Abstract
The observed increase in osteoarthritis (OA) of the knee as a result of an aging population and the obesity epidemic has led to a concomitant increase in the rates of total knee replacement (TKR), placing an additional financial and social burden on the ability of health care systems to control medical costs. Our study shows how a home-based, noninvasive biomechanical intervention reduced the rate of progression to surgery for a cohort of 237 patients with knee OA deemed eligible for TKR based on pre-established clinical selection criteria. Over the 24-month study period, 204 patients (86%) avoided surgery, with only 33 patients (14%, 95% confidence interval 82%–91%) progressing to a TKR with an average length of time to TKR of 324 days (ranging from 31 to 671 days). The application of this intervention provides health plans and provider networks managing patient care under financial risk arrangements an opportunity to realize significant cost savings without compromising quality of care or clinical outcomes.
Introduction
Osteoarthritis (OA) ranks as the 15th highest cause of the number of years lived with a disability. 1 OA is known to negatively impact the quality of life (QoL) in older adults by causing poor functional ability, pain-related distress, depression, and feelings of social isolation. 2 Both aging and rising rates of obesity are contributors to the increase in the prevalence of knee OA. 3 In the absence of therapies that are able to reverse the pathology of OA, patients are required to live with their chronic disability and pain.
Treatments for patients with knee OA aim to alleviate symptoms, provide joint stability, and postpone disease progression. 4 Standard treatments include arthritis education, structured land-based exercise programs, neuromuscular training, and dietary weight management. However, the efficacy of nonsurgical interventions for patients with knee OA is both limited and short term. 5 As a result, patients oftentimes choose surgery to alleviate their pain, and regain functionality and QoL. Data show that over half of the US adults diagnosed with knee OA will undergo a total knee replacement (TKR) as an end-stage solution. 6
In 2017, almost a million primary knee arthroplasties were performed in the United States 7 with no signs that the number of arthroplasties performed will abate. A study by Singh et al in 2019 estimates that by 2040, the number of arthroplasties will rise to ∼3 and a half million TKRs. 8 At the same time, because the average age for a TKR is falling, the prevalence of revision knee surgery is also expected to rise. By 1 estimate, there will be 700,000 revision surgeries performed by 2050. 9
Despite the favorable surgical outcomes, ∼20% of post-TKR patients continue to experience chronic pain and an equal number report that their expectations for a full recovery are unmet. 2,10 These projections show that the steep increase in TKR and revision surgeries will place an immense burden on the cost of health care, highlighting the urgent need for new nonsurgical approaches that more effectively manage OA symptoms of the knee. 9
Previous studies reported on a home-based, noninvasive biomechanical intervention that produced improved functionality and reduced pain symptoms in patients with knee OA. 12 –14 Another study published in 2013 by Bar-Ziv et al examined the changes in clinical outcomes among patients eligible for TKR treated with the same biomechanical device. 11 Although surgery avoidance was not the primary outcome of this investigation, the study showed that at 2 years, 2.5% of the study enrollees assigned to the intervention required TKR compared with 30% of the controls. Because of the relatively small sample size, the authors concluded that further research on a larger cohort of patients was required to validate the outcomes of the intervention.
In this study we examine how a home-based, noninvasive biomechanical intervention provided to patients with end-stage OA and eligible for TKR enabled the majority of patients to avoid surgery. In as much as the patients were enrollees of Heritage Provider Network (HPN), a nationally recognized physician network that provides care under financial at-risk contracts with its partner health plans, we believe the findings are of importance for other health care delivery systems such as accountable care organizations, operating under financial risk relationships.
Methods
In this retrospective study, we assessed the rates of surgery avoidance in patients deemed eligible for TKR who elected to receive a noninvasive biomechanical functional rehabilitation intervention. The study comprised a retrospective review of patient data and required no IRB approval. All personal health information [PHI] was de-identified. Clinical metrics were collected as secondary endpoints, namely changes in the levels of pain, function, and perceptions of QoL, as well as changes in spatiotemporal gait parameters.
HPN is a nationally recognized vertically integrated physician provider network that has been providing care management to patients in accordance with population health principles for almost 4 decades. In the areas in which it operates, HPN through its affiliated medical groups contracts with all the major US national health plans on a global budget for which HPN is financially at risk and responsible for the costs of patient care management that includes professional (physicians), institutional (hospital) services, durable medical equipment, and injectable pharmacy. Under its health plan's delegated assignment for utilization and quality management, treating physicians have the authority to make clinical decisions on patient care and assign patients to different treatment modalities.
