Abstract
Locusta migratoria is a useful model to carryout out laboratory investigations in vivo to study sickness behavior and infection of the central nervous system. Analogous anorectic behavior in rats and locusts, as well as the resemblance in the presence of anatomical and physiological barrier protecting the brain in both species and a large size of locust to inject/extract biomaterials, makes it a highly valuable tool to investigate molecular mechanisms of disease at an early stage of research.
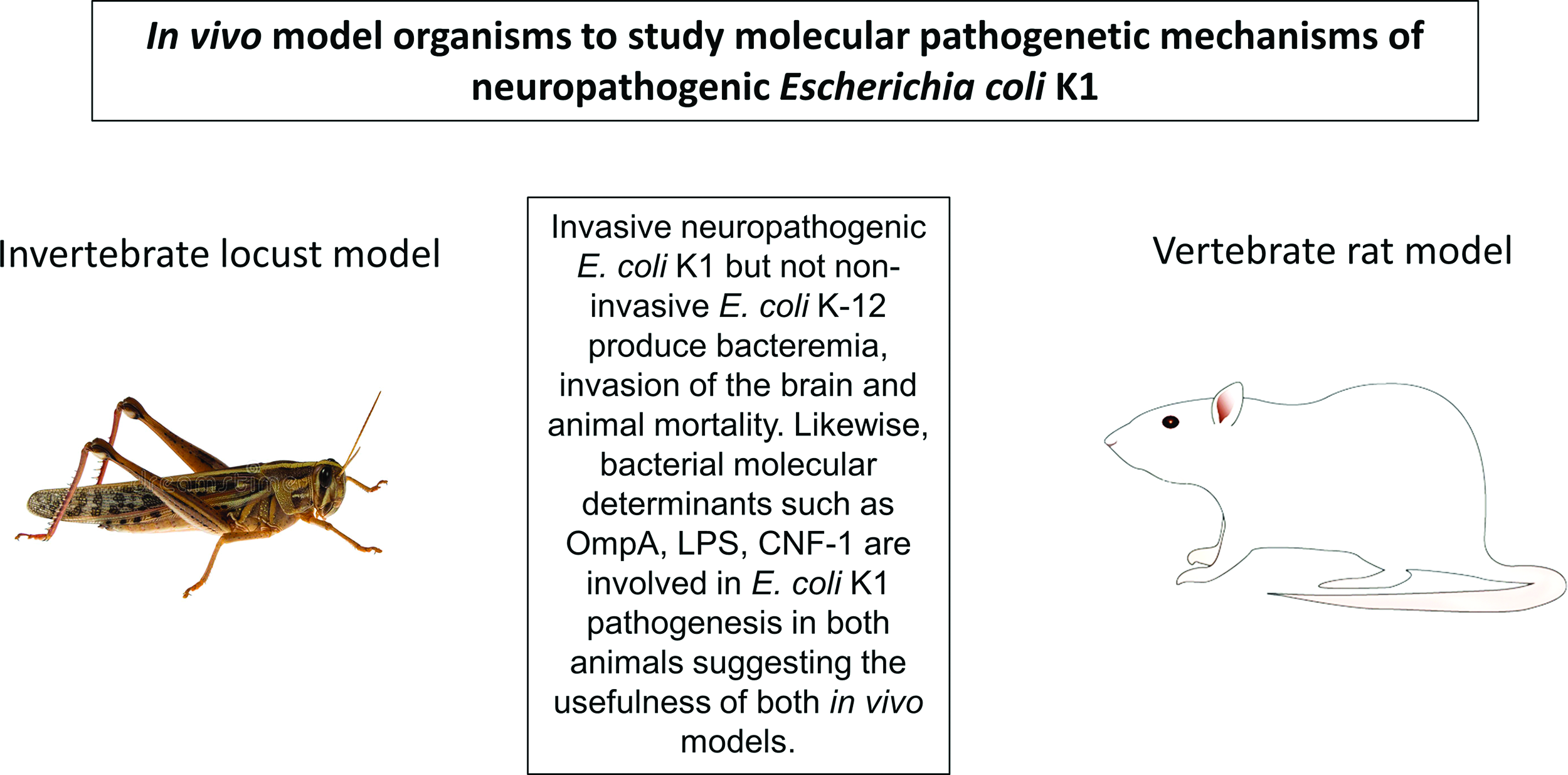
Invertebrates offer promising tools to investigate molecular mechanisms of microbial diseases in vivo. 1 In particular, the use of insects such as locusts offers several advantages, including legislative approval, cost-effectiveness, acceptance by the scientific community, and the public at large. Being pests, there are no ethical issues associated with their use for laboratories investigations in vivo. As whole organism approach is critical to validate findings arising from in vitro studies, insects such as locusts can provide useful model system prior to testing any leads in a vertebrate model system. Although insects such as Drosophila spp. are preferred, however given their small size, they may not be a feasible choice for experimentation in many laboratories. Larger insects such as locusts make it possible to carryout dissections, determine weight and size changes, feed selective/measured diet, collect hemolymph/feces, inject/extract materials, and quantitate other parameters with relative ease. Furthermore, insects such as locusts possess hemolymph–brain barrier (similar to the blood–brain barrier observed in vertebrates), ie, a physical barrier impeding the entry of microbial pathogens and/or large compounds circulating in the hemolymph into the brain.2,3 Notably, the requirement of molecules involved in bacterial pathogenesis in locusts and rats is significant. This was demonstrated using adult African migratory locusts, Locusta migratoria. 4 Locusts were injected with E. coli (2 × 106 colony forming units [c.f.u.] suspended in 20 µL) into the hemocoel of the locust abdomen by inserting the needle into the intersegmental membrane between two abdominal terga, and locusts mortality was recorded.2,3 When injected with neuropathogenic E. coli K1, the mortality rate was 100% within 72 hours, while nonpathogenic laboratory strain, K-12 produced less than 10% mortality. These findings are significant and clearly showed that invasive and noninvasive E. coli strains can be differentiated in vivo using invertebrate locust model, and these findings corroborated with the vertebrate rat model.5 Furthermore, the bacterial determinants such as lipopolysaccharide (LPS), outer membrane protein A (ompA), cytotoxic necrotizing factor-1, that are known to be responsible for