Abstract
Vaginitis is one of the most common reasons women access health care in the United States. Despite its prevalence and disruptive impact, it is frequently misdiagnosed and untreated, resulting in unnecessary patient discomfort, follow-up visits, and health care costs. This study presents a costs analysis of diagnostic testing technologies to demonstrate the potential of molecular tests to improve the value of care for women with vaginitis. This study tracks health care spending among women diagnosed with vaginitis and finds that nucleic acid amplification tests (NAATs) are cost-effective for the diagnosis of vaginal symptoms. Women who receive a NAAT on the day of their diagnosis have significantly lower 12-month follow-up costs compared to women who receive a direct probe test or those women who are clinically evaluated without the use of a molecular test. However, despite Food and Drug Administration approval, widely available molecular diagnostics have not been incorporated into clinical guidelines, and many payer policies fail to cover these tests. Greater utilization of NAAT for the diagnosis of vaginitis has the potential to improve the care of women seeking treatment for this prevalent condition and facilitate sexually transmitted infection testing without additional visits.
A brief update to this report titled “Optimizing Vaginitis Diagnosis to Reduce Health Care Costs in Nonpregnant Women Utilizing Molecular Diagnostics” was published on July 13, 2022 and is available at
Introduction
Vaginitis is one of the most common reasons for women to visit their primary care providers in the United States, responsible for 10 million office visits annually. 1 –4 It is defined as an inflammation or infection in the vagina that presents with various symptoms such as itching, burning, irritation, dyspareunia, vaginal odor, and abnormal vaginal discharge. 5 The 3 most common causes of vaginitis are bacterial vaginosis (BV), vulvovaginal candidiasis (VVC), and trichomoniasis (TV). 6,7 Of these, BV is the most common etiology. Although many cases are asymptomatic, women with both asymptomatic and symptomatic BV may experience serious health consequences such as an increased risk of contracting sexually transmitted infections (STIs), including human immunodeficiency virus, 8,9 and a high probability of preterm birth. 3 BV is more prevalent among Black, Hispanic, and Mexican-American women compared with non-Hispanic White women, which may explain some racial disparities observed for preterm birth. 10 –12 Vaginal symptoms related to vaginitis can result in absences from school or work, disruptions to sexual functioning, and a negative self-image 13 impacting women significantly, especially if undiagnosed or untreated. The annual cost of treating symptomatic BV has been estimated at $1.3 billion. 3
To improve patient outcomes, and take advantage of available testing technologies, it is imperative to improve diagnosis and treatment of patients with vaginitis at the initial presentation of symptoms. This study examines health expenditures following a diagnosis of vaginitis and compares the cost by the diagnostic modality used at the first medical encounter. The hypothesis is that women receiving nucleic acid amplification tests (NAATs) will have reduced total health care costs resulting from lower vaginitis-related heath expenditures. Identifying cost savings can potentially add value to women's health care for patients and payers, thus reducing the overall population-level burden of vaginitis. To the authors' knowledge, this is the first study to analyze health care utilization and diagnostic testing costs from the payer perspective in the 12 months following a diagnosis of vaginitis.
Background
Currently, the most popular recommended practice of diagnostic testing for vaginal symptoms is a potassium hydroxide (KOH) test and microscopic examination of fresh samples of the discharge. A formal diagnosis of BV is made when at least 3 of the following 4 Amsel criteria are met: homogenous, thin, white-gray discharge that smoothly coats the vaginal walls; more than 20% clue cells on saline microscopy; vaginal fluid pH >4.5; and a positive KOH whiff test. 14 Though microscopy has been a mainstay diagnostic approach, particularly in the case of TV, it has a lower sensitivity than alternative testing modalities. 15 Furthermore, it has a lower reliability as it is subject to variation in the collection, preparation, and technical proficiency of the examiner, and the potential self-treatment by the patient prior to evaluation makes visualization of the yeast challenging. In this analysis, the clinical evaluation (CE) subgroup is defined as women who received either a microscopy or had no evidence of testing at the office visit on their index date.
