Abstract
Introduction:
Effective blinding is especially challenging in randomized controlled trials (RCTs) involving psychedelics due to the inherent alterations in consciousness that these compounds induce. In this systematic review and exploratory analysis, we aim at synthesizing the methodologies used in RCTs involving classic psychedelics and identify procedures that can help minimize unblinding and bias.
Methods:
We completed a literature search that included prospective RCTs published between 1963 and January 2023, in which participants were randomized to receive either a classic psychedelic or placebo.
Results:
A total of 1402 papers were included in the initial search. After eligibility criteria were applied, 50 papers from 48 clinical trials were included. Most studies were double blinded (n = 34), used a within-subjects design (n = 32 studies), and included inert placebos (n = 35). The majority of studies did not report on the integrity of blinding procedures (n = 34 studies); however, in studies that did, blinding was unsuccessful.
Conclusion:
To improve blinding and lower expectancy effects, we suggest incorporating active placebos and utilizing dose response or active comparator study designs.
Introduction
The term “psychedelic” derives from the ancient Greek words meaning “mind” (psyche) and “manifesting” (delos). 1 Classic psychedelics include psilocybin, dimethyltryptamine (DMT; found in ayahuasca brew), lysergic acid diethylamide (LSD), and mescaline, which are all agonists at the serotonin 2A receptor. 2
Scientific interest in psychedelic compounds began >100 years ago when the German toxicologist Louis Lewin learned of ritualistic use of the peyote cactus by indigenous tribes in Mexico. 3 Mescaline, the psychotropic compound of the peyote cactus, was synthesized in 1918 by the chemist Ernst Spaeth. 3 Clinical research with mescaline was conducted in healthy volunteers and the state it induced was studied as a model of psychosis. 3
Research into psychedelic compounds increased dramatically when Albert Hoffman discovered LSD in 1943. 4 Research on LSD's psychotomimetic properties and potential to modify and potentially enhance psychotherapy flourished until almost all research was halted in 1970 with the passing of the Controlled Substances Act.4,5 The past decade has seen a resurgence of research investigating the use of psychedelic compounds for various psychiatric conditions. 6 This revival of research in psychedelic compounds is spurred by unmet needs in regard to the treatment of mental illnesses. 2
Psychiatric illness is widespread and debilitating, with both depression and anxiety in the top 25 causes of disease burden globally. 7 Although there are treatment options available for these conditions, such as psychotherapy and antidepressant medications, these options are not effective for everyone. 8 For example, approximately one in three people diagnosed with either Major Depressive Disorder or an anxiety disorder do not respond to first-line treatments and will develop treatment-resistant mental illness.9,10
Further, standard pharmacological antidepressant treatments are limited by significant delay in their onset (i.e., most take 2 to 6 weeks to produce a therapeutic effect), and many cause undesirable side effects.2,9 In randomized control trials (RCTs), psychedelics have shown promise for producing rapid acting and sustained therapeutic effects in patients suffering from various psychiatric illnesses, including cancer-related anxiety and depression, treatment-resistant depression, and alcohol use disorder.11–19
However, there are challenges in using standard clinical trial approaches to evaluate psychedelics, as they are unique from other classes of pharmaceuticals in that they significantly alter one's thoughts, perceptions, and feelings in a time-limited fashion. 1
Despite the promise psychedelics show in the treatment of mental illness, the significance of findings in recent studies are difficult to interpret due to methodological challenges, including challenges with adequate blinding, high expectation biases, and nocebo effects. 20 To date, the methodology used in RCTs involving psychedelic compounds is diverse with a wide range of placebos used, including both inert and active placebos.
Active placebos refer to control interventions that mimic physiological effects of the experimental compound without any suspected therapeutic benefit. 21 In addition to a placebo comparator, some studies also include active comparator groups, which refer to non-psychedelic interventions that may have a known therapeutic or significant effect.13,22–24 The methodological challenges outlined have limited the widespread generalizability of the available research.
As psychedelics gain attention as potent mental health treatments with promising results, it is crucial to optimize the methodology used in psychedelic RCTs to both ensure the veracity of findings and maximize the potential therapeutic benefits of these compounds. In this narrative review, we aim at synthesizing the placebo controls and study design methodology used in RCTs of classic psychedelics, and of identifying the procedures that lead to the minimization of bias and unblinding.
