Abstract
Background
: Sentinel lymph node (SLN) imaging is an established tool, while axillary reverse mapping (ARM) for early breast cancer is still evolving. Lymphedema is a vexing complication that can be avoided if ARM node is preserved.
Objective
: Our primary objective is to study the factors influencing ARM node identification in Positron emission tomography (PET) -reported node negative early breast cancer patients, and our secondary objective is to study the incidence of lymphedema in 18Fluoro Deoxy Glucose node negative (i.e., noncancerous node) breast cancer patients undergoing axillary lymph node dissection (ALND) by preserving the ARM node.
Materials and methods
: Forty-eight arms of 45 breast cancer pooled patients with FDG negative nodes underwent sentinel lymph node, SLN imaging using the 99mTc Sulfur colloid (99mTc SC, filtered), prior to surgery to rule out microscopic nodal disease. All patients were subjected to ARM on the day of surgery prior to mastectomy ± ALND.
Results
: Factors (technical and clinicopathological) affecting ARM node identification were analyzed. Colloid preparation was optimized based on heating temperature, duration of heating, radiochemical purity, labeling efficiency, and particle size. Ideal parameters were identified. 18 patients underwent both mastectomy and ALND. 99 SLN were harvested. ARM nodes were successfully identified in all patients. 98 percent of ARM nodes were found at locations different from breast draining nodes, i.e., SLN. No significant differences were observed between the incidence of SLN metastases and clinico pathological features.
Conclusions
: ARM is yet to be recognized as an imaging tool in routine practice. This study shows that standardization of technique can successfully avoid adverse factors that can affect ARM node identification. Only 2 out of 45 patients (4%) developed lymphedema, which is quite encouraging, indirectly validating the hypothesis that arm draining node is different from nodes draining the breast; hence, preserving ARM node can avoid lymphedema.
Introduction
With the availability of better staging techniques like FDG PETCT and sentinel lymph node biopsy (SLNB), recent concept includes surgical de-escalation of the axilla in breast cancer. Using PET alone, studies have proven that disease-free survival is high in early stage breast cancer patients with PET negative nodes.1 As predominant lymphatic drainage pathway from the breast cancer is toward the axilla ( Fig. 1), SLNB is routinely used in preoperative staging. It is far superior to FDG PETCT in identifying low volume nodal metastases. Subjecting early breast cancer patients to axillary lymph node dissection (ALND) exposes most of them to unnecessary morbid outcomes, such as arm lymphedema, nerve dissection leading to axillary numbness, and shoulder abduction deficits.2 Kim et al. have studied the role of ALND in patients with clinical node negativity (cN0).3 They found that 70–80% patients have pathologically free nodes (pN0). Instead of relying on clinical methods alone, FDG PETCT is a well-established and reliable screening tool that can be used to select patients for ARM node preservation during ALND.

Graphical representation of breast draining lymph nodes.
Lymphedema occurs due to skeletonizing of vessels and disruption of arm lymphatics lying in and around the SLN in axillary region during ALND. Reverse mapping of axilla forms the basis of ARM procedure. Mapping depicts the migration of 99mTc SC from webspaces through lymphatic channels toward axilla to reach the “ARM node.” This ARM node/s belongs to lateral axillary group, i.e., humeral or deltopectoral nodes and invariably confined to three different locations ( Fig. 2).4 ARM is a novel concept in preventing limb edema in treated breast cancer patients using surgery or external beam radiotherapy.

Lymphatic drainage of upper limb with rectangular area highlighting the location of ARM nodes.
Based on the hypothesis that arm draining node is different from nodes draining the breast, we set out to study the onset of lymphedema in patients with preserved ARM node/s. Although evidence for and against ALND is mounting, retaining the arm draining node may benefit low risk group of early breast cancer patients. As concept of ARM and ARM node detection is relatively new, studies are very few. With this background, we set out to study the factors influencing ARM node identification in FDG PET node negative early breast cancer patients. Our second objective was to determine the incidence of lymphedema in this group of patients.
