Abstract
Some 3 million people in the United States have atrial fibrillation (AF). Without thromboprophylaxis, AF increases overall stroke risk 5-fold. Prevention is paramount as AF-related strokes tend to be severe. Thromboprophylaxis reduces the annual incidence of stroke in AF patients by 22%–62%. However, antithrombotics are prescribed for only about half of appropriate AF patients. The study team estimates the economic implications for Medicare of fewer stroke events resulting from increased thromboprophylaxis among moderate- to high-risk AF patients. The decision model used considers both reduced stroke and increased bleeding risk from thromboprophylaxis for a hypothetical cohort on no thromboprophylaxis (45%), antiplatelets (10%), and anticoagulation (45%). AF prevalence, stroke risk, and stroke risk reduction are adjusted for age, comorbidities, and anticoagulation/antiplatelet status. Health care costs are literature based. At baseline, an estimated 24,677 ischemic strokes, 9127 hemorrhagic strokes, and 9550 bleeding events generate approximately $2.63 billion in annual event-related health care costs to Medicare for every million AF patients eligible for thromboprophylaxis. A 10% increase in anticoagulant use in the untreated population would reduce stroke events by 9%, reduce stroke fatalities by 9%, increase bleed events by 5%, and reduce annual stroke/bleed-related costs to Medicare by about $187 million (7.1%) for every million eligible AF patients. A modest 10% increase in the use of thromboprophylaxis would reduce event-related costs to Medicare by 7.1%, suggesting a compelling economic motivation to improve rates of appropriate thromboprophylaxis. New oral anticoagulants offering better balance between the risks of stroke and major bleeding events may improve these clinical and economic outcomes. (Population Health Management 2014;17:159–165)
Introduction
T
By itself AF is an important risk factor for stroke; overall, without thromboprophylaxis, individuals with AF have a 5-fold increased risk of ischemic stroke. 3,5 This risk is not uniform across AF patients; subpopulations have annual stroke rates that range from less than 2% to more than 18%. 6 Prevention is paramount as AF-related strokes tend to be severe, are more likely to recur, and have higher mortality than strokes that are not AF-related. 7,8
Meta-analyses have demonstrated that use of anticoagulants (eg, warfarin) and antiplatelet agents (acetylsalicylic acid, or ASA) reduces the risk of stroke by 22%–62% or more in patients with AF. 9,10 Because the net benefit of thromboprophylaxis is related to the underlying risk of stroke, stroke risk classification schemes—such as the CHADS2 scoring algorithm—have been developed to facilitate risk stratification and selection of antithrombotic prophylaxis based on patient-specific risk of stroke. 6 Published guidelines for stroke prevention in AF patients recommend treatment with ASA for patients with relatively lower stroke risk (lower CHADS2 scores) and warfarin for patients with relatively higher stroke risk (higher CHADS2 scores). 11,12
Indeed, the incidence of AF-related stroke has declined in the last decade, in part because of the use of antithrombotic agents. Nevertheless, the World Health Organization estimates that the aging of the population and the increasing incidence of AF will increase the stroke burden—in terms of the number of disability-affected life years related to stroke as well as the health system costs associated with stroke events—in the decade to come. 13
Despite the effectiveness of antithrombotic agents and their increased use since the early 1980s, 2,14,15 anticoagulation in the form of warfarin is prescribed for only about half of appropriate AF patients. 16,17 Long-term use of warfarin in clinical practice is hampered by drug and food interactions, slow onset of action, requirement for close patient monitoring and regular dose adjustments, and individual variability in metabolism. 18,19 Further, it is the nature of antithrombotic agents to increase bleeding risk; thus, the use of any thromboprophylaxis must balance the benefit of stroke prevention against the concomitant risk of bleeding. 20 Nevertheless, the underuse of antithrombotic prophylaxis in appropriate patients with AF suggests that, although significant reductions in stroke incidence have been achieved, more strokes may be prevented with wider use of thromboprophylaxis. 16 The goal of this study is to quantify this opportunity; specifically, to estimate the potential economic implications for traditional Medicare (Parts A and B) of fewer stroke events resulting from increased use of thromboprophylaxis monotherapy among AF patients at moderate-to-high risk of stroke events.
Methods
A decision-analytic model was used to assess the annual economic burden of ischemic and hemorrhagic strokes resulting from underuse of antithrombotic agents in a hypothetical Medicare population of patients with AF who are candidates for thromboprophylaxis monotherapy. Decision analyses are commonly used to estimate the financial stream of consequences related to the uptake and diffusion of health technologies. 21 Increasingly, decision analyses are sought out by those who manage and plan health care budgets, such as administrators of national or regional health care programs or private insurance plans.
The model begins with a hypothetical, age-representative population of 1 million Medicare patients with AF (Figure 1). 15 Inputs for the baseline case (Table 1) were set to approximate current clinical treatment patterns for use of antithrombotic monotherapy for patients with AF and to represent the age distribution of a Medicare cohort. Specifically, the model assumes that patients with AF are managed clinically either with no thromboprophylaxis (45%), ASA (10%), or warfarin (45%), a distribution that is consistent with studies of current management practices. 16 Stroke risk (based on CHADS2) and stroke risk-reduction are adjusted for age, comorbidities, and anticoagulation/antiplatelet status. The stroke prevention benefits of thromboprophylaxis are balanced against the risk of bleeding in the model, specifically intracranial hemorrhages and major extracranial bleeds that require hospitalization and/or blood transfusion.
