Abstract
The goal of the initiative was to evaluate the impact of an innovative practice model on identification of unmet vaccination needs and vaccination rates. This was accomplished through a prospective, multisite, observational study in 8 community pharmacy practices with adults receiving an influenza vaccine with a documented vaccination forecast review from October 22, 2015 through March 22, 2016. When patients presented for influenza vaccinations, pharmacists utilized immunization information systems (IIS) data at the point of care to identify unmet vaccination needs, educate patients, and improve vaccination rates. The main outcome measures were the number of vaccination forecast reviews, patients educated, unmet vaccination needs identified and resolved, and vaccines administered. Pharmacists reviewed vaccination forecasts generated by clinical decision-support technology based on patient information documented in the IIS for 1080 patients receiving influenza vaccinations. The vaccination forecasts predicted there were 1566 additional vaccinations due at the time patients were receiving the influenza vaccine. Pharmacist assessments identified 36 contraindications and 196 potential duplications, leaving a net of 1334 unmet vaccination needs eligible for vaccination. In all, 447 of the 1334 unmet vaccinations needs were resolved during the 6-month study period, and the remainder of patients received information about their vaccination needs and recommendations to follow up for their vaccinations. Integration of streamlined principle-centered processes of care in immunization practices that allow pharmacists to utilize actionable point-of-care data resulted in identification of unmet vaccination needs, education of patients about their vaccination needs, a 41.4% increase in the number of vaccines administered, and significant improvements in routinely recommended adult vaccination rates.
Introduction
V
HP, Healthy People; HPV, human papillomavirus; Tdap, tetanus, diphtheria, pertussis.
Tragically, these shortfalls in vaccination coverage are associated with significant morbidity and mortality. Statistics from the Centers for Disease Control and Prevention (CDC) are highlighted in the following list to emphasize the profound impact vaccine-preventable diseases have on population health in the United States. 5
According to the CDC: • More than 200,000 people are hospitalized because of influenza each year. • Between 3000 and 49,000 people die from influenza and its complications each year. • As many as 400,000 hospitalizations and 19,000 deaths can be attributed to pneumococcal disease each year. • Nearly 1 million people will be infected with herpes zoster (Zoster) each year and some will have painful complications that persist for years. • Human papillomavirus (HPV) causes more than 27,000 cases of cancer in women and men each year.
In addition to the morbidity and mortality highlighted in the list, vaccine-preventable diseases cause a significant economic burden, costing the US health care system an estimated $26.5 billion annually. 6
The health and well-being of communities across America are at risk given the current gaps in vaccination coverage. It is unlikely the Healthy People 2020 goals (Table 1), which are set to improve the health of all Americans, will be met unless effective changes in health care delivery are implemented. 7 Innovative solutions are the key to solving this critical public health issue. Compelling evidence indicates that evidence-based, patient-centered, team-based care that includes seamless communication between and among patients, pharmacists, physicians, and other providers improves the quality of care and population health. 8
Project IMPACT (IMProving America's Communities Together) Immunizations, like other successful Project IMPACT initiatives before it, 9 –16 was designed to implement and evaluate principle-centered processes of care that engage patients in meaningful ways at the point of care, facilitate pharmacists practicing at the top of their license within their scope of practice, provide seamless opportunities for pharmacists to collaborate and communicate with other health care providers, and create consistency and predictability in delivering care that improves vaccination rates and enhances population health in the United States.
Objective
The primary objective of Project IMPACT Immunizations during the October, 2015 to March, 2016 time frame was to evaluate how implementing an innovative practice model that provides the pharmacist access to a patient's vaccine history at the point of care impacts the pharmacist's ability to identify unmet vaccination needs and increase vaccination rates for routinely recommended adult vaccinations.
The authors hypothesized that: 1. A significant number/percentage of people presenting to the pharmacy for an influenza vaccination would have additional unmet vaccination needs. 2. With clinical decision-support tools available to the pharmacist at the point of care, the pharmacist would be able to identify unmet vaccination needs and educate people about their unmet needs. 3. When educated at the point of care, people with unmet vaccination needs will take advantage of the opportunity for additional vaccinations.
Methods
Setting
Project IMPACT Immunizations was implemented in 8 community pharmacy practices in Washington State. A variety of community pharmacy practices types and geographic locations were selected to participate in the project to ensure the model can be successfully implemented across a credible spectrum of pharmacy practice environments (Table 2).