To confirm the absence of nodal metastases, all patients were thoroughly screened clinically and underwent whole body FDG PETCT prior to SLN imaging and biopsy for accurate nodal staging of disease. PETCT was critically interpreted to look for any PET positive axillary nodes close to the usual location of ARM node.4 , 5
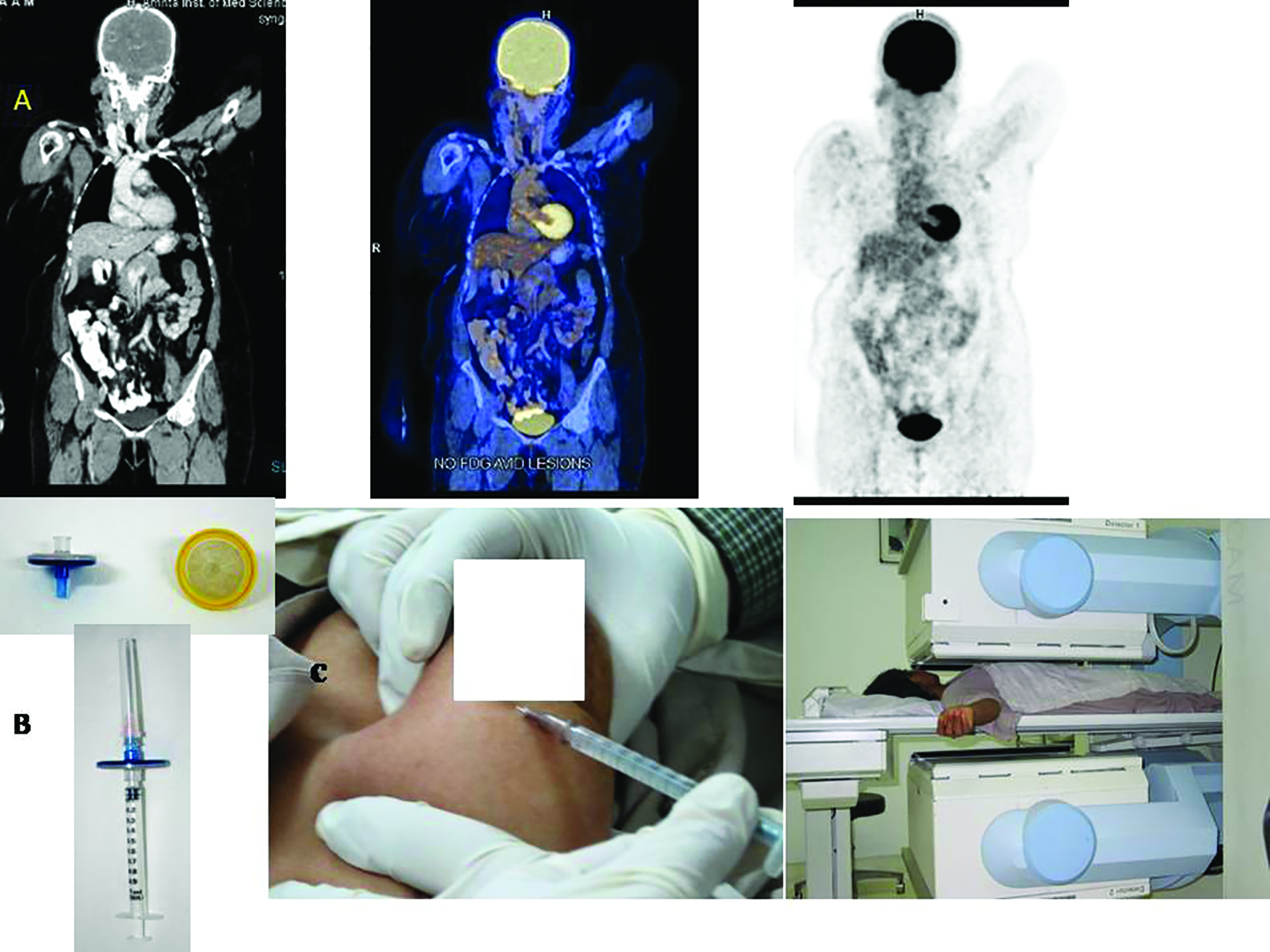
(A) Whole body FDG PETCT (head to mid thigh)—from left to right Contrast Enhanced CT, fused and PET only images showing no nodal or distant metastases. (B) Millipore filter and 1 ml syringe fitted with Millipore for reducing particle size. (C) 99mTc sulfur colloid (filtered) injections (4 in number) for SLN imaging. Patient positioned under Gamma camera during imaging.