Methods
The protocol for this study was registered with PROSPERO (CRD42020185100). This review is reported in accordance with PRISMA guidelines.
Search strategy and selection criteria
PubMed was electronically searched on May 15, 2020 using the following search terms: (psychedelic* OR hallucinogen* OR psilocybin OR ayahuasca OR DMT OR dimethyltryptamine OR mescaline OR peyote OR LSD OR lysergic acid diethylamide) AND (mood* OR major depress* distress* OR depress* OR PTSD* OR post-trauma* OR post trauma* OR posttrauma* OR anxiety* OR schizophren* OR bipolar) AND (placebo OR sham OR control OR controlled) AND (treatment OR intervention OR trial OR).
We included all prospective controlled trials in which individuals were given a placebo in the context of a trial for a classic psychedelic (psilocybin, mescaline, DMT, LSD, ayahuasca, peyote). Retrospective studies, microdosing studies, and studies examining the effects of psychoactive compounds not identified as classic psychedelics (e.g., 3,4-methylenedioxy-methamphetamine [MDMA], ketamine, benzodiazepines) were excluded. There were no exclusion criteria related to age, gender, or psychiatric diagnosis. The initial search was run on May 15, 2020. The search was repeated and updated on January 17, 2023.
Data extraction
Two reviewers (N.S. and B.D.M.J.) independently screened all titles and abstracts and removed irrelevant articles. Two reviewers (N.S. and B.D.M.J.) independently screened the full text of relevant papers for the initial search. Two reviewers (A.W. and C.R.W.) completed the updated search. A.W., N.S., and S.M. extracted data from the studies for the qualitative and quantitative analyses.
Data analysis and presentation
Google sheets was used to analyze results through analysis of descriptive statistics and to generate figures. Data on placebos used are presented in a stacked bar chart (Fig. 2). Data on the study design, blinding, and demographics are presented in tabular format (Tables 1 and 2).
Study Design Methods in Psychedelic Randomized Controlled Trials Including Blinding Assessment, Study Design, and Blinding Type
Patient Demographics in Psychedelics Randomized Controlled Trials
DMT, dimethyltryptamine; LSD, lysergic acid diethylamide.
Results
The initial search yielded a total of 1053 records. After the search was repeated and duplicates were removed, 374 records were added. One thousand three hundred forty-six records were excluded after a review of titles and abstracts. Thirty-two records were excluded after a review of their full texts. This resulted in a total of 50 papers from 48 clinical trials5,11–19,23–62 (Please refer to the Supplementary Table S1 for a full list of the papers included). Please refer to Figure 1 for an outline of the study selection process. The majority of psychedelic RCTs used LSD (n = 19) and psilocybin (n = 21) (Fig. 2). There were three RCTs of DMT and five RCTs of ayahuasca.
PRISMA flow diagram.
In this review, in most cases we included only one paper from a clinical trial to avoid redundancy. In two cases, we included two papers derived from the same clinical trials as they discussed aspects of study design and blinding not present in the original paper.11,13,17,27 However, for the quantitative analyses, information from each clinical trial was included only once to avoid redundant information.