In recent years, commercially available molecular diagnostic tests have emerged as a viable alternative to CE and literature suggests that they are superior to standard methods of CE. 16 The analysis conducted here focuses on 2 such tests, NAAT and direct probe tests (DPs). NAATs have been shown to have superior sensitivity (98.7%), specificity (95.9%), and negative (92.9%) and positive (96.9%) predictive power than DP for diagnosis of vaginitis. 17,18 The 3 causative agents of BV are Gardnerella, Atopobium, and Prevotella. NAAT tests use amplified probes that can detect the relative levels of these bacterial species. Earlier generation molecular tests such as DP were less sensitive and not specific and relied on direct probe binding to detect the levels of these agents. 19,20 In general, molecular testing allows the clinician to detect not only the presence of specific microbes but also to determine their ratio in the vaginal microbiome. Presence alone does not provide adequate specificity, potentially resulting in a false positive diagnosis and, therefore, potential overtreatment.
Misdiagnosis can increase health care costs resulting from unnecessary, incorrect, or prolonged treatment, as well as the need for continued follow-up visits and testing for recurring vaginal symptoms.
Methods
Data
This analysis was conducted using the IBM MarketScan Commercial Claims and Encounters Database, which contains claims for medical and pharmacy expenditures for a subset of employer-provided fee-for-service and managed care health insurance plans in the United States. Medical claims, outpatient pharmacy claims, and enrollment information are linked together using unique enrollee identifiers. All required study variables were based on relevant International Classification of Diseases, 9th and 10th Revision, Clinical Modification and procedure coding system (ICD-9-CM, ICD-10-CM, and ICD-10-PCS) codes, Current Procedural Terminology 4th edition (CPT-4) codes, Healthcare Common Procedure Coding System codes, and National Drug Codes. The paid amount on the claims includes both the insurer-paid and patient out-of-pocket portions.
The data were de-identified and certified to be fully compliant with US patient confidentiality requirements set forth in the Health Insurance Portability and Accountability Act of 1996. Institutional Review Board approval was not required.
Study design
Women between the ages 18 and 64 years with at least 1 nondiagnostic medical claim (not laboratory or radiology) with a diagnosis code for vaginitis (Appendix A1) in an outpatient setting between January 1, 2016, through December 31, 2018, were selected from the data. The initial encounter was used to set the date of first vaginitis diagnosis (referred to as the index date). Patients were required to have at least 12 months of continuous enrollment with medical and pharmacy benefits before (defined as the baseline period) and after (defined as the follow-up period) the initial encounter. Women with health care costs for baseline or follow-up that totaled at or above the 99th percentile were excluded from all analyses.
