Abstract
Purpose:
To determine the efficacy of brolucizumab in patients with aflibercept resistant neovascular age-related macular degeneration (AMD).
Methods:
This case series retrospectively collected data from October 2019 to March 2020 of 13 patients detailing aflibercept-resistant AMD. Patients received at least three doses of 2 mg/0.05 mL aflibercept on a 4-week dosing schedule before switching to three doses of 6 mg/0.05 mL brolucizumab on a 4-week schedule. Central subfield thickness (CST) and visual acuity (VA) were measured before and after each drug’s administration.
Results:
The mean baseline VA was logMAR 0.85 [0.65], which worsened to logMAR 0.86 [0.53] after aflibercept. The VA improved to a mean of logMAR 0.70 [0.44] with brolucizumab, but this fell short of significance (0.70, 95% CI: 0.40-1.00; P = 0.16). The mean baseline CST was 354.62 [93.79] μm, and it worsened to 369.31 [116.61] μm after aflibercept administration. Following the brolucizumab treatment, the mean CST significantly improved to 290.46 [89.84] μm (290.46, 95% CI: 228.21-352.72; P = 0.03). After brolucizumab use, there was no observed occlusive retinal vasculitis in this case series.
Conclusion:
Brolucizumab significantly improved CST, but the VA improvement fell short of significance. Three individuals experienced inflammation following brolucizumab use, but there were no observed occlusive retinal vasculitis. While brolucizumab may not be the first option to treat AMD, it holds value for patients who have been unresponsive to other anti-vascular endothelial growth factor agents.
Introduction
Age-related macular degeneration (AMD) is a late-onset, chronic eye disease that affects the central retina and is a leading cause of blindness in elderly patients.1 The global estimated prevalence of AMD is 8.69% of the population.2 Neovascular AMD, in particular, is present in 10–15% of all AMD cases and can quickly progress to blindness if left untreated.3 The identification of vascular endothelial growth factor (VEGF) in 1989 presented the opportunity for targeted therapy of neovascular AMD. In October 2019, the US Food and Drug Administration (FDA) approved the anti-VEGF drug brolucizumab to treat neovascular AMD.4 Three other anti-VEGF drugs have been FDA approved for intraocular treatment of elevated VEGF, including pegaptanib, ranibizumab, and aflibercept.5 The HAWK and HARRIER phase 3 clinical noninferiority trials confirmed that patients treated with brolucizumab had similar visual acuity (VA) post-treatment compared to patients treated with aflibercept. VA was stable in more than 50% of eyes treated with a dosage of 6 mg of brolucizumab every 12 weeks for 48 weeks. Aflibercept’s FDA approval is for 2 mg every 8 weeks, while the treatment regimen with brolucizumab allows for a lower number of required injections.6 Treatment with brolucizumab has also been shown to exhibit more favorable anatomical outcomes compared to treatment with aflibercept. At week 40 of clinical trials involving both drugs, brolucizumab had a superior drying effect as 61.0% of brolucizumab-treated eyes were completely dry without subretinal or intraretinal fluid as opposed to only 35.0% of aflibercept-treated eyes.7
Clinical trials of brolucizumab have also found evidence of intraocular inflammation and retinal vasculitis following treatment. By week 48 in the HAWK and HARRIER trials, 2.7–5.3% of patients treated with 6 mg of brolucizumab experienced intraocular inflammation compared to 0.3–0.8% of patients treated with aflibercept.8 After FDA approval of brolucizumab, there have also been reports documenting retinal vasculitis following the intravitreal administration of brolucizumab.9 , 10 However, there are many patients with neovascular AMD who fail first line treatment and continue to have persistent fluid after 1 year of treatment with 2 mg of aflibercept.11 There have been numerous studies demonstrating the effectiveness of aflibercept in anti-VEGF treatment-resistant cases of neovascular AMD, but little research has been conducted on the efficacy of brolucizumab in such cases.12–14
The purpose of this retrospective data analysis is to study the effect of brolucizumab on vision and anatomical outcome of patients with neovascular AMD who are resistant to aflibercept and to record any brolucizumab-related adverse events.
Methods
A retrospective review of patient charts with neovascular AMD was conducted at Springfield Clinic Eye Institute in Springfield, IL, and the Retina Macula Institute in Torrance, CA, between October 1, 2019 and March 4, 2020. Ethical approval for this study was waived by the Springfield Committee for Research Involving Human Subjects (SCRIHS) because it is a retrospective analysis of deidentified patient charts. An informed patient consent was not sought for the present study since it was a retrospective examination of patient charts. Data of 13 eyes from 13 different subjects were included in the analysis. All patients were first administered at least three doses of 2 mg/0.05 mL of aflibercept, with each dose being 4 weeks apart. They were switched to brolucizumab once found to be resistant to aflibercept. Resistance to aflibercept was defined as either the presence of intraretinal fluid 28 days after the third monthly injection of aflibercept was administered or if the interval between doses could not be extended from 4 weeks to 8 weeks secondary to loss of efficacy with longer intervals. They were then given three doses of 6 mg/0.05 mL brolucizumab on a 4-week dosing schedule. Data were collected post third injection to evaluate the response to change in therapy. Changes in vision at baseline and post-treatment were found by measuring the central subfield thickness (CST) and the logarithm of the minimum angle of resolution (logMAR) as a measure of VA. The differences in CST and VA before and after the administration of each drug were compared, and statistical significance was determined using a paired t-test with the significance level set to 0.05. While no formal long-term data were included in this study, all patients continue to be monitored in the eye clinics.