Internal Review Board (IRB) approval: not applicable. Being a conventional investigation done for early breast malignancies (SLN imaging, and biopsy and ARM), no separate IRB approval was sought.
Patients consent: Patients have given their consent for both investigations (SLN imaging, biopsy and ARM).
Materials and Methods
Inclusion criteria
Recently diagnosed early breast cancer patients, i.e., TIS, T1, T2, T3 with N0 nodes, were selected. Patients with cytological/histological confirmation of primary malignancy.
Exclusion criteria
Advanced breast cancer, i.e., T4, any N or M disease. Patient with synchronous/metachronous malignancies. Patients with recurrent breast carcinoma. Patients with previous surgeries of the upper limb, axilla, supraclavicular, or cervical region. Post-neoadjuvant chemotherapy.
45 consecutive early breast cancer patients scheduled for radical mastectomy were recruited for the study at various selected centers. Study was conducted from January 2018 to 2019. All patients were explained about the study procedure and gave their written consent to undergo all the three procedures (PETCT, SLN, and ARM)
Fig. 3.
PETCT Procedure
Whole body FDG PETCT for staging was performed in euglycemic status. 0.1 mCi (millicurie)/kg body weight of FDG was injected intravenously. Sixty minutes later, PETCT (with oral and IV contrast) was performed (head to mid-thigh level at seven or eight bed positions) on a 16 slice Biograph (Siemens) PETCT system. Images were interpreted by senior nuclear medicine physician, and SUV Max (standardized uptake value maximum) of each lesion was documented. All were female patients with a median age of 48.5 years (20-74 years).
ARM Pilot Study
Labeling procedure
Freshly prepared 99mTc Sulfur colloid (99mTc SC) is recommended for the investigation. Non-radioactive SC is available as a freeze-dried cold kit. Each reconstituted reaction vial (10 mL) can be used as a multidose preparation. Each vial of sulfur colloid has three components: (1) component A (reaction) vial; (2) component B; (3) component C. Each kit contains the sterile nonpyrogenic, nonradioactive ingredients necessary to label 99mTc Technetium for diagnostic use. Freshly eluted Sodium Pertechnetate (Na 99mTcO4) Injection in isotonic saline is mixed with these components. Component A vial contains 0.5 mL solution of 0.3 N hydrochloric acid, and component B vial contains 1 mL solution of gelatin/hemaccel, thiosulfate, and perrhenate. Component C has 1 mL of phosphate buffer. Desired radioactivity dosage is added to component A. 40 mCi of Na99mTcO4 is needed to obtain 1 mCi of 99mTc SC Filtered (in 0.9% of sodium chloride) after filtration. 0.5 mL of vial B is added to the reaction vial after withdrawing equal volume of air from the vial. Vial is kept in boiling water bath for 3-5 minutes. After removing the vial from the water bath, cooled to room temperature. 0.5 ml of component C was transferred to the reaction vial and mixed well. Once the final preparation is completed, activity for ARM is dispensed in 1 mL disposal syringe and transferred to the operation theater in lead-lined syringe carrier. This aseptic preparation of radioactive colloid is undertaken in the hospital radio pharmacy as per good manufacturing practice.
