Abstract
Obesity-related comorbidities (ORCs) cause significant economic and clinical burdens for people with obesity and the US health care system. A reduction in weight at the population level may reduce incident ORC diagnoses and associated costs of treatment. The aim of this work is to describe obesity burden in the United States through the prevalence and direct treatment costs of ORCs, as well as the clinical and economic value of 15% weight loss in a population of adults with obesity. The IQVIA Ambulatory US electronic medical record database was used to create a cohort (7,667,023 individuals 20–69 years of age, body mass index of 30–50 kg/m2), utilized to characterize the prevalence of 10 ORCs. Direct treatment costs were collected from literature reports. A risk model was leveraged to estimate the number and cost of additional ORC diagnoses over 5 years from baseline through two scenarios: stable weight and 15% lower body weight at baseline for all members of the population. Prevalence, incidence, and cost data were scaled down to a representative subset of 100,000 individuals. In 2022, the annual treatment costs for all 10 ORCs exceeded $918 million for the representative cohort. In a stable-weight scenario, these costs were estimated to increase to ≈$1.4 billion by 2027. With 15% lower body weight at baseline, $221 million in cumulative savings was estimated, corresponding to $2205 in savings/patient over 5 years. Consequently, weight loss in this population may correspond to significantly reduced numbers of incident ORC complications translating to substantial cost savings.
Plain Language Summary
Obesity and related health problems can cause strain for people with obesity and the health care system. Weight loss might help reduce this strain. This report modeled how costs would change if body weight were 15% lower in 100,000 people with obesity. It was determined that this would reduce obesity-related health problems and save around $221 million in health care costs over 5 years. This study suggests long-term weight loss would improve the health and health care costs of people with obesity.
Introduction
A body mass index (BMI) of 30 kg/m2 or higher in adults may be an indication of obesity. 1 Obesity is associated with a decreased life expectancy of approximately 5–20 years, depending on the severity of the disease and other associated comorbid disorders. 1,2 People with obesity are at increased risk of long-term comorbidities, including type 2 diabetes (T2D), hypertension, depression, and 13 different cancer types. 3,4
In addition to health-related consequences, people with obesity may experience increased social burdens such as social stigma, lower productivity, and overall reduced quality of life. 5 –9 Moreover, the estimated global economic impact of obesity annually is $2.0 trillion. 10 This corresponds to 2.8% of the global gross domestic product. Individuals with obesity and their families may also face higher economic burden. In the United States, it has been estimated that relative to an adult with normal weight, an 18-year-old adult with obesity may amass excess lifetime medical costs from ≈$2,700 to ≈$12,800. 11 These excess lifetime costs were predicted to exceed $38,000 by age 60 in adults with obesity.
There appears to be a complex relationship between the metabolic, psychological, social, and behavioral factors that contribute to the development of obesity in adults and children. 3 These complexities contribute to the challenge of effectively addressing the increasing prevalence of obesity. Current options for the treatment of obesity are limited to lifestyle interventions focused on diet and exercise, surgical interventions, and pharmaceutical interventions. 12 Therapies based on lifestyle and behavioral interventions may provide moderate efficacy, but often have poor long-term success. 12,13 Alternatively, surgical interventions are both invasive and applicable to a limited population due to issues of accessibility and suitability, and thus are incapable of meeting the needs of people with obesity on a wide scale. 12,13 Pharmaceutical interventions pose a convenient and accessible alternative for the effective treatment of people with obesity, but few of these pharmaceutical treatments have been proven to consistently enable weight loss reduction >15%. 12
In the absence of effective and accessible prevention and treatment options, obesity rates have nearly tripled over the last five decades, nearing pandemic proportions. 14,15 In the United States, more than one third of the adult population is living with obesity. 16 The national population of people with obesity has been projected to increase to up to 1 in 2 adults, or 48.9% of the population by 2030. 17 It is therefore important that steps are taken to understand the scope of obesity-related comorbidities (ORCs) and their related economic and clinical burdens on people with obesity and the health care system. To that end, this study has leveraged electronic medical records (EMRs) to generate a representative cohort of people living with obesity in the United States and characterized the obesity-related clinical and economic burden of this group. In addition, the potential impact that a 15% lower body weight, across all individuals in the described cohort, may have on the number of incident ORC diagnoses and the direct treatment costs was assessed over a 5-year period.
