Abstract
Since the outbreak and spread of COVID-19 in Japan, we have been investigating and anticipating the possibility of anti-inflammatory effects in combination therapy with antibacterial and antiviral agents. Minocycline is an antibacterial agent that has been validated for its anti-inflammatory effects and has a broad antibacterial spectrum. In the present study, we hypothesized the possibility of combination therapy with minocycline and favipiravir for COVID-19. Here, we validated our hypothesis based on the results of preliminary basic experiments and clinical trials. Large-scale clinical trials of minocycline and favipiravir combination therapy are warranted.
Dear Editor,
Tetracyclines have been reported to be potential candidates for the treatment of severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) infection.1 Favipiravir (FVP) was developed as an influenza drug, but during the current coronavirus disease 2019 (COVID-19) pandemic, it has been given for compassionate use because of its antiviral effect in inhibiting the RNA-dependent RNA polymerase of SARS-CoV-2.2 However, no standard treatment for COVID-19 has yet been established.
Tetracyclines inhibit bacterial protein synthesis by binding to the 30S subunit of the ribosome. Among them, minocycline (MIN) and doxycycline are orally absorbed more than 90% and have a broad antibacterial spectrum. Thus, it may be effective against many bacterial infections that may complicate COVID-19 patients. Moreover, MIN may be effective in inhibiting the main protease of SARS-CoV-2.3 MIN inhibits its intracellular signaling pathway by inhibiting molecules located downstream of Toll-like receptor 4,4 which is activated in COVID-19,5 and suppresses proinflammatory cytokine/chemokine production.
In preliminary experiments, results showed that FVP also suppressed inflammatory cytokine/chemokine production in lipopolysaccharide-stimulated THP-1 cells (Figure 1).
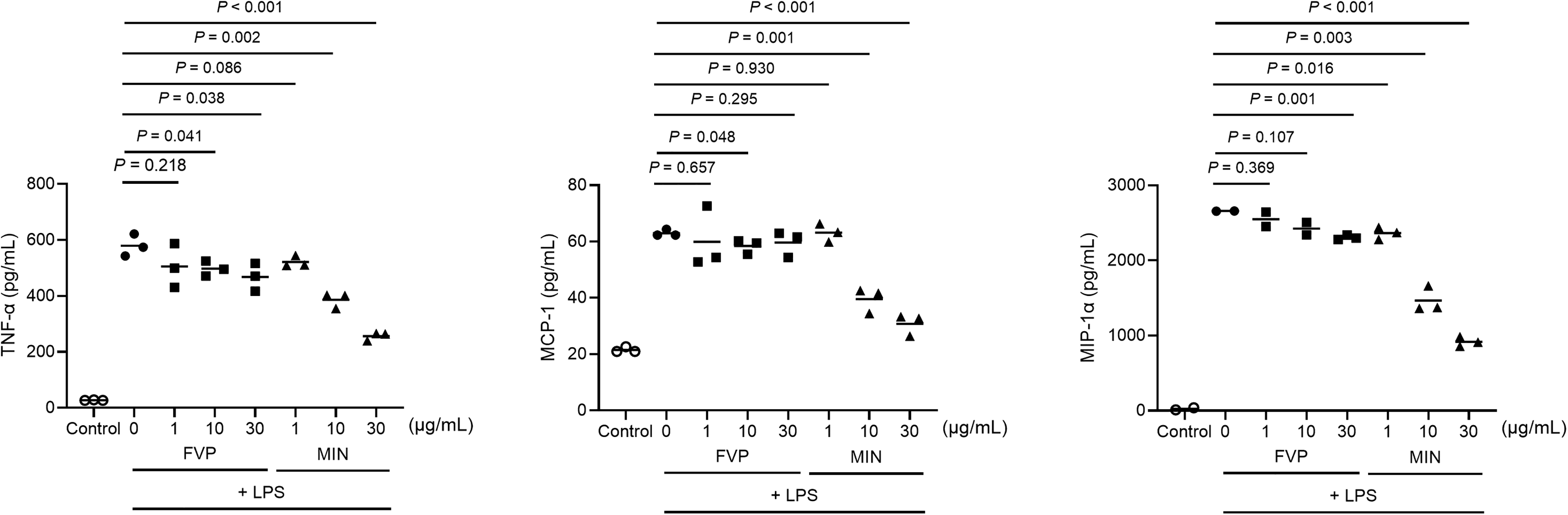
Effects of FVP and MIN on cytokine and chemokine release in LPS-stimulated THP-1 cells. THP-1 cells were incubated with or without LPS (0.05 µg/mL). The LPS-stimulated THP-1 cells were then treated with 0, 1, 10, or 30 µg/mL of FVP or MIN for 4 hours. Then, the TNF-α, MCP-1, and MIP-1α concentrations in the supernatants of the LPS-stimulated THP-1 cells were determined simultaneously using multiplex bead immunoassays. Horizontal bars indicate the means. Statistical analysis (two-tailed unpaired t-test) was performed by GraphPad Prism version 9.2.0 software (GraphPad Software, San Diego, CA, USA). Values with a P < 0.05 were considered to be significantly different. FVP, favipiravir; LPS, lipopolysaccharide; MCP-1, monocyte chemotactic protein 1; MIN, minocycline; MIP-1α, macrophage inflammatory protein-1 alpha; TNF-α, tumor necrosis factor alpha.
In our clinical study, the combination of FVP and MIN suppressed IL-6 production (measured by multiplex bead immunoassays) in the serum of COVID-19 patients compared to FVP alone. Furthermore, the time for COVID-19 patients to become SARS-CoV-2 polymerase chain reaction (PCR) negative was also shortened in the FVP and MIN combination group.3 The combined use of FVP and MIN may effectively accelerate the negative results of SARS-CoV-2 PCR because they exhibit antiviral effects through different mechanisms. We believe that FVP inhibits the RNA-dependent RNA polymerase of SARS-CoV-2, and MIN inhibits the main protease of SARS-CoV-2, thereby creating an additive effect (Figure 2).