Filtration of colloids
Filtration of the final radioactivity preparation is mandatory to reduce the particle size of colloids for uniform distribution through lymphatic channels and lodging in lymph nodes. A 33 mm diameter sterile single use, syringe filter with a 0.22 µm pore size having hydrophilic polyethersulfone membrane is used. It is marketed as Millex from Merck Millipore Ltd, Tullagreen, Carrigtwohill, Co, Cork, Ireland, Catalogue number SLGP033RS. It is a fast flow low binding filter with a filtration area of 4.5 cm2.
Estimation of radiochemical purity (RCP)
RCP values were determined at different time intervals after filtration of RP using paper chromatography. A drop of 99mTc-SC was placed on the bottom of a strip of Whatman 3MM chromatography paper. The strip was placed in a test tube of 85% methanol, taking care to keep the 99mTc-SC spot above the solvent line. A rubber stopper was placed on top of the tube to maintain equilibrium. Once the solvent front reached the top of the strip, the strip was then removed from the tube and cut in half. Both the top and bottom portions of the strip were measured separately in a well counter, and the activity was recorded. According to the United States Pharmacopeia, 92% bound and above has satisfactory RCP.
Identification of radioactive counts by gamma probe
Of the various intraoperative probes used for minimally invasive surgeries like SLNB and parathyroidectomies, the most widely used type is the general-purpose “gamma” probe, designed for counting of radionuclides emitting gamma rays. Major requirement of a gamma probe is to provide clear instantaneous and cumulative radioactive counts. ARM was performed using a cordless handheld battery-operated gamma probe (Seno Rx, marketed by World of Medicine, Germany). Probe was set at 140-kilo electron volt for 99mTc photopeak. The tip of the probe was covered with a sterile wrapper during surgery. After injection, the probe is placed at 35° angulation and traced along the injected arm from the site of injection to trace the lymphatic channels. The colloid transit is almost instantaneous, thus no time should be lost in tracing the lymphatic flow. The ARM node is identified as the node/s with at least 10 times the background radioactive counts (as shown in the display screen and high audio alarm emitted from the gamma probe). Counts are recorded per unit time with the probe in the operative field over the node. A background tissue count is also recorded with the probe pointing away from the injection site/adjacent organ, etc.

Demonstrates Sentinel node imaging - Static and SPECTCT images, displaying four quadrant injections and identification of axillary group of sentinel node on the right side.
A pilot study for ARM was separately conducted previously that enrolled 10 cases of breast cancer apart from the proposed study patients, posted for mastectomy. This was based on the major concern with SLNB that care must be undertaken to avoid failure to identify the ARM node. During this initial learning curve in our institution, technical factors that can interfere with ARM node identification were studied. Time taken to identify ARM node from the time of web space injection was also noted. (a) Labeling efficiency, (b) RCP, (c) colloid particle size, (d) volume/dose of radioactivity in syringe, and (e) best method of using the gamma probe by surgeons during preoperative ARM were determined.
SLN/ARM procedure
Prior to day of surgery, pooled patients were scheduled for SLN imaging followed by biopsy on the subsequent day. Less than 1 mCi of 99mTc SC (filtered) was prepared in 0.4 mL volume. Four peri areolar (intradermal) injections were administered (0.1 mL volume at each site). Injected area was massaged gently. Immediate static followed by SPECTCT imaging was performed using a dual head ECam gamma camera. Number and location of hot node/s (SLN) were tabulated. SLN was marked over the skin using indelible ink, and gamma probe counts from axilla (over the site of SLN) were also noted ( Fig. 4).
On the day of surgery, before anesthesia induction, the counts over the SLN skin marking were rechecked with gamma probe (to check for loss of counts due to radioactive decay) and documented. ARM procedure was undertaken prior to mastectomy ± ALND. For ARM procedure, after induction, less than 1 mCi of 99mTc SC (filtered) was injected in the second/third web spaces of ipsilateral hand as a single or equally divided dosages. Immediately after the web space injection, the arm draining lymphatic channels were mapped from the site of injection. This was done either by taking the readings (on display screen) of radioactive counts emanating from lymph channels or based on audio alarm by moving the gamma probe across the fore arm and upper arm in a linear fashion. The ARM node was identified as a node showing high counts (more than 10 times the background) situated in one of the following three locations, as shown in Figure 2, as reported in our previous study4:
lateral to the subscapular artery pedicle,
above the second intercostobrachial (ICB) nerve, and
just below the axillary vein.
