Abstract
Recombinant zoster vaccine has been recommended by the US Advisory Committee on Immunization Practices (ACIP) for the prevention of herpes zoster (HZ) in immunocompetent adults aged at least 50 years since 2018. In January 2022, this was extended to immunodeficient/immunosuppressed adults aged at least 19 years. Key study objectives were to assess specialists’ knowledge of the ACIP HZ vaccination recommendations, their attitudes toward HZ vaccination, and HZ vaccination practices/barriers. This cross-sectional, web-based survey (conducted in March 2022) included US dermatologists, gastroenterologists, infectious disease specialists, oncologists, and rheumatologists who treat patients with psoriasis, inflammatory bowel disease, human immunodeficiency syndrome, solid tumors/hematological malignancies, and rheumatoid arthritis, respectively. Although most of the 613 specialists correctly identified the ACIP HZ vaccination recommendations for adults aged at least 50 years (84%) and immunodeficient/immunosuppressed adults aged at least 19 years (67%), only 29% knew that recombinant zoster vaccine is recommended for individuals who have previously received zoster vaccine live, and only 18% knew all current ACIP recommendations. For patients with the diseases listed, 84% of specialists thought that HZ is a serious risk, 75% that HZ vaccination is extremely/very important, and 69% were extremely/very likely to recommend HZ vaccination. Only 36% administer vaccines themselves, mainly because patients receive vaccinations from others. Barriers to vaccination included more urgent/acute issues, insufficient time, and lack of patient motivation/willingness. Full knowledge of the ACIP HZ vaccination recommendations among the surveyed specialists was low. There may be a need to educate specialists to improve adherence to these recommendations.

Introduction
Herpes zoster (HZ), which is characterized by a vesicular rash (typically on the trunk or face), results from the reactivation of latent varicella-zoster virus infection. 1 Some patients can suffer HZ complications, including postherpetic neuralgia (chronic pain following rash resolution) and HZ ophthalmicus (which can affect all ophthalmical structures and potentially lead to vision loss). 1,2 Approximately 1 in 3 people will develop HZ at some point during their life, 3 with the incidence increasing with age from approximately 5 per 1,000 person-years (PY) in adults aged 50 to 54 years to 11 per 1,000 PY in those aged at least 85 years. 4 Other HZ risk factors include immune-altering conditions (e.g., bone marrow/stem cell transplant, human immunodeficiency virus [HIV], rheumatoid arthritis [RA], cancer, inflammatory bowel disease [IBD], and psoriasis), immunosuppressants, and chemotherapy. 5
Recombinant zoster vaccine (RZV) has been recommended by the US Advisory Committee on Immunization Practices (ACIP) for the prevention of HZ and related complications in immunocompetent adults aged at least 50 years since January 2018, 6 primarily based on two phase 3 trials in immunocompetent adults. 7,8 At this time, the ACIP made a preferential recommendation for RZV over zoster vaccine live (ZVL) and also recommended RZV for adults who had previously received ZVL, 6 which is no longer available in the United States. 9 In January 2022, the ACIP also recommended RZV for immunodeficient/immunosuppressed adults aged at least 19 years, 10 based on studies in patients who had undergone autologous stem cell transplantation 11 or renal transplant 12 and those with HIV 13 or cancer. 14,15 The recommendation includes hematopoietic cell transplant recipients, solid organ transplant recipients, and patients with cancer, HIV, or autoimmune/inflammatory conditions/treatments. 16
Specialists treating immunocompromised/immunosuppressed adults are now in a position to recommend/prescribe/administer HZ vaccination to these patients and, along with every member of the care team, play a crucial role in adult vaccination and in promoting public health. A web-based questionnaire was developed and administered to various specialists (dermatologists, gastroenterologists, infectious disease specialists, oncologists, and rheumatologists), with the primary objectives being to describe specialists’ knowledge of HZ, HZ risk, and HZ vaccination; attitudes toward HZ risk and vaccination; and vaccination practices and barriers. Secondary objectives were to identify approaches, practices, and guidelines used when recommending/prescribing/administering vaccines and to explore differences in specialists’ knowledge and attitudes toward HZ vaccination.
Materials and Methods
Study design
In this cross-sectional, web-based survey (GSK study identifier: VEO-000280), physicians in the United States who were part of M3’s Global Research (M3) physician panel were invited to participate by M3 through emails, which were sent out in waves. The panel is derived from multiple sources—including individual opt-ins, other M3 services, and peer referrals—and is updated annually to confirm that panel members are representative of US physicians in terms of age, gender, geography, and year of graduation from medical school. The web-based questionnaire, which was developed using best practices for instrument development 17 by RTI Health Solutions to address the study objectives, as well as to describe the study participants and characteristics of their clinical practice, was self-completed at the physician’s convenience. A feasibility assessment (to confirm the planned eligibility criteria and appropriate response ranges; n = 50) and cognitive pretesting (to evaluate the questionnaire and ensure the items, response options, and recall periods were understandable and easily answered; n = 5) were conducted before fielding the questionnaire. The questions in the final questionnaire are detailed in Supplementary Text S1.
Study population
Physicians with the following specialties were randomly recruited from a physician panel by M3, screening specifically for those who treat adult patients with diseases of interest: psoriasis for dermatologists, IBD for gastroenterologists, HIV for infectious disease specialists, solid tumors or hematological malignancies for oncologists, and RA for rheumatologists. The study aimed to include 600 specialists (150 dermatologists, 125 gastroenterologists, 100 infectious disease specialists, 125 oncologists, and 100 rheumatologists). Once individual specialist targets had been met, no new respondents were allowed (although in-progress surveys could be finished). Once all targets had been met, the survey was closed to new and in-progress responses.