Results
Demographics
There were a total of 13 patients treated with brolucizumab after failing to have notable improvement or continued intraretinal fluid following the use of aflibercept. The mean age was 78 years (SD: 10.83 years), and five of them were women (38.46%). Prior to treatment with aflibercept, three patients were treated with other anti-VEGF agents. Two patients received both bevacizumab and ranibizumab, while four patients received only bevacizumab.
Visual acuity
Figure 1 and Table 1 illustrate the changes in VA values after applying aflibercept and brolucizumab. Before the patients were treated with aflibercept, the mean baseline VA was 0.85 (SD: 0.65). Afterward, the mean VA worsened to 0.86 (SD: 0.53). The postaflibercept treatment point served as the baseline for prebrolucizumab. Following the use of brolucizumab, the mean VA improved to 0.70 (SD: 0.44); however, this did not meet statistical significance (0.70, 95% CI: 0.40–1.00; P = 0.16). Upon careful review of the individual patient data, seven patients either worsened or had no change in VA, while the remaining six patients had an improvement in VA from a mean value of 0.90 to 0.46 (0.46, 95% CI: 0.26–0.67; P = 0.055).

Change in mean logMAR VA at each interval.
Summarize results and outcomes for the patients
Central subfield thickness
Figure 3 and Table 1 highlight the alterations in CST for the patients. The mean CST before patients received aflibercept was 354.62 μm (SD: 93.79 μm). After the use of aflibercept, the mean CST worsened to 369.31 μm (SD: 116.61 μm), but this increase did not meet statistical significance (369.31, 95% CI: 298.84–439.77; P = 0.69). After brolucizumab was applied, the mean CST significantly improved to 290.46 μm (SD: 89.84 μm) (290.46, 95% CI: 228.21–352.72; P = 0.03). Figure 2 is a representative OCT illustrating substantial improvement in pigment epithelial detachment (PED) size from baseline. Closer examination of individual CST data revealed that three patients had their CST decrease by less than 20 μm, and two patients ended with a worse CST after receiving brolucizumab as compared to before. The average CST of the other eight individuals significantly decreased from 383 μm to 251.63 μm (251.63, 95% CI: 199.69–303.56; P = 0.015).
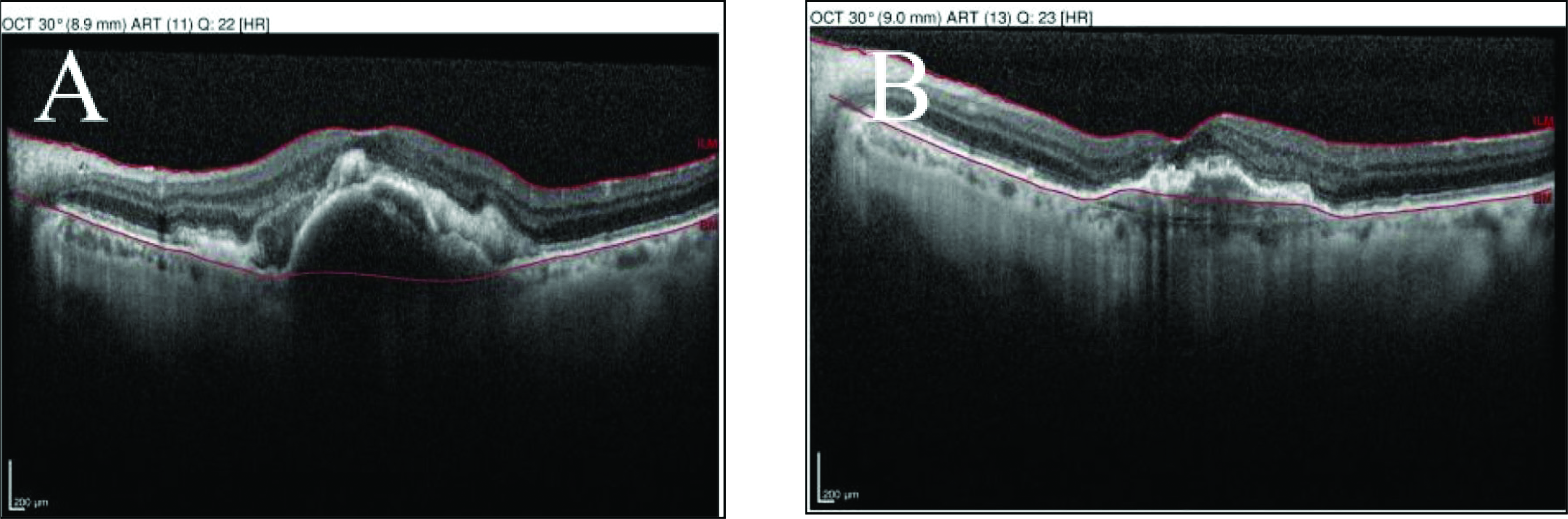
(A) An 82-year-old male’s optical coherence tomography showing substantial subretinal hyperreflective material and large PED before receiving brolucizumab. (B) Four months after starting brolucizumab treatment, the patient had less subretinal hyperreflective material and a reduction in the size of the PED.

