI in patients with NTG. A significant myopic shift occurred after OMDI administration (p< 0.01). Three patients (2.9%) had a myopic shift ≥−0.75 D.
Kanamori etal. measured the spherical equivalents of patients before and after the administration of OMDI.4 In addition, the patients were divided based on OMDI administration into the first administration group, the added group, and the switching group.4 The spherical equivalents at baseline and after administration were −4.70 ± 3.06 and −4.73 ± 3.09 D in the first administration group (n = 62), –6.57 ± 5.04 and –6.85 ± 5.21 D in the added group (n = 7), and –5.62 ± 2.97 and –5.27 ± 3.30 D in the switching group (n = 13), respectively; in all groups, there was no difference before and after OMDI administration. In the current study, all patients were previously untreated and then administered OMDI; after administration, the mean refraction showed a statistically significant myopic shift. The mean change in refraction from baseline to the first visit was –0.10 D, and the mean increase in myopia was only 3% compared to that at baseline, which was not considered clinically significant. However, three patients (2.9%) developed a myopic shift ≥–0.75 D. The baseline refractions of patients who had a myopic shift were –9.0, –7.0, and0 D. Myopia after OMDI administration tended to develop in patients with high myopia.
The proportion of patients with a myopic shift as an adverse effect after the administration of OMDI is small (Table2).3,5 In previous reports, the incidence rates of myopic shifts following naïve monotherapy were 0.6%5 and 1.3%3; however, it was higher, at 2.9%, in this study. It is possible that the proportion of patients with myopic shift differed because of the small number of patients in this study compared with that in previous reports.3,5 In a report by Miki etal., the refractive change was unclear because they provided no definition of a myopic shift.5 In a study by Nakazawa etal., a myopic shift was defined as having a refractive change ≥−0.75 D, occurring undeniably after the OMDI administration, and it was deemed as a myopic shift by the physician. They reported a myopic shift in only 1.1% of patients,3 whereas we reported it in 2.9% of patients. Furthermore, their study did not describe the detailed course of myopic patients.
Frequency of myopic shifts after the administration of omidenepag isopropyl
In the current study, there were three patients with myopic shift ≥0.75 D after the administration of OMDI. In one of these three patients, the administration was discontinued; myopia resolved early after the administration was discontinued, and the myopic shift may have been influenced by OMDI. The myopic shift in the other two patients who continued the administration improved at 6 months and 9 months. Because of the progression in these two patients, the myopic shift after the administration of OMDI is expected to be transient.
In the multiple regression analysis, no factors were associated with refractive changes. These results indicated that it is difficult to predict the cases in which myopic shifts will develop.
It has been reported that the cornea is thickened by OMDI administration.6 In previous reports, the mean cornea thickening was 10.7–17.8 μm.6 In this study, the effect on the cornea before and after the administration of OMDI was unclear because of the lack of central corneal thickness measurement.
This study also compared keratometric readings before and after OMDI administration for the four patients with myopic shifts and found no difference; therefore, the corneal curvature was not considered a factor. Although the level of corneal thickening differs from case to case, it is improbable that corneal thickening affected the refraction.
Ciliary muscle tension may be related to myopia development. Particularly at ≥40 years of age, with the appearance of presbyopia, the weakening of the action of the ciliary muscle is possibly a contributing factor.
We reviewed a few reports1 , 15–17 to analyze the mechanism of action of myopia development following OMDI administration. In an investigation of guinea pigs, adenylyl cyclase activation increased scleral cyclic adenosine monophosphate (cAMP) levels. It thereby promoted myopia development through the inhibition of genes that promote collagen synthesis and extracellular matrix fibrosis.15 Furthermore, compared with the control group, the EP2 agonist butaprost administration group developed myopia, and the cAMP levels increased in the sclera.16 OMDI, which is an EP2 agonist similar to butaprost, may thus cause a myopic shift through the same mechanism mentioned earlier. Myopic shifts were transient in three patients in this study. Another explanation could be pseudomyopia, which is the temporary development of myopia caused by the ciliary muscle becoming tense and the lens thickening. It is undeniable that ciliary tone is involved because OMDI binds to and stimulates EP2 receptors as a mechanism of IOP-lowering action,1 and EP2 receptors are also distributed in the ciliary body.17
This study has some limitations. Considering the retrospective nature of the study, blurred vision was deduced only from the medical records, and determining compliance to administration of OMDI was difficult to guarantee. The number of cases was smaller than that of previous studies.3,5 Finally, as this was a preliminary retrospective study, future studies will be required to validate these results as well as to determine the efficacy of OMDI in other patient groups and compare its efficacy to those of other antiglaucoma agents.
In conclusion, in this study, we retrospectively investigated the myopic shift after the administration of OMDI in patients with NTG. Myopia developed in 2.9% of the patients. Because refraction gradually returned to <±0.75 D of baseline at follow-up, we concluded that the myopic shift was transient; nonetheless, careful follow-up is necessary just after administration.
Footnotes
Acknowledgments
None.