Eligible specialists were required to be licensed and board-certified physicians currently practicing in the United States; their primary specialties could include dermatology, gastroenterology, infectious diseases, oncology (or hematology/oncology), or rheumatology; they must have treated at least one adult patient with the specified diseases in the last three months; worked more than 20 hours per week on average for patient care; possessed the ability to complete the survey in English; and provided informed consent. Physicians who had previously participated in cognitive pretesting were excluded, as were those whose primary practice was located in states that prohibit monetary compensation for participation in observational studies involving physicians (namely Vermont, Minnesota, or Massachusetts).
Objectives
The primary objectives were to describe specialists’ knowledge of HZ, the risk of HZ in their patient population, vaccination for the prevention of HZ; attitudes toward HZ risk and vaccination (HZ and other) in their patient population; and vaccination (HZ and other) practices and barriers. Secondary objectives were to identify approaches, practices, and guidelines used when prescribing/recommending/administering vaccinations (HZ and other) in at-risk patients and to explore differences in specialists’ knowledge and attitudes relating to HZ vaccination.
Data analysis methods
Physician responses are summarized as counts and/or percentages for categorical variables and as means and standard deviations (SDs) for continuous variables.
Multivariable logistic regression was used to explore factors associated with correctly identifying all approved indications for RZV; correctly identifying all current ACIP recommendations for HZ vaccination; and being extremely/very likely to recommend HZ vaccination to adults with the disease of interest. Models were also developed for knowledge of the current ACIP recommendations for HZ vaccination for adults who had previously received ZVL and the current ACIP recommendations excluding the former. Initially, univariate logistic regressions, including the dependent and candidate variables, were used to assess the association between these variables and the outcome of interest. Next, multivariable logistic regression models were developed using backward selection to select final independent variables (with P < 0.1). Certain variables were selected for inclusion in the final models based on expected clinical relevance. Further details about the models are available in Supplementary Text S2.
All analyses were performed using SAS 9.4 statistical software (SAS, Cary, NC).
Ethics
The study was approved for exemption by an RTI institutional review board and was completed in accordance with the Declaration of Helsinki.
Results
Study population
Of 11,041 health care providers (HCPs) who were invited to participate, 752 accessed the link. Of these, 88 were ineligible and 51 were eligible and gave consent but did not complete the survey because the quotas were already met. Therefore, 613 physician specialists completed the survey during March 4 to March 28, 2022 (150 dermatologists, 126 gastroenterologists, 101 infectious disease specialists, 125 oncologists, and 111 rheumatologists). Most specialists were male (65%), White (55%), aged 30 to 59 years (78%); worked in group private practice (43%) or academic medical centers (25%); and had at least 5 HCPs in their practice (75%) (Supplementary Table S1). Most were in urban (49%) or suburban (46%) settings, with 5.1% rural. Specialists treated a mean (SD) of 107 (96) adults per week, a mean of 37% of whom had the disease of interest (Supplementary Table S1). Specialists reported having seen a mean (SD) of 17 (33) cases of HZ overall and 8.1 (21) among patients with the disease of interest within the past year (Supplementary Table S2).
Knowledge of HZ risk factors, incidence, and vaccination
Overall, 77% of specialists correctly identified all five HZ risk factors (58% of oncologists to 92% of infectious disease specialists), with immunosuppressive medications being identified by 94%, bone marrow or solid organ transplant recipients by 92%, chronic conditions by 90%, HIV by 90%, age at least 50 years by 88%, and cancer by 88% (Fig. 1). However, only 32% of specialists selected the correct incidence rate of HZ in the United States (around 5 cases per 1,000 PY) (Supplementary Fig. S1).
