Abstract

Introduction
In collaboration with the Jefferson College of Population Health, the Access to Equitable Carrier Screening Coalition (AECSC) hosted a Multi-Stakeholder Virtual Roundtable Summit on June 25, 2020 to identify the challenges/barriers to optimal carrier screening, discuss potential solutions, and generate recommendations for future clinical and economic approaches, messaging, and research. The following report on the summit proceedings is intended to elevate the conversation around increased access to expanded carrier screening (ECS) and disseminate relevant information across a broad range of stakeholders.
Overview
In the United States today, genetically-related conditions account for a disproportionately high number of infant and early childhood deaths and avoidable neonatal intensive care unit admissions. In fact, genetic disorders – and congenital malformations indicative of genetic syndrome – contribute to neonatal mortality in 30%–50% of neonatal and infant deaths. 1
Being a carrier for an inherited condition has lifelong implications and health sequelae for both the carrier and subsequent generations. For this reason, population-specific genetic carrier screening was introduced in the United States more than 40 years ago to help identify couples at risk for having children with autosomal recessive or X-linked recessive genetic conditions. For example, Tay-Sachs disease carrier screening was offered to individuals of Ashkenazi Jewish ancestry and sickle cell disease testing was offered to individuals of non-Hispanic Black/African American descent. Today's unprecedented social, cultural, and demographic shifts (including increased usage of in vitro fertilization [IVF] and gamete donors to achieve pregnancy), in combination with greater racial and ethnic diversity among individuals and their reproductive partners, make a compelling case for broader, more equitable carrier screening to guide reproductive life planning decisions.
Advances in technology and genomic sequencing over the past decade have made it possible to assess the risk for multiple serious conditions with a single test. Access to such carrier screening can provide targeted information to individuals to guide critical reproductive decisions. Ideally, ECS should be available to all individuals of reproductive age, regardless of ethnicity, during the preconception or prenatal periods so as to ameliorate the exacerbation of health inequities and disparities.
The goal of ECS testing is to improve health outcomes for newborns, young children, and adult carriers, and to increase autonomy for prospective parents with respect to shared decision-making regarding reproductive options. To that end, the AECSC was formed (in 2019) to promote access to and availability of ECS by means of: Building relationships and providing education on the utility of ECS testing. Facilitating communication and information sharing. Collaborating with all stakeholders to ensure that ECS is covered by payers.
Recognizing the challenges to universal implementation of ECS, AECSC is committed to promoting equity and thoughtful conversation around ethical questions, clinical guidelines, advantages and disadvantages, and reimbursement policy.
Virtual Roundtable Participants
Proceedings from the Multi-Stakeholder Virtual Roundtable Summit
A primary goal of good obstetrical care is to identify pregnancies that are at higher risk for maternal or fetal morbidity and mortality. Inherited and spontaneous genetic risks affect pregnancy outcomes and, according to recent statistics, birth defects are the leading cause of infant death in the United States. 2
Typically, clinicians first assess inherited risks by inquiring about a patient's family history and/or assigning ethnicity based on their own observation or a patient's self-report, and then recommend carrier screening in accordance with current screening guidelines (Figure 1). Unfortunately, methods used for assessing ethnicity frequently lead to inaccurate assumptions and there is mounting evidence that family history is an inadequate predictor of birth outcomes. In fact, up to 80% of genetic risks are not detectable via family history. 3 As our society becomes increasingly multi-ethnic, 4 many carriers will be missed if testing is limited to certain ethnic groups.

Current carrier screening guidelines. ACOG (691) is the Committee Opinion relative to carrier screening. Number 691. American College of Obstetricians and Gynecologists. March 2017; reaffirmed 2020. Source:
Although ethnicity-based carrier screening is “recommended,” it is nearly impossible to execute with consistent accuracy. Analyses reveal substantial discrepancies between self-reported ethnicity and genetic ancestry. 5 Moreover, there are no recommendations regarding management of the increasing number of individuals who define their ethnicity as 2 or more races, individuals who are adopted, or individuals who are otherwise uncertain of their ethnicity.