Study design
Project IMPACT Immunizations was a prospective, multisite, observational study. Pharmacists at each pharmacy practice site that participated in the study were trained to implement the innovative practice model using the following principle-centered approach: • Patient requests an influenza vaccination from the pharmacy • Pharmacist accesses the patient's vaccination history from the State immunization information system (IIS) using a technology interface ( • The technology interface provides clinical decision support for the pharmacist by generating a patient-specific vaccination forecast at the point of care. The vaccination forecast uses current ACIP recommendations to make a prediction of what vaccines are due for the individual patient based on the information that is documented in the patient's IIS record. • Pharmacist reviews the technology-generated patient-specific vaccination forecast • Pharmacist assesses the patient at the point of care • Pharmacist uses his/her clinical judgment to determine which forecasted vaccinations should be recommended to the patient based on the patient assessment (eg, pharmacist evaluates patient's documented IIS history to verify predicted patient needs, pharmacist evaluates medication use profile and patient self-reporting for documented previous administration to avoid potential duplications, pharmacist identifies patient needs including potential travel, pharmacist assesses patient for potential contraindications to forecasted vaccinations) • Pharmacist educates patient about his/her vaccination needs based on the pharmacist assessment • If patient consents, pharmacist administers appropriate vaccines • Pharmacist documents administered vaccines in the State IIS, thus sharing vaccination records with the patient's other health care providers who also have access to the IIS • If patient declines vaccination recommendations at the point of care, pharmacist recommends follow-up, including options for the patient to return at a later date or to receive the vaccination from his/her physician or other health care professional
The primary end points for this study were: • The number of times the pharmacist reviewed the vaccination forecast and educated the patient on his/her vaccination needs; • The number of unmet vaccination needs that were identified and met (administered) during the study period; and • The number and types of vaccines administered by the pharmacist.
The secondary end points for this study were: • The number of times a patient declined a forecasted vaccine at the point of care and the pharmacist recommended follow-up; • The number of times a forecasted vaccine was not recommended because a contraindication was identified by the pharmacist; and • The number of times the patient declined a forecasted vaccine because he/she says he/she already received it but it is not documented in the IIS.
IIS Background – Today, individuals of all ages are receiving vaccinations in a variety of settings that include medical practices, pharmacies, workplaces, travel clinics, and others. Statewide IIS, formerly referred to as immunization registries, have become the single consolidated resource for recording patient immunization events from a variety of data sources, including medical records, health information exchanges, dispensing systems, and others. IIS are confidential, population-based, computerized databases that provide participating providers an efficient way to record all administered vaccination doses. When utilized appropriately, IIS can support timely immunizations, consolidation of records, and enhanced provider efficiency.
Vaccination Forecasts Utilized by the Pharmacists in the Study - When the pharmacist accessed the State IIS, the technology interface utilized a clinical decision-support model based on current ACIP recommendations to generate a patient-specific vaccination forecast. The forecast identified the vaccines due that day for the individual patient based on the patient's documented vaccination history in the State IIS. Vaccines routinely recommended by ACIP for adults (ie, pneumococcal; tetanus, diphtheria, and pertussis [Tdap]; Zoster; HPV) were forecasted for patients who met the age requirements and had no documented history of receiving the vaccine in the IIS. In addition to the routinely recommended vaccines, the clinical decision-support technology also forecasted vaccines if the patient had initiated a vaccine series for hepatitis A, hepatitis B, or measles, mumps, rubella (MMR) but had no documented history of completing the series in the IIS.
Data collection
After institutional review board approval and clinical trials registration (NCT02584036), data collection began on October 22, 2015 and continued until March 22, 2016. This gave participating pharmacists a 6-month window during influenza season to implement the innovative practice model. Data were collected in the IIS and aggregate de-identified data were reported to the researchers. The data reported to the researchers included all administered vaccines that were documented by any health care professional in the IIS for the duration of the evaluation period. Data collection, use, and management procedures were compliant with the patient confidentiality provisions of the Health Insurance Portability and Accountability Act.
Data analysis
To be included in the evaluable population for data analysis, the following criteria must have been met: • The patient was at least 18 years of age; • The individuals were patients of participating pharmacies, and were eligible for and received an influenza vaccine at a participating pharmacy site during the study period (October 22, 2015 through March 22, 2016); and • The pharmacist documented reviewing the patient's vaccination forecast.