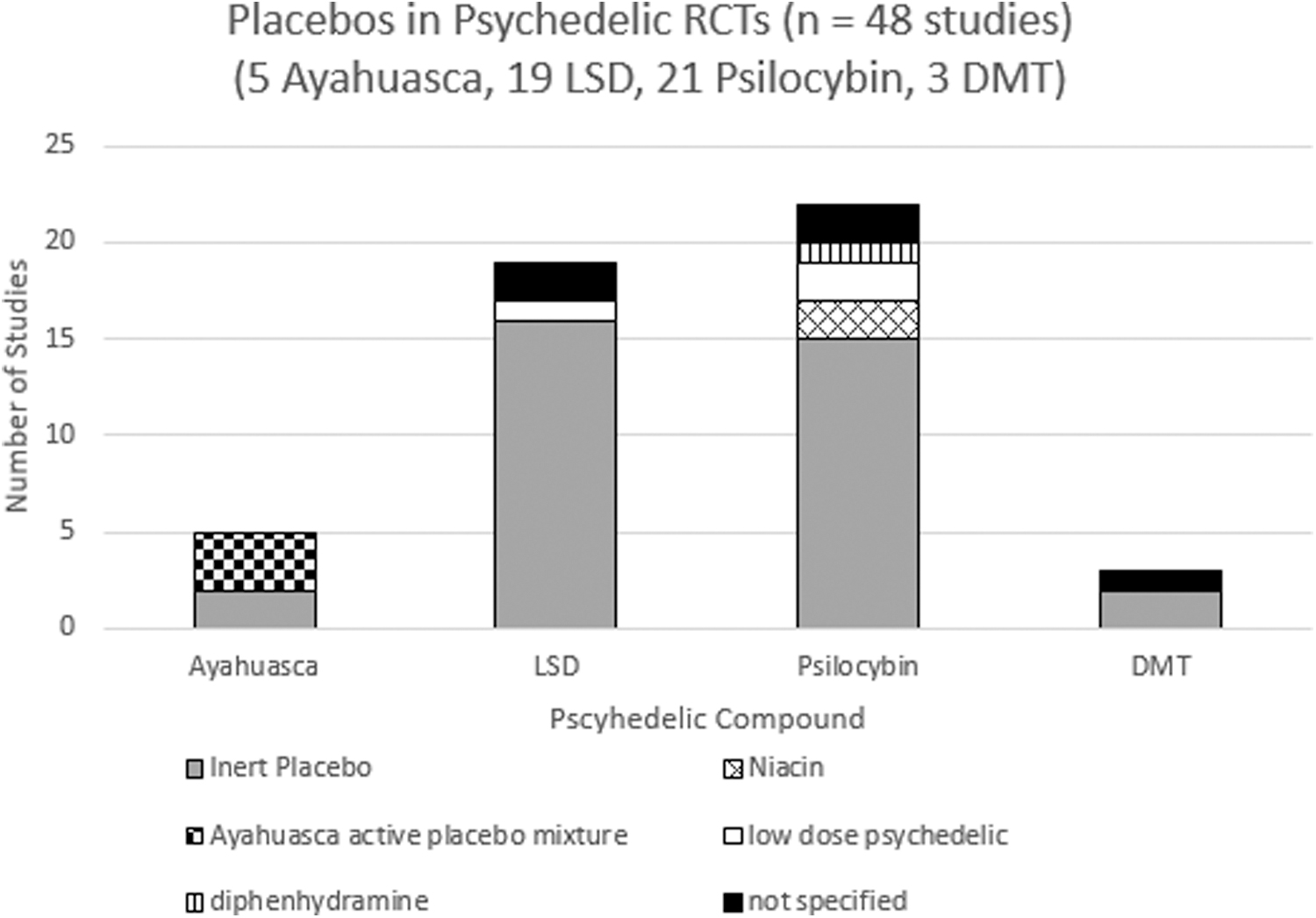
Placebos used in Psychedelic RCTs. Ayahuasca (3 studies used ayahuasca active placebo mixture, 2 studies used inert placebos); LSD (2 not specified, 1 study used low-dose psychedelic, 16 studies used inert placebos); psilocybin (15 studies used inert placebos, 2 studies used niacin, 1 used diphenhydramine, 2 studies used low-dose psychedelics, and 2 did not specify); DMT (1 did not specify, 2 used inert placebos). DMT, dimethyltryptamine; LSD, lysergic acid diethylamide; RCTs, randomized controlled trials.
Placebos used
The majority of studies (73%) used inert placebos. Inert placebos included the following: water (4 total: LSD = 2, ayahuasca = 1, psilocybin = 1), saline IV (9 total: LSD = 5, psilocybin = 2, DMT = 2), raspberry syrup (2 total: LSD = 2), empty capsule (3 total: psilocybin = 3), lactose, mannitol, or ethanol (15 total: psilocybin = 9, LSD = 5, ayahuasca = 1), alcohol solution (1 total: LSD = 1), and bitter lemon mixture (1 total: psilocybin = 1).