A recent analysis examined the limitations of ethnicity-based carrier screening guidelines in a large (>93,000) pan-ethnic cohort of individuals undergoing ECS for >90 conditions. 5 The researchers concluded that, even in the context of medical care, self-reported ethnicity is an imperfect predictor of genetic ancestry and carrier risk. For reasons ranging from demography to how individuals and/or providers complete test requisition forms, carrier screening based on ethnicity leads to inequitable medical care. A more desirable approach to carrier screening guidelines would entail broadened criteria (eg, 1 in 100 carrier frequency in at least 1 ethnicity) and inclusion of X-linked conditions to provide comprehensive fetal genetic risk assessment.
Current carrier screening guidelines and policies
The rationale for carrier screening is not to focus on specific disorders but rather to make an individual's carrier status known. The current professional guidelines for carrier screening (Figure 1) introduce inconsistencies in fetal risk assessment. For example, all current guidelines fail to account for nondisclosure of ethnicity and/or family history of inherited conditions, which can occur because of an associated stigma in certain cultures; and individuals of many ethnicities (eg, East Asian, South Asian, Hispanic) fall outside of the American College of Obstetricians and Gynecologists (ACOG) guidelines for ECS.
Unfortunately, there are conditions outside the guidelines that are more – or nearly – as prevalent as those within the guidelines. Today, pan-ethnic screening (ie, screening patients without regard for ethnicity) is recommended for only 2 conditions – cystic fibrosis (CF) and spinal muscular atrophy. CF is the most common monogenic disorder among non-Hispanic Whites of Northern European descent. One newborn is affected out of every 2500 to 3500 Caucasian births. 6 However, severe conditions such as 21 OH-deficient congenital adrenal hyperplasia and GJB2-related non-syndromic hearing loss and deafness that have a higher prevalence in certain ethnicities than does CF are not included in screening recommendations.
Contrast this with the less rigorous current screening recommendations for certain conditions for which the risk of an affected newborn is much higher. For instance, the guidelines consider it “acceptable” to perform carrier screening for Fragile X syndrome if patients request it. Fragile X syndrome is the most common inherited cause of learning problems and intellectual disabilities in the United States. 7 Because it is an X-linked condition, males that inherit a variant FMR1 gene from their mother are almost always affected. The female carrier prevalence is estimated to be as high as 1 in every 130–250 across all ethnicities; 1 in every 4000 male children, and 1 in every 8000 female children, are variably affected with the condition.
ACOG recommendations include screening women of Cajun or French-Canadian descent for Tay-Sachs, a complete blood count for all women, and hemoglobin electrophoresis for women of Southeast Asian, West Indian, and/or Mediterranean ancestry.
The first successful community-based genetic screening initiative for recessive disorders was launched in the 1970s for the condition known as
Principally, screening identifies carrier status of those individuals who are at risk for having a child who is affected with a genetic disease. Ideally, testing would occur in the preconception period – a time when meaningful information can be used by prospective parents to guide reproductive planning based on their personal values. The reality is that carrier screening occurs most often in the early prenatal period.
A joint statement of the American College of Medical Genetics and Genomics (ACMG), ACOG, National Society of Genetic Counselors, Perinatal Quality Foundation, and the Society for Maternal-Fetal Medicine suggests characteristics of conditions that should be considered in designing ECS panels, including: cognitive disability, effect on quality of life, need for surgical or medical intervention, and a prenatal diagnosis that may result in (1) improved perinatal outcome and immediate care of the neonate and/or (2) prenatal education of parents regarding special needs care after birth. 9
An ACMG Carrier Screening Policy Statement takes into account disorders of a nature such that most “at-risk” women and their partners would consider having prenatal diagnosis. Patients must provide consent when adult-onset disorders are included. The policy specifies that causative gene(s), mutations, and mutation frequencies should be known for each disorder so that residual risk can be communicated to individuals whose tests are negative. 10 Clinical association between the mutation(s) detected and the severity of the disorder should be validated.
