Abstract
Purpose:
To assess the effectiveness and tolerability of a new latanoprost 0.005% benzalkonium chloride (BAK)-free nanoemulsion.
Methods
: A nonrandomized multicenter study was conducted between January 2017 and October 2017 in 15 ophthalmological clinics from Argentina. This study enrolled 113 patients with primary open angle glaucoma or pseudoexfoliative glaucoma, who had been under treatment with any BAK-preserved latanoprost ophthalmic solution 0.005% as monotherapy for at least 6 months, switch to administer latanoprost BAK-free nanoemulsion. Primary outcome was intraocular pressure (IOP) lowering efficacy at 4, 8, and 12 weeks after treatment switch. Secondary outcomes included clinical signs and symptoms of ocular surface damage (Ocular Surface Disease Index (OSDI), conjunctival hyperemia, Schirmer I test, tear film break-up time (BUT), corneal epithelial fluorescein staining, and tear meniscus height).
Results:
In total, 103 patients (198 eyes) concluded the study. Mean IOP values between baseline and at week 12 were 14.63 (95% confidence interval (CI), 14.25 to 15.01) and 13.65 (95% CI, 13.32 to 13.98). After a 12 weeks of treatment switch, mean Ocular Surface Diseases Index score decreased by 11.53 units (95% CI, 15.18 to 7.88; p < 0.0001), and the number of eyes with conjunctival hyperemia decreased by 27.8% (95% CI, 17.93 to 37.64; p < 0.0001). Mean break-up time increased by 1.75 seconds (95% CI, 1.37 to 2.12; p < 0.0001) 12 weeks after treatment switch, and the number of eyes with corneal staining decreased by 19.7% (95% CI, 11.68 to 27.72; p < 0.0001).
Conclusions:
The new nanoemulsion maintains the decreasing intraocular pressure effect, with significant improvements in signs and symptoms of ocular surface disease.
Prostaglandin analogues (PAs) represent the first choice in the medical treatment of glaucoma, and latanoprost stands out for offering favorable balance in terms of intraocular pressure-lowering, efficacy, and tolerability.1–3 PAs are known to produce some no severe systemic adverse events and local adverse events such as conjunctival hyperemia, stinging, foreign body sensation, and eyelid and orbital changes (periorbitopathy associated with prostaglandins).4–6 These effects cannot be fully attributed to the active component of formulations, as there is an extensive body of literature supporting the inflammatory and toxic effects related to preservatives used in them.7–9
Benzalkonium chloride (BAK) is a commonly used preservative in ophthalmic formulations, although it has been shown to contribute significantly to ocular surface and deeper internal structures damage.10,11 BAK is an amphiphilic molecule that increases prostaglandin solubility in water as well as acts as a detergent disrupting the lipid film continuous layer, also affecting the mucin layer, losing tear film integrity, which is reflected in a diminish break-up time (BUT) and ocular surface toxicity signs in glaucoma patients who use BAK-preserved antiglaucomatous medication.8,9,12–15 It has been established a strong link between treatment tolerability and compliance.16,17 Adherence to treatment is particularly important in chronic conditions such as glaucoma, where patients typically lose peripheral vision and may completely lose vision unless consistent lifelong treatment is received.18,19
In recent years, different pharmacological strategies of developing preservative-free formulations avoiding BAK have been probed their “in-vitro and clinical” benefits in tolerability and efficacy in the treatment of glaucoma.20–25 Nevertheless, eliminating BAK from the formulation poses pharmacotechnical challenges. Considering that nanoemulsions are excellent formulations to deliver lipophilic drug substances to the eye, as they provide enhanced ocular penetration.26 So, we have developed an original pharmaceutical form of latanoprost 0.005% BAK-free nanoemulsion, and our purpose was to demonstrate that this new formulation has an equivalent ocular hypotensive efficacy to traditional BAK-preserved latanoprost 0.005% ophthalmic solutions and is able to improve signs and symptoms of ocular surface disorders in the treatment of primary open angle glaucoma.