Methods
A study population of 23.5 million adults representative of the US population was extracted from the IQVIA Ambulatory EMR (AEMR) real-world database of US EMRs by selecting individuals active after January 1, 2018. The most recent BMI record after this date was defined as the index date. The last available index date was November 29, 2021. From this study population, a cohort of 7,667,023 adults 20–69 years of age and with BMIs of 30–50 kg/m2 was used to evaluate the prevalence of 10 of the most common chronic ORCs (Fig. 1, Table 1), defined by diagnosis records for the ORCs at any time before the index date. The ORCs included in the analysis were T2D, asthma, sleep apnea, hip/knee osteoarthritis, heart failure, chronic kidney disease (CKD), hypertension, dyslipidemia, atrial fibrillation (AFib) and flutter, and unstable angina/myocardial infarction (UA/MI). Direct treatment costs, including costs for consultations, laboratory analyses, hospitalizations, procedures, and prescriptions, were derived from the literature 18 –28 to quantify the yearly direct treatment costs for any patient with any of the 10 associated comorbidities.
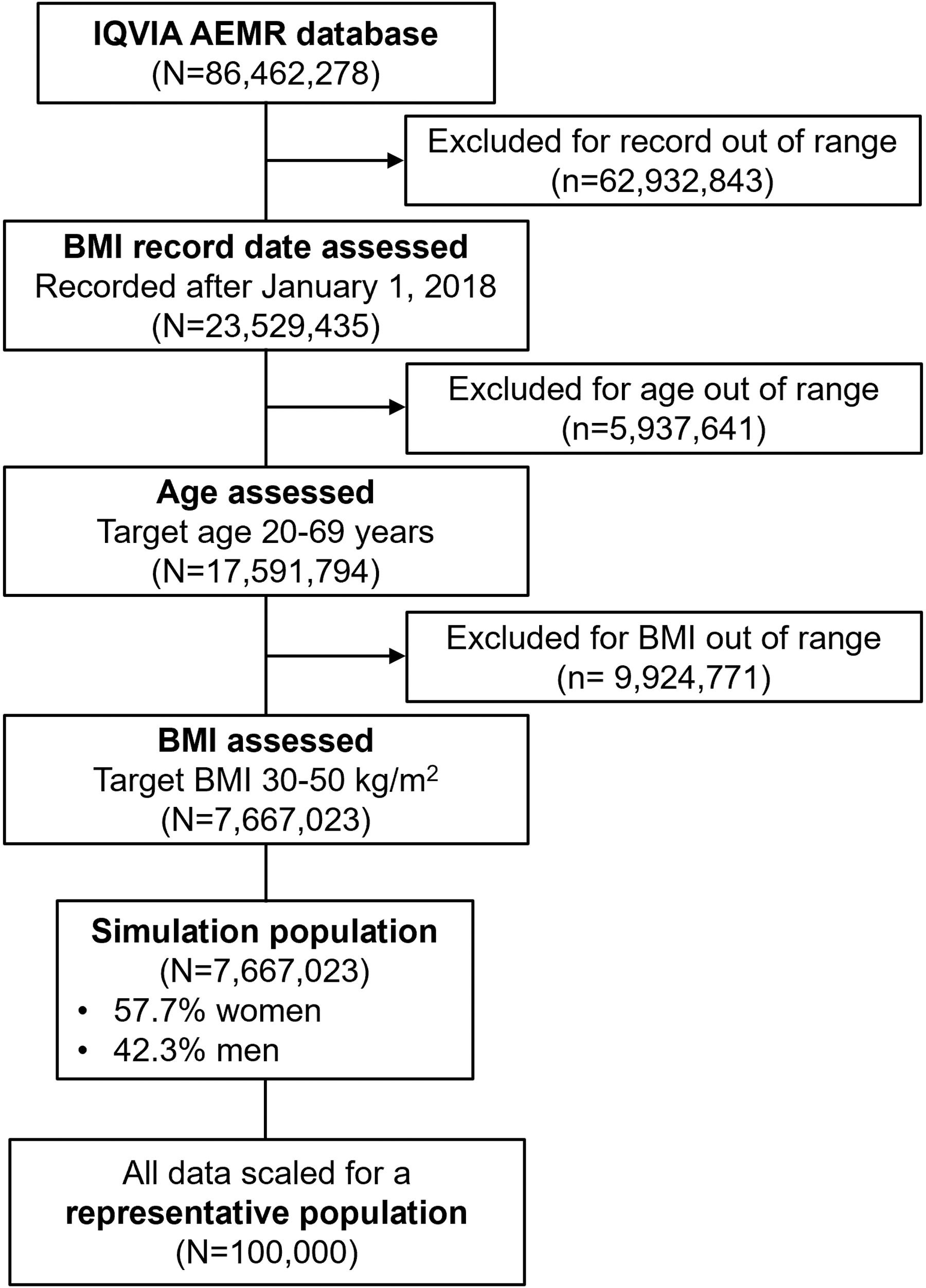
Study sample attrition diagram. AEMR, ambulatory electronic medical record; BMI, body mass index.
