Abstract
Abstract
Background:
Immunotherapy using engineered T cells expressing chimeric antigen receptor (CAR) is used in an increasing number of cases. The CAR molecule has an antigen-binding domain usually consisting of an antibody-derived VL and VH domains. The arrangement of VL and VH domains has been shown to affect CAR characteristics and functions of the CAR-transduced T (CAR-T) cells.
Objectives:
This study aims to clarify whether the relative arrangement of VL and VH domains in CAR affects the exhaustion status of CAR-T cells especially bearing herpes virus entry mediator (HVEM)-derived co-stimulatory signal domain (CSSD).
Method:
We generated CARs bearing the HVEM-derived CSSD and the VL and VH domains derived from HIV-1 broadly neutralizing antibody VRC01 in different arrangements (VL-VH or VH-VL) as a model system, and compared effector functions and characteristics of the CAR-T cells.
Results:
In the transduced T cells, the VL-VH CAR was expressed at higher levels and led to more potent effector functions than the VH-VL CAR. Phenotypic analysis showed that the VH-VL CAR-T cells contained two populations: one with higher CAR expression and CAR-mediated tonic signaling associated with more exhausted phenotype, and the other with the opposite characteristics. Interestingly, the VL-VH CAR-T cells, although expressing higher levels of CAR, exhibited lower CAR-mediated tonic signaling associated with lower percentage of exhausted population, which led to higher expansion upon cognate antigen simulation. These results suggested that the VL-VH CAR-T cells appeared to escape exhaustion induced by CAR-mediated tonic signaling despite the high CAR expression.
Conclusions:
The arrangement of VL and VH domains in VRC01-based CAR appears to play important roles in not only functions but also exhaustion status of CAR-T cells with HVEM-derived CSSD. Therefore, preclinical validation of the relative position of VL and VH domains combined with various CSSDs in CAR could assist in developing effective CAR-T cell-mediated immunotherapy.
Introduction
Immunotherapy using human T cells transduced with the chimeric antigen receptor (CAR) gene, so-called CAR-transduced T (CAR-T) cell therapy, is expected to become a useful therapeutic intervention against otherwise intractable diseases. For example, the CD19-targeted CAR-T cells demonstrated remarkable efficacy against B cell malignancy,1 and the clinical application has been started. On the other hand, constitutive tonic signaling in the absence of ligands is an increasingly recognized complication for synthetic fusion receptors including CAR and can be a cause of poor antitumor efficacy, impaired survival, and reduced persistence of the CAR-T cells in vivo. 2 Thus, it is necessary to analyze the relationship between CAR characteristics and CAR-T cell activities to further improve CAR-T cell technology.
CAR consists of an extracellular antigen-binding domain combined with an intracellular signal transduction domain. The first generation CAR-T cells that have CD3ζ as a signal transduction domain have been shown to often become anergic and fail to elicit a potent immune response.3 The second and third generation CAR-T cells have been developed by adding one and two co-stimulatory signal domains (CSSDs) in the CAR, respectively.4 These CAR-T cells could successfully mimic the T cell receptor-mediated signal transduction upon stimulation by cognate antigens, leading to proliferation and activation.5 This indicates that a signaling assumed by the CSSD is a key event for CAR-T cells to exhibit potent efficacy.4 So far, the CSSDs derived from various co-stimulatory molecules, such as CD28 and 4-1BB, have been employed for constructing CAR.6,7
Accumulating reports have suggested the roles of herpes virus entry mediator (HVEM) in effector function and memory development of CD8 T cells.8,9 In addition, tumor cells that express anti-HVEM single chain Fv (scFv) induce a potent proliferation and cytokine production of co-cultured T cells.10 These findings have suggested that HVEM serves as a potent co-stimulatory molecule in T cells. Thus, we have tested HVEM as a CSSD in CAR-T cells and demonstrated that the HVEM-derived CSSD could be useful for generating potent CAR-T cells.11
To construct the extracellular antigen-binding domain of CAR, the scFv composed of the light- and heavy-chain variable domains (VL and VH, respectively) derived from a monoclonal antibody against target antigens has usually been used.4,12,13 It has previously been shown that the arrangement of VL and VH domains could affect the characteristics of the scFv expressed in Escherichia coli, such as expression level, antigen binding, cross-reactivity, immunotoxin activity, and viral neutralization activity.14–20 It has also been reported that the artificial GD2 and CD3-bispecific antibodies could redirect cytotoxic T cells to tumor cells and the activity depended on the arrangement of VL and VH domains.21 In another study, the functions of the CAR-T cells targeted to C-type lectin-like molecule-1 for human acute myeloid leukemia cells have been compared by changing the arrangement of VL and VH domains in the context of CAR with 4-1BB-derived CSSDs.22 Thus, the arrangement of VL and VH domains in CAR could affect its expression levels and CAR-T cell funcions.21–24 However, it is still unknown whether the arrangement of VL and VH domains in the context of CAR with HVEM-derived CSSD could affect the CAR-T cell functions and exhaustion status in particular.