Methods
This study was a Phase IV, nonrandomized, prospective, open-label, single-arm, multicenter, 12-week study with monthly visits. This study was registered in clinicaltrials.gov under the identifier NCT03331770 and was designed and conducted according to the criteria set by the declaration of Helsinki and applicable local regulations (Resolution 1480/11 of the Argentine Ministry of Health). It was carried out in 15 medical centers of Argentina, and the study protocol was approved by the Ethics Committee of Glaucoma Research Foundation before the start of the study. A written informed consent was obtained from all patients prior to participation in this study.
This study began on January 6, 2017 and was completed on October 6, 2017. Patients were recruited by the director of the glaucoma department of each participant’s institution, by reviewing their clinical charts and accordingly the inclusion/exclusion criteria. This study enrolled 113 patients, of which 103 (198 eyes) finished the study. Participants were adult patients (≥18 years old) of either sex with primary open angle glaucoma or pseudoexfoliative glaucoma, who had been under treatment with any BAK-preserved latanoprost ophthalmic solution 0.005% as monotherapy for at least the previous 6 months. Additional inclusion criteria were corneal pachymetry between 520 and 580 µm and signed informed consent. Exclusion criteria were as follows: known allergy to any component of the nanoemulsion; secondary glaucoma or angle closure glaucoma; previous glaucoma surgery or trabeculoplasty within 1 year of the study; cataract surgery within 6 months of the study; a history of uveitis or intraocular inflammation; corneal alteration that interferes with tonometry; pregnancy, lactation, or intention to become pregnant during the study. Every patient who met inclusion criteria received the informed consent.
At the baseline visit (day 0), patients were switched from any BAK-preserved 0.005% latanoprost solution to the new BAK-free 0.005% latanoprost nanoemulsion with potassium sorbate as preservative (Louten® Emulsión, POEN S.A.U). A total of three bottles of the new product were freely given to each patient, delivery in every participant center, by the chief of glaucoma department. Dosing schedule with the new formulation was that the same patients had been using with BAK-preserved latanoprost solutions (one drop at night on each eye in one or both eyes).
As primary outcome intraocular pressure lowering efficacy was evaluated after 4, 8, and 12 weeks of treatment with BAK-free latanoprost, intraocular pressure was assessed on monthly visits that took place at the same time of the day. It was measured in millimeters of mercury (mmHg) using a calibrated Goldmann Applanation tonometer (GAT) after having applied topical anesthesia and fluorescein staining. The treatments were considered to have equivalent ocular hypotensive efficacy if the two-sided 95% confidence intervals (CIs) were within –1.5 and +1.5 mmHg from baseline mean at all times.
As secondary outcomes, clinical signs, and symptoms of ocular surface damage were evaluated, follow-up assessments were performed at 4, 8, and 12 weeks of BAK-free latanoprost treatment. Evaluation of ocular surface damage included Ocular Surface Diseases Index (OSDI) and biomicroscopy by slit lamp examination. Conjunctival hyperemia was graded using a 4-point scale ranging from 0 to 3 units (0 = none, 1 = mild, 2 = moderate, and 3 = severe). Additional evaluations included Schirmer I test (millimeters/5 minutes), BUT, and corneal epithelial fluorescein staining, which was graded using a three-point scale ranging from 0 to 2 units (0 = absence of keratitis, 1 = inferior punctate keratitis, and 2 = central punctate keratitis). Using the slit-lamp, the tear meniscus height was measured and graded on the lower eyelid, as 0 (diminished: <0.2 mm), 1 (normal: 0.2 mm), or 2 (increased: >0.4 mm). The participant researchers who made the clinical examinations were experienced ophthalmologist. Moreover, a uniform criterion was adopted for describing the characteristics of the ocular surface in a standardized and reproducible manner, based in part in the guidelines published by Bron et al.27 Also, every adverse event was recorded, and reasons for abandoning the protocol were also registered.