The active placebos included the following: a placebo mixture containing water, zinc, citric acid, caramel dye, and other components designed to simulate the organoleptic properties of ayahuasca (3 total: ayahuasca = 3), niacin (2 total: psilocybin = 2), diphenhydramine (1 total: psilocybin = 1), and low-dose psychedelics (3 total: LSD = 1, psilocybin = 2).11–16,18,28,29
Notably, there were 22 placebos used in psilocybin trials despite only 21 completed studies, because 1 study utilized both low-dose psilocybin (1 mg) and empty placebo pills as placebo controls. 13 Also, five studies did not specify what placebo was used.30,36,43,50,59 A summary of placebos used in RCTs with psychedelics is presented (Fig. 2).
Study design and blinding
The majority of psychedelic RCTs utilized a within-subjects design, with 32 studies using a within-subjects design and 16 studies using a between-subjects design (Table 1). The majority of studies were double blinded (n = 34). A few studies reported on the success of experimenter blinding, with 34 out of 41 (83%) not reporting any blinding assessments (Table 1). A few studies reported on the success of participant blinding, with 34 out of 48 studies (71%) not reporting on any blinding assessments (Table 1). A summary of study design and blinding in RCTs with psychedelics is presented (Table 1).
In regards to experimenter blinding, four studies reported quantitative information, and three studies reported qualitative information.12,14,16,28,29,41,57 All three studies reporting qualitative information reported that experimenters almost always knew whether the participant was receiving placebo or active treatment16,41,57 Three out of four studies reporting quantitative information also reported that experimenters almost always knew whether the participant was receiving placebo or active treatment with one study reporting 100% accuracy.12,14,28
In one study that used the ayahuasca placebo mixture, experimenters reported experiencing some difficulty identifying the participants group allocation and were incorrect for 5 out of 20 (25%) of participants. 29 A detailed recording of the experimenter blinding assessments can be found in Supplementary Data S1.
Nine studies reported quantitative information on participant blinding assessments.12,14,17,23,26,28,29,33,55 Two studies did not report on participant blinding in the active and placebo group separately12,29: one ayahuasca study described that overall, 13 out of 20 (65%) subjects were able to correctly identify whether they received placebo or ayahuasca 29 ; and one psilocybin study described that following the first dosing session, 93.6% of participants were able to correctly identify whether they received placebo or psilocybin, and following the second session, 94.7% correctly identified whether they received placebo or psilocybin. 12
The remaining seven studies reported on blinding success in the active group and placebo group separately.14,17,23,26,28,33,55 Altogether in these seven studies, 65 out of 65 (100%) subjects correctly identified that they were in the active group, and 67 out of 72 (93%) subjects correctly identified that they were in the placebo group.14,17,23,26,28,33,55 All five participants who incorrectly guessed that they were in the active group were participants with treatment-resistant depression in an ayahuasca study that used a placebo mixture designed to mimic the organoleptic properties of ayahuasca. 17
Five studies only reported qualitative information on participant blinding.16,25,38,52,57 Three out of these five studies reported that most subjects were able to easily ascertain what group they were in.16,25,57 One of these five studies stated that several subjects (healthy volunteers) mistook the lowest dose of DMT (0.05 mg/kg) for placebo. 38
One of these five studies discussed the difficulty participants (healthy volunteers) had in discerning between the very low dose (45 μg/kg) and low dose (115 μg/kg) psilocybin, as well as the medium dose (215 μg/kg) and high dose (315 μg/kg) psilocybin. 52 However, this study did not mention anything about difficulty discerning between active sessions and placebo sessions. 52
Discussion
In this review, 48 studies and 50 articles were included, ranging from 1963 to 2023. In this discussion, we comment on blinding, expectancy and placebo and nocebo effect, placebos used, study design, and how the demographics of participants in psychedelic RCTs could be contributing to blinding difficulties. To our knowledge, this is the second review to examine the use of placebo in RCTs with classic psychedelics.
The first review by Nayak et al. was recently published in the Journal of Clinical Psychiatry and explores many similar features such as blinding and placebos used. 63 Our review is consistent with the findings of Nayak et al. and with many of their novel and well thought out recommendations on addressing issues with psychedelic RCTs.