Because HPN pays all claims for services rendered to patients (excluding oral pharmacy), HPN provides an ideal value-based environment (similar to bundled payment programs and Accountable Care Organizations 10 ) to assess population health care practices, in particular the clinical efficacy and quality outcomes of innovative approaches to high-cost medical and surgical procedures, such as TKR surgery.
To assess the benefits of the biomechanical intervention, we examined as the primary endpoint of the study the progression to surgery over a 24-month follow-up period for each of the 237 patients who met the eligibility criteria for TKR and who received the nonsurgical intervention during a period between March 2018 and March 2019. Over the same time period, a group of 294 patients who met eligibility criteria for TKR and chose a surgical intervention constituted the control group. As a secondary endpoint, as already noted, we collected clinical metrics for 1 year for patients receiving the intervention.
The eligibility criteria for TKR comprised a combination of the following: 2 professional claims related to knee pain, radiological confirmation of knee OA, subjective knee pain >3 months impacting the QoL, no reliance on assistive devices to walk indoors, and <2 falls in the past year.
Patients who met the eligibility criteria were offered the option of surgical intervention or the biomechanical intervention through HPNs established utilization management program. Patients who were receiving standard physiotherapy, corticosteroid, or hyaluronidase knee injections at the time of the assignment continued on these therapeutic modalities as per their physician directives. Patients who were treated with the noninvasive biomechanical intervention were referred to physiotherapists trained in the application and use of the modality (See Biomechanical intervention section below).
Outcomes
The primary outcome measure of this study was the avoidance of TKR surgery rates at the end of a 2-year follow-up for patients selecting the biomechanical intervention and for control patients selecting surgery from the time of their enrollment into the study. Secondary outcomes included the measurement in the changes in clinical symptoms at 12 months after initiation of the intervention. Although cost-saving determination was not an endpoint of this study, given that HPN is the payor for all care services for its members, the cost savings from TKR avoidance were estimated based on Medicare reimbursement for both professional and institutional services and are included in the Discussion section.
All patients underwent a computerized gait analysis test and completed 2 validated patient-reported outcome measures (PROMs) to assess pain, function, and QoL. These were completed during the initial evaluation (baseline assessment), then repeated at 3, 6, and 12 months into the treatment.
Trained physical therapists (clinicians) used a pressure walkway gait analysis system (Zeno, ProtoKinetics LLC, PA) to measure spatiotemporal gait parameters. 15 Patients were instructed to walk barefoot at a self-selected speed over a 4.3 meters pressure mat with 2 meters before and after the measurement area to allow for sufficient acceleration and deceleration time outside of the measurement area. Each gait test included 4 lengths from which the mean values were calculated for each parameter.
The parameters recorded include velocity (cm/s), step length (cm), and single-limb support (SLS) phase (% gait cycle). Step length and SLS were calculated for the less and more symptomatic limbs, respectively. For patients with bilateral OA, the more symptomatic limb was determined by the limb with the lower SLS at baseline, whereas for patients with unilateral OA, the more symptomatic limb was the limb that was reported with OA.
Patients were asked to complete 2 gold-standard PROMs for knee OA. The first was the Western Ontario and McMaster Universities Osteoarthritis Index (WOMAC) that was used to evaluate changes in patients' perception of pain and function. 16 WOMAC contains 24 visual analogue scale (VAS) questions that can be divided into 3 subcategories (Pain: 5Q, Functional Limitation: 17Q, and Stiffness: 2Q). Results range from 0 to 100 mm, in which 0 mm indicates no pain or limitation in function and 100 mm indicates the most severe pain or limitation in function.
The second was the Short-Form 36 (SF-36) QoL questionnaire. 17 Results range between 0 and 100, with 0 indicating the worst QoL and 100 indicating the best QoL. The total score is made up of 8 subcategories that make up 2 summarizing scores: Physical Component Summary (PCS) and the Mental Component Summary (MCS). These reflect the patients' physical/mental condition, respectively.
Patients were provided education on their clinical condition and instructed to use the biomechanical device daily, typically starting with 30 minutes a day while performing their daily activities at home or work, and gradually building up to 2–3 hours a day while increasing their activity levels.