To evaluate the change in vaccination rates for the vaccines from baseline to the end of the study, investigators needed to determine the number of patients due for each vaccine based on ACIP recommendations at baseline. Investigators then determined how many patients were up-to-date with the vaccine at baseline and compared that to the number of patients up-to-date at the end of the study. To determine if there were any changes in the primary end points from the baseline measurement to the 6-month study end point, a series of McNemar tests comparing related proportions for paired pre- and post-implementation data (α = 0.05) were performed to test for a difference for each vaccine. Clustering effects of sites were ruled out using preliminary comparisons on demographics before performing McNemar tests. For secondary outcomes and demographic data, descriptive statistics were reported.
Results
Study population
During the 6-month study period, the pharmacists documented reviewing vaccination forecasts for 1080 patients who received influenza vaccinations in the pharmacies. Of the 1080 patients, 54.3% were female, with an average age of 62.5 years.
Number of additional vaccines due at the time of influenza vaccination
According to the vaccination forecasts generated by the clinical decision-support technology for the 1080 study patients, there were 1566 additional vaccines that also were due at the time the patient was receiving the influenza vaccination. For every patient who received an influenza vaccination in this study, an additional 1.45 vaccines were forecast by the technology.
Pharmacist assessment of additional vaccines due at the time of influenza vaccination
The pharmacists reviewed the 1566 vaccines that were due according to the vaccination forecasts generated by the clinical decision-support technology (Fig. 1). By assessing the patient's history and consulting with the patient, pharmacists identified and documented 36 contraindications and determined these patients were not eligible for vaccination. In addition to the contraindications identified, pharmacists identified 196 forecasted vaccines that were declined by the patient because the patient self-reported they had already received the forecasted vaccine. As a result of the pharmacists' assessments, there were a total of 1334 unmet vaccination needs in patients who were eligible for vaccination.