However, our review adds to the literature as it covers trials up to January 17, 2023 whereas their review only covers trials up to April 30, 2021. 63 Our review also makes novel suggestions for how to assess blinding and expectancy and makes suggestions for novel active placebos that can be used to minimize unblinding. In addition, our review goes into more detail on how unique study designs can be used to alleviate expectancy effects and unblinding. Lastly, our review describes demographics as a possible contributor to unblinding, which is not discussed in their review.
Blinding
Successful blinding occurs when participants do not know whether they receive placebo or active treatment. This uncertainty ensures that expectancies are equally distributed across both the active arm and placebo arm, and that placebo effects in each arm are equally distributed. 64 In psychedelic RCTs to date, the blinding of both participants in the placebo group and active group is highly unsuccessful with virtually all participants able to correctly identify which group they were in.14,16,17,23,25,26,28,29,33,55,57
Unsuccessful blinding can significantly decrease confidence in research findings, as the effects of the active treatment cannot be distinguished from expectancy effects and magnified placebo effects. 64
The majority of studies in our review did not report on blinding. Given that assessment of blinding is imperative to determine the validity of results, more psychedelic RCTs should report on blinding in a systematic and quantitative fashion. Bang's blinding index (BI), a variation of the kappa coefficient, appears to be a reliable method to assess blinding success.65,66
Bang's BI was used in two clinical trials and applied retrospectively to multiple studies.66,67 Bang's BI appears to be particularly useful, as it allows researchers to detect both blinding and response bias. Bang's BI will result in a value between −1 and 1, with 0 indicating completely unsuccessful blinding, 1 indicating perfect blinding, and values closer to −1 indicating opposite/incorrect group allocation and possible response bias.65,66
To utilize Bang's BI, it is recommended that blinding be reported separately for the active and placebo arms. Reporting blinding for arms separately is also useful, as blinding for active and placebo arms is not always the same. There were two studies in our review that reported blinding as a whole rather than for each group separately, which made it difficult to discern the efficacy of blinding in each group.12,29
In addition, it is recommended that blinding be reported on either a 3-point scale (“active,” “placebo,” “don't know”) or a 5-point scale with varying levels of confidence in participants' choice of active or placebo (see Kolahi et al. for a detailed description of the 5-point scale).65,66
It is clear that reporting of blinding needs to be improved in clinical trials with psychedelic compounds, but it is also important to note that the paucity in reporting on blinding is not unique to clinical trials with psychedelic compounds. Lin et al. found that only 11 out of 154 (7%) double-blind RCTs of antidepressants reported on blinding, and Fergusson et al. found that only 7 out of 97 (7%) of general medicine trials reported on blinding.68,69
The struggle maintaining successful blinding is also not unique to trials with psychedelics, although trials with psychedelics are likely faced with the greatest amount of unblinding. Lin et al. also found that in nine studies of antidepressants, including 1177 patients, 52.2% of participants correctly guessed they were in the active group and 71.63% participants correctly guessed they were in the placebo group. 68
Fergusson et al. found that in seven general medicine trials, five trials had imperfect blinding, one trial did not remark on blinding success, and one trial reported successful blinding with no data to support the claim. 68
Expectancy and placebo and nocebo effects
Placebo effects refer to beneficial outcomes that result from patient expectation rather than the treatment.70,71 Nocebo effects refer to negative outcomes that result from patient expectation rather than from the treatment.70,71 Clinical trials with psychedelic compounds are especially susceptible to placebo and nocebo effects as they are met with high patient expectation, resulting from substantial amounts of coverage of psychedelics in the popular media and a ritualized procedure for psychedelic administration.71,72
This susceptibility to the placebo and nocebo effect is further amplified by the aforementioned blinding challenges in psychedelic trials, that is, placebo effects resulting from patients randomized to the active group being aware of their group allocation experiencing additional benefits driven by expectation; nocebo effects resulting from patients randomized to the placebo group being aware of their group allocation experiencing negative outcomes from disappointment.
We found that no study in our review assessed for expectancy in participants. Given the high potential for placebo and nocebo effects mentioned earlier, expectancy should be assessed in clinical trials with psychedelic compounds. The Stanford Expectations Treatment Scale (SETS) appears to be a reliable method to assess expectancy. 73
This scale was tested and developed through use in three studies at Stanford Hospital and was used in a clinical trial looking at smoking cessation.73,74 This scale consists of two subscales: positive and negative expectancy to allow for assessment of both placebo and nocebo effects. 73
In addition, if the concern for nocebo effects is elevated, such as in therapeutic trials working with at-risk psychiatric patients, we suggest using study designs where all participants are guaranteed the active treatment (ex., Crossover designs or designs with an open label phase) to mitigate potential nocebo effects from participant disappointment.