Biomechanical intervention
AposHealth® is an FDA cleared class I medical device for patients with knee OA. It is a biomechanical device (Apos system) combined with a treatment methodology used by patients during their normal daily activities. The AposHealth device comprises convex, adjustable biomechanical elements that are placed under the hindfoot and forefoot regions of each foot through a platform in the form of a shoe (Fig. 1). The biomechanical device combines 2 principles: first, manipulation of the center of pressure and modification of the ground reaction forces to change the forces acting upon the respective joint. 18 –25 By setting the position of the 2 pods, the clinician can redistribute the load away from the affected knee area and modify the way a patient walks.
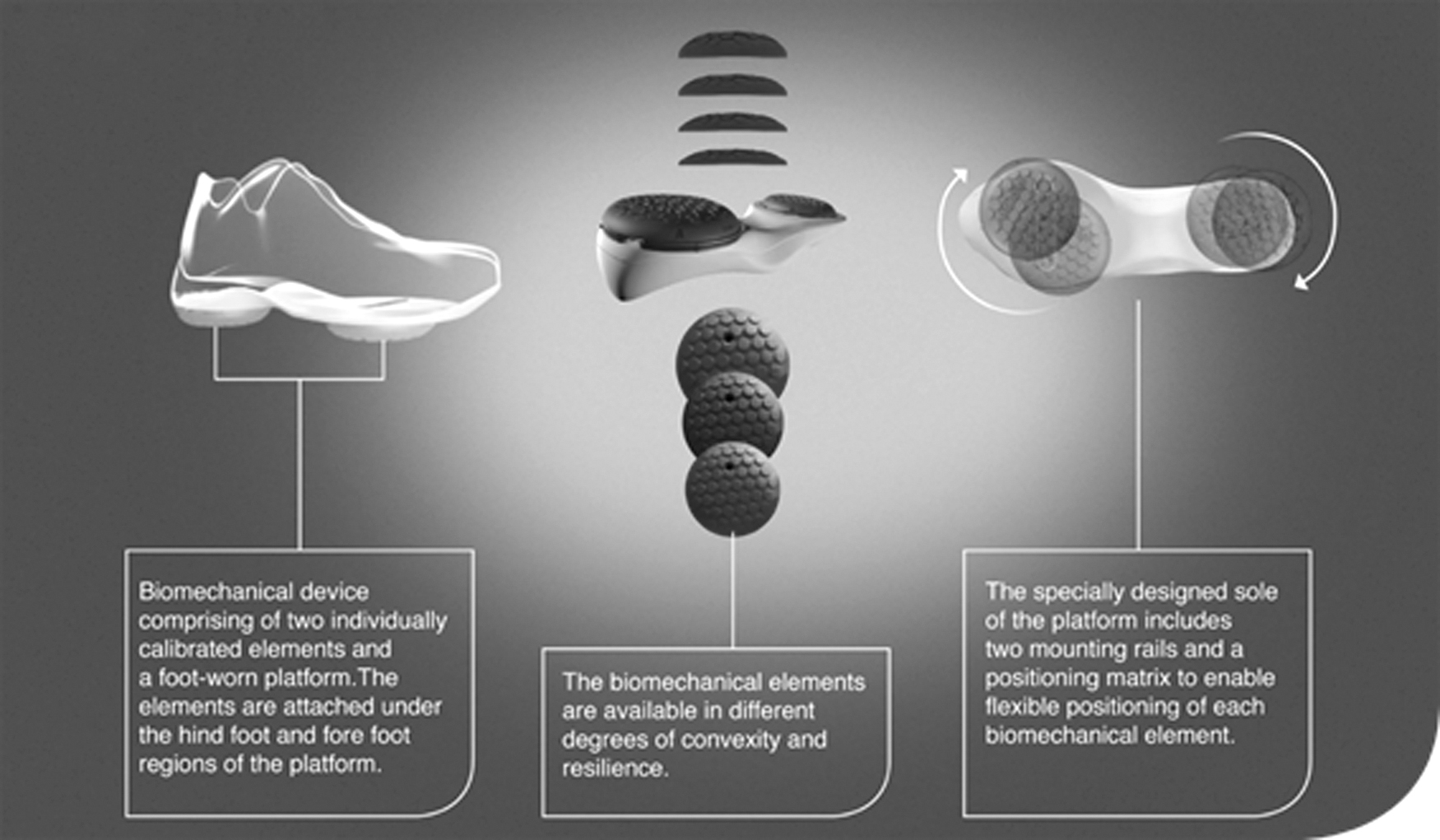
The biomechanical device. Image was provided by the company and was approved to be used.