Patient Demographics and Clinical Characteristics
Data include the patient characteristics at baseline.
AFib, atrial fibrillation; BMI, body mass index; CKD, chronic kidney disease; ORCs, obesity-related comorbidities; T2D, type 2 diabetes; UA/MI, unstable angina/myocardial infarction.
A UK-derived risk model was previously developed to follow people with obesity, with 13% intentional weight loss, to assess any change in risk of 10 ORCs compared with those with lower, stable BMI. 29 In this study, an extension of that risk model was used to estimate the association between weight change +/−20% and the future risk of 13 ORCs. 30 This risk model was adapted to the US population using the simulation population (N = 7,667,023) to estimate the yearly incidence of the 10 ORCs over a 5-year period. Treatment costs for each ORC were applied to the estimated incidence of each ORC to predict the yearly increase in clinical and economic burdens of obesity within this time frame. The risk model was again used to estimate the number of incident ORC diagnoses that could be prevented if the baseline body weight of all individuals in the study cohort was 15% lower, achieved through weight loss with lifestyle, surgical, or pharmaceutical intervention, compared with individuals with stable weight. No weight restrictions were imposed on either group during the 5 years following the baseline period. All prevalence, incidence, and cost data were scaled down to a representative subset of 100,000 people and used for further analyses. The direct treatment costs were estimated for each scenario (cohort with 15% lower body weight vs. original cohort) and the value of weight loss was defined using the expected yearly direct treatment costs (assumed to be stable over 5 years from baseline) for the prevented ORC complications and the cumulative cost savings over a 5-year time frame (Fig. 2).
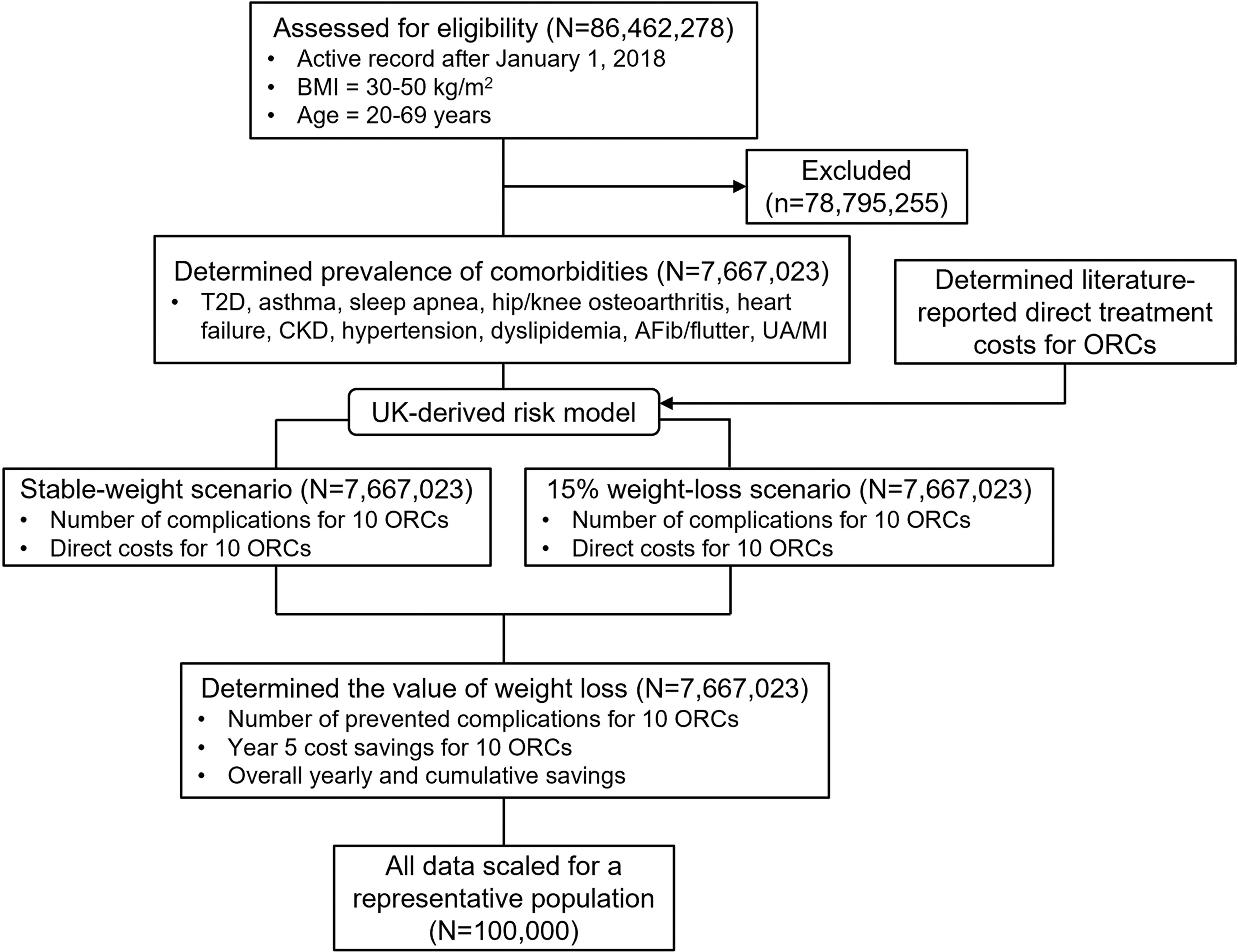
Study design diagram. AFib, atrial fibrillation; BMI, body mass index; CKD, chronic kidney disease; ORCs, obesity-related comorbidities; T2D, type 2 diabetes; UA/MI, unstable angina/myocardial infarction.
Results
For the cohort of 7,667,023 people with BMI of 30–50 kg/m2 and 20–69 years of age, 57.7% were female and 42.3% were male. The prevalence of the 10 analyzed ORCs among the cohort ranged from 1.7% to 52.4% (Table 1). The ORCs with the highest prevalence were hypertension (52.4%) and dyslipidemia (46.9%), and the ORCs with the lowest prevalence were AFib/flutter (2.2%) and UA/MI (1.7%). Direct treatment costs for each ORC were extracted from the literature, with yearly costs ranging from $1767 to $58,460 per patient complication (Fig. 3). CKD and T2D were among the most expensive ORCs, with yearly costs of $58,460 and $11,071, respectively. By contrast, the lowest yearly costs were incurred with sleep apnea and hip/knee osteoarthritis, which amounted to $1767 and $2728, respectively.