Change in mean CST at each interval.
Adverse events
Our patients were observed on average for 87 days after receiving the first brolucizumab dosage. During this time, two additional doses of brolucizumab were administered, and they were monitored for signs of harmful effects due to the use of brolucizumab. Three people developed intraocular inflammation after treatment with brolucizumab. One developed white keratic precipitates ( Figure 4), while the others experienced a more typical intraocular inflammation with white cells in the anterior chamber. One of these patients was continued on brolucizumab, while the other two were switched back to aflibercept. Furthermore, one patient had a subconjunctival hemorrhage. None of the patients in the study had occlusive retinal vasculitis at the conclusion of this study.

A slit lamp examination demonstrates one subject with intraocular inflammation after the use of brolucizumab with white keratic precipitates deposited on the cornea.
Conclusions
This study evaluated the effectiveness of brolucizumab in neovascular AMD patients who were not responsive to aflibercept and switched over to brolucizumab. The study’s mean age of 78 is comparable to the mean age of 80 for the entire AMD patient population. Analysis of the VA and CST data was conducted before and after treatment as measures of efficacy. For VA, there was a trend toward improvement that did not reach statistical significance as the study was insufficiently powered to determine a significant change in VA given the relatively small sample size. In contrast to the improvement in VA, the improvement in CST did reach statistical significance following the use of brolucizumab (290.46, 95% CI: 228.21–352.72; P = 0.03). There was no relationship found between changes in CST and changes in VA.
This retrospective review suggests that brolucizumab may be a useful alternative for AMD patients who have worsened or have not responded to other VEGF inhibitors. Currently, there are limited treatment options for such patients. This may be related to the molecular properties of brolucizumab, which is a single-chain antibody fragment inhibitor of all forms of VEGF-A and can be concentrated to higher doses due to its high solubility and small molecular weight.4
Brolucizumab use is not without controversy given the well-documented side effects, including inflammation, retinal vasculitis, and significant irreversible vision loss with some cases of retinal vasculitis.4 All anti-VEGF drugs are reported to have some inflammatory response, and brolucizumab is reported to have an even higher rate of inflammation (6%) for certain subgroups with baseline antibodies to the drug.6,15,16 Additionally, there have been recent case reports of occlusive retinal vasculitis following intravitreal brolucizumab.4,17 In retinal practice, reports of higher rates of intraocular inflammation and especially reports of occlusive retinal vasculitis have curtailed the use of this novel anti-VEGF agent. However, in patients who have failed other anti-VEGF agents, there are limited therapeutic options, and brolucizumab may improve CST and trends toward improvements in VA. Given the greater risk of adverse effects, brolucizumab may not be a first-line agent for the clinician, but it may have a place in the retina specialist’s armament.
Patients with chronic recalcitrant exudative macular degeneration are the most difficult to treat, often with a combination of residual fluid, subretinal fibrosis, and atrophy of the retina, resulting in photoreceptor cell loss. Given that any of the changes associated with chronic recalcitrant exudative macular degeneration can result in poor long-term visual prognosis, it is not surprising that the patients in this study showed suboptimal response in regard to visual acuity.
The main limitation of our study is the limited sample size, and short longitudinal follow-up. This study was designed to provide data on the initial response to change in therapy. While no specific follow-up analysis is included in this study, the patients continue to be monitored in clinic. In the 6 month period directly following the study, two of the three patients who developed inflammation were switched to another anti-VEGF agent. All other patients were managed with brolucizumab and maintained their anatomic improvement with stable visual acuity from the end of the study period. As shown with many medications, efficacy has the possibility to wane over time. However, the early indications are encouraging in aflibercept nonresponders and offer a new treatment option to patients with recalcitrant chronic neovascular AMD. Further longitudinal studies with larger sample sizes will help evaluate the long-term safety of brolucizumab and identify the subset of patients that are more likely to benefit from receiving this drug.
All other authors of the paper certify they have no affiliations with or involvement with any organization or entity with any financial interest (such as honoraria, educational grants, participation in speakers’ bureaus, membership, employment, consultancies, stock ownership, or other equity interest, and expert testimony or patent-licensing arrangements) or nonfinancial interest (such as personal or professional relationships, affiliations, and knowledge or beliefs) in the subject matter or materials discussed in this manuscript.
