Abstract
Background:
Antiretroviral therapy (ART) is free in Brazil and indicated for all people living with HIV since December 2013 regardless of CD4 T cell count. The benefits of rapid ART include less risk of AIDS and non-AIDS conditions to the individual as well as lower transmissibility (Undetectable = Untransmittable). However, ART introduction may take months after diagnosis as an immediate treatment it is not a priority in many places.
Method:
We evaluated the time to loss of follow-up (LTFU) of 589 patients diagnosed in up to 6 months, stratified according to the time between diagnosis and ART initiation, as same week (SW), for those starting ART within 7 days of diagnosis, intermediate (I) between 8 and 30 days and late treatment (LT), for those treated after 31 days. Patients without ART withdrawal in the last 100 days were considered LTFU. Analysis was performed using the Cox proportional-hazards model.
Results:
The 589 consecutive patients admitted were stratified as SW 18% (n = 105), intermediate 20% (n = 119) and LT 62% (n = 365). The LTFU outcome was not different among strata (hazard ratio 1.02, 95%CI 0.71-1.46). However, LTFU was higher among non-Whites (hazard ratio 1.85, 95%CI 1.07-3.20) and transwomen (hazard ratio 2.97, 95%CI 1.35-6.55). Cox regression analyses adjusted for strata were associated for transwomen to stratum SW (hazard ratio 6.90, 95%CI 1.64-28.97) and for non-Whites to stratum LT (hazard ratio 2.34, 95%CI 1.30-4.22).
Conclusion:
Our study suggests that the rapid antiretroviral treatment is feasible in public units, without increases in LTFU for cases treated in the same week. However, special attention should be given to transwomen and non-Whites with public policies aimed at reducing racial and gender inequalities.
Introduction
WHO recommends “treating everyone,” antiretroviral therapy (ART) for all people living with HIV (PLWH), regardless of CD4 T cell count, based on evidence from clinical trials and observational studies, showing that early use of ART had resulted in better clinical outcomes compared to delayed treatment.1
The ambitious goal 90‐90‐90 proposed by the UNAIDS (Joint United Nations Program on HIV/AIDS) in 2014 aimed to interrupt the AIDS epidemic in 2030. It would be feasible if, in 2020, 90% of PLWH knew their diagnosis, 90% of them received ART continuously and 90% of them maintained viral suppression.2 Advances toward this goal varied across different countries, and overall, at the end of 2019, an estimated 81% of PLWH knew their status, 67% were receiving ART, and 59% had achieved HIV suppression. Although the number of deaths was the lowest in the last 20 years (690,000 HIV-related deaths in 2019), it is estimated that 1.7 million new infections still occur annually.3 The assumption that using voluntary universal anti-HIV testing and immediate ART would reduce HIV incidence and mortality was suggested in mathematical models since 2009.4
The Brazilian AIDS Program since the end of 2013 advocates the immediate start of ART, with the objective of reducing transmission.5 Free access is guaranteed for all PLWH, a right granted by the 1988 Federal Constitution, to ensure access to health goods and services in a universal, equal and integral way, through the Brazilian Unified National Health System (SUS). Financing has been a constant concern and per capita public health expenditure has been reduced in recent years.6
In the years that followed, studies showed the benefits of earlier ART initiation at any CD4 T cell count with lower risk to AIDS and to non-AIDS outcomes7,8 and that treatment could be a prevention strategy that helps to control the HIV epidemic (TasP, treatment as prevention).9–11
WHO strongly recommended that people with no contraindication to ART initiation, especially for people with very low CD4 T cell count, should be offered rapid ART initiation, including the option of same day initiation. The rapid onset of ART is defined as within 7 days after the diagnosis of HIV.12 Our study evaluated the feasibility in a public service of the strategy of starting ART within a week of diagnosis (same week) and its impact on patient’s loss to follow-up.
Materials and Methods
We conducted a retrospective study in an Infectious Diseases Outpatient Clinic in Santo André, metropolitan area of São Paulo, Brazil, which offers open and free access to HIV tests and treatment. The study was approved by the institutional ethical committee (CAAE: 69927317.8.0000.00590) and all participants provided informed consent.