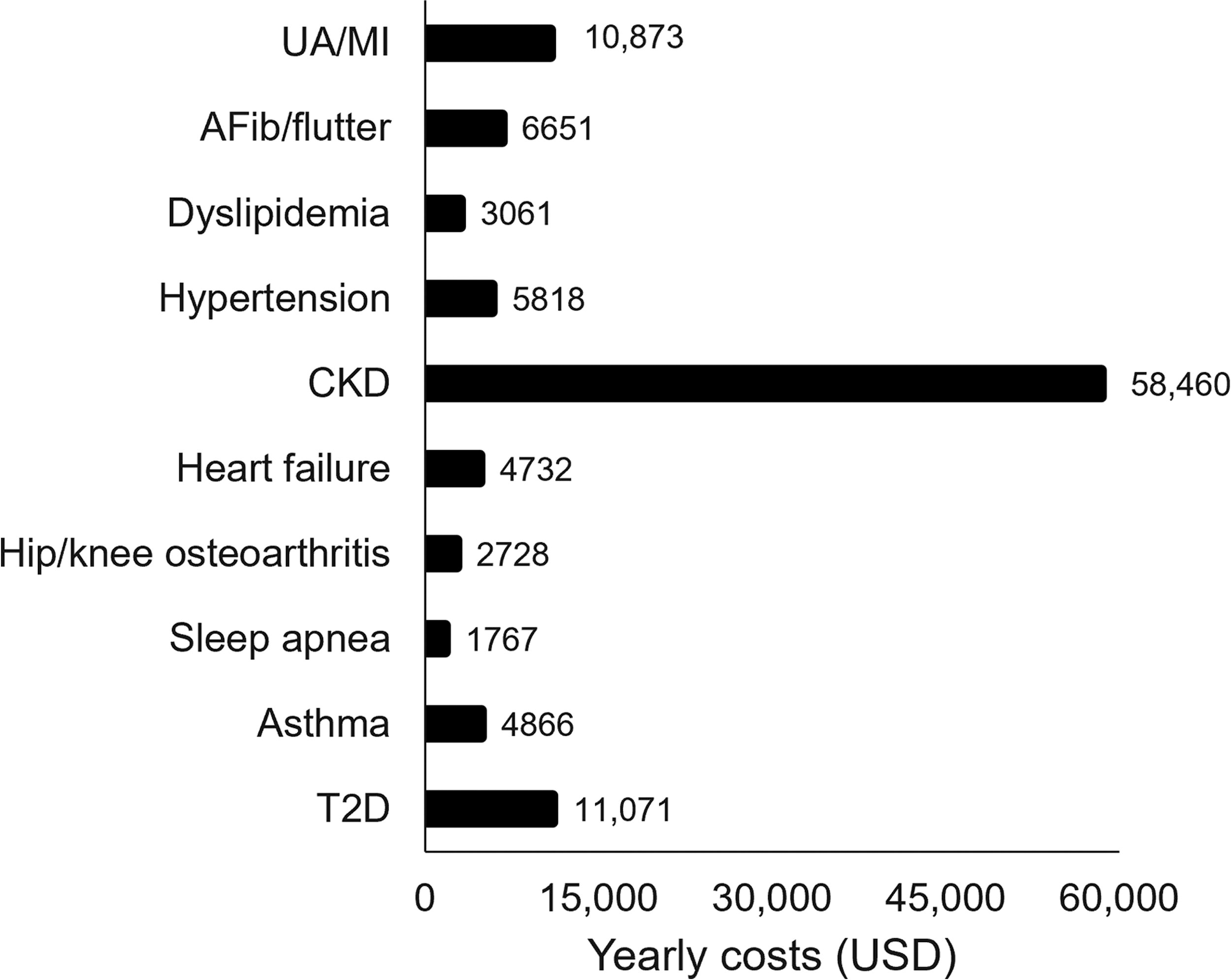
Direct treatment costs in US dollars of the 10 ORCs for an individual. Costs were extracted from the literature and reported as yearly sums. AFib, atrial fibrillation; CKD, chronic kidney disease; ORCs, obesity-related comorbidities; T2D, type 2 diabetes; UA/MI, unstable angina/myocardial infarction; USD, US dollars.
The number of complications and the direct costs for each ORC were determined for the simulation cohort of 7,667,023 individuals at baseline (2022) and subsequently estimated over a 5-year period (2027) using a UK-derived risk model. 29 These data are summarized in Table 2 for the representative population of 100,000 individuals. At baseline, costs ranged from $8.9 million to $305 million with the highest expense incurred for hypertension, followed by CKD. Direct costs associated with CKD were predicted to nearly double in the next 5 years, reaching $511 million from approximately 8700 complications (including almost 5000 new complications) and accounting for more than one third of total costs. Secondary drivers of cost include T2D and hypertension, each contributing more than 7000 new complications and more than $43 million in direct costs in the next 5 years. The total cost of treatment was determined as the sum of all direct costs, exceeding $918 million in 2022. In the following 5 years, the total cost of treatment was predicted to exceed $1.4 billion, which is more than 1.5 times the baseline value.