In order to investigate this issue, we generated the CAR bearing VL and VH domains derived from an antihuman immunodeficiency virus type 1 (HIV-1) Env antibody VRC01 in different arrangements, namely, VL-VH and VH-VL, combined with the HVEM-derived CSSD as a model system and compared the effector functions and exhaustion status of the CAR-T cells expressing the respective CAR molecules. The results showed that the VL-VH CAR was expressed at higher levels than the VH-VL CAR, leading to enhanced effector functions and killing activities against the HIV-1 Env-expressing cells. Further analysis indicated that the VL-VH CAR-T cells contained a smaller proportion of exhausted cells than that of the VH-VL CAR-T cells. In addition, the VH-VL CAR-T cells contained two distinct populations of the cells: one expressing relatively high levels of the CAR associated with higher CAR-mediated tonic signaling and more exhausted phenotype and the other expressing lower levels of the CAR associated with lower CAR-mediated tonic signaling and less exhausted phenotype. Interestingly, the VL-VH CAR-T cells exhibited less exhausted phenotype associated with lower CAR-mediated tonic signaling than the VH-VL CAR-T cells, which led to higher proliferation upon cognate antigen simulation. The results suggested that the VL-VH CAR-T cells appeared to escape exhaustion induced by CAR-mediated tonic signaling despite the high CAR expression. Taken together, the arrangement of VL and VH in the extracellular domain of the CAR appears to play important roles in not only functions but also exhaustion status of CAR-T cells with HVEM-derived CSSDs. For developing potent and effective CAR-T cell-mediated immunotherapy, it may be necessary to properly design the extracellular structure including the arrangement of VL and VH domains in CARs with various CSSDs.
Materials and Methods
Ethics approval and consent to participate
This study was conducted according to the principles of the Declaration of Helsinki and with the approval of the Scientific Ethics Committee of the Dokkyo Medical University. Written informed consent was obtained from all subjects after oral explanation of this study.
Cell culture
Minimum essential medium (MEM), Dulbecco’s modified eagle medium (DMEM), and Roswell Park Memorial Institute (RPMI) medium (Thermo Fisher Scientific, Waltham, MA) were supplemented with 10% FBS (Thermo Fisher Scientific), 2 mM glutamine (Thermo Fisher Scientific), 10 U mL−1 penicillin, and 10 µg mL−1 streptomycin (Thermo Fisher Scientific) and named as M10, D10, and R10, respectively. Chinese hamster ovary (CHO) cells expressing GFP (CHO-GFP) or both HIV-1 Env and GFP (CHO-Env-GFP) were maintained in M10 supplemented with nonessential amino acid (NEAA) containing 5 mg mL−1 blastcidine (BSD).11 To prepare target cells for stimulation of CAR-T cells, subconfluent CHO-GFP cells or CHO-Env-GFP cells on a 150 mm culture dish were cultured in the presence of 10 µg mL−1 mitomycin C (Tokyo Chemical Industry) for 3 h. Cells washed with PBS were harvested with a cell scraper (Corning, Corning, NY). Single cell suspension was obtained through a 70 µm nylon cell strainer (Corning). 293FT cells were cultured in D10, and Jurkat E6.1 cells obtained from European Collection of Authenticated Cell Cultures through DS Pharma were maintained in R10. Human peripheral blood mononuclear cells (PBMCs) from healthy donors were prepared by Ficoll-Paque (GE Healthcare Life Sciences, Pittsburgh, PA) density gradient and cultured in AIM-V (Thermo Fisher Scientific) supplemented with 5% FBS, 10 mM HEPES (complete AIM-V) overnight to remove adherent monocytes. The monocyte-depleted PBMCs were used for transduction experiments. All the cells were grown at 37°C with 5% CO2.