Patients diagnosed within 6 months without previous use of ART were included in the study. Admission to the service was performed by the multidisciplinary team. The service prioritized the collection of exams and medical appointments of suspected cases of acute or symptomatic HIV infection (AIDS), as well as, cases with syphilis at the rapid test, or with suspicion of another sexually transmitted infection. However, universal collection and medical consultation within 7 days after admission was adopted in August 2018. The decision to start or postpone ART was at the discretion of the infectious disease physician.
The treatment initiation stratified the cases into three strata, according to the time between diagnosis and the ART initiation as early treatment, called same week (SW), within 7 days of diagnosis, intermediate (I) cases between 8 and 30 days, and late treatment (LT) if more than 31 days from diagnosis. Patients were categorized as a loss to follow-up (LTFU) if without withdrawal antiretrovirals in the last 100 days, according to medication dispensary electronic database SICLOM (Logistic Control System for Medicines of Brazil) in April 2020.
Data were obtained from clinical records and electronic databases, managed at a RedCap database. Anonymized data related to the study were retrieved and descriptive and statistical analyses were performed in Stata version 14.2 (Stata Corp LLC, College Station, Texas, USA). The results of continuous variables are described as medians, with the 25th and 75th percentiles (IQR), 95% confidence interval (CI 95%), and a significant level of P < .05, two-tailed, was applied to all analyses. Variables from time to treatment initiation strata were compared using the Kruskal–Wallis test for continuous variables and chi-squared (χ2) or Fisher’s exact tests for categorical variables, as appropriate, and Cox proportional-hazards analyses were performed to assess associations to the LTFU. Survival time was defined as the time (days) from the date of onset of ART to the date of the last ART withdrawal, censored in April 2020. To evaluated strength of associations, we evaluated (unadjusted) demographic and clinical variables, as sex (male), gender (transwomen), men who have sex with men (MSM), race (nonwhite), degree of education (college degree), age (years), viral load > 100,000 copies/mL, and late diagnostic (CD4 below 200 cells/mm3) at admission. Covariates significantly associated (
Results