Sample size was calculated based on the primary end point at 12 weeks. Initial calculation determined that a sample size of 195 eyes would provide a power of 90% or greater at the 0.05 significance level to ensure that the limits of a two-sided 90% CI exclude a difference in means of more than 1.5 (common SD, 4.5). Assuming a 10% dropout rate, a sample size of 215 eyes, efficacy data, and signs and symptoms of dry eye syndrome are presented as mean and CI. The chi square test was used for categorical variables. For ordinal variables, two-tailed repeated measures ANOVA with Bonferroni correction was used. All tests were two tailed, and the significance was set at p < 0.05. All statistical analyses were conducted using SPSS statistical software, version 21 (SPSS Inc). The data from each center were collected in excel files, and two authors of this work (MdP and SP) processed them.
Results
Safety evaluations
A total of 113 patients from 15 ophthalmology centers were enrolled, and 103 patients (198 eyes) finished the study according to the protocol (flow chart is shown in Fig. 1). Six patients (5.3%) discontinued treatment with BAK-free latanoprost because of ocular itching, increased tearing, blurred vision, strange body sensation, dry eye, or allergic reactions. Four patients were lost to follow-up. Neither nonocular treatment emergent adverse events nor unexpected ocular adverse events were reported. Reported treatment emergent adverse events were eye itching, epiphora, blurred vision, foreign body sensation in eye, dry eye, or allergy.
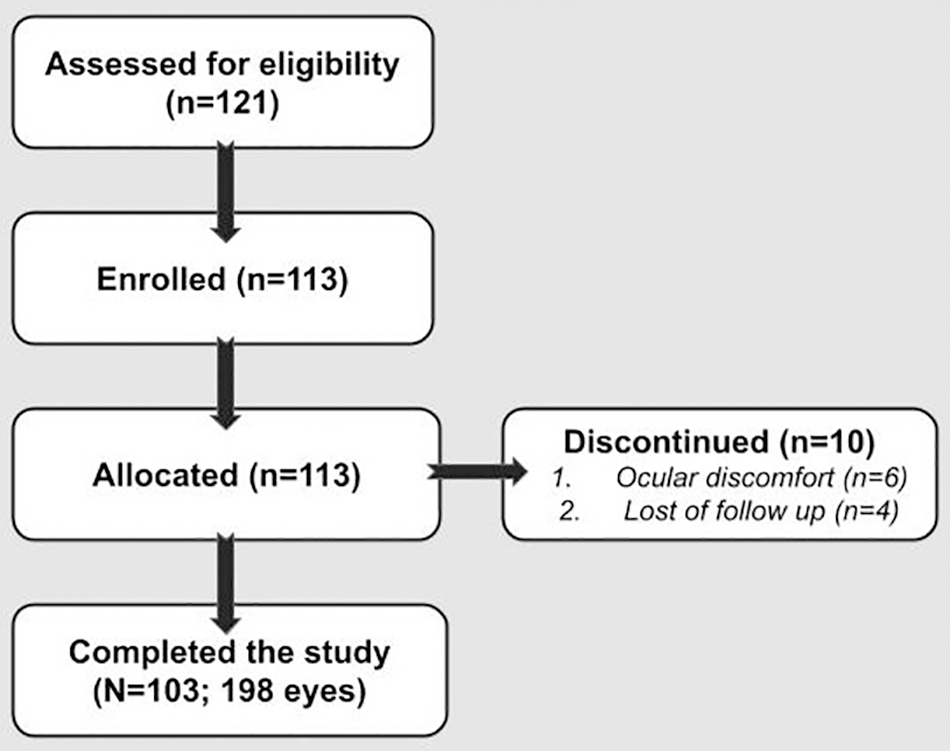
TREND flowchart of the latanoprost 0.005% BAK-free nanoemulsion clinical trial (n = number of patients).