Vector constructions
The cDNA fragment encoding the antigen-binging domain with the VH-VL arrangement was artificially synthesized using Genscript (Piscataway, NJ) from an anti-HIV-1 Env (clone: VRC01).25 A series of PCR reactions were carried out by using KOD-Plus-Ver.2 (TOYOBO) for making the cDNA fragment encoding the VRC01-drived antigen-binding domain with the VL-VH arrangement (Supplementary Fig. S1). The GGGGS linker sequence was added to the 5′ end of the VL region by PCR with primers VL-GS-F1 and VL01-GS-R, yielding the VL-GS fragment. Then, the XbaI restriction site and the leader sequence were added to the VL-GS fragment by PCR with primers VL-GS-F2 and VL01-GS-R, yielding the SP-VL-GS fragment (Supplementary Fig. S1A). The GGGGS linker sequence and the EcoRI restriction site were added to the 5′ and 3′ end, respectively, of the VH region by PCR with primers GS-VH-F and GS-VH-R, yielding the GS-VH fragment (Supplementary Fig. S1B). The assembly PCR was carried out by using the GS-VH and the SP-VL-GS fragments as templates with VL-GS-F2 and GS-VH-R primers (Supplementary Fig. S1C). The purified DNA fragment was incubated with Ampli Taq (Thermo Fisher Scientific, Waltham, MA) at 72°C for 10 min to add A tails. The A-tailed DNA fragment was ligated with the pGEM-T easy vector (Promega, Madison, WI). The sequence was verified by using a BigDye Terminator v3.1 cycle sequencing kit (Thermo Fisher Scientific). To construct the CAR-expressing lentiviral vectors, XbaI/EcoRI digested VRC01 scFv DNA fragments (VH-VL or VL-VH) and each of EcoRI/BamHI digested DNA fragments encoding signal sequences derived from HVEM and CD3ζ were ligated into XbaI/BamHI digested pTK643-EF1a-IRES-GFP/BSD.11
Preparation of the vector virus and HIV-1
The vector lentivirus was prepared as described previously11 with some modifications. Briefly, 293FT cells were cultured at 80-90% confluency on the 150 mm dish (IWAKI, Shizuoka, Japan) coated with poly-
Transduction with lentiviral vectors
Two million Jurkat E6.1 cells were seeded in each well of the six-well plate and inoculated with 1 mL of unconcentrated vector virus preparation in the presence of polybrane (8 µg mL−1). The cells were centrifuged at 5,500 rpm for 3 h at 22°C to enhance viral infection. After removal of the supernatant, the cells were cultured at 37°C in a CO2 incubator for 48 h. The culture medium was replaced with the media supplemented with 10 µg mL−1 BSD. Thereafter, transduced cells were maintained in the media with 10 µg mL−1 BSD until the following assays were performed.
Human primary CD4+ and CD8+ T cells were isolated from monocyte-depleted PBMCs by using a FACS Aria II cell sorter (BD Biosciences, Franklin Lakes, NJ) with antihuman CD3-APC, CD4-PE, and CD8-PE/Cy7 antibodies (all from Biolegend, San Diego, CA), and purity of more than 95% was routinely achieved. The purified CD8+ T cells were activated with anti-CD3/CD28 beads (Thermo Fisher Scientific) at the bead-to-cell ratio of 3:1 in complete AIM-V supplemented with 40 U/mL recombinant human IL-2 (obtained from Dr. Maurice Gately through NIH AIDS Reagent Program). On the next day, activated CD8+ T cells were transduced with concentrated recombinant lentivirus by using the plates coated with Retronectin (TAKARA, Shiga, Japan) according to the manufacturer’s instructions. The cells transduced with the vector expressing only GFP were used as a control. After incubation for 48 h, the supernatant was replaced with complete AIM-V supplemented with 300 U mL−1 of recombinant human IL-2. The anti-CD3/CD28 beads were removed at 48 h after changing the media. Thereafter, the cells were cultured for 16-18 days by replenishing fresh media containing IL-2 (300 U mL−1) every 2 to 3 days.
Flow cytometry
For flow cytometry, the centrifuged cells were resuspended in the fluorescence-activated cell sorter (FACS) buffer (PBS containing 2% FBS and 0.02% sodium azide) containing the antibodies and incubated on ice for 30 min otherwise indicated. After washing with ice-cold FACS buffer, the cells were fixed with 1% paraformaldehyde/PBS and analyzed by using a flow cytometer (FACS Calibur or LSRFortessa X-20; BD Biosciences). The data were analyzed using the FlowJo software (BD Biosciences). The following antibodies used for flow cytometry were obtained from Biolegend, unless it is indicated otherwise. The CAR expression on the transduced Jurkat E6.1 cells was analyzed with an anti-c-myc tag antibody (Santa Cruz Biotechnology, Dallas, TX) and an antimouse Immunoglobulin(Ig)s-PE antibody (Agilent Technologies, Santa Clara, CA) as primary and secondary antibodies, respectively. The activation of the vector-transduced Jurkat E6.1 cells in the co-culture assay was analyzed with antihuman CD3-APC and CD69-PE antibodies. The CAR expression on the vector-transduced primary CD8+ T cells was analyzed with the biotinylated anti-c-myc tag antibody followed by streptavidin-PE (TONBO biosciences, San Diego, CA). For evaluating exhaustion of the CAR-T cells, flow cytometry was performed with antihuman PD-1-APC and antihuman LAG-3-PE/Cy7 antibodies. For detecting CD3z phosphorylation in CAR-T cells, the vector-transduced primary CD8+ T cells were stained with the biotinylated anti-c-myc tag antibody followed by streptavidin-PE. Stained cells were fixed at 37°C for 10 min in Fix Buffer I (BD) and permeabilized at 4°C for 30 min in Perm Buffer III (BD). Permeabilized cells were incubated with the anti-CD247(pY142)-PE antibody (BD) at room temperature for 30 min.