The universal treatment of all PWLH was instituted by the Brazilian National STI/AIDS Program in December 2013, hence the study enrolled 589 patients admitted from December 2013 to April 2019, stratified according to the time between diagnosis and the ART initiation; 18% (105/589) initiating within a week (same week), 20% (119/589) between 8 and 30 days (intermediate), and 62% (365/589) after 30 days, late treatment stratum. Figure1 shows the distribution of cases according to the year of inclusion in the study and strata.
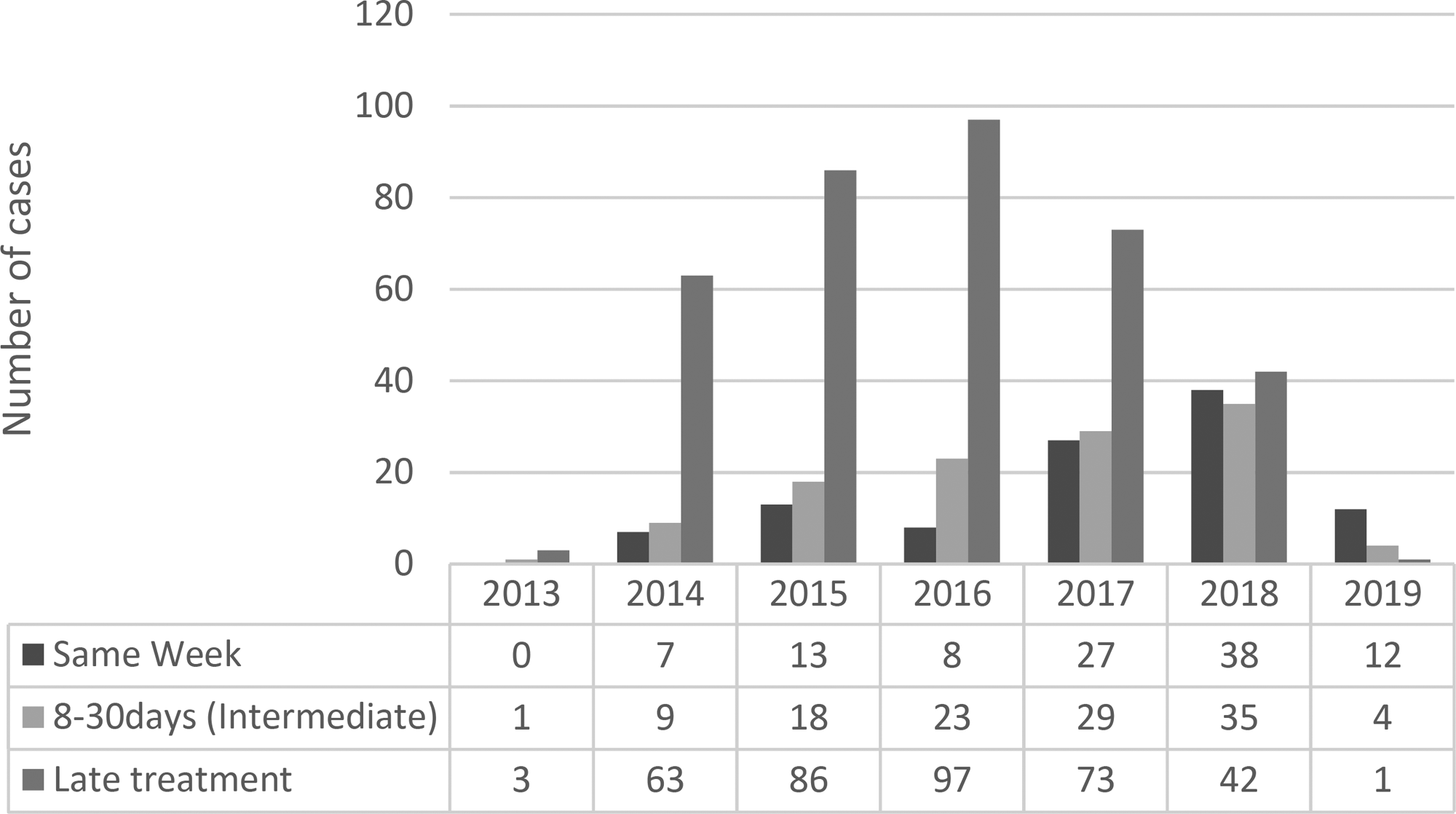
Year of inclusion of study cases according to treatment initiation after as same week, intermediate and late treatment strata.
The majority of patients (79%, 465/589) had HIV infection diagnosed by a third-generation rapid diagnostic test (RDT3) as part of routine testing. In 1% (6/589) of cases, initially nonreactive RDT3 was diagnosed in the acute/early phase of HIV infection, 5/6 of those are in the same week group. The fourth-generation rapid diagnostic test (RDT4, HIV-combo, Abbott®), when available, allowed start treatment immediately in 2 cases, confirmed with the viral load as cases of early infection. Another 4 acute infections were diagnosed only by molecular biology (viral load—VL), during which time RDT4 was not available.
Table1 shows the demographic and laboratory data at admission according to treatment initiation strata. Most were young (median 31 years old), white (55%), male (83%), 74% of them are MSM, generally with only basic education, and 18% with a completed college degree. There were no statistical differences between the groups. Late diagnosis, defined as a CD4 count of fewer than 200 cells/mm3 at diagnosis, was more frequent among heterosexuals (either male or female) 31% (71/223) as compared to 12% (42/360) among MSM (p < 0.001).
Socio-demographic and laboratory data at admission stratified according to the time between diagnosis and initiation of antiretroviral therapy.
MSM, men who have sex with men; CD4 T cell count presented as absolute value (cells/mm3) and dichotomized as above or below 200 cells/mm3. Viral Load presented as Log10 of the number of copies/mL (c/mL) and dichotomized as above or below 100,000 c/mL. Values are presented as number of cases and proportion. Continuous variables are expressed as the median and interquartile range (IQR 25th-75th). Variables were compared using Kruskal–Wallis test for continuous variables and chi-squared (χ2) or Fisher’s exact tests for categorical variables, as appropriate.
Table2 shows parameters of patients linking to clinical service, the period between the events involved in the linking process that impact the inclusion of cases in different stratum, as the time between diagnostic and admission, time from diagnostic and first scheduled appointment and from diagnostic and ART initiation.