Figure 1 presents how the cohorts were constructed. A 12-month look-back period was used to establish baseline characteristics of the cohorts and review gynecologic history. Exclusion criteria included evidence of prior vaginitis based on diagnosis codes, or NAAT testing for Candida vaginitis (CV), TV, or BV based on procedure codes during the baseline period, and an initial encounter in an inpatient setting; women with diagnosis of malignancy also were excluded. These exclusions limited the sample to patients newly diagnosed with vaginitis in an outpatient setting, excluding those with significant comorbidities or recurrent disease, removing women with extreme outlier costs in the baseline and follow-up period.
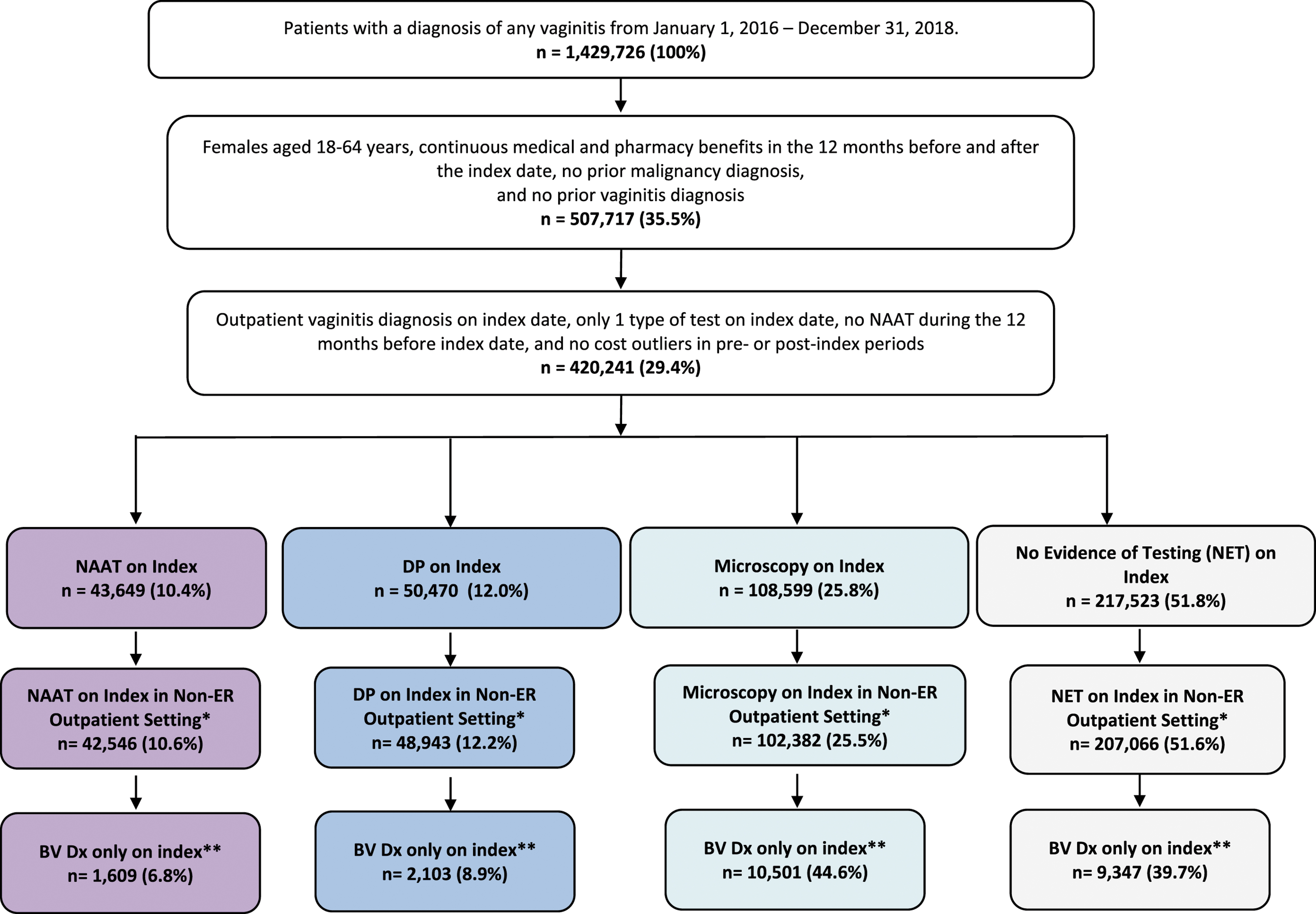
Patient attrition. *Proportions are calculated among all patients with non-ER outpatient diagnosis (N = 400,937). **Diagnosis of vaginitis and other specified bacterial agent/infection (ICD-9-CM 041.89 or ICD-10-CM B9689) and no diagnosis of Candida vaginitis, trichomoniasis, or Chlamydia Trachomatis/Neisseria gonorrhea on index date. Proportions are calculated among all patients with non-ER outpatient diagnosis and BV only (N = 23,560). *** NAAT includes Current Procedural Terminology codes 87661, 87481, 87511. DP includes 87480, 87510, 87660, and Microscopy includes 87205, 87210, 87220, 83986, 82120, 87905, 87970, 87071, 87073, 87075, 87076, and 87077. BV, bacterial vaginosis; DP, direct probe; Dx, diagnosis; ER, emergency room; ICD-9-CM, International Classification of Diseases, Ninth Revision, Clinical Modification; ICD-10-CM, International Classification of Diseases, Tenth Revision, Clinical Modification; NAAT, nucleic acid amplification test.
The sample of women meeting these criteria were then classified into one of 3 cohorts based on the test they received: NAAT, DP, or CE, which included women who had microscopy and those with no evidence of testing. Patients with evidence of more than 1 type of test on initial encounter (ie, NAAT and microscopy) were excluded from the analysis. The cost analysis sample was limited to women diagnosed in the office setting, n = 400,937, as only a small portion of women were diagnosed in the emergency room (ER) setting (<5%).
Provider type on initial encounter (OB-GYN, primary care, other specialist, other/unknown/missing) and type of vaginitis diagnosis on the index date (CV, TV, BV, Chlamydia trachomatis/Neisseria gonorrhea, or nonspecific vaginitis codes, and evidence of telemedicine on initial encounter) were captured.