Number and Cost of ORCs over a 5-Year Time Frame Following 15% Weight Loss of All Individuals in the Cohort of 100,000
Data include the number of complications and direct costs at baseline, complications and direct costs in the stable and 15% weight-loss scenarios, number of prevented complications and cost savings in a 15% weight-loss scenario at year 5, and cumulative 5-year cost savings for the representative cohort of 100,000 individuals.
AFib, atrial fibrillation; CKD, chronic kidney disease; ORCs, obesity-related comorbidities; T2D, type 2 diabetes; UA/MI, unstable angina/myocardial infarction; USD, US dollars.
Next, the representative cohort (N = 100,000) was assessed in a scenario in which body weight was 15% lower in all individuals at baseline and the number and cost of incident complications following a 5-year period were compared with those of the stable-weight scenario. The number of prevented complications and cost savings at year 5 were compared for all ORCs (Table 2). The greatest cost savings were predicted to be accrued by the avoided T2D complications, numbering 3000 (37%) prevented incident diagnoses and valued at $101.9 million in cumulative savings by year 5. The reduced number of new CKD diagnoses would save almost $60.5 million over 5 years. Significant reductions were predicted for hypertension and dyslipidemia complications, representing relative reductions in incidence of about 20% and 14%, respectively, and accumulating cost savings of $27 million and $11 million over 5 years, respectively.
Finally, the total value of cost savings for all ORCs was determined for each year in the 5-year window for the representative cohort of 100,000 individuals (Supplementary Fig. S1, Table 2). Yearly savings were predicted to increase by more than $14 million with each consecutive year. The cumulative total direct treatment cost savings for all 10 ORCs were estimated to reach nearly $220 million over a 5-year period from baseline. This would correspond to about $2205 in total savings per patient.
Discussion
Obesity and its related comorbidities are associated with a high economic burden on the US health care system. In this study, the prevalence and economic burden of 10 ORCs in a population of adults with obesity have been summarized. An evaluation of EMRs and claims from 2004 to 2013 reported the prevalence of 8 of the 10 ORCs investigated in this study, all of which closely agree with the values summarized in this study. 31 These consistencies support the accuracy of the prevalence data as extracted from the AEMR and its applicability to determining future costs.
The data presented in this study suggest that, without intervention, the economic burden of obesity and its related comorbidities will continue to grow. In 2022, the yearly direct treatment costs for 10 ORCs in a representative cohort of 100,000 individuals living with obesity were estimated to be more than $918 million. Over the following 5 years, without intervention, these costs were predicted to increase by 56% due to the diagnoses of additional ORCs for the individuals in this cohort. Tangential studies have established the burden of ORCs on the US health care system, finding that expenditures are higher for people with obesity, who develop cancer, 32 diabetes, 33 and asthma, 34 compared with those without obesity.
Intentional weight loss in people with obesity, in contrast with maintaining a stable weight, is associated with reduced incidence of ORCs. Among the cohort of 100,000 individuals, 15% lower body weight at baseline may be associated with a reduction in additional ORC diagnoses. The validity of these results is supported by previously reported evidence of the association between weight loss and reduced incidence of several ORCs, including sleep apnea, osteoarthritis, heart failure, CKD, hypertension, dyslipidemia, and T2D. 35 –37 The association was demonstrated to strengthen with duration of weight loss (up to 10 years) and the magnitude of weight loss (from 1% to >10% body weight loss). It can therefore be inferred that 15% weight loss maintained over the course of 5 years would correlate with a reduced incidence of ORC complications.