E. coli K1 pathogenesis in vivo using invertebrate rat model, were tested in the invertebrate locust model. The results demonstrated that E. coli K1 expressing the rough LPS was ineffective in producing locust infection that is similar to the noninvasive E. coli K-12.2,3 Similarly, the locust mortality was reduced significantly when injected with ompA-deletion mutant as well as cnf1-deletion mutant compared with the wild-type neuropathogenic E. coli K1.2,6 As bacteremia is a prerequisite to E. coli K1 meningitis, hemolymph was collected from the cuticle and arthrodial membrane at the base of the hind leg. At 48 hours of postinjection, bacteremia was significantly lower in locusts injected with ΔompA further confirmed validity of the invertebrate locust model to study E. coli K1 pathogenesis. Moreover, E. coli K1 expressing rough LPS and/or deletion of ompA did not invade the locust brain, and these findings are well-correlated with the mortality findings.2 Overall, these findings showed that bacterial determinants responsible for bacterial invasion of the brain and mortality between locusts and rats exhibited significant resemblance.2,5,6 It is evident that the similarities and the underlying molecular mechanisms support the use of locusts as a useful in vivo model to study E. coli pathogenesis and possibly other pathogenic microbes.
It is also interesting to note that locusts exhibit anorectic effect.7 When injected with laminarin (a storage glucan), locusts did not eat. A reduced dose of laminarin does not affect feeding behavior of locusts, suggesting that laminarin-mediated sickness behavior observed in locusts is likely through the release of serotonin. This is confirmed when anorectic effect of laminarin was reversed by injection of mianserin. This is consistent with findings observed in rats where mianserin and serotonin were shown to affect feeding behavior in rats.8 It is intriguing that sickness-induced anorexia could involve serotonin, which also acts as a diuretic hormone in some insects. As for vertebrates, it is not clear whether cytokines play a role in anorectic behavior of locusts; however, the similarities observed in locusts and rats are remarkable, and further studies are warranted to explore the underlying molecular mechanisms.
Taken together, the use of an in vivo model to study pathophysiology is necessary to determine the value of underlying molecular mechanisms. Though vertebrate models remained a preferred choice, given their physiological resemblance, insects such as locusts offer a valuable in vivo tool to undertake laboratory experimentations to investigate potential leads generated through in vitro experimentation, for their role in pathological events in selected diseases.
Footnotes
Acknowledgment
Authors are grateful to G. Goldsworthy for valuable and critical discussions.