Patient demographic characteristics were reported from the initial encounter, including age, region, urbanicity, and insurance type (Table 1). The following clinical characteristics were identified in the baseline period via relevant diagnosis, procedure, and medication codes on claims: infection, STI, substance abuse/dependence, hormonal contraceptive use, diabetes, and preventive care screenings (cervical cancer and breast cancer).
Demographic Characteristics of the Total Cohort
The percentages reported in the cells for index year are the shares of each testing modality over the entire sample each year.
CDHP/HDHP, consumer-directed health plan/high-deductible health plan; DP, direct probe; EPO/PPO, exclusive provider information/preferred provider organization; HMO, Health Maintenance Organization; NAAT, nucleic acid amplification test; NET, no evidence of testing; POS, point of service plan; SD, standard deviation.
Health care utilization and costs were compared in the baseline and follow-up period, including the initial encounter. At the initial encounter, average costs of NAAT and DP were calculated using only claims with nonzero payment amounts with codes corresponding to these tests. All-cause costs were defined as the sum of the payment on all claims regardless of diagnosis, including inpatient, ER, physician office, other outpatient services, laboratory services, and outpatient pharmacy. All-cause costs also included vaginitis and STI testing, including HIV. All-cause utilization and costs were measured separately for the 12-month baseline period, day of initial counter, and the 12-month follow-up period inclusive of the initial encounter.
Analyses and model
This analysis was descriptive in nature and all comparisons are unadjusted. Results are presented for the overall vaginitis cohort, and the subsample of women diagnosed with BV only at initial encounter. All analyses are stratified by diagnostic testing modality. The BV-only sample was analyzed to demonstrate the contribution of BV to VVC and as a robustness check to ensure that the results were not driven by any single etiology.
The cost analyses exclude women diagnosed in the ER. Means and standard deviations are presented for continuous variables. Counts and proportions are presented for categorical variables. t-Tests and chi-square tests were used to compare across testing cohorts, more specifically to the NAAT cohort.
A per-member per-month model was constructed to demonstrate the health care costs among the testing cohorts accounting for the cost of the newly introduced CPT code for the BV-specific NAAT (81513). All-cause spending among the 400,937 women who were diagnosed with vaginitis in a non-inpatient, non-ER setting was tabulated using 5 categories for each cohort: inpatient admissions, ER visits, physician office visits, vaginitis testing, and pharmacy costs. The 2021 Medicare fee schedule rate of $143 for CPT code 81513 was used for cost calculations. To arrive at the per-month cost per diagnosed member cost within each cohort, the percent of women with an encounter (admission, visit, or pharmacy fill) was multiplied by the mean cost of the encounter (Table 2) and divided by 12 months.
Vaginitis-Related Utilization and Costs 12 Months Following Diagnosis
Laboratory services category does not include vaginitis testing and STI testing.
Vaginitis test utilization and cost include culture claims.
DP, direct probe; ER, emergency room; NAAT, nucleic acid amplification test; OP, outpatient; Rx, prescription; SD, standard deviation; STI, sexually transmitted infections.
Results
Patient characteristics
There were more than 1.4 million women with a diagnosis of vaginitis identified from January 1, 2016, through December 31, 2018, of which 420,241 women satisfied the inclusion criteria. Of those, 10.4% received NAAT, 12.0% received DP, and 77.6% were evaluated clinically (25.8% had evidence of Amsel microscopy at the initial encounter while 51.8% had no evidence of testing) (Figure 1). On average, women who received molecular testing were 36.7 years old, 92.3% lived in urban areas, and 54.1% had exclusive provider information/preferred provider organization (EPO/PPO) health insurance, while women who were not tested were 39.5 years old, 88.1% lived in urban areas, and 51.1% had EPO/PPO health insurance (Table 1). During the study period, the proportion of patients who received NAAT testing increased (by 2.6 percentage points) while the proportion of patients receiving microscopy decreased (by 4 percentage points) (Table 1). Among the CE cohort, only 2.0% of women who received microscopy had concurrent claims for a smear, whiff test, and pH test, suggesting an incomplete Amsel assessment as all 3 tests are required to meet the criteria to properly evaluate BV. CE with microscopy is declining, from approximately 32% in 2015 to 20.2% in 2018, while no evidence of testing remains about the same. 21 A similar pattern of decline was observed in this analysis (Table 1).