Placebos
The only cases in which blinding of participants randomized to the placebo group was successful was when an active placebo was used. A placebo mixture designed to simulate only the organoleptic properties of ayahuasca, including color, taste, and gastrointestinal discomfort, was used in three studies.17,28,29 In one of these studies, 5 out of 15 participants in the placebo group guessed that they were in the active group. 17
In another study, 7 out of 20 participants incorrectly identified their group placement. 29 Developing an active placebo that has both a taste and color component and mimics some of the common physiological effects of psychedelics, such as elevated heart rate, headache, and fatigue, could create a more convincing placebo for LSD and psilocybin.75,76
To mimic this ayahuasca placebo for psilocybin and LSD, we have two suggestions. First, for psilocybin, we suggest a clinical trial with organic substances, that is, mushrooms with psilocybin. Similar to the ayahuasca brew, nausea and vomiting are one of the common side effects of psychedelic mushrooms. 77 A mushroom with no psychedelic compounds that mimics the taste, color, and side effects (nausea, vomiting, elevated heart rate, etc.) of the psychedelic mushroom could create a convincing placebo.
Second, to mimic the ayahuasca placebo for psilocybin or LSD, we suggest administering the psychedelic through a liquid. There was one study that used a bitter lemon liquid mixture as a placebo and used the bitter lemon liquid mixture with psilocybin in a powder form mixed in as the active group. 31
There was also one study done in the 1960s that administered LSD mixed in with raspberry syrup and used raspberry syrup only as the placebo. 25 As a placebo for either LSD or psilocybin, we suggest the use of a liquid with both a taste and color component along with the addition of an active placebo such as niacin, diphenhydramine, or a low-dose psychedelic to mimic physiological effects.
Another active placebo used is a low-dose psychedelic. Only three studies used a low-dose psychedelic as a placebo.13–15 Gasser et al. used 20 μg LSD as a placebo. All participants were able to tell whether they were in the active or placebo condition (despite 11 out of 12 participants having no experience with LSD). 14 It is possible that a slightly higher dose of LSD was needed to create a more convincing active placebo.
Although not present in the current review, in a study looking at microdosing LSD, the acute subjective and behavioral effects of 26 μg LSD were assessed. 78 It was found that 26 μg of LSD led to significant increases in drug-like effects and blood pressure but did not change mood, cognition, heart rate, or body temperature, 78 suggesting that such a dose could function as an active placebo with limited effects on clinical outcomes.
Both Carhart-Harris et al. and Goodwin et al. used 1 mg psilocybin as a placebo. Unfortunately, neither study reported on blinding, so we cannot comment on its effectiveness as a placebo. Interestingly, in the Carhart-Harris et al. study, the 1 mg psilocybin placebo was also utilized to balance expectation effects.
Subjects were told that they would receive psilocybin regardless of group allocation and the dosages of psilocybin were not revealed to participants.13,27 The use of low-dose psychedelics as placebos should be studied in more detail as they appear to mimic some of the properties of psychedelics at high doses, and could potentially reduce expectation bias.
Study design
In our review, there were no cases in which a subject in the active group thought they were in the placebo group. Due to the potent subjective effects of psychedelics, it is easy to imagine the extreme difficulty in maintaining the blind for the active group. Although there is no apparent method to successfully ensure participants receiving an active psychedelic believe they are receiving placebo, there have been some unique study designs implemented to lower expectancy effects.