Second, neuromuscular training is achieved by introducing controlled perturbation through the convex nature of the pods. Walking on a smaller base of support creates controlled microinstability that stimulates the proprioceptive system throughout the gait cycle. 22,26 –29
The clinician or trained physiotherapist calibrates the device individually to the patient after a thorough assessment comprising a medical history interview, computerized and visual gait assessment, assessment of PROMs, and a physical examination. When calibrating the device, the aim of the clinician is to obtain a desired functional alignment in which less pain is reported by the patient and gait patterns are more symmetrical. Patients are instructed to wear the device for 30–60 minutes a day while performing their daily activities at home or work. Patients are requested to return once every 3 months for follow-up appointments to reassess their overall condition, gait patterns, and PROMs and adjust the device calibration and the treatment program as required.
Statistical analysis
The statistical analysis hereunder was conducted by an independent biostatistician. Clinical outcomes data were analyzed using SPSS software version 27.0 with significance levels set at 2-sided P < 0.05. A Student's t-test was used to compare the baseline characteristics of patients who avoided surgery and patients who progressed to surgery. Data are presented as mean and standard deviations for spatiotemporal parameters and questionnaires (WOMAC/SF-36), followed by a 95% confidence interval (CI) for all times.
The repeated measures General Mix Model was used to assess changes over 4 time points by groups (patients who avoided surgery and patients who progressed to surgery). Lastly, we calculated the Outcome Measures in Rheumatology Clinical Trials (OMERACT)-Osteoarthritis Research Society International (OARSI) responder criteria for clinically significant improvement for each of the patients. 30 These stipulate an improvement either in the overall score or in the subscales of pain or function of at least 50% with a decrease of 2 cm on the VAS, or an improvement in both pain and function of at least 20% with a reduction of 1 cm on the VAS. 30
Results
Two hundred thirty-seven patients, 35% females and 65% males, who were eligible for TKR and who chose to receive the biomechanical intervention were each followed for a period of 24 months. Over the same enrollment period (March 2018–March 2019), 294 patients who chose to receive surgery comprised the control group. For the cohort of patients assigned to receive the biomechanical intervention, the baseline characteristics between patients who progressed to surgery and those who avoided surgery are summarized in Table 1.
Baseline Characteristics and a Comparison of Group Differences
Results are for patients who were treated with the noninvasive intervention showing the comparison between those who avoided surgery and those who progressed. Results are presented as mean (SD). Significance was set as P < 0.05.
GC, gait cycle; LS, less symptomatic; MCS, mental component summary; MS, more symptomatic; PCS, physical component summary; SF-36, Short-Form 36; SL, step length; SLS, single limb support.
The results show that over the 24-month study period, 34 patients who received the intervention (14%, 95% CI 82%–91%) progressed to a TKR. The average length of time to progress to TKR was 324 days (ranging from 31 to 671 days). Sixty-four percent of those who underwent TKR had their surgery within 12 months after the initiation of the intervention. The remainder underwent TKR after they had been using the intervention for longer than 12 months. In addition, 27 patients (11%) termed their insurance coverage and were disenrolled from HPN, and 5 patients (2%) were deceased.
Of the 294 patients in the control group who chose TKR surgery, 259 (88%) received a knee replacement.
With respect to the clinical outcomes measurements, for the 172 patients who chose the biomechanical intervention and who completed the program, 138 (88%) had clinical data at 3 months, 111 (65%) patients had clinical data at 6 months, and 52 (30%) patients had clinical data at 12 months.
The General Mixed Model that includes repeated measures from 4 visits showed a significant reduction in WOMAC pain (P < 0.001) and WOMAC function (P < 0.001) after 12 months of treatment. It is estimated that pain decreased by 19.6 points (36%) at the end of year 1, and functional disability decreased by 16.4 points (34%). There was a significant increase in the SF-36 overall score by 5.4 points (10%) at 1 year (P < 0.001). Likewise, the PCS increased significantly by 5.6 points (13%) after 12 months of treatment (P < 0.001). No significant changes in MCS were noted.