Health care costs and utilization
Among women diagnosed in a non-ER setting, overall health care costs were lowest among women in the NAAT cohort. Women who received a NAAT had significantly lower 12-month costs than those who received a diagnosis based on DP and CE ($7660 vs. $8189 and $8232, respectively; both P < 0.001) (Figure 2). The 12-month follow-up costs for women diagnosed with BV only on the index date with a NAAT were $7102 (SD = $11,447), while claims for women in the DP and CE cohorts reflect 12-month costs of $7567 (SD = $11,548) and $7217 (SD = $11,337), respectively. However, the differences were not statistically significant. In all cohorts, the highest proportion of costs were observed in other outpatient services and outpatient pharmacy. Pharmacy costs for women receiving NAAT were $11 less compared with the DP cohort and $271 more compared with the CE cohort (Figure 2).
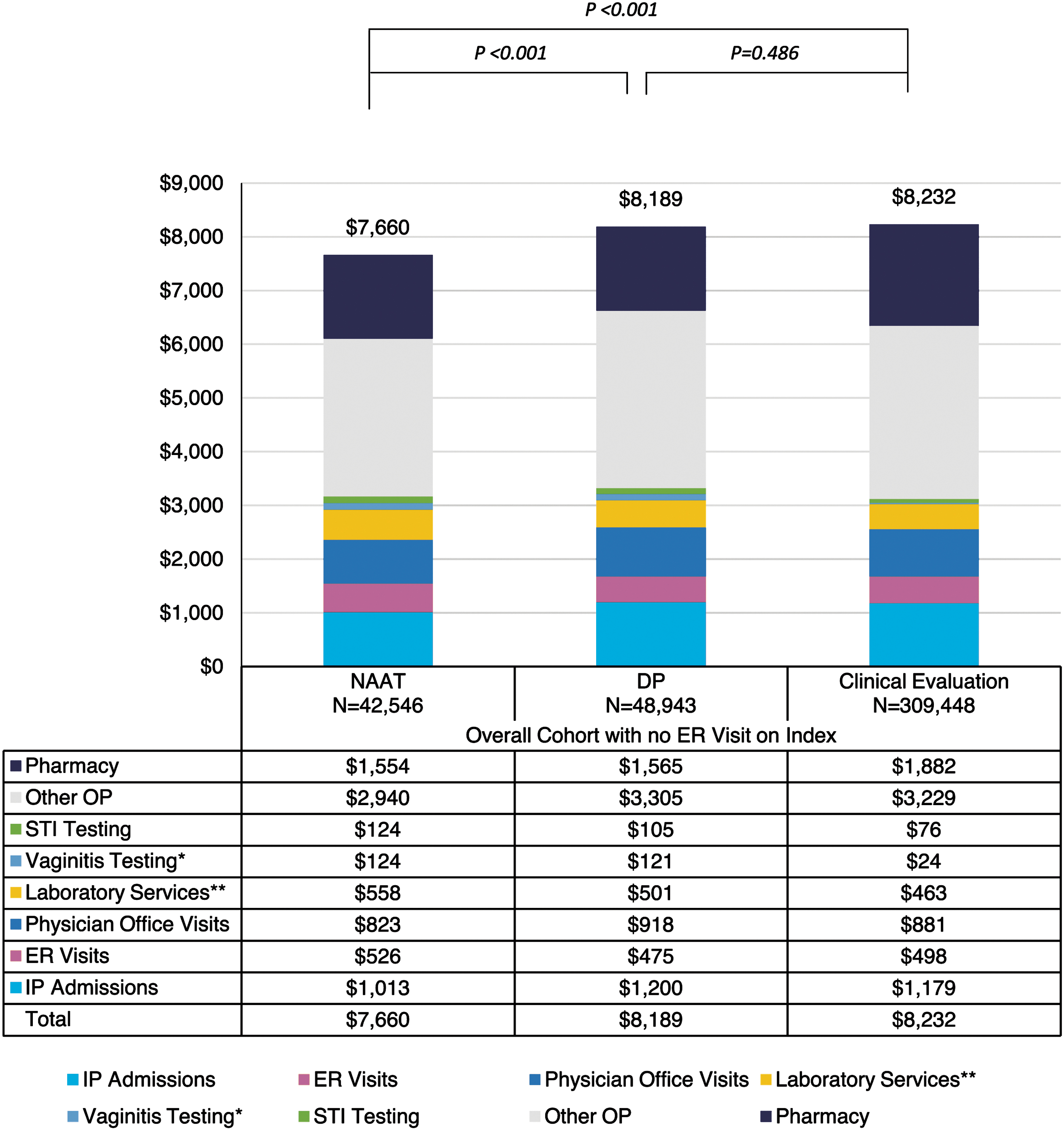
All-cause costs over the 12-month follow-up period for the overall population diagnosed in the non-ER outpatient setting. *Vaginitis testing includes NAAT and DP for Candida vaginitis, trichomoniasis, and bacterial vaginitis, and microscopy. **Laboratory testing includes tests other than vaginitis and STI testing. In the overall cohort, total costs in molecular testing were significantly lower than clinical evaluation (P < 0.05). DP, direct probe; ER, emergency room; IP, inpatient; NAAT, nucleic acid amplification test; OP, outpatient; STI, sexually transmitted infection.