Western blot
Cells (5 × 106) were washed once with ice-cold PBS, resuspended in 150 µL of RIPA buffer (10 mM TrisHCl [pH 7.4], 1% NP-40, 0.1% sodium deoxychorate, 0.1% SDS, 0.15 M NaCl, and 1 mM EDTA) supplemented with Complete Mini (Roche, Mannheim, Germany) and 1 mM PMSF and incubated on ice for 30 min. The cell lysate cleared by centrifugation (20 µL) was mixed with the NuPAGE LDS sample buffer (Thermo Fisher Scientific) and 0.1 M dithiothreitol and boiled at 95°C for 5 min. The boiled samples were separated by electrophoresis in a NuPAGE 10% Bis-Tris gel (Thermo Fisher Scientific) and transferred to an Immobilon P membrane (Millipore, Burlington, MA). The membrane was blocked with the blocking buffer (5% nonfat dried milk and 0.1% Tween-20 in Tris-buffered saline [TBS]) for 30 min. The blocked membrane was incubated overnight at 4°C with the anti-Actin antibody (I-19) or anti-CD3ζ antibody (F-3) (Santa Cruz Biotechnology, Dallas, TX) diluted in a blocking buffer. The membrane was then incubated for 1 hour with antigoat IgG-HRP (Millipore) or antimouse IgG-HRP (GE Healthcare UK Ltd., Buckinghamshire, England) diluted in a blocking buffer. The membrane was washed three times with 0.1% Tween-20/TBS after all antibody incubation steps. Immunoreactivity was visualized with Lumi-Light PLUS (Roche) as a substrate and detected with Light-Capture II (ATTO, Tokyo, Japan). The band intensity was quantified using ImageJ (National Institute of Health, MD).
Co-culture assay for effector functions of the CAR-T cells
Activation and IL-2 secretion of the CAR-transduced Jurkat E6.1 cells were determined by co-culturing with target cells (CHO-GFP or CHO-Env-GFP). Target cells (1 × 105) were seeded in each well of a 96 well flat-bottom plate. CAR-transduced Jurkat E6.1 cells (2 × 105) were added and co-cultured for 24 h. On the next day, the culture supernatants including the cells were harvested and centrifuged to separate cells and cell-free supernatants. IL-2 secretion into the supernatant was measured using Human IL-2 ELISA MAX Deluxe (Biolegend) according to the manufacturer’s instructions. The remaining cells were used to determine CD69 expression by flow cytometry.
To determine cytotoxicity of the CAR-T cells, target cells (CHO-GFP or CHO-Env-GFP, 1 x 104) were seeded in each well of 96 well flat-bottom plate. The CAR-T cells were added at different effector-to-target ratios (see also in figure legend) and co-cultured in phenol red-free R10 with NEAA. After 24 h of incubation, the cell-free supernatant was collected by centrifugation. Lactate dehydrogenase release in the supernatant was assayed using CytoTox 96 nonradioactive cytotoxicity assay (Promega). The cytotoxicity (% lysis) was calculated according to the manufacturer’s instructions. In cases of unequal percentage of CAR-transduced cells in the cultures, un-transduced T cells were added to ensure that both the number of CAR+ T cells and the total number of T cells remained consistent across the CAR-T cell groups. The collected supernatants were also used to determine IL-2, TNF-α, and IFN-γ secretion using human IL-2, TNF-α, and IFN-γ ELISA MAX Deluxe (all from Biolegend) according to the manufacturer’s instructions.
Co-culture for anti-HIV-1 activity of the CAR-T cells
CD4+ T cells isolated from the same donor of CD8+ T cells, which have been used for generating CAR-T cells, were activated with anti-CD3/CD28 beads at the bead-to-cell ratio of 3:1 in complete AIM-V supplemented with 40 U mL−1 IL-2. The anti-CD3/CD28 beads were removed at days 3 and cultured for 9 days with replenishing fresh media containing 40 U mL−1 IL-2 every 2–3 days. Activated CD4+ T cells (1 × 105) were exposed with HIV-1 (NL4-3 strain) at MOI = 0.1 for 3 h. After removal of unbound viruses, the virus-infected cells were cultured overnight. HIV-1-infected CD4+ T cells were co-cultured for 3 days with CAR-T cells at the E:T ratio of 1:1, 0.1:1, and 0.01:1. Cells were harvested and stained with antihuman CD8-PE/Cy7 (Biolegend) as described above. Intracellular HIV-1 p24 expression was detected using Intracellular Fixation and Permeabilization Buffer Set (eBioscience) with anti-HIV-1 p24 antibody KC57-RD1 (Beckman Coulter) according to the manufacturer’s instructions. The levels of HIV-1 p24 secreted into the supernatant of the co-culture were determined by the HIV-1 p24 ELISA Kit (RIMCO CORPORATION, Okinawa, Japan).