Role of pan-ethnic ECS in improving quality of care and health outcomes
Carrier screening for multiple genes associated with life-limiting and/or cognition-impairing conditions can have profound effects for individuals, their families, and communities. Population-level access to pan-ethnic ECS enhances and optimizes clinical utility that can lead to a more predictable genetic risk profile for improving infant health and well-being.
ECS uses sequencing and other technologies to evaluate a large number of genes implicated in inheritable disorders, allowing for more comprehensive assessment of carrier status and fetal risk in our increasingly multi-ethnic society and geographic population distribution. Importantly, the technologies confidently identify pathogenic variants (ie, variants that cause disease) in hundreds of genes. Full gene sequencing enables analysis of all variants in a gene, and targeted next-generation sequencing (NGS; amplicon sequencing) enables analysis of variants in specific genomic regions.
For the foregoing reasons, ECS provides valuable information for counseling couples at preconception and early pregnancy, including preimplantation genetics implications in certain situations.
The ideal approach to screening – and the goal of the Coalition – is to highlight the societal benefit for pan-ethnic ECS in light of an increasingly diverse population. As shown in Figure 2, current recommendations that exclude most ethnicities result in unacceptably large numbers of missed affected pregnancies. A pan-ethnic approach to screening would remove inconsistencies and improve detection rates across ethnicities. With a core panel of conditions that have a carrier screening frequency of ≥1 in 100 in any ethnicity, a pan-ethnic ECS approach would substantially increase the identification of affected pregnancies and greatly reduce the number of missed affected pregnancies in every ethnic group.
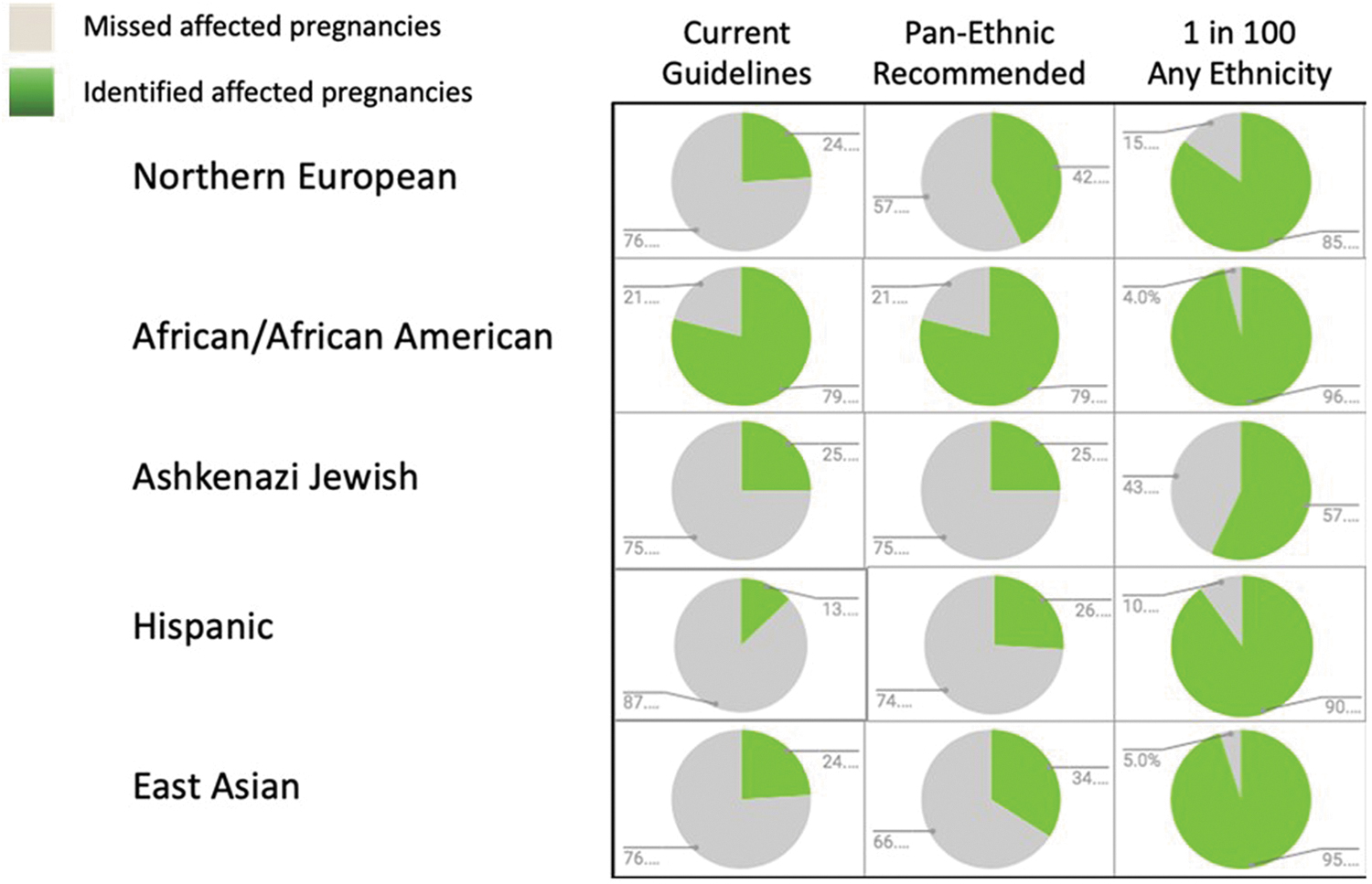
Current guidelines create disparities in care across ethnic groups. Color images are available online.
Additional screening may be warranted based on family history or specific ethnicity.