18 patients required ALND based on PET positive nodal disease. During axillary dissection, the hot SLN/s and ARM node location were rechecked using gamma probe to ensure that both lie at different locations and do not coincide. Radioactive counts from both these nodes were reassessed using the gamma probe. The hot SLN/s were harvested, and ex vivo counts were checked and sent for histopathological examination. ARM node and its surrounding lymphatics were left untouched. Surgeon was requested to direct the probe away from activity surrounding the areolar injection (four equidistant injection sites) while checking for SLN counts. Counts were recorded per unit time with the probe in the operative field, over the node before excision (in vivo) and after excision (ex vivo). A background tissue count was also recorded.
Intraoperatively, two patients displayed high radioactive counts from closely placed breast and arm draining nodes (coinciding SLN and ARM nodes, which could not be differentiated based on count statistics); hence, both nodes were harvested. After successful excision of SLN node, the axillary bed was checked for residual activity. Thus, after removal of one SLN node, another hot node may still be present below or close to it, which may be missed if background radioactivity check was not performed.
After completion of mastectomy with or without ALND, excised SLNs were labeled and sent to pathology for interpretation. The nodes were cut longitudinally into two halves; half were frozen for immediate examination, and the other half were embedded in paraffin, sectioned, and examined with microscopy after hematoxylin and eosin staining and immunohistochemical staining.
Lymphedema assessment and follow-ups
All patients were recommended to undergo follow-up visits at specified time intervals (3, 6, 9, and 12 months post-operatively for the first year and then every half year thereafter) for arm circumference measurements or appearance of any cancer-related disease recurrence. Patient’s appreciation of symptoms related to lymphoedema was collected. Arm circumference was measured at eight locations on both limbs using a measuring tape. Standard locations measured were midmetacarpal, the wrist, forearm (at 5 and 10 cm above the wrist), elbow joint, and in the arm (at 15, 10, and 5 cm above the elbow). All measurements were performed by the same health personnel. Lymphedema was considered when there was a 2-cm or greater increase in ipsilateral arm measurements compared with contralateral arm measurements at any of the above-mentioned locations on ipsilateral limb. The median follow-up period was 26.7 (range, 24-30 months posttreatment) months.
Quality of life (QoL) assessment was done based on a set of questions distributed to each patient at each follow-up under the lymphatic filariasis specific QoL questionnaire that is based on 2009, Institute of Applied Dermatology (IAP), Kasaragod, Kerala, India. Patients’ response was scored under different headings (mobility, selfcare, usual activities, disease burden, psychological health, pain/discomfort, and social participation). Each section has various sub-questions, which need to be scored as follows: 4, 3, 2, 1, 0, and 0 (no problem, mild, moderate, severe, unanswered, or not relevant). The overall response given by each patient was charted. Scoring was done as per the IAP recommended guidelines. Overall QoL = total score (4× no. of questions answered) × 100. The domain score was calculated as follows: total score on domain/4 × number of questions answered in the domain) × 100. Maximum domain score is 100. Higher the score depicts better QoL. Similarly, all questions may not be relevant to every patient, so the percentage analysis is done based on the queries answered in each domain.
Data collection and statistics
All clinical data were analyzed using SPSS (IBM Corp., Armonk, NY) 24.0 software. The data obtained from the RCP testing were summarized as mean ± SD and analyzed using an unpaired t test. Samples within the ideal range for sentinel node lymphoscintigraphy were summarized as a percentage with corresponding 95% confidence interval (CI) and compared between heating time and collection times using the χ2 test. The nonparametric Kolmogorov–Smirnov (KS) test was used to compare the distribution of particle sizes between subgroups defined by the amount of heating time and sampling time.