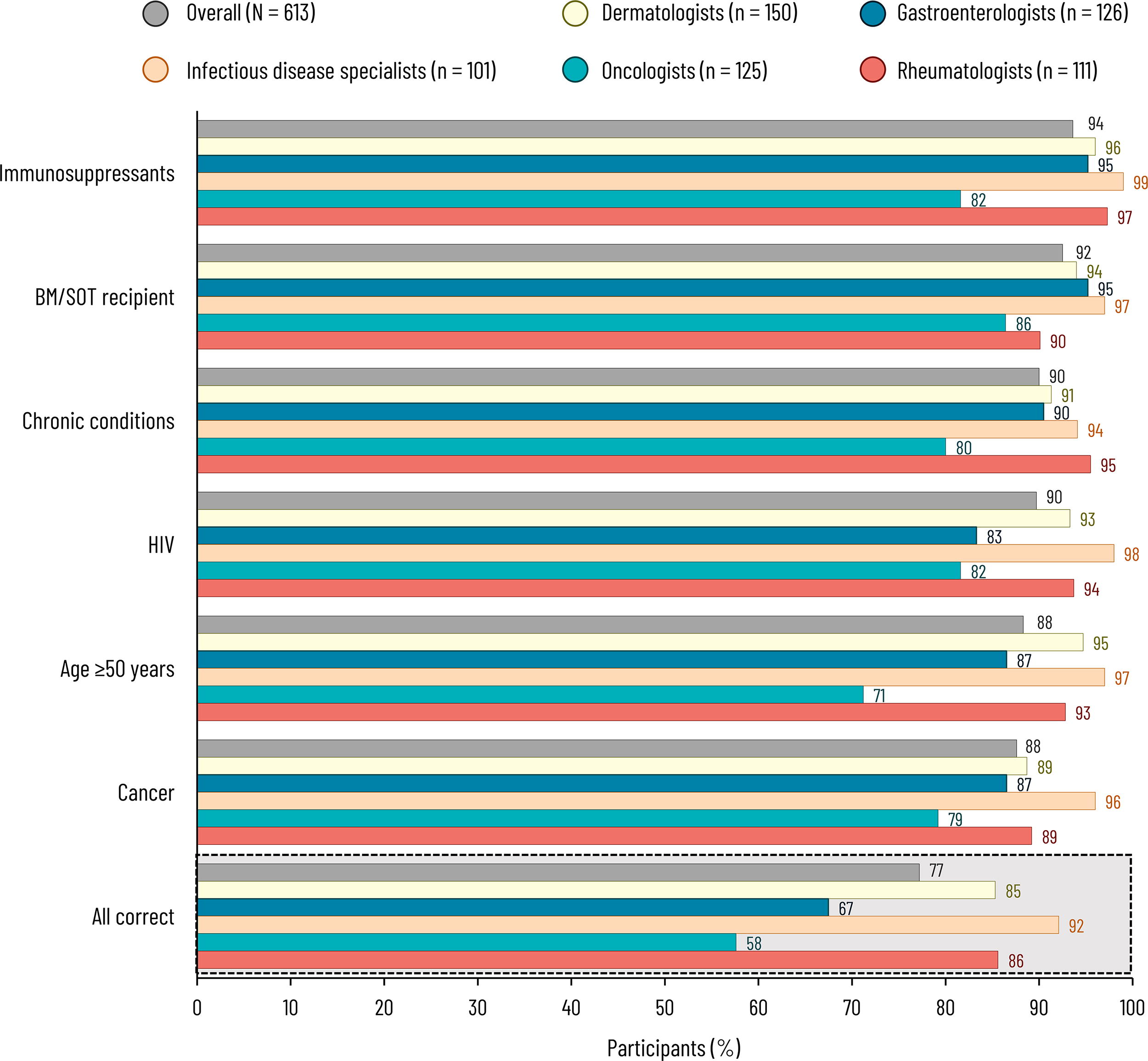
Responses to “Which of the following are risk factors for developing or contracting herpes zoster?” (Q16). Specialists were asked to select all that applied (all available options were correct) or could answer “none of the above” or “I do not know”, which were selected for 0.3% and 1.3% (overall), 0% and 2.7% (dermatologists), 0% and 0.8% (gastroenterologists), 1.0% and 0% (infectious disease specialists), 0.8% and 2.4% (oncologists), and 0% and 0% (rheumatologists), respectively. BM, bone marrow; HIV, human immunodeficiency virus; Q, question; SOT, solid organ transplant.
Only 43% of specialists correctly identified all approved indications for RZV (23% of oncologists to 55% of rheumatologists) (Fig. 2). Overall, 84% correctly identified the approved indication for the prevention of HZ in adults aged at least 50 years. Fewer (76%) identified the approved indication for the prevention of HZ in adults aged at least 18 years who are or will be at increased risk of HZ due to immunodeficiency or immunosuppression caused by known disease or therapy. However, 15% thought that RZV was approved for all adults for the prevention of HZ; and 19% and 23% thought that it was approved for the prevention of primary varicella infection in adults aged at least 18 and at least 50 years, respectively.
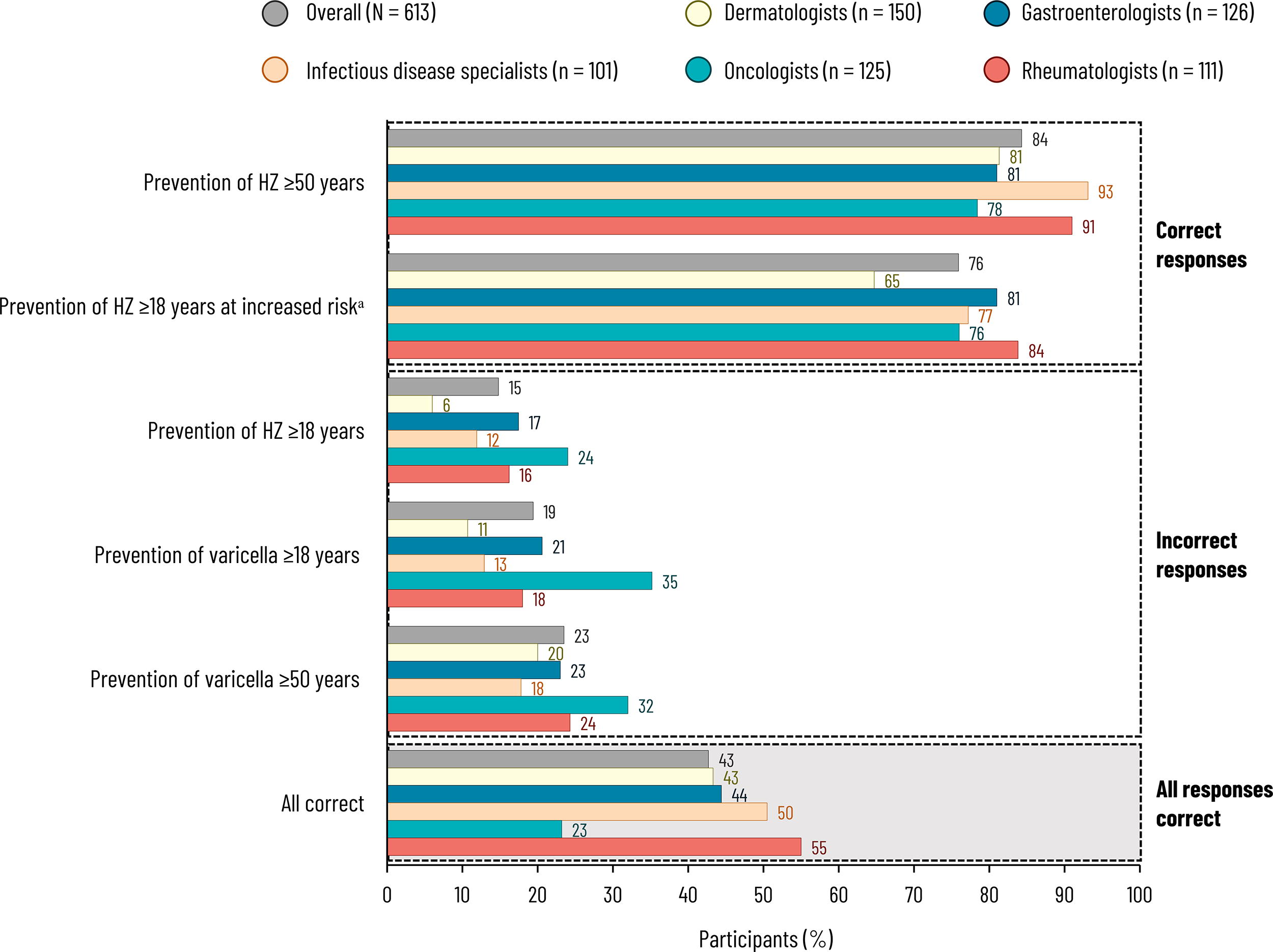
Responses to “According to the prescribing label, what are the approved indications for herpes zoster vaccination?” (Q17). Specialists were asked to select all that applied or could answer “none of the above” or “I do not know”, which were selected for 0.5% and 7.7% (overall), 0.7% and 14% (dermatologists), 0.8% and 10% (gastroenterologists), 0% and 2.0% (infectious disease specialists), 0.8% and 6.4% (oncologists), and 0% and 2.7% (rheumatologists), respectively. aExact wording: “Prevention of herpes zoster in adults aged 18 years and older who are or will be at increased risk of herpes zoster due to immunodeficiency or immunosuppression caused by known disease or therapy”. HZ, herpes zoster; Q, question.
Only 18% of specialists correctly identified all ACIP HZ recommendations (11% of oncologists to 33% of infectious disease specialists) (Fig. 3). Overall, 84% correctly identified the 2018 recommendation 6 to administer RZV to adults aged at least 50 years. Fewer (67%) identified the 2022 recommendation 10 for immunocompromised/immunosuppressed adults aged at least 19 years. Only 29% knew that RZV is recommended for adults who have previously received ZVL.