A significant reduction in future treatment costs and substantial cumulative savings was estimated over 5 years in the representative population with 15% lower body weight at baseline. These results are supported by a similar modeling study that estimated the cost reduction associated with lowered obesity prevalence in international populations, ranging from $500 million to almost $12 billion in savings annually. 38 Importantly, the cost savings detailed in this study are a reflection of reduced direct treatment costs through prevention of future ORCs, and do not take into account the savings associated with resolution of ORCs present at baseline through weight loss. The overall savings may therefore be underestimated through this analysis.
Limitations
This study does not estimate the effect of weight loss over time, but rather the burden that may be reduced in a scenario in which 100,000 patients with obesity had a 15% reduction in body weight at baseline. Some limitations of this study are related to the use of real-world data from the IQVIA AEMR database. This is not a claims database, and therefore, continuous enrollment could not be achieved. In addition, because the existence of a BMI record is the main inclusion criterion when extracting data, this analysis may either capture people with a high BMI, but without obesity, or fail to capture all people with obesity, introducing a bias. EMR data sets are skewed toward patients who seek health care for various health problems. People with obesity may experience ORCs that are not recorded in their records. Furthermore, this database is chiefly composed of ambulatory patients, and thus patients with the most severe diseases may not be captured.
Further limitations may be attributed to the methods used in this study. The risk model used in this analysis was derived from UK EMRs, and the patient lifestyle and ORC incidence may differ in the United States. The risk model was developed using data from as early as 2001, 29 during which time no efficient pharmaceutical treatment was available. Consequently, a majority of the patients included in the original data set relied upon lifestyle changes to achieve weight loss. Pharmaceutical treatments for obesity may provide advantages to weight loss that lifestyle changes alone cannot achieve. Consequently, it is possible that the number of prevented incident ORCs predicted by the UK risk model, and thus the value of weight loss, is underestimated in this analysis. Alternatively, a 15% weight loss achieved by lifestyle changes alone would likely require a significant improvement in dietary quality or a considerable increase in physical activity and may have health benefits that pharmacologically induced weight loss cannot provide. In that case, the value of pharmacologically induced weight loss would be overestimated in this study. The clinical impact determined in this study may be further limited because the model does not account for factors such as mortality.
Regarding health care costs, some of the comorbidities included in this analysis (eg, CKD) are classified by stages of progression, which may incur a range of costs. In addition, the costs were assumed to remain stable over 5 years, which does not truly reflect the cost of health care in the United States. The estimated treatment costs may therefore have some level of uncertainty. Additionally, only direct treatment costs are included, which do not take into account indirect costs such as loss of income due to medical leave, reduced productivity at work or home, or the cost of child care required as a result of treatment. 39 Conversely, as this analysis only captures incident diagnoses and the associated costs, any potential saving due to weight loss having beneficial effect on preexisting comorbidities was not captured.
Conclusions
As of the time of this publication, the clinical and economic burdens of obesity are significant. In the absence of highly effective and accessible treatment options, these burdens will continue to grow as the population of people with obesity age and comorbidity incidence consequently increases. Currently, options for surgical 40 or pharmaceutical interventions in combination with lifestyle changes 41 are available, which may deliver up to 15% weight loss. This study has shown that having 15% lower body weight among all members of a population of 100,000 individuals with obesity at baseline may effectively prevent a significant number of ORCs over the coming years, resulting in substantial economic savings.
Footnotes
Acknowledgments
The authors acknowledge the medical writing assistance of Amy Ryan, PhD, of PRECISIONscientia, Yardley, PA, which was supported financially by Novo Nordisk Inc., Plainsboro, NJ, in compliance with international Good Publication Practice guidelines. The final version was approved by all authors.
Authors’ Contributions
All authors were involved in study design, data analysis, and interpretation. All authors revised the article content and approved the final version.
Author Disclosure Statement
J.T. is an employee and shareholder of Novo Nordisk Inc and has received support from Novo Nordisk for research, medical writing, fees associated with article publication, travel, and registration fees for conferences/meetings. V.S. is an employee of Novo Nordisk A/S and has received funding for data access and medical writing support from Novo Nordisk. L.R. is an employee and shareholder of Novo Nordisk A/S and has received funding for data access and medical writing support from Novo Nordisk.
Funding Information
All data collection, data analysis, and article development were funded by Novo Nordisk A/S.
Supplementary Material
Supplementary Figure S1
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