One possible solution is to employ a dose-response study design testing both various doses of psychedelics and placebo. Six studies included in our review used this approach.24,26,33,38,51,53 In the Griffiths et al. study, all subjects received the following: placebo and 5, 10, 20, 30 mg/kg psilocybin for a total of five sessions. Participants were randomized to receive psilocybin in either ascending or descending dosages. 53
However, placebo was given in a quasi-random scheduling to make it more difficult for participants to discern both the psilocybin dosage and the placebo session. 53 This method demonstrated potential to blind research staff, as none of the eight blinded therapists involved in drug administration were able to identify the ascending/descending schedule of drug administration. 53
However, the Griffiths et al. study did not assess participant blinding nor expectancy, so we are unable to comment on the effectiveness of this method for participant blinding. In another dose-response study, all subjects received the following: placebo, 25, 50, 100, 200 μg LSD, and 200 μg LSD+ketanserin. 33 Although no subjects in the 100 or 200 μg LSD arm thought they received placebo, some thought they may have received 50 or 25 μg LSD. 33
In the placebo group, no subjects thought they had received 100 or 200 μg LSD, but some thought they may have received 25 μg LSD or 200 μg LSD+ketanserin. 33 The use of dose response designs could help improve blinding and lower expectancy effects. In addition, the use of a psychedelic along with serotonin blockers such as ketanserin can be useful to improve blinding and lower expectancy effects as patients can be told that they will be getting a psychedelic.
Another approach to decrease expectancy effects is to use active comparator groups in addition to placebos.13,23,24,54 Two studies used multiple active comparator groups.23,55 In the Gouzoulis-Mayfrank et al. study, a between-subjects design was implemented and participants received either placebo, psilocybin,
Unfortunately, the Gouzoulis-Mayfrank et al. study did not assess blinding. In the Gouzoulis-Mayfrank et al. study, participants received either placebo or psilocybin, but were told they could also receive d-amphetamine or MDE. In this study, all six participants in the psilocybin group correctly identified that they received psilocybin.
55
Two out of six participants in the placebo group experienced minor psychological effects and thought they might have been in the MDE or
In another study, participants received all of the following: LSD, MDMA,
Two studies used one active comparator group.13,24 In the Carbonaro et al. study, subjects received the following for a total of five dosing sessions: 10, 20, and 30 mg/kg psilocybin, dextromethorphan, and placebo. Interestingly, to minimize expectancy effects, subjects were told that they could receive either placebos or 1 of 38 different psychoactive drugs, including psilocybin and dextromethorphan. 24 Unfortunately, neither expectancy nor blinding was assessed in this study. 24
In the Carhart-Harris et al. study, subjects were randomized to one of two arms: psilocybin or escitalopram. 13 There were two dosing sessions spaced 3 weeks apart. 13 Subjects in the psilocybin arm received 25 mg psilocybin during dosing sessions and were instructed to take placebo tablets daily in the 3 weeks after each of their dosing sessions.13,27
Subjects in the escitalopram arm received 1 mg psilocybin during two dosing sessions and were instructed to take 10 mg escitalopram tablets daily for the 3 weeks between dosing session 1 and 2 and 20 mg escitalopram daily for 3 weeks after dosing session 2.13,27 This unique design utilizes both an empty capsule as a placebo for escitalopram and 1 mg psilocybin as a placebo for 25 mg psilocybin, which is helpful for allowing comparison of psilocybin to an established antidepressant.
Because of the aforementioned difficulties with blinding and inferring causality in psychedelic RCTs with placebos, comparisons of psychedelics to current treatment methods may be more appropriate than placebos. Comparing psychedelics with current established pharmacological and psychotherapeutic treatment methods could help balance expectancy between groups, improve blinding, and lead to more valid and pragmatic results.
Demographics and recruitment
Available information on patient demographics is limited, as some studies did not report specific demographic information. Out of the 48 studies included in this analysis, 11 did not report on mean age16,34–38,40,42,43,48,59; and 2 did not report on sex.40,59 Only nine studies reported information on ethnicity.11–13,15,18,19,24,30,32 Only 16 studies reported adequate information on education level for our analysis.11,13,18,24,28,30,32,41,42,44,46,49,50,53,54,60
The demographics of participants in RCTs with classic psychedelics is homogeneous, consisting of mainly white (77.8%), college educated (79.1% college students or collect graduates), and young participants (Table 2). Interestingly, in two studies, all participants were either physicians or psychologists.54,55
In addition to highlighting the need to recruit a more diverse population, these participant demographics may also influence blinding efficacy. It is possible that this highly educated participant population is more aware of the science and phenomenology of psychedelics, making them more challenging to blind, contributing to the blinding challenges in RCTs with psychedelics thus far.