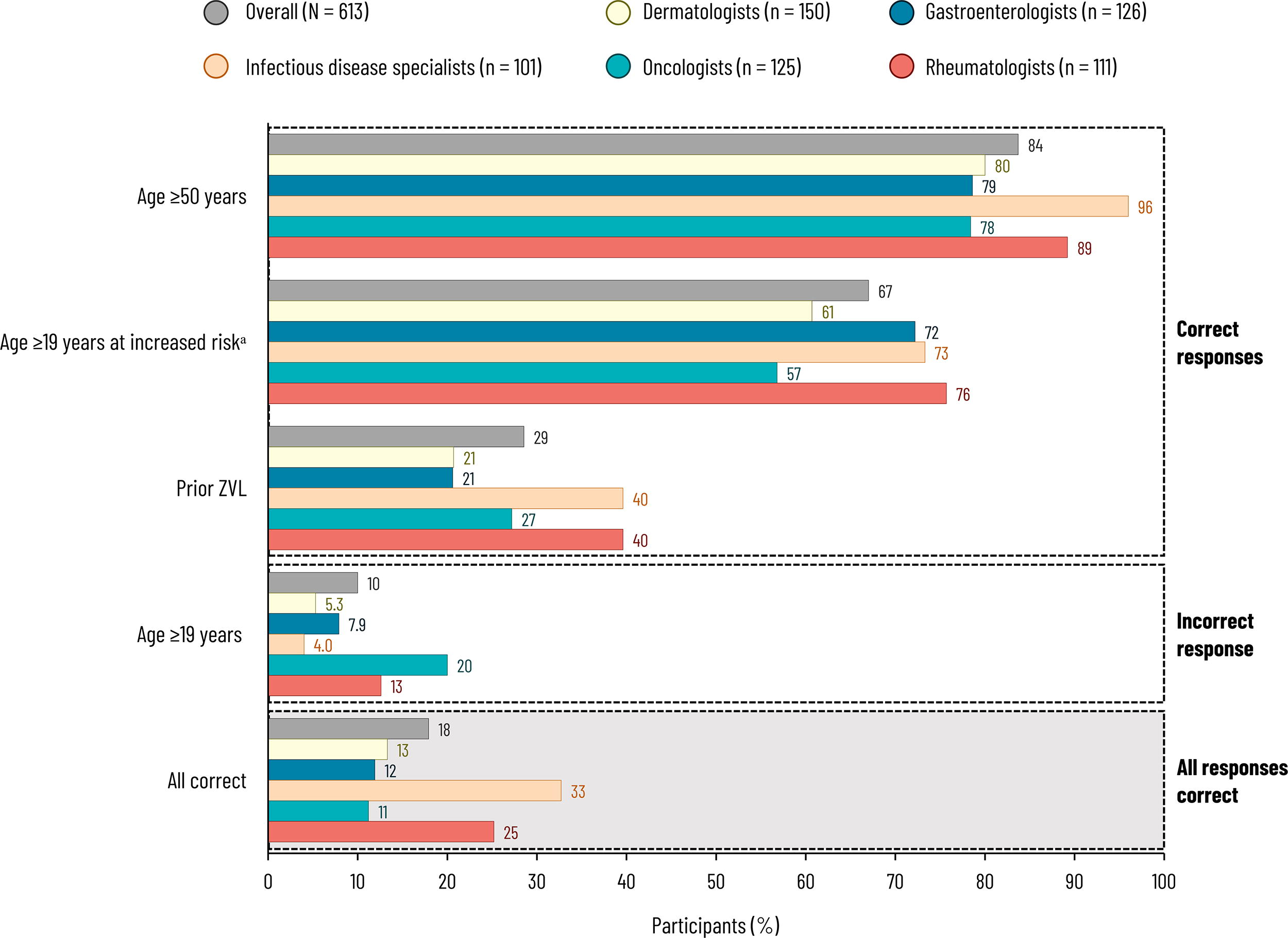
Responses to “To your knowledge, what are the current ACIP recommendations for herpes zoster vaccination in adults?” (Q18). Specialists were asked to select all that applied or could answer “none of the above” or “I do not know”, which were selected for 0.2% and 10% (overall), 0% and 17% (dermatologists), 0.8% and 13% (gastroenterologists), 0% and 1.0% (infectious disease specialists), 0% and 13% (oncologists), and 0% and 3.6% (rheumatologists), respectively. aExact wording: “Recommended for adults aged 19 years and older with immunocompromising diseases or taking immunosuppressive treatments”. ACIP, Advisory Committee on Immunization Practices; Q, question; ZVL, zoster vaccine live.
Attitudes toward HZ risk and vaccination
Most specialists agreed that HZ was a serious risk to patients with the disease of interest (84% overall; 67% of dermatologists to 95% of rheumatologists) (Fig. 4A) and felt that it was extremely/very important to vaccinate against HZ in these populations (75% overall; 59% of dermatologists to 86% of rheumatologists) (Fig. 4B). Among 607 who recommend/prescribe/administer vaccines to adult patients, 69% were extremely/very likely to recommend HZ vaccination to adults with the disease of interest (50% of dermatologists to 81% of rheumatologists) (Fig. 4C).
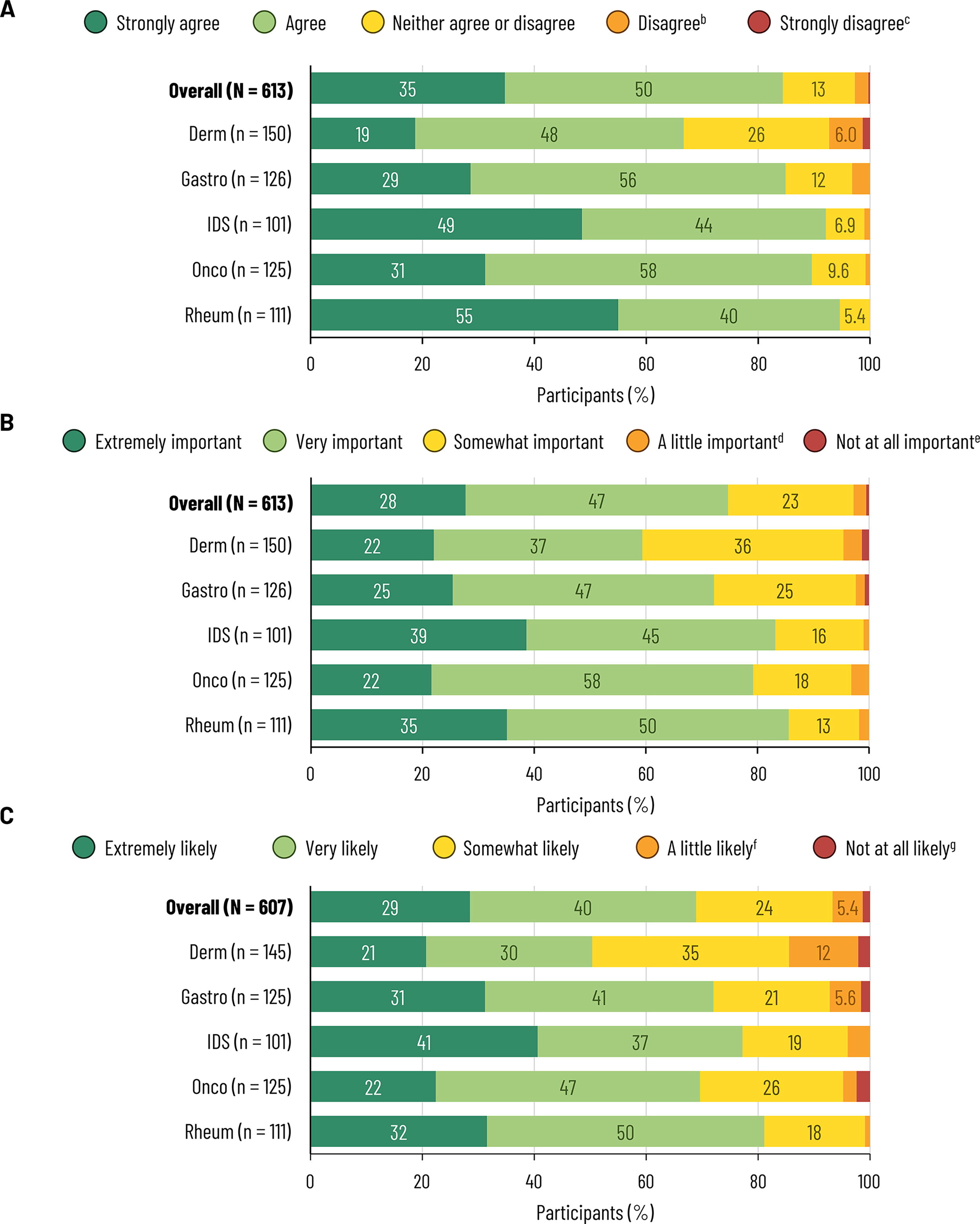
Responses to
On average, specialists estimated that they had initiated a conversation about any vaccination with 61% of their adult patients with the disease of interest over the past year falling to 42% for HZ vaccination (Supplementary Table S2). The most common reasons for not initiating conversations about HZ vaccination were that there are more urgent/acute issues to discuss (65%), patients have already been vaccinated (61%), lack of time during appointments (43%), lack of patient willingness/motivation (33%), and that other HCPs (e.g., primary care physicians [PCPs], pharmacists) discuss HZ vaccination with these patients (27%) (Supplementary Fig. S2).
Vaccination practices and barriers