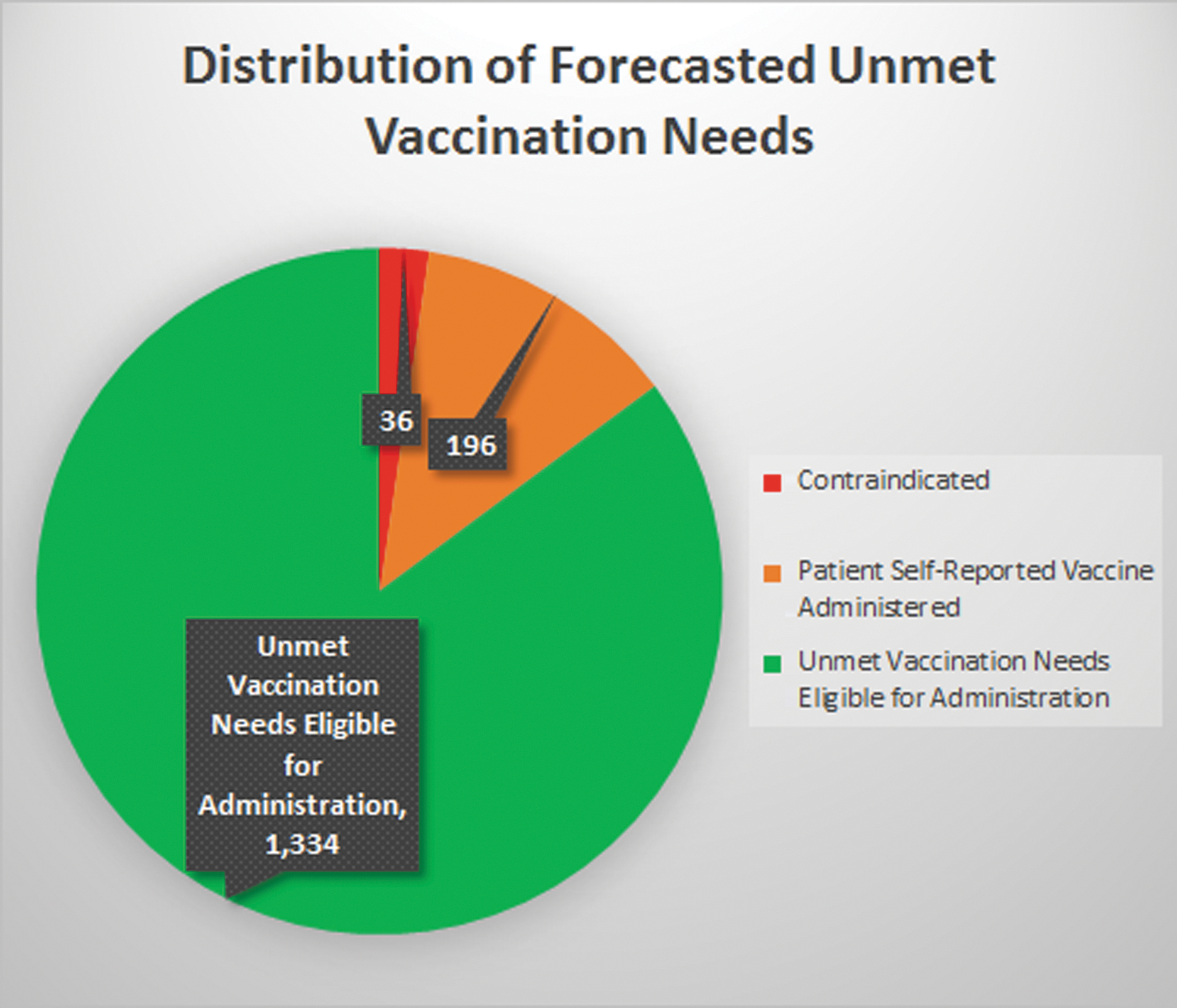
Distribution of forecasted unmet vaccination needs.
Unmet vaccination needs eligible for vaccination
A breakdown of the 1334 unmet vaccination needs in patients who were eligible for vaccination during the study period can be found in Table 3. The majority (92.3%) of the unmet vaccination needs in patients who were eligible for vaccination were for vaccines that are routinely recommended by ACIP for adults (ie, pneumococcal conjugate vaccine (PCV), pneumococcal polysaccharide vaccine (PPSV), Tdap, Zoster, HPV). A small percentage (8.7%) of patients also were due for an additional dose of hepatitis A, hepatitis B, or MMR because their IIS history indicated they had initiated the series of doses but had not yet completed the series (Table 3).
HPV, human papillomavirus; MMR, measles, mumps, rubella; PCV, pneumococcal conjugate vaccine; PPSV, pneumococcal polysaccharide vaccine; Tdap, tetanus, diphtheria, pertussis.
Unmet vaccination needs resolved
Of the 1334 unmet vaccination needs identified in patients who were eligible for vaccination, one third were resolved during the 6-month study period. Of the 447 unmet vaccination needs that were resolved during the 6-month study period, 426 (95.3%) were administered by the pharmacist at the point of care, while <5% were resolved at a patient-initiated follow-up visit (Table 3).
In the remainder of cases, pharmacists documented that they educated the patient about his/her unmet vaccination needs at the point of care but the patient chose to decline the additional vaccines recommended that day at the time of their influenza vaccine. In these cases, the pharmacist recommended that the patient follow up for their needed vaccines, referring them to other providers as needed.
Total number of vaccines administered
The total number of vaccines administered during the study included 1,080 influenza vaccines plus 447 additional non-influenza vaccines, equaling a total of 1527 total vaccines. The 447 additional vaccines administered as a result of using this innovative process of care resulted in a 41.4% increase in the number of vaccines administered (Fig. 2).