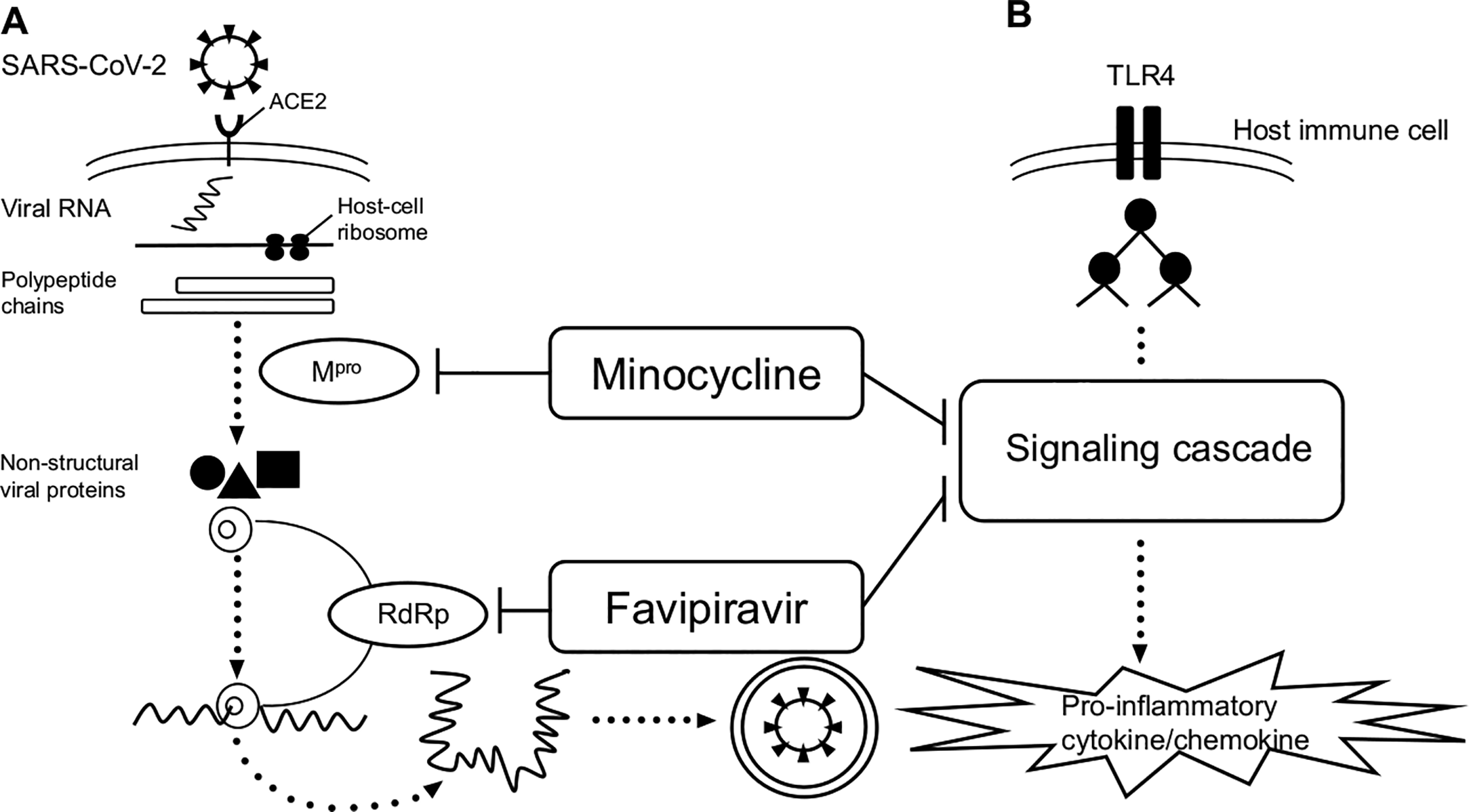
Hypotheses on the mechanisms of antiviral and anti-inflammatory effects of favipiravir and minocycline on SARS-CoV-2 infection. (A) Once the viral RNA enters the cell, the host’s ribosome converts the viral RNA into long proteins. The virus creates protease enzymes, which uses to cut up the long strands into functional proteins. Replication and transcription proteins emerge from a complex in the host vesicle. RdRp generates viral RNA and RNA that is translated by the host into new viral proteins. The new virus is packaged and released. (B) TLR4 recognizes lipopolysaccharides and other PAMPs and DAMPs on the cell surface, and signals are transmitted by a signaling cascade of molecules comprising the intracellular transduction pathway to produce inflammatory cytokines and chemokines. The hypothesis is that minocycline inhibits Mpro, favipiravir inhibits RdRp, and both of them inhibit TLR4 signaling, thereby suppressing inflammatory cytokine and chemokine production. ACE, angiotensin-converting enzyme; DAMP, damage-associated molecular pattern; Mpro, the main protease of SARS-CoV-2; PAMP, pathogen-associated molecular patterns; RdRp, RNA-dependent RNA polymerase; SARS-CoV-2, severe acute respiratory syndrome coronavirus 2; TLR, toll-like receptor.
Based on the above clinical trials and basic data, FVP and MIN have antiviral activity against SARS-CoV-2 and inhibit the excessive production of inflammatory cytokines associated with COVID-19 severity, suggesting that the combined administration of FVP and MIN may have more additive effects than FVP alone. Further validation in a large number of patients is warranted for clinical trials.