In the 12-month follow-up period, inpatient admissions were rare (<10% of women) while between 26% and 29% of the overall sample had an ER visit (Table 2). Nearly all patients had non-ER outpatient medical services and outpatient prescriptions (Table 2). All differences are statistically significant.
Among the overall sample of women, including those diagnosed in an ER setting, women in the NAAT cohort received the highest rate of STI testing with 86.4% of women being tested compared to 68.3% and 55.2% for the DP and CE cohorts (Table 3).
Clinical Characteristics of the Cohorts
DP, direct probe; NAAT, nucleic acid amplification test; NET, no evidence of testing; SD, standard deviation; STI, sexually transmitted infections.
Cost of treatment model
To further demonstrate the utility of a NAAT test to diagnose BV, a cost calculation was performed to examine the health care expenses per month per diagnosed member (in a non-inpatient, non-ER setting) across the 3 diagnostic approaches: CE, DP, and NAAT (Figure 3).
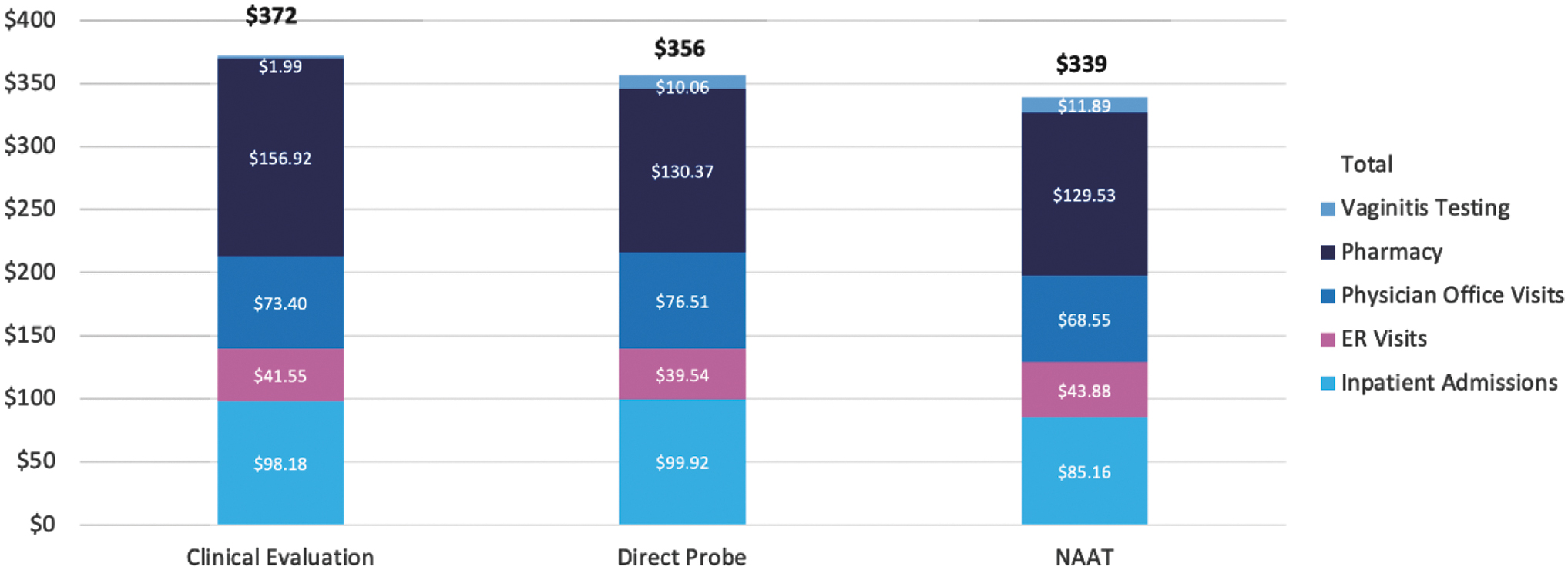
Cost per member per month by diagnostic cohort. ER, emergency room; NAAT, nucleic acid amplification test.