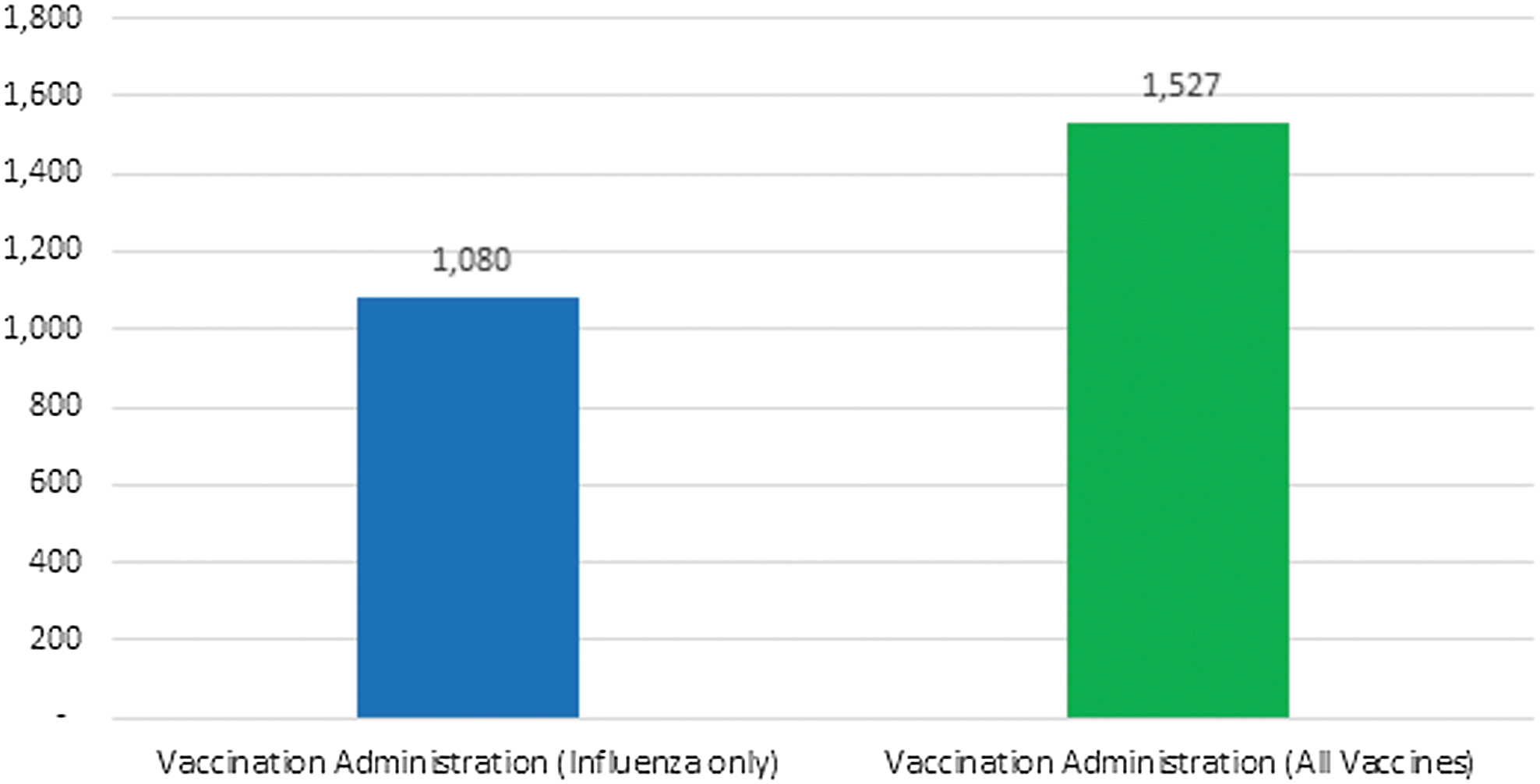
Number of vaccinations administered (influenza compared to all vaccines).
Change in vaccination rates for routinely recommended vaccines
Table 4 reports the number of patients who were up-to-date with each routinely recommended vaccine at baseline and how many eligible patients were up-to-date at the end of the study. Vaccination rates for PCV, PPSV, Tdap, Zoster, and HPV all increased from baseline to the end of study. The change in vaccination rates from baseline to the end of the study was statistically significant for PCV, Tdap, and Zoster.
Statistically significant.
HPV, human papillomavirus; PCV, pneumococcal conjugate vaccine; PPSV, pneumococcal polysaccharide vaccine; Tdap, tetanus, diphtheria, pertussis.
Discussion
Adult vaccination rates are low, significant opportunities to identify and resolve unmet vaccination needs exist, and changes in the health care system are required to achieve improved population health. The National Vaccine Advisory Committee calls on all health care professionals to take the following steps to ensure their adult patients are fully immunized. 17
1. Assess the immunization status of all patients at every clinical encounter.
2. Strongly recommend vaccines that patients need.
3. Administer needed vaccines or refer patients to a vaccination provider.
4. Document administered vaccines.
The Project IMPACT Immunizations innovative care model embraces these steps. During this study, pharmacists implemented the innovative practice model for 1 segment of their practice – patients requesting an influenza vaccine from the pharmacy.