Intraocular pressure
No patient had IOP values higher than 20 mmHg while using BAK-free latanoprost nanoemulsion. Mean IOP values at week 0, 4, 8, and 12 were 14.63 (95% CI, 14.25 to 15.01), 14.00 (95% CI, 13.65 to 14.35), 13.73 (95% CI, 13.38 to 14. 07), and 13.65 (95% CI, 13.32 to 13.98), respectively. After 12 weeks of treatment with BAK-free latanoprost nanoemulsion, IOP difference with respect to week 0 was –0.98 mmHg (95% CI, –1.37 to –0.54; p < 0.0001) ( Table 1). Although differences in IOP were statistically significant at all times with respect to week 0, the 95% CIs were within –1.5 and +1.5 mmHg from baseline mean at all times. These results indicate that there are no significant differences between treatments for changes in the primary outcome.
Latanoprost 0.005% BAK-free nanoemulsion: intraocular pressure and ocular surface parameters at different time-points
Results: (95% CI). Abbreviations: IOP, intraocular pressure; OSDI, Ocular Surface Disease Index; BUT, break up time; two-tailed repeated measures ANOVA.
Clinical signs and symptoms of ocular surface damage
Statistically significant differences were found between the mean OSDI score obtained at weeks 0, 4, 8, and 12 (Table 1). After 12 weeks of treatment with BAK-free latanoprost nanoemulsion, the mean OSDI score decreased by 11.53 units (95% CI, –15.18 to –7.89; p < 0.0001) (Table 1). The result represents a statistically significant change from baseline and measurable clinical improvement in OSDI score.
Analysis of conjunctival hyperemia showed that after 12 weeks of treatment with BAK-free latanoprost nanoemulsion, the number of eyes with conjunctival hyperemia decreased by 27.8% (95% CI, 17.93 to 37.64; p < 0.0001; 128 eyes showing conjunctival hyperemia at week 0 vs 73 eyes with conjunctival hyperemia at week 12). Furthermore, as from week 8 after treatment switch, no patient presented with severe hyperemia ( Fig. 2).

Bar graphs of conjunctival hyperemia, corneal epithelial fluorescein staining, and tear meniscus height. (A) Conjunctival hyperemia degree at baseline, 4, 8, and 12 weeks from the switch of treatment expressed by number of eyes. (B) Corneal staining degree at baseline, 4, 8, and 12 weeks from the switch of treatment expressed by number of eyes. (C) Tear meniscus height degree at baseline, 4, 8, and 12 weeks from the switch of treatment expressed by number of patients.
Mean Schirmer I test values obtained at weeks 0, 4, 8, and 12 are shown in Table 1. After 8 and 12 weeks of BAK-free latanoprost treatment, there was a statistically significant increase in tear volume compared to week 0 (p < 0.01 for 0 vs 8; p < 0.001 for 0 vs 12). After 12 weeks of BAK-free treatment, the mean tear volume increased by 1.19 mm (95% CI, 0.47 to 1.90; p < 0.001).
Results of BUT measurements obtained at weeks 0, 4, 8, and 12 are shown in Table 1. At 4, 8, and 12 weeks after treatment switch, the mean BUT obtained with BAK-free latanoprost was significantly higher than the mean BUT at week 0 (p < 0.001). Mean BUT increased by 1.75 seconds (95% CI, 1.24 to 2.25) 12 weeks after treatment switch (Table 1).
Corneal epithelial fluorescein staining revealed an inverse proportionality between corneal staining and time elapsed since treatment switch (p < 0.001). Twelve weeks after starting treatment with BAK-free latanoprost, the number of eyes with corneal staining decreased by 19.7% (95% CI, 11.68 to 27.72; p < 0.0001; 61 eyes presenting corneal staining at week 0 vs 22 eyes with corneal staining at week 12) (Fig. 2).
Finally, tear meniscus height was assessed. The tear meniscus height was decreased at baseline in 29 patients (28.2%), and 12 weeks after treatment with BAK-free latanoprost, it was improved and only 23 (22.3%) patients remained with a decreased meniscus height. This represents a decrease in 5.8% (95% CI, –6.04 to 17.69), which did not reach statistical significance (Fig. 2).