Table 2 summarizes the estimated mean from the General Mixed Model for pain, function, and QoL from baseline to 1 year. Table 3 summarizes the estimated mean from the General Mixed Model for the gait metrics. All gait metrics demonstrated a significant trend of improvement over time except for SLS.
General Mixed Model Outcomes for Patients Receiving the Biomechanical Intervention
Results are presented as estimated mean from the General Mixed Model (see) [95% CI]. Significance was set as P < 0.05.
MCS, mental component summary; PCS, physical component summary; SF-36, Short-Form 36; WOMAC, Western Ontario and McMaster Universities Osteoarthritis Index.
Changes in Spatiotemporal Gait Changes Over Time for Patients Receiving the Biomechanical Intervention
Results are presented as estimated mean from the General Mixed Model (SD) [95% CI]. Significance was set as P < 0.05.
GC, gait cycle; LS, less symptomatic; MS, more symptomatic; SL, step length; SLS, single limb support.
To further understand the differences in the patients assigned to the intervention who progressed to TKR and those who avoided TKR, we compared the clinical data for the 2 cohorts. The data showed that patients who “failed” the biomechanical intervention and progressed to TKR presented with a significantly higher pain and functional disability at their baseline presentation. Pain was measured at 53.4 (23.3) compared with 62.3 (24.2) for the nonsurgical patients versus patients referred for TKR, respectively (P = 0.04). Similarly, functional disability was 46.2 (25.6) compared with 55.8 (23.1) in the 2 groups, respectively (P = 0.04). There were no differences in SF-36 total score and PCS and MCS subcategories and no differences in gait characteristics.
The results of the General Mixed Model show that there was no interaction effect between the patients who were assigned to the intervention and did not receive TKR and those that did, namely, changes in the clinical outcomes already mentioned over time were similar in both groups.
Discussion
This study conducted over a period of 2 years evaluated as a primary endpoint the prevention of TKR in patients with knee OA who met eligibility criteria for surgery using a noninvasive biomechanical intervention. All patients were eligible for TKR and would have been referred for surgery if they had not been provided with an option to use the intervention.
Our results show that 86% of the patients enrolled into the study and who received the intervention avoided knee replacement surgery at 2 years. In addition to surgical avoidance, the clinical outcomes of the biomechanical intervention at 1 year show a significant improvement, including an estimated 36% reduction in pain, a 34% reduction in disability, and a 13% increase in physical aspects of QoL, as well as a significant improvement in gait patterns after the intervention. With regard to the patients who received the intervention and progressed to surgery, the study shows that they had higher pain and disability level at baseline, namely had more symptomatic disease, than those patients who avoided surgery.
Given that OARSI guidelines for nonsurgical management of knee OA show a limited therapeutic effect for other noninvasive interventions, such as other types of biomechanical interventions, strengthening exercises and training, injectables, and self-management, 5 the results of this study holds meaningful promise as a treatment modality with regard to the future management of the OA of the knee in patients eligible for TKR.
Previously published studies that examined the biomechanical intervention treatment used in this study reported a positive effect with respect to clinical outcomes and improved gait patterns. 11 –14,31 The improvement in clinical outcomes showed that the recommended Minimal Clinically Important Difference was met, namely a 17%–22% reduction in pain 32 and a 7.8 points increase in PCS. 33 In addition, 67% of the patients met the OMERACT-OARSI criteria for clinically significant outcomes. 30 The improvement in gait velocity over time documented with this biomechanical intervention has substantial positive implications for patients. 34
Recommended walking speeds for pedestrian crosswalks are typically between 3.0 and 3.5 feet/s, corresponding to velocities of 91 to 107 cm/s. Patients in this study showed that their average gait velocity of 80.9 cm/s before the intervention increased to 90.3 cm/s after 3 months of treatment, providing patients with additional confidence in their ability to safely cross most street intersections. 35 Furthermore, a study by Studenski et al has shown a correlation between walking speed and longevity in older adults. Authors found that survival increases across the full range of gait speeds, with significant increments per 10 cm/s. For a 70-year-old patient, a 10 cm/s increase in gait velocity corresponds to the rise of 1–2 years longevity. 34
Over the past 2 decades, the United States has embraced the principles of population health management 36 as a means of lowering the cost of care while optimizing both quality and clinical outcomes. Medical and technological innovations such as the biomechanical intervention presented in this study that reduce costs while at the same time providing patients with subjective clinical improvement offer health care delivery systems operating under population health guidelines a significant advantage.