Number of days for linking parameters stratified according to time from diagnosis to initiation to antiretroviral therapy
ART, antiretroviral treatment; Continuous variables presented as the median and interquartile range (IQR 25th-75th). Statistical analyses performed using Kruskal–Wallis test.
The first VL available after ART initiation there was very variable and ranged from 1 to 1439 days (median of 48 days, IQR 32-104), but was obtained earlier in the SW group (median of 34 days, IQR 27-51) than in the Intermediate group (median of 49, IQR 29-100) or LT stratum (median of 56 days, IQR 34-113) (p < 0.001). Due to this variability, the time to achieve viral suppression was not evaluated in the study.
The period in ART (time from ART initiation to the last medication dispensed) was longer in the late treatment stratum (median of 1231 days, IQR 840-1604) when compared to same week (median of 789 days, IQR 519-1195) and to intermediate (median of 973 days, IQR 633-1373) (p < 0.001). Figure2 shows the Kaplan–Meier survival curve with the proportion of LTFU comparing same week, intermediate, and late treatment strata.

Kaplan–Meier survival curve with proportion of loss to follow up (LTFU) comparing treatment initiation as same week, intermediate and late treatment strata.
Table3 shows the unadjusted Cox regression model for available demographic and laboratory variables and of the three strata of time between diagnosis and ART initiation. Transwomen, MSM and nonwhite were variables with an association (p < 0.2) to LTFU and were further evaluated at an adjusted Cox regression (Table4).
Cox regression model: Unadjusted evaluation of the association of study variables to patients loss to follow up (LTFU) including treatment initiation strata (same week, intermediate, and late treatment), demographic and laboratory variables.
Time to antiretroviral (ARV) initiation from admission stratified as same-week, intermediate (8-30 days) or longer; MSM, men who have sex with men; CD4 T CD4 T cell count, VL: Viral load.
Values at admission.
Adjusted Cox regression analysis of loss to follow up (LTFU) according to time to ARV initiation.
Adjusted analysis of variables showing associations (p < 0.2) at unadjusted analyses. Time to antiretroviral (ARV) initiation from admission stratified as same week, intermediate (8-30 days) or longer; MSM, men who have sex with men.
Discussion
In this study, we documented the feasibility of immediate, or at least in the same week, ART introduction and evaluated the potentially deleterious impact of this policy in suppression rates and patient retention. Some factors limit early treatment, such as the loss of diagnostic opportunities, which can be suggested by the high percentage of late diagnosis, 19% in our studied group, 27% in Brazil in 2020 (Available from: http://indicadoresclinicos.aids.gov.br/, accessed April 23, 2021). Although this problem is multifactorial, it may be reinforced in our country at some units that do not routinely offer immediate HIV testing, and rather mischaracterize rapid testing by scheduling this test, leading to many losses of diagnostic opportunities. Unavailability of immediate or rapid collection of pre-ART admission exams, as the CD4 count test, in which the sample cannot be stored for more than 24 hours is another limitation to identify cases with advanced disease and new testing technologies, as CD4 Pima (Abbott) or another point of care technology,13 could favor the immediate ART initiation, without the need to return on another day, which may favor linkage to care14 and improve management of advanced disease cases.