Most specialists (94%) responded that they recommend vaccines to some adult patients, with 54% saying that they prescribe vaccines, and 36% that they administer vaccines, with 607 (99%) doing at least one of these (Supplementary Fig. S3). Among 393 who do not administer vaccines, the most common reasons were that patients receive vaccinations elsewhere (e.g., PCP, pharmacy, health department) (74%) or from other staff members (31%) (Supplementary Fig. S4).
Among specialists who had recommended/prescribed/administered vaccines to at least one adult in the past 3 months, 81%, 73%, and 66% responded that they had recommended/prescribed/administered influenza, HZ, and pneumococcal disease vaccines, respectively, to at least one adult in the past 3 months (Supplementary Fig. S5). For the subset of adult patients with the disease of interest, 74%, 64%, and 60% of specialists responded that they had recommended/prescribed/administered influenza, HZ, and pneumococcal disease vaccines, respectively, to at least one adult patient with the disease of interest in the past 3 months (Supplementary Fig. S6).
The mean numbers of HZ vaccinations recommended/prescribed/administered to any adult and adults with the disease of interest in the past year are provided in Supplementary Table S2.
The most frequently selected factors for deciding whether to recommend/prescribe/administer an HZ vaccine to an adult with the disease of interest were the level of immunosuppression or disease severity (91%), patient age (85%), and medical history (78%) (Supplementary Fig. S7).
The most common perceived barriers to HZ vaccination recommendation included insufficient time during visits, contraindications, and inadequate reimbursement (Supplementary Fig. S8). The most commonly perceived barriers to HZ vaccination administration included lack of patient motivation/willingness, lack of patient knowledge of HZ risk, and cost (Supplementary Fig. S9).
Approaches, practices, and guidelines used for vaccinations
Most specialists (77%) agreed that ACIP sets the standard for vaccine practices in the United States (Supplementary Fig. S10). Furthermore, specialists responded that they always (23%) or most of the time (50%) followed the ACIP recommendations when deciding on recommending/prescribing/administering vaccines to adults with the disease of interest (Supplementary Fig. S11). However, the