Stimulation of the CAR-T cells with the cells expressing the cognate antigen
The CAR-T cells (0.5 × 106) at days 18 were stimulated with mitomycin C-treated CHO-GFP or CHO-Env-GFP cells (1 × 106) in 2 mL of complete AIM-V supplemented with 300 U mL−1 IL-2. The cells were cultured for 7 days by replacing half of the culture media with the fresh media containing 300 U mL−1 IL-2 every 2 to 3 days. On days 7, the cells in half of the culture were stimulated again with mitomycin C-treated CHO-GFP or CHO- Env-GFP cells (1 × 106) in 2 mL of complete AIM-V supplemented with 300 U mL−1 IL-2 and cultured for 7 days. Flow cytometric analyses to determine CAR-T (CD3+GFP+) cell expansion were performed on days 7 and 14 after the initial stimulation.
Microscopic analysis of CAR cluster formation
The CAR-transduced primary CD8+ T cells at days 9 were stained with the biotinylated anti-c-myc tag antibody followed by streptavidin-APC (Biolegend) as described above. The CAR-T cell population with high CAR expression was sorted on FACS Aria II and seeded on a flat bottom 96 well cell culture plate. The sorted cells were observed using a BZ-9000 fluorescent microscopy system (KEYENCE, Osaka, Japan) with a 20× nonoil objective lens. The phase contrast and fluorescent images were taken and analyzed using ImageJ (National Institute of Health, MD).
Statistical analysis
Statistical analysis was described previously.11,28 The two-tailed Mann-Whitney U test and unpaired one-way or two-way analysis of variance (ANOVA) with the Bonferroni multiple comparison test were performed using GraphPad Prism (GraphPad Software, San Diego, CA). The P value of <.05 was considered statistically significant.
Results
Arrangement of VL and VH domains affects expression levels of CAR in human T cells
A pair of lentiviral vectors expressing the CAR bearing antigen-binding domains with different arrangements of the VL and VH domains derived from the anti-HIV-1 Env monoclonal antibody VRC01 were constructed (Fig. 1A and Supplementary Fig. S1). The viral titers for both CAR-expressing lentiviral vectors were very similar (Supplementary Fig. S2). The human T lymphocyte-derived Jurkat E6.1 cell line was transduced with either one of the VH-VL and VL-VH vectors and analyzed for the CAR expression. Whereas the transduction efficiencies of the VH-VL and VL-VH vectors determined by the percentage of GFP+ cells were similar to each other (Supplementary Fig. S3), expression levels of the VL-VH CAR on the surface of successfully transduced cells were significantly higher than those of the VH-VL CAR (Fig. 1B). Western blot analysis also revealed that the amount of the VL-VH CAR in the whole cell lysate was larger than that of the VH-VL CAR (Fig. 1C). Therefore, the arrangement of the VL and VH domains in the CAR appears for the determination of the level of cell surface expression of the CAR possibly through the effects on CAR synthesis or stability in the CAR-T cells.

Arrangement of VL and VH domains affects expression levels of CARs in human T cells.
Arrangement of VL and VH domains in the CAR affects antigen-dependent activation of the CAR-T cell line
The Jurkat E6.1 cells were transduced with the VL-VH or VH-VL lentiviral vector, and the successfully transduced CAR-T cells were co-cultured with the CD3- target cells expressing HIV-1 Env. Then, CD69 expression and IL-2 secretion of the CAR-T cells were determined by flow cytometry and ELISA, respectively. While no significant upregulation of CD69 in the CAR-T cells was observed upon co-cultivation with the control CHO-GFP cells, the cells co-cultured with CHO-Env-GFP cells showed significant levels of CD69 upregulation, indicating the antigen-dependent activation of the CAR-T cells (Fig. 2A and Supplementary Fig. S4). Consistently, IL-2 secretion by the CAR-T cells was increased upon co-cultivation with the antigen-expressing target cells (Fig. 2B). Antigen-dependent activation determined by CD69 upregulation and IL-2 secretion was higher for the VL-VH CAR than that of the VH-VL CAR (Fig. 2A and B), indicating that the arrangement of VL and VH in the antigen-binding domain of the CAR could affect the level of antigen-dependent activation of the CAR-T cells.

Arrangement of VL and VH domains in the CAR affects antigen-dependent activation of the CAR-T-cell line.