According to a report published by Blue Cross and Blue Shield in 2019, the price for a TKR episode of care (12 months of presurgery care, surgical procedure, and 60-day postsurgical care) ranged from $19,844 to $80,456, with an estimated market average cost of $42,415 in the California counties in which HPN operates. 37
For HPN, based on its proprietary professional and institutional contracts, the savings accrued by surgical avoidance after subtracting the costs of the biomechanical device and physiotherapy fees exceeded $8 million for the cohort of patients assigned to the biomechanical intervention, for at least 2 years. Given that 88% of HPN patients who met eligibility criteria TKR were referred directly for and received surgery, by improving awareness of the benefits of intervention among patients and providers, the potential savings to HPN is estimated to be double the amount.
By extrapolating these cost savings estimate, the implications for reducing costs on a national basis in the United States through health care delivery models that are driven by value rather than volume are substantial and self-evident.
It should be noted that within a care delivery system practicing population health, the biomechanical device aligns with the quality and outcome goals of orthopedic surgeons who are reimbursed by health plans or health care systems on a capitated basis, as opposed to a fee for service basis.
Some limitations to our study should be acknowledged. First, the results of this study looked at 2 years outcomes. To validate the clinical and quality outcomes and financial benefits, additional investigation is needed to evaluate longer term surgery avoidance among patients selecting a noninvasive biomechanical intervention. Second, the study used patients who chose surgery as the control group. Because of this, the possibility of a selection bias cannot be ruled out. Furthermore, because all patients had the right to receive surgical intervention under their health plan benefit, randomizing members against their choice was not possible.
Furthermore, because metrics on patients choosing surgery were not collected, we cannot exclude that the control group, although unlikely, had significantly more debilitating disease than the investigatory cohort. For these reasons, conducting a prospective blindly randomized control study of a cohort of patients who meet eligibility criteria for TKR and comparing the intervention with different noninvasive modalities are required. A third limitation is the studies inability to collect follow-up clinical metrics on all patients choosing the biomechanical intervention because of poor patient adherence to follow-up appointments at 6 and 12 months.
Incomplete collection of clinical metrics makes it difficult to make conclusive statements about the positive trends seen in pain reduction, reduced disability, improved QoL, and gait patterns. Although we used a repeated-measures General Mix Model to assess changes over time and accommodate for missing data, additional long-term studies should further investigate the promising improvements in clinical functionality such as pain reduction, reduced disability, QoL, and gait patterns. That said, for the primary outcome of the study, the avoidance of TKR, none of the patients were lost to follow-up.
Conclusions
Given the increasing prevalence of knee OA and the additional costs that will be incurred by the health care system, there is a critical need to consider new nonsurgical interventions for knee OA. This study is a retrospective analysis of the efficacy of a biomechanical intervention in a real-life setting. The results suggest that the noninvasive biomechanical functional treatment presented in this study is a viable nonsurgical option for patients who meet eligibility criteria for TKR. The application of this treatment in population health settings provides a distinct opportunity to health plans and provider networks who are managing patient care under financial risk arrangements.
Footnotes
Acknowledgments
The editor in chief wishes to acknowledge that he serves as an advisor to APOS Health, the creators of the technology discussed in this article. He also wants to thank the associate editor and the outside reviewers for their detailed reviews of the article. The authors would like to thank Dr. Younghoon Kim, Tim Terrio, Caryn Harris, Brian Finch and Connie Michaels for treating the patients and utilizing Apos.
Authors' Contributions
All authors take full responsibility for the entire manuscript content, integrity of the data and the accuracy of the data analysis. Study Concept and Design: ID, RM. Data collection: MF, CL, DL. Analysis and Interpretation of data: ID, MS. Original Drafting of the Manuscript: ID. Critical Revision of the Manuscript: ID, MH, CL, MS, RM.
The authors read and approved the final manuscript.
Author Disclosure Statement
No competing financial interests exist.
Funding Information
No external funding was received for this article.