This analysis illustrates that the use of a NAAT-based test as part of the initial diagnostic workup resulted in a 9% reduction in all-cause costs ($339 vs. $372) compared to CE; and a 5% reduction ($339 vs. $356) compared to DP. This suggests that it is possible to absorb the initial increased cost of NAAT testing, specifically the cost of the BV-specific NAAT (CPT 81513) at the patient's first encounter, and potentially reduce all-cause health care expenses in the 12 months following the initial diagnosis of vaginitis.
Discussion
Vaginitis, rarely discussed as a public health issue, causes more than 10 million office visits annually, and the cost of care for treating BV, specifically, is estimated to be nearly $1.3 billion. The burden to women includes pain and discomfort, psychosocial stress, workplace absenteeism, and time spent at appointments. A broad spectrum of assessment tools is available to clinicians, yet many appear to be under or improperly utilized. In the present analysis there is evidence of poor compliance with clinical practice guidelines (Amsel criteria), and a heterogenous approach to office diagnostics. With widespread availability of molecular testing, sensitive and specific diagnosis and treatment of vaginitis can be accomplished in 1 visit. Compared to CE, molecular tests offer high sensitivity and specificity that provide a precise treatment route. In addition to improved accuracy, recent evidence demonstrates that the combination of sensitive and specific laboratory testing as well as careful patient evaluation have the potential to reduce unnecessary follow-up visits and improve patient care. 22 Molecular testing corresponds to lower 12-month follow-up costs than CE, and it should be noted that between the 2 molecular testing modalities, NAAT testing is more cost-effective than DP. Patients receiving NAAT tests on their initial encounter experienced fewer follow-up visits than those receiving CE as well as those receiving DP.
Although microscopy with Amsel criteria continues to be included in the American College of Gynecologists (ACOG) guidelines, it may not be reasonable for health care providers to implement. Many providers may lack the training and/or equipment to conduct a full Amsel criteria assessment, and the claim analysis revealed that fewer than 5% of women in the microscopy cohort had evidence of meeting Amsel criteria (smear, whiff test, or pH test). Recent guidance from ACOG recommends NAAT as an alternative when a microscope is not available 5 and the National Academies of Sciences, Engineering, and Medicine states that while Amsel and Nugent methods are widely used, they are less specific and less sensitive than NAAT tests that detect and quantify the presence of contributing organisms. 23
Among the molecular testing options, NAAT has lower 12-month follow-up costs than DP ($7660 and $8189, P < 0.001). A key advantage of molecular testing is the ability to conduct STI testing simultaneously as women with vaginitis are at higher risk of contracting STIs. 24 Compared to DP, not only do women receiving NAAT have lower 12-month follow-up costs on average, but they also have significantly higher rates of STI testing. As such, NAATs improve the value of care for women who present with signs and symptoms of vaginitis compared to either DP or the current mix of CE.
Limitations
The limitations of this analysis included those shared in common with retrospective claims-based analyses. Administrative claims data were subject to coding inaccuracy and undercoding as well as lack of detail to understand the full picture of clinical conditions. This study was limited to individuals with employer-sponsored health coverage. Consequently, the results may not have been generalizable to individuals with other insurance, such as Medicaid or Medicare, or individuals without health insurance coverage. All analyses were unadjusted and, therefore, there may have been systematic differences between study cohorts or selection bias that may have accounted for differences found in study outcomes. Lastly, health care services and costs not covered by the insurance system (eg, over-the-counter medications) were not considered in this analysis. Over-the-counter pharmacy utilization and costs were not included in any analysis because they were not available in claims data.