Arrangement of VL and VH domains in the CAR also affects car expression levels and effector functions of the primary human CAR-T cells
Next, human primary CD8+ T cells were transduced with the VL-VH or VH-VL lentiviral vector for preparing CAR-T cells. Transduction efficiencies determined by the percentage of GFP+ cells were similar to each other (Supplementary Fig. S5). “Similar to the results of the Jurkat E6.1 T cell line (Fig. 1 and Fig. 2), the amount of the VL-VH CAR in the whole cell lysate of the transduced primary T cells was larger than that of the VH-VL CAR (Fig. 3A), Whereas both the VL-VH and VH-VL CAR-expressing T cells exhibited cytotoxicity against the HIV-1 Env-expressing target cells, and the cytotoxic activities of the VL-VH CAR-T cells were higher than those of the VH-VL CAR-T cells (Fig. 3B). Similarly, the levels of secreted cytokines, such as IL-2, interferon (IFN)-γ, and tumor necrosis factor (TNF)-α, were higher for the VL-VH CAR-T cells than for the VH-VL CAR-T cells (Fig. 3C). Similar results were also obtained when the HIV-1-infected CD4+ T cells were used as a target. Namely, the VL-VH CAR-T cells eliminated HIV-1-infected CD4+ T cells more efficiently than the VH-VL CAR-T cells (Supplementary Fig. S6 and Fig. 3D). Consistently, the levels of HIV-1 p24 in the co-culture supernatants were lower for the VL-VH CAR-T cells than for the VH-VL CAR-T cells (Fig. 3E). These data suggested that the arrangement of VL and VH in the antigen-binding domain could affect expression levels of the CAR and the effector functions in human primary CD8+ T cells as well.
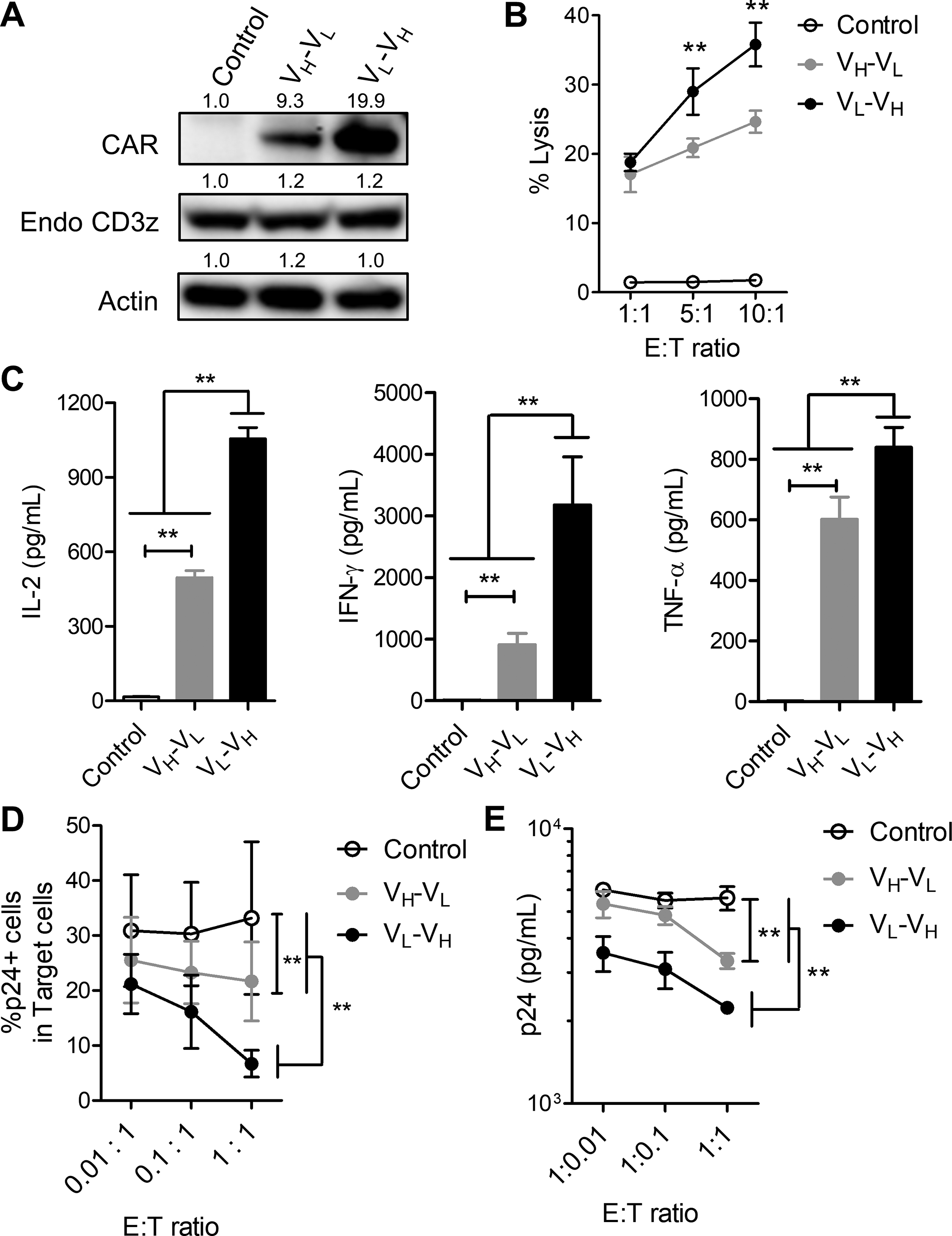
Arrangement of VL and VH domains in the CAR also affects CAR expression levels and effector functions of the primary human CAR-T cells.