On the other hand, different studies suggest that 10-50% of HIV transmission events occur during acute and early HIV infections (AEHI),15–18 period when antibodies are undetectable (acute infection) or inconclusive (early infection),18 being missed opportunities for rapid treatment. Although not the aim of the study, same week strategy allows targeting this population may contribute to point-of-care initiatives to diagnose and treat AEHI, reducing the time from HIV diagnosis to viral suppression.19,20 In our study, six were diagnosed as AEHI, RDT4 (HIV-combo, Abbott®) was performed in 2, allowing them to start treatment on the same day, while other cases had their treatment delayed while waiting for the results of the viral load. RDT4 can be an alternative, a point-of-care of low cost compared to RNA-PCR in the diagnosis of AEHI.21
Despite the limitation of the Brazilian public system (SUS), our study, conducted on rather typical infectious diseases and STD reference unit, demonstrates the feasibility in a real-world public health clinic to treat quickly, shortening the time for viral suppression, without impairing retention. Our study documents the lack of deleterious effect of immediate treatment, as suggested by some.22
Other groups have also documented a benefit and did not identify harm in speeding treatment initiation. However, a negative impact of this immediate treatment strategy has been suggested. The same day ART initiation for HIV-infected adults was studied in 13,038/32,290 South African records with analysis of routine data and was associated with 30.1% LTFU in comparison to 22.4% (n = 7,261), 19.8% (n = 6,534), and 21.9% (n = 5,534) among clients initiating ART 1-7 days, 8-21 days and ≥22 days after HIV diagnosis, respectively (p < 0.001). However, ART initiators on the same day were younger (p < 0.001), more likely women (p < 0.001), higher initial CD4 counts (p < 0.001) compared to clients who started ART later after the diagnosis of HIV.22 Distinct to our study, in which there was no statistical difference in median age (p = 0.3), CD4 T cell count at admission (p = 0.1) and sex, with a predominance of males (p = 0.04) between the treatment initiation strata. Different cultural and socioeconomic aspects might impact these strategies and need further evaluation and adaptation to local conditions. Probably having the patient understand and get involved in the treatment is central to any strategy outcome.
Pilcher etal. studied 39/86 patients who began ART within 24 hours in San Francisco, time to viral suppression (TVS) was significantly faster (median 1.8 months) among intervention-managed patients when compared with patients treated in the same clinic under prior recommendations for universal ART (4.3 months; p < 0.001) and LTFU was similar in the intervention (10.3%) and nonintervention groups (14.9%) during the study.23 Although our results are in line with their findings, the proper assessment of suppression was hampered by the lack of systematic collection of samples for viral load after the introduction of ART.
Coffey etal.23 evaluated long-term viral suppression rates for patients who initiate immediate ART through a RAPID program of San Francisco study. They found viral suppression (lower than 200 copies/mL) of 90% at last viral load, measurement over a median of 1.09 years.24
Gomillia etal. evaluated 16/47 with rapid ART initiation, defined as within 7 days. The median TVS decreased from 77 days (2.6 months) in nonrapid starters to 55 days (1.8 months) in rapid ART starters (p = 0.03).25
Another study conducted in Botswana suggests the feasibility, acceptability, and safety of rapid ART initiation in low- and middle-income country settings. One year after ART initiation, 1,472 of 1,627 individuals (90.5%, 95% CI 87.4-92.8) who linked in the standard ART period were in care and had a viral load of fewer than 400 copies/mL, compared with 578 of 631 (91.6%, 95% CI 88·1-94.1) in the rapid ART period (risk ratio 1.01, 95% CI 0.92-1.11).26
We need to consider that our study, as most references discussed, reflects a situation before the COVID-19 pandemic and adjustments accordingly may be necessary.27
Conclusion
Our study suggests that swift treatment, within the same week, is a feasible strategy in a typical Brazilian public health service, with no difference in the LTFU compared to postponing treatment. However, operational studies are needed to expand these observations and to adapt the strategies to local conditions. Attention should be given to transwomen and non-Whites including adaptations of public policies aimed at reducing racial and gender inequalities.
Footnotes
Acknowledgments
The authors would like to thank the Same week working group: Norberto Camilo Campos and Giselle Ibete Silva López-Lopes from Virology Center, Adolfo Lutz Institute, São Paulo, São Paulo, Brazil; Andreia Moreira dos Santos Carmo, Delma Aparecida Molinari, and Valeria dos Santos Candido from Santo André Regional Center, Adolfo Lutz Institute, Santo André, São Paulo, Brazil; Ana Carolina Gaidis, Ana Paula de Angelo Guimarães, Aurea Pereira Rodrigues, Debora Fernanda Vichessi, Deise Lopes Arantes, Elis Muriel Marques Monti, Guilherme Penteado Teixeira, Humberto Barjud Onias, Juliana Camargo Botter Zabosqui, Laura Ballesteros Bao, Luiz Eduardo Andrade Alkimim, Mara Mirna Ramires Moreno, Marcia Cristiane Roma, Mauricio Tavano Bacal, Olivia Olinda dos Santos, Tania Isabel Ferrazzo and Thiago Sanches Brumatti from Infectious Diseases Outpatient Clinic, Santo André Health Secretary, Santo André, São Paulo, Brazil.