Results
Technical factors affecting the identification of ARM node
The labeling efficiency was determined, and ideal colloid particle size of radiopharmaceutical was ascertained for ARM procedure in our laboratory. Boiling of freshly prepared 99mTc SC at 100°C for 3-5 minutes duration was found to produce ideal colloid size with optimum count statistics. Due to heterogeneity of colloids, a membrane filter (0.22 µm, Millex) was used to obtain a uniform size of majority of colloid particles. After cooling the labeled sulfur colloid, filtration was performed to obtain the final product. The optimum dosage to achieve a good count rate during ARM surgery was studied. Differing dosages were tried (0.5, 1, 2, and 3 mCi) with no significant change in dosage of 0.5 and 1 mCi; higher doses were found to produce count saturation during probing. Procedure was standardized, and good count statistics was obtained (using gamma probe) when less than 1 mCi of 99mTc SC in a volume of 0.4 mL was injected.
For the pilot study, 10 patients were analyzed for the standardization of ARM technique. Average RCP values (±SD) for 99mTc-SC with 5 minutes heating were 98.4 ± 2.0% and 98.1 ± 1.6% for 0 hour and 6 hours, respectively (n = 10) ( Table 1). Average RCP values for 99mTc SC with 3 minutes heating were 98.4 ± 3.8% and 96.6 ± 3.4% for 0 hour and 6 hour, respectively (n = 10). Median particle sizes for the 3 minutes heating at 0 and 6 hours were found to be 24 and 35 nm, respectively. Median particle sizes for the 5 minutes heating at 0 and 6 hour were 29 and 27 nm, respectively. The proportion of particles was similar between the different heating temperatures (91% for 3 minutes heating at 0 hour and 89.1% for 5 minutes heating at 0 hour, P = 0.1854). Heating of SC for the average time of 3.6 minutes ± 30 seconds with labeling efficiency of >88% and particle size of 50-70 nm was found to be ideal for successful ARM identification in our study.
Colloid particle sizes and radiochemical purity checked at different time points and duration of heating in the pilot study for 10 patients
Mean ± standard deviation for labeling efficiency using 99mTc SC was 95.5. RCP difference pre- and post-filtration of the 99mTc SC preparation was –1.5 ± 1.6%. Particle size was found to be 30-50 nm in 89% patients. Nine percent of patients had admixture of small and larger colloid particles. Preparation was finally filtered to achieve homogenous colloid distribution. We did not filter colloids in remaining 2% patients, and the size of particles was >150 nm, resulting in poor lymphatic transport through the arm (no counts obtained) perceived by gamma probe even at 75 minutes post-injection. However, ARM node was identified in all patients. Deft movement of the gamma probe over the upper limb in the proximal upward direction immediately after web space injection was recommended. Ensure that the direction of probe is away from injection site (to avoid count interference). Average time taken for the identification of ARM node over skin surface with gamma probe was 51 ± 06 seconds (post web space injection).
To summarize the technical factors, most important factor that can affect the identification of ARM node was found to be particle size while using 99mTc SC. Standardization of technique and skill of surgeon are also factors that can affect node identification.
Clinicopathological factors and ARM node identification
22% of our patients were less than 35 years of age, majority being multiparous (nulliparous:multiparous:uniparous = 5:80:15%). Histologically, infiltrating ductal cancers were predominant followed by ductal Ca in situ, infiltrative lobular and other rarer types like papillary, lymphoma (69:18:08:05%). As per FDG PETCT findings, the T stage categorization was 45% in stage I, 33% under stage II, while 22% were designated under stage III with tumor sizes ranging between 0.5 and 6.5 cm, median 2.8 cm. 59% of patients were hormone positivity in our series ( Table 2).