Arrangement of VL and VH domains in the CAR affects exhaustion status of the CAR-T cells
To examine the effects of VL-VH arrangements in the antigen-binding domain of the CAR on exhaustion status of the CAR-T cells, we have compared the VL-VH and VH-VL CAR-T cells for expressions of the inhibitory receptors, such as lymphocyte activation gene 3 (LAG-3) and programed cell death 1 (PD-1). Flow cytometric analysis revealed that the LAG-3+/PD-1+ cells, which represent terminally exhausted population, were induced in the VH-VL CAR-T cells but not in the VL-VH CAR-T cells at days 9 (Fig. 4A) and 16 (Fig. 4B) of the culture period. A recent study has suggested that clustering of CAR molecules on the cell membrane might induce strong tonic signaling, which in turn induces CAR-T cell exhaustion.7 To examine whether tonic signaling has been associated with CAR-T cell exhaustion, the levels of phosphorylation in tyrosine 142 in CD3ζ of the CAR were analyzed by flow cytometry. As CD3ζ phosphorylation was not elevated in nontransduced population of the control T cells and the CAR-T cells with different VL-VH arrangements (Supplementary Fig. S7), CD3ζ phosphorylation was specifically occurred in the CAR-T cells (Fig. 4C). The level of CD3ζ phosphorylation was higher in the VH-VL CAR-T cells than in the VL-VH CAR-T cells at days 9 and 16 (Fig. 4C). These data suggested that the strength of tonic signaling was associated with the level of CAR-T cell exhaustion. Since poor proliferation upon stimulation with the cognate antigen is one of the hallmarks of the T cell exhaustion,29,30 we next examined CAR-T cell proliferation upon cognate antigen stimulation. When the CAR-T cells were stimulated with the mitomycin C-treated antigen-expressing cells, the VL-VH CAR-T cells proliferated more vigorously than the VH-VL CAR-T cells (Fig. 4D). Thus, these data showed that the VH-VL CAR-T cells appeared to be exhausted at higher level than the VL-VH CAR-T cells, suggesting that arrangement of VL and VH in the antigen-binding domain of the CAR affects exhaustion status of the CAR-T cells.

Arrangement of VL and VH domains in the CAR affects exhaustion status of the CAR-T cells.
The VL-VH CAR-T cells resist exhaustion despite the high CAR expression
Next, we examined the characteristics of the CAR-T cell with different VL-VH arrangements in more detail during the culture period. It was noted that while the VL-VH CAR was stably expressed at a high level during the culture period, the VH-VL CAR expression at day 9 became bimodal and that the proportion of the cells with the lower VH-VL CAR expression level enlarged at day 16 (Fig. 5A and B, upper panels). Flow cytometric analysis revealed that the population of the CAR-T cells with the higher levels of VH-VL CAR expression contained much larger percentage of the LAG-3+/PD-1+ cells compared to the other population with the lower levels of VH-VL CAR expression (Fig. 5A and B, lower panels). Immunostaining analysis revealed that the T cells expressing the VH-VL CAR at higher levels indeed had clustering of the CAR molecules on the cell membrane (Fig. 5E, middle panels). Interestingly, the VL-VH CAR expressed at high levels without causing significant T cell exhaustion was also shown to form CAR clusters on the cell membrane (Fig. 5E, lower panels). There was no significant difference in the number of CAR-T cells with the CAR cluster between the VH-VL CAR-T cell and the VL-VH CAR-T cell (Fig. 5E, right graph). We next examined the level of CD3ζ phosphorylation in each CAR-T cell population (Fig. 5F). The VH-VL CAR-T cell with lower CAR expression did not increase the level of CD3ζ phosphorylation in the CAR, whereas the one with higher CAR expression significantly increased the level of CD3ζ phosphorylation in the CAR during the culture period. Interestingly, the level of CD3ζ phosphorylation in the CAR was slightly increased in the VL-VH CAR-T cell which expressed the CAR at high level but was significantly lower than that in the VH-VL CAR-T cell which expressed the CAR at high level. Thus, the VL-VH arrangements in the context of the CAR construct used in this study may allow the T cells to evade tonic signaling-induced CAR-T cell exhaustion despite the high CAR expression.

The VL-VH CAR-T cells resist exhaustion despite the high CAR expression.