Genetic testing for a broad spectrum of severe inherited conditions should be accomplished via a genomic sequence analysis. Theoretically, the cost of a test panel should be similar irrespective of the number of genes included. For example, a core panel might include, at a minimum, the following prevalent conditions:
21-hydroxylase-deficient congenital adrenal hyperplasia (CYP21A2)
Alpha thalassemia (HBA1)
Canavan disease (ASPA)
Carnitine palmitoyltransferase II deficiency (CPT2)
Congenital disorder of glycosylation type la (PMM2)
Cystic fibrosis (CFTR)
Duchenne muscular dystrophy
FKTN-related disorders (including Walker-Warburg syndrome) (FKTN)
Fragile X syndrome (FMR1)
GJB2-related DFNB1 non-syndromic hearing loss and deafness (GJB2)
Hb beta chain-related hemoglobinopathy, including sickle cell disease (HBB)
Hereditary fructose intolerance (ALDOB)
Medium chain acyl-CoA dehydrogenase deficiency (ACADM)
Pendred syndrome (SLC26A4)
Phenylalanine hydroxylase deficiency (PAH)
Pompe disease (GAA)
Smith-Lemli-Opitz syndrome (DHCR7)
Spinal muscular atrophy (SMN1)
Tay-Sachs disease (HEXA)
USH2A-related disorders (USH2A)
Wilson disease (ATP7B)
Molecular testing is not the optimal screening approach for every condition. For example, mean corpuscular volume and hemoglobin electrophoresis remain the gold standard testing for beta hemoglobinopathies.
ECS has enabled detailed analyses of carrier frequencies for many diseases. Westemeyer et al recently presented outcomes from a large cohort of individuals receiving ECS using NGS for up to 274 genes. 10
Several results illustrated the benefits of a pan-ethnic approach to carrier screening. For example, 81.6% (4434/5435) of identified carriers of 1–8 Ashkenazi Jewish (AJ) diseases had no reported AJ ancestry. In the case of CF, 44% (6260/14,229) of carriers identified had a variant that is not included in a standard genotyping panel. Assuming random pairing across the study population, the authors estimated that 1/175 pregnancies would be affected by a disorder in the largest (274 gene) panel (Table 1). The authors observed that offering an ECS panel to all individuals recognizes the diversity in contemporary US populations and leads to substantially increased identification of at-risk couples. They also appropriately noted some cautions and limitations (eg, variants of uncertain significance [VUS]).