Real-time point-of-care access to IIS vaccination histories and clinical decision-support technology made it feasible for pharmacists to efficiently assess the vaccination status of 1080 patients requesting an influenza vaccination during the study. According to the vaccination forecasts generated by the technology interface, there were 1566 additional vaccines due in the 1080 patients receiving an influenza vaccination. These results support the hypothesis that many adults, even those seeking an influenza vaccine, are unaware of their vaccination needs and emphasizes the importance of assessing each patient's immunization status on a consistent basis to avoid missed opportunities to vaccinate. Furthermore, according to the CDC, the general US population is far less likely to be vaccinated (Table 1) than participants who were in the study population (Table 4).
Although the clinical decision-support technology streamlined the process, it is important to recognize that a critical component of the process of care was the pharmacist assessment of the technology-generated vaccination forecasts. By engaging with the patient and using their clinical judgment (eg, pharmacist evaluates patient's documented IIS history to verify predicted patient needs, pharmacist evaluates medication use profile and patient self-reporting for documented previous administration to avoid potential duplications, pharmacist identifies patient needs including potential travel, pharmacist assesses patient for potential contraindications to forecasted vaccinations), pharmacists were able to identify if the patient had a contraindication or if the patient self-reported they had already received the forecasted vaccine. The pharmacist assessments minimized the potential for adverse events associated with contraindications and avoided potential duplications of therapy by identifying if the patient self-reported already receiving the vaccine.
Once the pharmacists conducted their assessments, they had the opportunity to educate patients about unmet vaccination needs and make evidence-based recommendations to the patients at the point of care. Research suggests that a recommendation from a health care professional is the strongest predictor of adults getting vaccinated. 17 –21 During Project IMPACT Immunizations, one third of patients eligible for an ACIP-recommended vaccine (447/1334) who received a recommendation from the pharmacist about their unmet vaccination needs were up-to-date by the end of the 6-month study period. Because the pharmacists conducted thorough vaccination needs assessments and educated the patients at the point of care, 447 patients received vaccines that they may have been unaware they needed.
Notably, 95.3% (426/447) of the patients' unmet vaccination needs that were resolved during the study occurred at the point of care, meaning the patient accepted the pharmacist's recommendations and received the additional vaccine(s) at the same time they received their influenza vaccine. This result highlights the value of incorporating this process of care into the pharmacy workflow, allowing pharmacists to maximize opportunities to vaccinate at the point of care.
The majority of the unmet vaccination needs that were identified and resolved during Project IMPACT Immunizations were for routinely recommended adult vaccinations (ie, pneumococcal, Tdap, Zoster, HPV). In addition to the routinely recommended adult vaccinations, there were 103 cases in which a patient had received at least 1 dose of a multidose series of hepatitis A, hepatitis B, or MMR but did not have documented evidence that they had completed the recommended series of doses. These vaccines are administered as a multidose series where the first dose provides some protection against the disease and the additional dose(s) maximize the level of protection. Pharmacists had the opportunity to educate the 103 patients who were under-vaccinated for hepatitis A, hepatitis B, and MMR about the need to complete the vaccine series to maximize their protection. Unvaccinated or under-vaccinated individuals are at risk for disease and they also can spread disease to others in the community. Therefore, identifying and resolving unmet vaccination needs not only has a positive impact on the individual patient's health but also makes a positive contribution to the health of the population in the community.
In those cases in which the patient made the decision to decline the additional vaccine recommendations at the point of care, the pharmacists still had the opportunity to educate the patient about his/her unmet vaccination needs and provide recommendations for appropriate follow-up and referral. These types of pharmacist recommendations can improve people's awareness and contribute to population health.
Vaccines administered by the pharmacists during Project IMPACT Immunizations were documented in the IIS. Pharmacists were actively contributing valuable information to the patient's IIS vaccination record so the information could be shared with the patient's other health care providers who also have access to the IIS. Recording accurate information in the patient's IIS record is critical to ensure continuity of care. The Project IMPACT Immunizations model of care embraced patient-centered care and the concept of the immunization neighborhood, wherein pharmacists were actively involved in collaborating, coordinating, and communicating with other immunization stakeholders to meet the vaccination needs of their patients. 22
By implementing the Project IMPACT Immunizations process of care, pharmacists not only improved their patients' awareness of their vaccination needs, they resolved unmet vaccination needs and improved vaccination rates in their patient population, thereby addressing prevention, one of the most formidable public health challenges. Prevention of disease through vaccination helps minimize the burden of vaccine-preventable diseases in communities. Ultimately, the goal of prevention through vaccination is to minimize the number of cases of vaccine-preventable diseases, decrease the number of hospitalizations and deaths, and lower the costs associated with vaccine-preventable diseases.