Discussion
This study shows that the intraocular pressure lowering efficacy of a new BAK-free formulation of latanoprost in nanoemulsion was noninferior than BAK-preserved latanoprost eyedrops. Moreover, additional benefits were observed about clinical signs and symptoms of the ocular surface. Up to our knowledge, this is the first study reporting with an original nanoemulsion of BAK-free latanoprost.
BAK is a well-known substance used as preservative in the ophthalmic drug industry, which causes disruption of the tight junctions of the corneal epithelium, an effect that has led to BAK being considered an enhancer of drug penetration into the anterior chamber.28 Likewise their potential toxicity, it was also historically reported, as Wilson et al. published in 1975, emphasizing the need to investigate the effects of BAK in the ocular surface, as well as the necessity of developing other preservative substances.29 Wilson et al. have found in his in vivo experiments a “drying effect” of BAK, in rabbits and humans corneas. Since then, many studies have shown that the chronic use of BAK in ophthalmic solutions leads to a decrease in density of goblet cells, resulting in decreased mucin production and tear film stability and an increase in inflammatory cells infiltration on the conjunctival stroma, developing a concomitant chronic disease, in the ocular surface.16,17,30
Since the seventies to this new millennium, new drugs with antiglaucoma topical formulations have been discovered with a simultaneous growing of the knowledge about the importance of the ocular surface health. Symptoms of ocular surface disease can severely affect the quality of life of people.31 Ocular surface problems, as a glaucoma topical side effect, decrease the patient’s compliance to the glaucoma treatment and also the efficacy of the treatment.18 Because of that, ophthalmologist and the pharmacological industry take care about not only the hypotensive effect of a glaucoma treatment but also their chronic tolerability. Many studies confirm that BAK-free glaucoma treatments are better than BAK-preserved, reporting the noninferiority results.20–25,28,29 The present study provides evidence that BAK is not required in the formulation to maintain the efficacy of latanoprost. BAK-free latanoprost nanoemulsion is equally effective in lowering intraocular pressure, while it improves ocular surface parameters.
The original pharmacological formulation was used in this phase IV clinical study; it was an approved product by different regulatory offices in many Latin American countries, which is commercially available since 2017. This latanoprost BAK-free nanoemulsion is a colloidal system of two liquid phases of oil dispersed in water of small size droplets (50–250 nm) stabilized by surfactants, enhancing their pharmacokinetics properties to deliver the lipophilic prostaglandin analogue (latanoprost 0.005%), with increased ocular permeability and bioavailability. Potassium sorbate may meet the present need for alternative preservatives that are gentle to the ocular surface, as has been previously published,32 and compatible for use in multidose BAK-free prostaglandin analogue formulations. Long-term adverse effects have not been reported from the use of potassium sorbate, and the replacement of BAK by potassium sorbate has a beneficial effect on the ocular surface, with a significant improvement in tolerability and dry eye signs and symptoms in patients, without affecting intraocular pressure lowering efficacy. Furthermore, the nanoemulsion formulation is thermostable and provides additional benefits, since it avoids cold chain and can be stored at a temperature of up to 30°C for up to 2 years. All this could improve treatment compliance and help preserve patient’s quality of life.
There is an interesting development, which was published by Lallemand et al. in 2012,33 regarding a preservative-free cationic nanoemulsion of latanoprost 0.005%. It was proved their safety and tolerance in animal models.34 Their exploratory efficacy, tolerance, and safety in humans result from a phase II study that was published in 2017, only in a nonindexed PubMed journal, up today.35 Recently (2020), an experimental study was published evaluating the pharmacokinetic and pharmacodynamic properties of a travoprost nanoemulsion in rabbits.36 In the same study, they also evaluated safety and efficacy outcomes in a short term, obtaining promising results. After performing a complete online database research, until February 2021, there are no more published studies about nanoemulsion BAK-free with prostaglandin analogues for ophthalmic use, and the product evaluated in this work was the only one commercially available and approved in different countries.