Combined At-Risk Couple Rate and Affected Pregnancies Expected Across and Within Racial/Ethnic Groups by 1 of 274 Disorders
For α-thalassemia, individuals with the genotype α-/αα (silent carriers) were excluded. Those with αα/–, α-/α- and α-/– genotypes were included in calculating the carrier frequency. Inclusion of the α-/α- genotype may have overestimated the combined projected affected pregnancy rate.
Source: Westemeyer et al. 11
In- a 2019 study of 1391 couples at risk of having a pregnancy affected by 1 or more of 176 conditions, Johansen et al found that among those screened before becoming pregnant, 77% planned or pursued actions to avoid having affected offspring. Among those screened during pregnancy, 37% elected prenatal diagnostic testing for that pregnancy. The authors concluded that results of ECS informed couples' reproductive decision-making and led to altered pregnancy management that reduced the risk of having affected offspring. 12
The increasing number of genes evaluated in ECS panels also has implications for health outcomes in the lives of heterozygous individuals (eg, cancer, neurological disorders). With benefit of early identification via ECS, these individuals can be monitored as they mature. There is much yet to be learned about the genetic causes of disease and the role of heterozygosity in nonreproductive clinical situations.
Preconception ECS screening and awareness
The goal for prepregnancy carrier screening is to provide individuals/couples with information to assist in their understanding of reproductive risk for disease, family planning, and consideration of reproductive options that reduce the risk of an affected pregnancy (eg, IVF with preimplantation testing for single gene disorders, donor gametes, avoidance of conception, adoption).
There is significant variation in the presentation of certain conditions (eg, age at onset, degree of severity), and variants may have incomplete penetrance (ie, individuals may remain asymptomatic throughout their lives). For these and other reasons, there is general agreement that pretest genetic counseling be considered before implementing ECS in some cases.
Clinicians must provide clear, objective, nondirective counseling that ensures patient understanding of the benefits, limitations, and costs of various options. However, there are some limitations such as VUS that may not necessarily provide accurate information on significant infant, childhood, or adult risk. Fortunately, VUS are not typically included on carrier screening results reports, thus eliminating confusion regarding their significance during counseling.
The Association of Nurse Practitioners in Women's Health position statement on prepregnancy carrier screening calls for:
Establishing policies within the health care setting that protect autonomy and confidentiality regarding carrier screening.
Capturing an accurate family history and utilizing a validated prepregnancy assessment screening tool.
Establishing policies within the health care setting for individuals and couples who are identified as high risk and/or genetic condition carriers. 13
Informed decisions require pretest education that includes: increased autonomy and options to reduce the risk of disease, limitations, Genetic Information Nondiscrimination Act information, costs, and statistics. Care must be taken to ensure each patient's understanding that carrier screening will not identify all patients who are at risk for the screened conditions. Partner testing is recommended for autosomal recessive conditions.
The long-term health implications for carriers themselves – not just the affected child - must be included in genetic counseling. There are variable health implications for carriers of certain recessive conditions; for example, a carrier of DMD (Duchenne muscular dystrophy) is at risk for cardiomyopathy and a positive test result should trigger an echocardiogram of the woman's heart.
Unfortunately, it sometimes can take several years – and multiple specialist visits and diagnostic tests – to diagnose a child with a rare genetic condition. The knowledge acquired from carrier screening can reduce anxiety as well as saving substantial costs associated with years of time in a diagnostic odyssey. Of couples who opt for ECS during the preconception period, 77% pursue further actions to reduce the risk of an affected pregnancy. 12
Counseling limitations and challenges
Although some resources are available (eg, Joint Statement on ECS, ACOG Committee Opinion 693), there is a lack of comprehensive information regarding posttest counseling and best practices (eg, flow charts indicating partner/no partner, timing in pregnancy, concurrent partner, fetal test late in pregnancy).
Regardless of test results, risks remain for every hereditary disease and future testing may be advisable. For instance, residual risks may be associated with a negative test, and reproductive risks may fluctuate if partners change over time. Every effort must be made to maintain the availability of test results from one pregnancy to another.