Opportunities/implications for practice
Pharmacists are important members of the interprofessional health care team who are ideally positioned to contribute to the solution of this public health problem. With more than 86% of Americans living within 5 miles of a community pharmacy, pharmacists are one of the most accessible health care providers in the US health care system. 23 As one of the most accessible health care providers, pharmacists have unparalleled access to people in communities across the country. If pharmacists can identify an efficient way to assess the immunization status of their patients at the point of care, they will have numerous opportunities to identify and address unmet vaccination needs in their patient population.
Even motivated, pro-vaccine patients who present to the pharmacy requesting an influenza vaccine are under-immunized. This study's data show that there was an average of 1.45 additional vaccines forecast as being due for each patient who requested an influenza vaccination. Project IMPACT Immunizations demonstrated that when pharmacists had IIS data and clinical decision-support resources, they were able to identify additional vaccination opportunities and, as a result of using the innovative process of care, increase the number of administered vaccines by 41.4%. Although the process of care was implemented in pharmacies building off of influenza vaccination encounters alone, the principle-centered approach to this innovative practice model could be replicated, allowing pharmacists to identify unmet vaccination needs for other segments of the population who seek pharmacy services. By building on this model and extrapolating it across America, pharmacists can continue to improve the nation's health by addressing unmet vaccination needs and improving vaccination rates. Understanding how this innovative practice model's results can be effectively translated into practices across the United States merits further investigation.
Limitations
Project IMPACT Immunizations was a pilot project implemented in 8 community pharmacy practices in Washington State to test an innovative practice model, so broad generalizability in practices across the United States may require further study. The 6-month study period limited the evaluation of the number of patients declining additional vaccinations at the point of care who may have responded to a structured follow-up system to receive the needed vaccinations over a longer period of time. Study methods limited the evaluation to just the population of patients who were already receiving influenza vaccinations, so understanding the effect of this model in the total population will require further evaluation. A larger, controlled evaluation could identify possible differences in population, process, provider, and practice types that could not be discerned in a prospective, observational pilot.
Conclusion
Gaps in vaccination coverage pose a significant risk to the health and well-being of our communities and innovative solutions are needed to solve this critical public health issue. The results from Project IMPACT Immunizations validate that pharmacists can be part of the solution. When pharmacists implemented the Project IMPACT Immunizations innovative practice model that enabled them to conduct comprehensive vaccination history reviews at the point of care, they were able to identify a significant number of unmet vaccination needs, educate 1080 patients about their vaccination needs, increase the number of vaccines administered by 41.4%, and improve vaccination rates for routinely recommended adult vaccinations. Exploring how to successfully integrate streamlined principle-centered processes of care that allow pharmacists and other health care providers to utilize actionable point-of-care data to effectively engage and educate patients to improve vaccination rates in practices across the United States will help us to
Footnotes
Acknowledgments
The authors wish to acknowledge Aaron Bonham from the Propel Centre for Population Health Impact at the University of Waterloo for his contributions to statistical analysis; Scientific Technologies Corporation for providing technology and implementation support to the pharmacists; the pharmacist-led care teams in each of the Washington State pharmacy practice sites for their dedication to improving people's health; the Washington State Pharmacy Association for their efforts to advance immunization practice and for providing state-level support in the project. The author received the following financial support: Project IMPACT Immunizations was conducted by the APhA Foundation with financial support from Merck and Co., Inc.
Author Disclosure Statement
Mr. Bluml, Dr. Brock, Dr. Hamstra, and Ms. Tonrey declared the following conflicts of interest with respect to the research, authorship, and/or publication of this article: Mr. Bluml is employed by the APhA Foundation. Dr. Brock served as a consultant to the APhA Foundation throughout the duration of Project IMPACT Immunizations. The authors have no other conflicts of interest.