The present clinical trial was finished in October 2017. After that, principal efforts were done to approve regulatory issues and legal aspects in different Latin American and Caribbean countries. Recently, during the longer lock down that underwent Argentina due to the COVID-19 emergency (since middle of March until December 2020), our group reviewed the final report of this clinical trial with the purpose of sharing results with the scientific community. The positive findings in this study after using a nanoemulsion BAK-free latanoprost treatment include a significant reduction in OSDI score, from moderate to mild, which suggests an improvement in dry eye symptoms. Also, switch to BAK-free latanoprost nanoemulsion significantly improved BUT. Furthermore, conjunctival hyperemia significantly decreased after BAK-free treatment. This decrease was mainly seen on severe and moderate hyperemia. Also, the number of eyes with corneal fluorescein staining decreased after treatment switch. These results suggest that BAK can increase OSDI by disrupting tear film, increasing conjunctival inflammation, and directly damaging the corneal epithelium, as many studies have previously described.20–23,25
Latanoprost acts as a prostaglandin F2 analogues to decrease intraocular pressure, principally interacting with the EP3 receptor, besides their action in others receptors, as EP1, RP2, EP3, and EP4.37 Our interpretations of the present results led us to hypothesize that the decreased conjunctival hyperemia is associated with an “encapsulation effect” of the active principle in the nanoemulsion, avoiding their interaction with the prostaglandin receptors allocated in the conjunctival tissue. In this way, nanocarriers leave the drug in their specific pharmacological bio-phase, without conjunctival inflammation as side effect. Although to confirm that, an experimental study must be developed.
In the present study, tear meniscus height increased after BAK-free treatment, but this increase did not reach statistical significance. Schirmer I test results significantly improved under BAK-free treatment, but this improvement was not clinically significant. This can be explained by the fact that study participants had good test results at the baseline visit (day 0), and that Schirmer I test measures basal and reflex secretion and has low sensitivity for detecting small changes in lacrimal production.
Adverse events were generally mild and self-limited. Six patients withdrew from the study due to local (ocular) expected adverse events. No serious treatment-related adverse events were reported.
The current study is subjected to limitations. The first limitation is the lack of a control arm. The use of control group or control arm in clinical trials has a key role in isolating the effects of the medical product from those caused by other factors. The second one is a performance bias because it is an open-label study, in which both the patient and investigator know the study medication. This knowledge generates a prejudiced idea about its efficacy both positively or negatively, distorting the results, especially when symptoms or clinical signs are measured without sufficiently objective and not very reproducible definitions. The third limitation is associated with the subjective evaluation of the ocular surface. Even when the participant ophthalmologists were experienced physicians, and they were also trained specifically for standardizing the ocular surface evaluation, with accepted clinical methods, all of them still have been subjective measurements.
In conclusion, the nanoemulsion of BAK-free latanoprost 0.005% has proved its capability of maintaining the intraocular pressure-lowering effect, adding benefits for the ocular surface. It will be interesting to perform a prospective randomized controlled comparative study with other BAK-free prostaglandin analogues, in a non-nanoemulsion formulation, comparing safety, efficacy, and objective measurements of the ocular surface.
MdP and MSP: design of the work, data collection, data analysis and interpretation, drafting the article, critical revision of the article, and final approval of the version to be published.
All coauthors have reviewed and approved of the manuscript prior to submission.
This manuscript has been submitted solely to this journal and is not published, in press, or submitted elsewhere.
Footnotes
Acknowledgments
The authors wish to acknowledge the contribution of Julia Brown Arnold, MD; Fernando Cataldi, MD; Gabriela Catallini, MD; Nora Chiari, MD; Juan Dalmagro, MD; Graciela Gonzalez, MD; Ignacio Lischinscky, MD; Celina Logioco, MD; M. Angélica Moussalli, MD; Pablo Nahun, MD; Federico Lucio Ribola, MD; and Delia Sivori, MD, and also of Rodrigo M. Torres, MD, PhD, for his scientific advisor.