most common approaches or processes to inform decision-making were clinical judgment (97%), patient request (84%), and non-ACIP guidelines from professional organizations (80%), while only 32% used a state immunization information system, 43% standing order protocols, and 46% patient questionnaires about vaccinations (Supplementary Fig. S12). Results were generally similar across specialties, but infectious disease specialists were much more likely than other specialists to use ACIP recommendations (94% vs 26% to 46%), Centers for Disease Control and Prevention (CDC) immunization schedules (87% vs 45% to 67%), and standing order protocols (65% vs 27% to 47%). When asked specifically about HZ vaccination, results were similar to those for vaccinations in general (Supplementary Fig. S13 and Supplementary Fig. S14). Most specialists reported using non-ACIP professional guidelines relevant to the specialty to inform HZ vaccine decision-making (Supplementary Fig. S15).
Less than one-third of the 607 specialists who recommend/prescribe/administer vaccines to adults reported that they stock HZ vaccine at their practice (30% overall; 7.6% of dermatologists to 60% of infectious disease specialists). Among those who do not stock HZ vaccine, the most common reason was that most of their patients received HZ vaccination elsewhere (72%) (Supplementary Fig. S16).
Factors associated with knowledge and likelihood of recommending HZ vaccine
By multivariable logistic regression, factors that were significantly associated with knowledge of the approved RZV indications included identifying age at least 50 years as a risk factor for HZ (adjusted odds ratio [aOR] 2.87), rheumatologist (vs dermatologist) (1.79), and rural (vs urban) location (0.32) (Supplementary Table S3). Factors significantly associated with knowledge about the current ACIP HZ vaccination recommendations included identifying age at least 50 years as a risk factor for HZ (aOR 9.23), specialist age 50 to 59 or 60 to 69 (vs up to 39) years (2.13 and 4.33, respectively), infectious disease specialist or rheumatologist (vs dermatologist) (2.43 and 2.18, respectively), and stocking HZ vaccine (2.10). Results for knowledge about the recommendation to give RZV to adults who previously received ZVL and results excluding this factor are shown in Supplementary Table S3.