The reality is that not all pregnant women can be reached for pretest and posttest genetic counseling as required by some payers. This is a concern, especially for women with difficulties in accessing care (eg, those living in rural and ethnic minority communities). The genetic counseling community believes that the need for counseling varies according to the specific situation. A National Society of Genetic Counselors task force is focused on how to better distinguish individuals who would most benefit from formal genetic counseling (ie, those who might face adverse outcomes without genetic counseling) from those who might do as well with other forms of education that do not require a licensed genetic counselor.
Pan-ethnic ECS costs and potential cost savings
Economic considerations of pan-ethnic ECS include the cost of testing, and patient and provider time (pretest and posttest counseling). The psychological burden associated with a positive test and its implications (eg, anxiety, social stigma) also must be taken into account.
There are a number of areas in which pan-ethnic ECS has potential for reducing costs. For instance, high yearly and lifetime costs of managing severe conditions are often mitigated by alternative reproductive options that reduce the risk of having an affected pregnancy. 14 Most importantly for families and caregivers, the diagnostic odyssey might be shortened when ECS results are known, thereby avoiding the costs of unnecessary testing and treatment and streamlining care.
When compared with panels that screen for fewer conditions, lab costs for pan-ethnic ECS are similar and relatively inexpensive relative to costs for affected individuals. For instance, yearly costs for DMD = $23,920–$54,27015; lifetime costs for familial dysautonomia = $1,758,000 for an average life expectancy of 15 years. 16
Costs for children with genetic disease are higher than those for children with chronic conditions – particularly in the first year after diagnosis – because of high rates of hospitalization and specialist visits. 17 Although pediatric genetic disease inpatient admissions account for only 3%–4% of all admissions, they account for 11%–46% of costs based on charges per discharge. 15 “The amortized cost of carrier screening across the population for hundreds of genetic diseases is now far less than the cost of treating an affected child.” 18
A modeling study of 1 million couples with the 14 most common recessive disorders via NGS detected more carriers and decreased incidence of affected pregnancies by 61%, thereby reducing lifetime treatment costs of the screened disorders by $275 million (66%) vs no screening. Compared to the typical genotyping strategy, NGS yielded savings of $19 million as well as preventing 13% of affected births. 16 The authors concluded that the savings in treatment of genetic disorders are greater than the costs associated with genetic screening.
Consider the following examples:
Caused by a trinucleotide repeat in the FMR1 gene, Fragile X syndrome is a condition that causes a range of developmental problems including learning disabilities and cognitive impairment that are evident beginning at age 2. Most males with Fragile X syndrome have mild to moderate intellectual disability, and about one third of affected females are intellectually disabled. 7 One model predicted that population-wide screening for Fragile X would detect 80% of affected fetuses and would be cost-effective at $14,858 per quality-adjusted life year. The screening would be cost-effective up to $140 per test. 19
Each type of leukodystrophy – a group of rare, progressive, metabolic, genetic diseases – is caused by a specific gene abnormality that leads to abnormal development or destruction of the white matter (myelin sheath) of the brain, 20 spinal cord, and often the peripheral nerves. An 8-year retrospective study of 64 patients revealed the potentially extreme economic hardships associated with inherited leukodystrophies. 21 Although the average number of diagnostic laboratory tests per patient was 20, up to 42 tests were required for some. The average cost per test was $4200 (range $357–$15,611) in 2013 dollars. Fifty-three percent of patients were diagnosed in 20 or fewer tests; testing was less successful in reaching a diagnosis with 25 or more tests. Although costs associated with clinician/specialist visits, examinations/procedures, and attempts to manage undiagnosed conditions were not included in the study, the overall cost of care for patients with these conditions can exceed $1 million (average $107,000).
Pan-ethnic ECS provides timely, actionable information about genetic carrier status to patients at a reasonable cost. With respect to economies of scale, the ACOG Committee on Genetics acknowledges that the cost of carrier screening for individual conditions may be higher than the cost of testing with commercially available ECS panels. It advises that “when selecting a carrier screening approach, the cost of each option to the patient and the healthcare system should be considered.” 22
Key Discussion Points
Timing and access
Today, the vast majority of women in America who receive carrier screening are tested during pregnancy and for very few conditions (eg, CF). Women contemplating pregnancy or in early pregnancy and their partners need better understanding of their risks for inherited conditions. A vigorous effort should be made to increase preconception testing and to support good communication between frontline providers and specialists.