Conclusion
This review explores placebo controls employed in RCTs with psychedelic compounds from 1963 to 2023. Most studies in our review did not report on blinding or expectancy. For future studies, we suggest reporting on both blinding and expectancy in a systematic and quantitative fashion, such as through the use of Bang's BI or the Stanford Expectations Treatment Scale.65,66,73 We also suggest reporting blinding of the placebo group and psychedelic group separately.
Inert placebos are generally unsuccessful in maintaining blinding in RCTs with psychedelics. Therefore, we suggest the use of active placebos. The most successful active placebo was an ayahuasca placebo mixture designed to simulate the organoleptic properties of ayahuasca, including taste, color, and gastrointestinal discomfort.11,28,29
We suggest mimicking this ayahuasca placebo mixture for LSD and psilocybin by either conducting trials with organic substances (psychedelic mushrooms) for psilocybin or administering psychedelic compounds through a liquid formulation. We also suggest studying the use of low-dose psychedelics as active placebos.
Low-dose psychedelics as placebos can also be used to balance expectancy effects between groups, as participants can be guaranteed that they will be receiving the psychedelic compound in all study arms.13,27
Third, we suggest using two different study designs to reduce expectancy effects. We suggest either employing active comparator groups that are not psychedelics or testing multiple different doses of psychedelics. We also suggest exploring the use of a serotonin blocker (e.g., ketanserin) alongside a psychedelic to improve blinding and lower expectancy. 33
Lastly, we found that the participant populations in RCTs with psychedelics thus far have been mainly white and college educated. This participant population may be knowledgeable on the phenomenology of psychedelics, making them more difficult to blind and contributing to the blinding challenges in psychedelic RCTs thus far.
Footnotes
Authors' Contributions
A.W. conducted the updated search; reviewed studies and collected data; created figures and tables; and wrote the manuscript. N.S. conducted the original search; reviewed studies and collected data; and assisted in writing and editing of the manuscript. B.D.M.J. conducted the original search; reviewed studies and collected data; and assisted in writing and editing of the manuscript. R.J.Z. assisted in writing of the manuscript. S.M. assisted in writing of the manuscript. M.A.S. assisted in writing of the manuscript and the review of studies. D.M.B. participated in the conceptualization of the study design and assisted in writing and editing of the manuscript. Z.J.D. participated in conceptualization of the study design and assisted in writing and editing of the manuscript. C.R.W. participated in conceptualization of the study design and assisted in writing and editing of the manuscript. A.W. conducted the updated search and guided through the study review and the data analysis process.
Author Disclosure Statement
R.J.Z. is a post-doctoral fellow in the NYU Langone Psychedelic Medicine Research Training program funded by MindMed. M.A.S. has financial investments in the following companies: CMPS, ATAI, FTHWF, REUN, CYBN, NUMIF, and MNMD. D.M.B. receives research support from the Canadian Institutes of Health Research (CIHR), National Institutes of Health—US (NIH), Brain Canada Foundation and the Temerty Family through the CAMH Foundation and the Campbell Family Research Institute. He received research support and in-kind equipment support for an investigator-initiated study from Brainsway Ltd. and he was the site principal investigator for three sponsor-initiated studies for Brainsway Ltd. He received in-kind equipment support from Magventure for investigator-initiated studies. He received medication supplies for an investigator-initiated trial from Indivior. He has participated in an advisory board for Janssen. He has participated in an advisory board for Welcony, Inc.
Z.J.D. has received research and in-kind equipment support for an investigator-initiated study through Brainsway, Inc., and Magventure Inc., and industry-initiated trials through Magnus, Inc. He also currently serves on the scientific advisory board for Brainsway, Inc. His work has been supported by the National Institutes of Mental Health (NIMH), the Canadian Institutes of Health Research (CIHR), Brain Canada and the Temerty Family, Grant and Kreutzcamp Family Foundations. C.R.W. is supported by the Brain and Behavior Research Foundation Young Investigator Grant and Wellcome Leap Multi-Channel Psych program. He also reports consulting fees from Goodcap Pharmaceuticals, Inc. The other authors have no disclosures.
Funding Information
No funding was received for this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