Factors that were significantly associated with being extremely/very likely to recommend HZ vaccination to adults with the disease of interest were identifying immunosuppressive medications as a risk factor for HZ (aOR 4.14), gastroenterologist, rheumatologist, or infectious disease specialists (vs dermatologist) (2.58, 2.51, and 2.11, respectively), having recommended/prescribed/administered HZ vaccine in past 3 months to at least one adult with the disease of interest (2.79), stocking HZ vaccine (2.29), and outpatient clinic (vs group private practice) (2.01) (Supplementary Table S3).
Discussion
This survey was administered to 613 specialists who care for adults at increased risk of HZ due to immunodeficiency or immunosuppression caused by disease or therapy to better understand their knowledge, attitudes, and practices regarding HZ vaccination.
Only 43% of specialists correctly identified all RZV indications, although this was mainly because specialists (particularly oncologists) chose incorrect answers (e.g., for varicella prevention) in addition to correct responses. Rheumatologists were most likely to know the RZV indications, and this specialty was significantly associated with correct responses to these knowledge questions in multivariable analysis, as was working in a nonrural location (although it should be noted that only 5.1% of specialists worked in rural locations).
Full knowledge of the current ACIP recommendations for HZ vaccination 6,10 was even lower, with only 18% of specialists identifying all correct responses. This was mainly because only 29% knew that patients who have previously received ZVL are recommended to receive RZV. Factors that were significantly associated with knowledge of all ACIP HZ vaccine recommendations in multivariable analysis included being an infectious disease specialist or rheumatologist, being aged 50 to 69 years, and stocking HZ vaccine. Overall, these results indicate that most specialists could benefit from additional education on the ACIP recommendations for HZ vaccination.
For patients with the disease of interest, dermatologists were least likely to agree that HZ is a serious risk, think that it is extremely/very important to vaccinate against HZ, and be extremely/very likely to recommend HZ vaccination. This may be because the risk of HZ is lower among patients with psoriasis than the other diseases of interest, 5 but suggests that there may be an opportunity to educate dermatologists, especially for patients on biological therapies, systemic steroids, Janus kinase inhibitors, or combination systemic treatments. 18 –20
Infectious disease specialists were more likely than the other specialists to agree that ACIP sets the standard for US vaccine practices, always/mostly follow ACIP recommendations for patients with the disease of interest, and use ACIP recommendations to inform decision-making. The other specialists were more likely to use clinical judgment, patient requests, or other professional guidelines than ACIP recommendations to inform decision-making. While these are generally similar to the ACIP recommendations, there can be a time lag between ACIP recommendations being published and incorporated into other professional guidelines, highlighting the importance of HCP awareness of new ACIP recommendations.
Overall, 65% of specialists reported that one reason for not discussing HZ vaccination with their patients with the disease of interest was that there are more urgent or acute issues to discuss, with 43% citing insufficient time during appointments. Alternative approaches to improve HZ vaccination uptake among immunocompromised/immunosuppressed adults could include clinical decision support systems that provide prompts about vaccination in particular patients and the involvement of other care team members, for example, pharmacists and PCPs. Pharmacists have been recognized as playing an important role in recommending and administering vaccines. 21 Pharmacists can also help to educate the public about the efficacy and safety of vaccines, thus reducing vaccine hesitancy. 22 –24 A recent meta-analysis of mainly US studies found that pharmacist involvement (as advocates and/or immunizers) could improve immunization rates. 25 Furthermore, recent claims database studies have reported that 92% of claims for initial RZV doses were from pharmacies 26 and RZV completion rates were higher in pharmacy vs medical claims databases (73% vs 49% at 6 months). 27
Various previous surveys on knowledge and attitudes toward HZ vaccination in the United States have been carried out. Those conducted before the 2018 recommendations 6 mostly included PCPs and generally indicated that PCPs recognized the burden of HZ 28,29 and were likely to recommend HZ vaccination, 28 –32 but that HZ vaccination rates were suboptimal 33 –35 and there was a lack of awareness of and/or adherence to ACIP guidelines. 31,34 Results from a survey of HIV care providers in 2008–2009 were similar, with 89% agreeing that HZ was a serious disease in patients with HIV and 73% that HZ vaccination was important. 36 Although 75% did not administer HZ vaccinations to their patients with HIV, 36 this was likely because at that time, only ZVL was available and the ACIP recommendation then was not to give ZVL to people with clinical manifestations of HIV, but there was no specific guidance for people with HIV without clinical manifestations. A 2020 survey (carried out between the two latest recommendations 6,10 ) reported that PCPs and pulmonologists recognized the importance of HZ vaccination for patients with chronic obstructive pulmonary disease aged 50 years and over. 37 Another 2020 survey found that approximately one-third of PCPs were already recommending RZV to immunocompromised patients aged 18–49 years. 38
Although specialists treating immunocompromised patients are now in a position to recommend/prescribe/administer HZ vaccination, approximately 27% of specialists in the current survey believed that recommending, prescribing, or administering HZ vaccination was not within their purview, assuming that PCPs or pharmacists would address this matter with patients. Moreover, a significant proportion of specialists (74%) cited patient receipt of vaccines elsewhere as the reason for not administering vaccines themselves. Only 30% of specialists reported stocking HZ vaccine, and of those who did not, 72% attributed this decision to their patients receiving HZ vaccination elsewhere. A similar trend was observed among PCPs in a prior study, with 55% reporting vaccine stocking, while 77% referred patients to pharmacies for HZ vaccination. 29 Although specialists have an important role to play in HZ vaccination for immunocompromised patients, other members of the care team (e.g., pharmacists and PCPs) can also play an important role, provided they are aware of the new ACIP HZ guidelines. 10 Therefore, fostering better coordination of care across HCPs is imperative. This coordination could be facilitated through state information systems (as well as e.g., electronic health records, immunization information systems, vaccine management software) that incorporate reminders and recalls for vaccinations, a resource that was utilized by only approximately one-third of specialists in the current survey.