If a provider evaluated Amsel or Nugent criteria in the CE but did not bill for it, that patient would have been erroneously included in the no testing cohort. These assignments highlight the heterogeneity of practice and imprecise diagnostic criteria for evaluating bacterial vaginitis.
Conclusion
This analysis demonstrates that the 12-month follow-up costs for women evaluated with NAAT at the initial encounter are lower than for DP and CE cohorts. NAAT is an effective method of evaluating vaginal symptoms to diagnose BV, TV, and candidiasis. Increased utilization of NAAT for the diagnosis of BV would improve the value of care for women who otherwise would have received CE or DP. Increased STI testing with NAAT represents an additional improvement in the value of care. This testing improves both detection and treatment of disease.
Access to NAAT testing will only be possible if commercial payers adopt positive coverage determinations that include NAAT for the assessment of infectious causes of vaginitis. The current empiric practice and mixed CE approaches may initially offer lower diagnostic costs, but these potential savings erode over time, increasing costs and adding challenges for patients, providers, and payers.
Footnotes
Acknowledgments
The authors thank Ellen Thiel of IBM Watson Health for her data interpretation and Shooshan Danagoulian, PhD, Department of Economic, Wayne State University, for her technical review of the manuscript.
Author Contribution Statements
Dr. Kong and Mr. Jenkins wrote this manuscript. Ms. Troeger reviewed the literature and the data analysis to help develop this article. Dr. Kim of IBM Watson Health performed the data collection and analysis. Dr. London provided subject matter, clinical insight, and editorial review.
Author Disclosure Statements
Hologic, Inc. is the manufacturer of assays that detect the organisms discussed in the paper. Ms. Troeger is a full-time employee, with salary, benefits, and stock at Hologic, Inc. IBM Watson Health (Dr. Kong and Dr. Kim) received research funding from Hologic, Inc. to perform the data collection and analysis. Mr. Jenkins received funding from Mary Ann Liebert, Inc. for the development and writing of this paper. Dr. London has been a consultant to Hologic, Inc in the past but did not receive funding for this work.
Funding Information
Hologic paid for research services from IBM Watson Health. This paper was made possible by a grant from Hologic, Inc. to Mary Ann Liebert, Inc., publishers.
Appendix
Description of Diagnosis Codes
Code type
Code
Description
Group
ICD-9-CM Dx
112.1
Candidiasis of vulva and vagina
CV
ICD-9-CM Dx
131.01
Trichomonal vulvovaginitis
TV
ICD-9-CM Dx
616.10
Vaginitis and vulvovaginitis, unspecified
OTHER
ICD-10-CM Dx
A5402
Gonococcal vulvovaginitis, unspecified
CTNG
ICD-10-CM Dx
A5602
Chlamydial vulvovaginitis
CTNG
ICD-10-CM Dx
A5901
Trichomonal vulvovaginitis
TV
ICD-10-CM Dx
B373
Candidiasis of vulva and vagina
CV
ICD-10-CM Dx
N760
Acute vaginitis
OTHER
ICD-10-CM Dx
N761
Subacute and chronic vaginitis
OTHER
ICD-10-CM Dx
N762
Acute vulvitis
OTHER
ICD-10-CM Dx
N763
Subacute and chronic vulvitis
OTHER
ICD-10-CM Dx
N7689
Other specified inflammation of vagina and vulva
OTHER
ICD-9-CM Dx
098.2
Gonococcal infection, chronic, of lower genitourinary tract
CTNG
ICD-9-CM Dx
098.0
Gonococcal infection (acute) of lower genitourinary tract
CTNG
ICD-9-CM Dx
099.53
Chlamydia trachomatis infection of lower genitourinary sites
CTNG
ICD-10-CM Dx
A541
Gonococcal infection of lower genitourinary tract with periurethral and accessory gland abscess
CTNG
ICD-9-CM Dx
04189
Other specified bacterial infections in conditions classified elsewhere and of unspecified site, other specified bacteria
BV
ICD-10-CM Dx
B9689
Other specified bacterial agents as the cause of diseases classified elsewhere
BV