Discussion
The CAR-T cell-mediated immunotherapy is expected to become useful therapeutic intervention against diseases with impaired cellular immunity such as malignant diseases. In order to improve the technology, it is important to design an optimal CAR molecule with maximal efficacy. For this purpose, newer generations of CARs are being developed by modifying its components such as CSSD. For example, a recent study from our group demonstrated that the CSSD derived from HVEM could be more useful than previously used CD28- or 4-1BB-derived CSSD.11 Although there is a previous study which compared different arrangements of VL and VH in the context of CAR with 4-1BB-derived CSSD,21–24 it has been unknown how the arrangement of VL and VH affects CAR-T cell functions and exhaustion status in particular in the context of the CAR bearing the HVEM-derived CSSD.
In order to investigate this issue, we constructed the lentiviral vectors for expressing the CAR bearing VL-VH or VH-VL derived from an anti-HIV-1 Env antibody and the HVEM-derived CSSD as a model system. Analysis of the human T cell line and primary T cells transduced with these vectors demonstrated that the levels of cell surface expression were higher for the VL-VH CAR than the VH-VL CAR apparently reflecting the different abundance in the cells. As to scFv molecules, it was previously reported that the arrangement of VH and VL could affect its expression levels or stability.14–18 It is possible that the level of synthesis or stability of the VL-VH CAR is higher than the VH-VL CAR in our system, although the detailed mechanism is unknown yet. Another issue is that the CAR with different VL and VH arrangement might have different binding affinities against the antigen. To clarify this, it may be necessary to perform the biophysical analysis for both VL-VH and VH-VL scFvs in the future. This might be able to reveal precise mechanisms of the differences in the potencies of the CAR-T cells with different VL and VH arrangements.
In our system, the VL-VH CAR-T cells were shown to be more potent than the VH-VL CAR-T cells in terms of their functions, such as antigen-dependent activation and cytotoxicity against the target cells. In our previous study comparing different CSSDs, the levels of CAR expression and CAR-T cell functions were correlated.11 Therefore, it is possible that the difference in the CAR-T cell functions is simply caused by the quantitative difference in the expression levels of CAR molecules. However, it is unlikely because the exhaustion status of the CAR-T cells was also affected by the arrangement of VL and VH in the CAR. It is thought that tonic signaling mediated by the CAR molecule is responsible for induction of CAR-T cell exhaustion.2 A previous study has shown that the CAR-T cells expressing high levels of CAR exhibited exhausted phenotype and that this phenotype had been rescued by experimentally reducing CAR expression.31 It has also been suggested that clustering of the CAR molecules on the cell membrane could induce tonic signaling, which leads to exhaustion of CAR-T cells.7,31 In consistent with the previous reports, our data indicated that the levels of VH-VL CAR expression were correlated with the strength of the CAR-mediated tonic signaling and the proportion of exhausted cells. Interestingly, the VL-VH CAR expressed at higher levels than the VH-VL CAR did not significantly induce CAR-mediated tonic signaling and lead to CAR-T cell exhaustion although it formed clusters on the cell membrane. In addition, the high level of VL-VH CAR expression was not associated with the increase in the exhausted cells (Fig. 5A-D). Thus, the abundance of the VL-VH CAR molecules by itself may not be responsible for induction of the exhausted phenotype. It has been reported that framework regions within certain CAR scFv are sufficient to induce tonic signaling.7 Thus, it is possible that the VL-VH arrangement in the context of the CAR construct used in this study might be hard to convey the CAR-mediated tonic signaling which induces CAR-T cell exhaustion. Homology modeling of the VL-VH and VH-VL CARs suggested the distinct structural differences in the antigen binding domain of the CAR molecules (Supplementary Fig. S8). This difference might also determine the level of tonic signaling.
Although the VL-VH arrangement appeared to be more useful than the opposite arrangement in our system, the limitation is that the results of this study could not directly be applied for other CAR constructs.21–24,32 However, the evidence showed in this study might provide useful information for designing potent CARs and for developing CAR-T cells, which could evade exhaustion. It would be desirable to compare different CAR constructs with both arrangements of VL and VH domains combined to various CSSDs at preclinical stage for generating effective CAR-T cells against various targets.
Conclusion
In summary, this study demonstrated that the arrangement of VL and VH in the antigen-binding domain could affect expression levels of the VRC01-based CAR with HVEM-derived CSSD as well as the functions and exhaustion status of the CAR-T cells by modulating CAR-mediated tonic signaling. This could provide useful information for designing optimal CARs and for developing effective CAR-T cell-mediated immunotherapy.
Footnotes
Acknowledgments
We thank Yuki Shinozaki, Satomi Yoshida, and Kayoko Kobayashi for technical and secretarial assistance; Yasuko Nonaka for cell sorting; and Takashi Namatame for DNA sequencing. We also thank members of Department of Microbiology, Dokkyo Medical University, for critical discussion of the paper and their input and assistance.