These findings underscore the importance of interdisciplinary collaboration and awareness of evolving vaccination guidelines among HCPs. Improved communication and shared responsibility among specialists, PCPs, and pharmacists can enhance the accessibility and adherence to essential vaccines, ultimately leading to better population health outcomes.
Strengths and limitations
As this study had limited inclusion/exclusion criteria and enrolled a heterogeneous sample of US specialists, the results should be generalizable to US specialists who provide care for adults at increased risk of HZ owing to immunodeficiency or immunosuppression caused by disease or therapy. However, selection bias is a potential limitation because specialists had to belong to the physician panel and be willing to participate. Response bias is also possible, as the survey results represent reported rather than observed practice. Although specialists could not go back and change earlier answers, they could have paused the survey and looked up answers, hence the survey results could overestimate knowledge. It is also possible that they could have provided answers that indicated better practice than reality. Furthermore, we recognize that, although our survey elicited responses to a range of key aspects, it was not totally comprehensive. For example, we did not include the ACIP recommendation to give HZ vaccination regardless of prior HZ disease 6 in our question on the ACIP recommendations, and this has previously been flagged as an opportunity for enhancing knowledge. 37
The overall response rate for the survey was low (5.6%), but this was partly because quotas per specialty were set. In addition, most specialists worked in group private practices or academic medical centers, hence these results may not be generalizable to other work environments. Finally, this study focused on specialists, but other HCPs play a vital role in vaccinations, including pharmacists, nurse practitioners, physician assistants, and PCPs. Future research could focus on their knowledge and practice about HZ vaccinations for adult patients at increased risk of HZ due to immunodeficiency or immunosuppression.
Conclusions
This study assessed the knowledge, attitudes, and practices of various specialists related to HZ vaccination in adults at risk of HZ due to immunodeficiency or immunosuppression caused by disease or therapy. Although 77% of specialists thought that ACIP sets the standard for US vaccine practices, only 18% knew all the current ACIP recommendations for HZ vaccination. Some specialists (27%) thought that they did not need to discuss HZ vaccination with their immunocompromised patients because other HCPs (e.g., pharmacists, PCPs) fulfill this role. In addition, the most common reason for not stocking or administering vaccines was that patients receive vaccinations elsewhere. There may be opportunities to improve adherence to ACIP recommendations for HZ vaccination in at-risk populations by educating specialists to increase awareness and by ensuring that other care team members (e.g., PCPs, pharmacists) are aware of the latest ACIP HZ vaccination recommendations, as they can often identify at-risk patients and recommend or administer HZ vaccination themselves.
Footnotes
Acknowledgments
The authors thank Tina Valentino (GSK) for her help with the interpretation of the results. The authors also thank Business & Decision Life Sciences Medical Communication Service Center for editorial assistance, article coordination, and writing support, on behalf of GSK. Jenny Lloyd (Compass Healthcare Communications Ltd., on behalf of GSK) provided medical writing support.
Authors’ Contributions
Study design: D.S., C.S., N.S., M.R., and S.P. Data collection/generation: C.S., M.R., and D.G. Data analysis and interpretation: D.S., C.S., N.S., M.R., D.G., and SP. All authors participated in the development of this article and in its critical review, with important intellectual contributions. All authors had full access to the data and gave approval before submission. All authors agreed to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved. The work described was carried out in accordance with the recommendations of the International Committee of Medical Journal Editors for the conduct, reporting, editing, and publication of scholarly work in medical journals.
Author Disclosure Statement
D.S., N.S., and S.P. are employed by and hold shares in GSK. C.S., M.R., and D.G. are employed by RTI Health Solutions, which received funding from GSK for the development of the study materials, the conduct of the study, and analysis and interpretation of the study results. The authors declare no other financial and nonfinancial relationships and activities.
Funding Information
GSK funded this study (GSK study identifier: VEO-000280) and was involved in all stages of study conduct, including analysis of the data. GSK also took in charge all costs associated with the development and publication of this article.
Data Availability
For requests for access to anonymized subject-level data, please contact corresponding author.
Supplementary Material
Supplementary Text S1
Supplementary Text S2
Supplementary Figure S1
Supplementary Figure S2
Supplementary Figure S3
Supplementary Figure S4
Supplementary Figure S5
Supplementary Figure S6
Supplementary Figure S7
Supplementary Figure S8
Supplementary Figure S9
Supplementary Figure S10
Supplementary Figure S11
Supplementary Figure S12
Supplementary Figure S13
Supplementary Figure S14
Supplementary Figure S15
Supplementary Figure S16
Supplementary Table S1
Supplementary Table S2
Supplementary Table S3
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