Clinicopathological and SLN characteristics’ along with arm circumference and quality of life scores
45 females with PET negative nodes and TIS, T1, T2, and T3 staging for breast cancer, aged between 20 and 72 years, median age 48.5, were studied. The most common site of tumor was the upper outer quadrant accounting for 72.0% of cases. Predominantly, the tumors were of solid type (95.2%), whereas both solid and cystic type of lesions were seen in rest of the cases on histology. Multicentricity of breast primary was identified congruently by PET and CT part of PETCT with contrast) alongwith histology in 97.8% patients. No significant differences were found between the incidence of SLN metastases and clinicopathological features, including age, clinical T and N staging, and histological type. However, we did see a positive correlation between the number of positive axillary lymph nodes and clinicopathological data, which was also confirmed by other studies.6 Clinicopathological factors in early primary breast malignancies, however, do not contribute significantly to the ARM node identification.
Out of 45 patients who underwent mastectomy at different centers, 18 required additional ALND based on FDG PETCT/per operative axillary basin examination. All SLN identified via subareolar breast injections were located in axilla. None of the patients demonstrated any internal mammary hot node on SLN imaging. Location of the ARM node was fairly constant in 84% of the cases and invariably was found within the area bounded by the subscapular vessels medially, the ICB nerve inferiorly and the axillary vein superiorly. Data from our previous publication showed only 0.1% of ARM node harbored metastatic disease in early breast cancer,4 ( Fig. 5). This encouraged us to retain the ARM node in this study to look for lower incidence of lymphedema as envisaged. ARM node and SLN were identified in all patients during surgery (100%). 99 SLNs draining breast were counted and harvested. Only 2% of harvested SLN in patients who underwent ALND harbored metastatic cells. Detection of ARM node is, however, not dependent on the tumor type, age of patient, or stage of the disease as shown by our study.

ARM procedure and gamma probing used for identifying the ARM node.
98% of ARM nodes were found at locations different from breast draining SLN. Under gamma probe guidance, ARM nodes were preserved, while SLN nodes were harvested. Two patients with overlapping high radioactive counts in axillary nodes (ARM/SLN) were harvested for biopsy confirmation. ARM nodes in these two patients were reported to be negative on biopsy. Follow-up FDG PETCT showed no disease progression.
There was no significant difference in arm circumference in 89% patients at 24-30 months post-treatment. Only two patients (4%) developed lymphedema in this series (those displaying a difference of more than 2 cm from contralateral arm were considered to suffer from lymphedema). The incidence of lymphedema was not higher in those who underwent mastectomy and ALND when compared with those who underwent mastectomy alone (P = 0.003). QoL assessment scores were high with a range of 167-182, median 172. Most of the patients were comfortable and demonstrated no great difficulty in managing their daily activities post-operatively.
Discussion
Studies have demonstrated that ALND was associated with 7–77% risk of lymphedema,7,8 and SLNB, although only a few axillary nodes removed, was also associated with 3–13% risk of lymphedema.9 Our study showed only 4% developed lymphedema at 24-30 month follow-up, which is far less than those quoted in literature.8 This indirectly confirms the theory that by preserving the lymphatic channels and arm draining node, one can reduce the occurrence of lymphedema. Many authors have reported the benefit of preserving ARM nodes10; however, caution needs to be exercised in selecting the right cases.
However, maximum effort has been undertaken to accurately stage the patients before preserving the ARM nodes, as ARM node could harbour microscopic metastases that may lead to nodal recurrence.11 , 12 In this study, by performing SLNB and ARM techniques, one could help distinguish ARM from SLN nodes. This proved useful in staging and reducing unnecessary ARM node resection. Moreover, we could use the ARM technique to establish the location of ARM node and differentiate arm draining node (ARM node) from breast draining SLN nodes in PET negative nodal disease, thereby ruling out the presence of nodal metastases.
Conclusions
ARM is yet to be recognized as an imaging tool in routine practice. Standardization of ARM technique is mandatory for the success of the procedure. Clinicopathological factors do not contribute significantly to the ARM node identification in early breast cancers. This study indirectly substantiates the hypothesis that arm draining node is different from nodes draining the breast; hence, preserving ARM node can avoid lymphedema. Choosing the right patient for ALND with the combination of FDG PETCT, ARM, and SLNB can make a great difference in clinical outcome of early breast cancers.