Appropriate preconception pan-ethnic ECS should be accessible for all women regardless of ethnicity or economic status, and strategies should be developed to promote and facilitate effective testing of partners. Partners are 2 distinct individuals – often with different insurance companies and benefits. For optimal effectiveness, pan-ethnic ECS requires consistency in coverage (ie, payers, test panels, and benefits) for both partners.
Multiple challenges – guidelines, parameters, approach, education
There was consensus among participants regarding major challenges, including current guidelines that create disparities in care across ethnicities and that rely on often inaccurate self-reported ethnicity. Although achieving consistency among screening tests (eg, number of conditions per panel, testing methodology) should be a high priority, the near-term aim should be to ensure access to pan-ethnic ECS so that all partners – regardless of ethnicity – can reduce their risk of having affected pregnancies.
The chief advantage of pan-ethnic ECS lies in its ability to identify more carriers of, and pregnancies affected by, serious inherited conditions with maximum efficiency. The participants acknowledged several issues that must be resolved in conjunction with broadening the implementation of pan-ethnic ECS, including: selection of, and multi-stakeholder agreement on, conditions for inclusion in test panels; selection of approach (sequencing vs. genotype); reporting requirements for pathogenic mutations; and education of patients and providers.
It might be useful to consider how other countries approach ECS. For example, the Israeli population genetic screening program for reproductive purposes includes all known severe diseases that are relatively frequent in a specific population (carrier frequency at or above 1:60 and/or disease frequency at or above 1 in 15,000 live births). 23 The carrier screening program is free of charge and offers testing according to disease frequency in the different groups within the population.
Participants pointed out the need to address concerns about test sensitivity versus predictive value – a worry that detecting increasingly rare conditions may do more harm than good. They agreed that a clearer understanding of the positive and negative predictive value of pan-ethnic ECS is needed.
Other important aspects of the discussion included:
Role of clinicians. Downstream cost avoidance issues must go beyond the purview of obstetricians/gynecologists and become part of the family practice and internal medicine armamentarium. With better data, clinicians can help to shorten the trajectory toward a correct diagnosis.
Role of payers. A joint statement among payers regarding coverage of pan-ethnic ECS is critical. The information payers need can be distilled into the “4 Cs” – Consensus among experts, Clarity (a simple construct), Communication (effective method and message), and Cost.
It would be helpful to payers to have more data on the number of rare conditions included in/identified by universal pan-ethnic screening (eg, panel with 38 vs. 100 conditions). In general, the rarer the disease, the less cost-effective the test. Even if 100% of rare condition carriers are identified, the psychological impact on women and their partners is considerable.
Payer discussions must consider costs that will be covered in both the near and the long term.
Role of genetic counseling. Preconception testing and counseling must be standard of care for all conditions on ECS test panels; for example, communication regarding the prevalence of Fragile X that is perpetuated in families is of particular importance. Two goals would benefit the Fragile X community: (1) avoiding the diagnosis by means of informed decisions made prior to conception, and (2) education on how to deliver news of results indicating that the patient is a carrier.
Patients are rarely willing to pay for access to an alternative path (eg, IVF with preimplantation testing for single gene disorders) without coverage by insurers. Payers and foundations could help by making digital tools accessible to provide pretest and posttest awareness and counseling.
Recognition of genetic counselors as independent providers could greatly improve patient access to these services.
Genetic Alliance's
Call to Action and Next Steps: Overcoming Challenges to Broad Implementation of Pan-Ethnic ECS
Now is the time to address a variety of barriers to optimizing carrier screening in the United States. Solutions to some important questions will require collaboration among multiple stakeholders. For example:
Continue to collect clinical data and build the evidence base regarding reproductive outcomes associated with pan-ethnic ECS use.
Resolve the ongoing debate as to which conditions should be included on pan-ethnic ECS panels.
Encourage laboratories to provide information on how they calculate residual risk. Pan-ethnic ECS panels may include rare conditions with unknown detection rates and carrier frequencies, making calculation of residual risk difficult. A woman may have a positive family history for a single gene disorder, but the family-specific pathogenic variants may not be detected on some commercially available panels that do not use a full gene sequencing method.
Other issues can be effectively addressed by specific groups:
Bring national professional guidelines into alignment with the science
ACOG, ACMG, and other relevant organizations should work toward adopting consistent, actionable guidelines for pan-ethnic ECS with the goal of a single set of recommendations.
Standardize tests and establish best practices for the laboratories performing them
There are many different ECS panels available, but not all screen for the same condition(s) and/or variant(s). An accurate clinical assessment is best achieved when the same test is used by both the patient and partner.
A sophisticated laboratory and level of testing are critical factors. Currently, laboratories vary widely in what they include on their ECS panels and often refer to identical components by different names. Of the 15 laboratories offering ECS, tests include 14–1800 conditions and 40–1500 genes with overlap in relatively few genes. Some laboratories use nonmolecular tests and others use sequencing. A “gold standard” should be established.
The associated lack of best laboratory practices results in quality control issues that lead to wastefulness and inefficiency. In order to achieve consistent, standardized care, a minimum ECS panel must be established. A joint statement from relevant professional societies that identifies conditions to be included on a “minimum panel” could lead to better coverage by payers.
Broaden clinician awareness
Clinicians who manage the care of patients during the preconception and early prenatal periods must become more familiar with the full range of implications of positive and/or negative ECS results. Only 50% of US counties have a genetic counselor. Broad-based education regarding pan-ethnic ECS must be provided to primary care physicians and general internists as well as obstetricians and nurse practitioners. Provider education should include the preferred test methodology (sequencing provides a more comprehensive assessment compared to genotyping), and the rationale for preconception and prenatal screening as well as guidance on efficient testing of partners - particularly when testing occurs during a pregnancy.
Giving the patient a test kit for the partner may help reduce the turnaround time and ensure that the same panel is used for both individuals. Posttest counseling for individuals with positive results is critical and patients with a positive family history should be referred for genetic counseling.
Bring payers into agreement on coverage for pan-ethnic ECS testing and the conditions that should be covered
Broadened access to pan-ethnic ECS is vitally important for prospective parents, families, communities, and, ultimately, for society as a whole. Summit participants strongly recommended that specific stakeholder groups consider taking immediate action as follows:
Footnotes
Acknowledgements
Thanks to the following Summit participants for their contributions to the content and editing of this proceedings paper: Haywood Brown, MD (Vice President, Institutional Equity at the University of South Florida System; Associate Dean, Morsani College of Medicine for USF Health; Executive Medical Director, Access to ECS Coalition); Lorraine Dugoff, MD (Department of Obstetrics and Gynecology, University of Pennsylvania); Rachel Gorham-Fidino, MSN, WHNP-BC, AGN-BC, DNP-S; Bryce Mendelsohn, MD, PhD: Medical Geneticist.
Author Disclosure Statement
Janice L. Clarke, RN, is employed by the Jefferson College of Population Health as its Senior Medical Writer and received no compensation from the Coalition.
Participant Disclosure Statements
Some members of the Access to Equitable Carrier Screening Coalition are involved in genetic testing, including: Invitae, Myriad Genetics, Natera Inc., Progenity, Thermo Fisher Scientific, and SEMA4. Roundtable participants were offered compensation by the AECS Coalition for time spent preparing for and attending the meeting. Drs. Haywood and Mendelsohn declined the offer of payment. Roundtable participant Haywood Brown, MD is Executive Medical Director of the Access to Equitable Carrier Screening Coalition. Roundtable participant Kenny Wong, MS, CGC is Chair of the Access to Equitable Carrier Screening Coalition.
Funding Information
Jefferson College of Population Health of Thomas Jefferson University received funding support from the Access to Equitable Carrier Screening Coalition to organize and convene the Virtual Roundtable Summit and to document the meeting proceedings detailed in this manuscript. The content and opinions expressed herein are those of the roundtable participants.