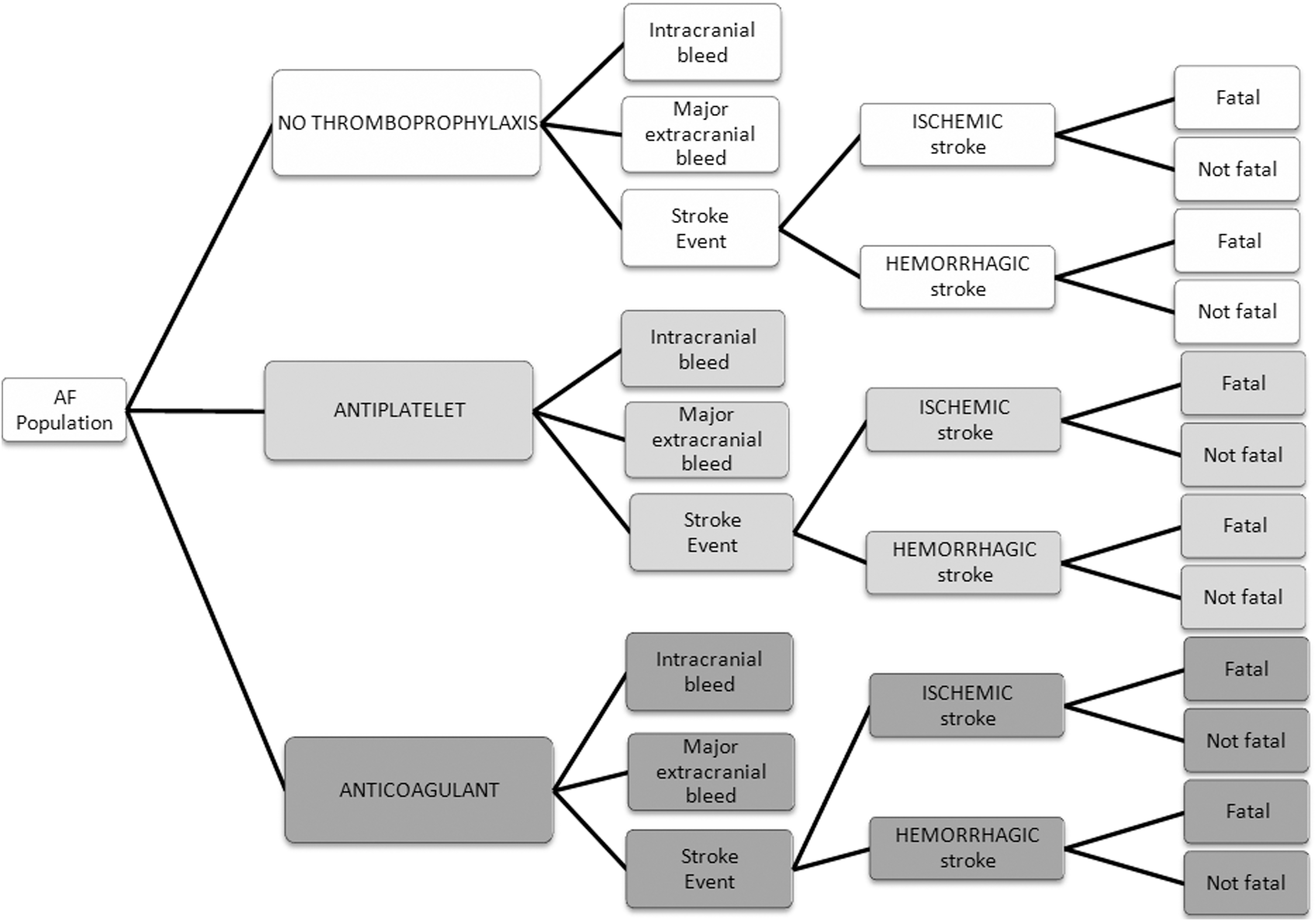
Model schematic. AF, atrial fibrillation.
Estimates of ICH event rates supported by the most recent data available.
AF, atrial fibrillation; AHRQ, Agency for Healthcare Research and Quality; HCUP, Healthcare Cost and Utilization Project; ICH, intracranial hemorrhage.
At baseline the model estimates the number of ischemic and hemorrhagic stroke events that might occur in each therapeutic cohort over a 1-year time horizon, as well as the number of those events that would result in fatality. In addition, the model estimates the annual stroke- and bleed-related health care costs (2011 US$) to Medicare, per million AF patients, based on the number of forecasted events and fatalities. A subsequent Increased Use analysis assesses the impact of a modest increase in the use of thromboprophylaxis on the incidence of stroke and bleed events, as well as the health care costs to Medicare associated with those events.
Results
Using baseline assumptions, Table 2 displays the model estimates of the numbers of ischemic strokes, hemorrhagic strokes, and major bleed events that will generate an estimated $2.63 billion in stroke/bleed-related health care costs to Medicare annually for every million patients with AF who are eligible for thromboprophylaxis monotherapy.
2011 US$.
AF, atrial fibrillation; ASA, aspirin/antiplatelet therapy.
A second analysis evaluated a 10% increase in the use of oral anticoagulants among the untreated population (ie, 35% of appropriate AF patients receive no thromboprophylaxis; 10% are prescribed ASA; 55% are prescribed warfarin), keeping all other variables constant. Table 3 displays the model estimates of the numbers of ischemic strokes, hemorrhagic strokes, and bleed events for this scenario. Together, these events will generate an estimated $2.44 billion in annual stroke/bleed-related health care costs to Medicare.
2011 US$.
AF, atrial fibrillation; ASA, aspirin/antiplatelet therapy.
Thus, the model (Figure 2) projects that a 10% increase in warfarin use from the untreated population will reduce total (ischemic and hemorrhagic) stroke events by 9%; reduce total stroke fatalities by 9%; increase bleed events by 5%; and reduce annual stroke/bleed-related costs to Medicare by about $187 million (7.1%) for every million patients with AF (Figure 3).
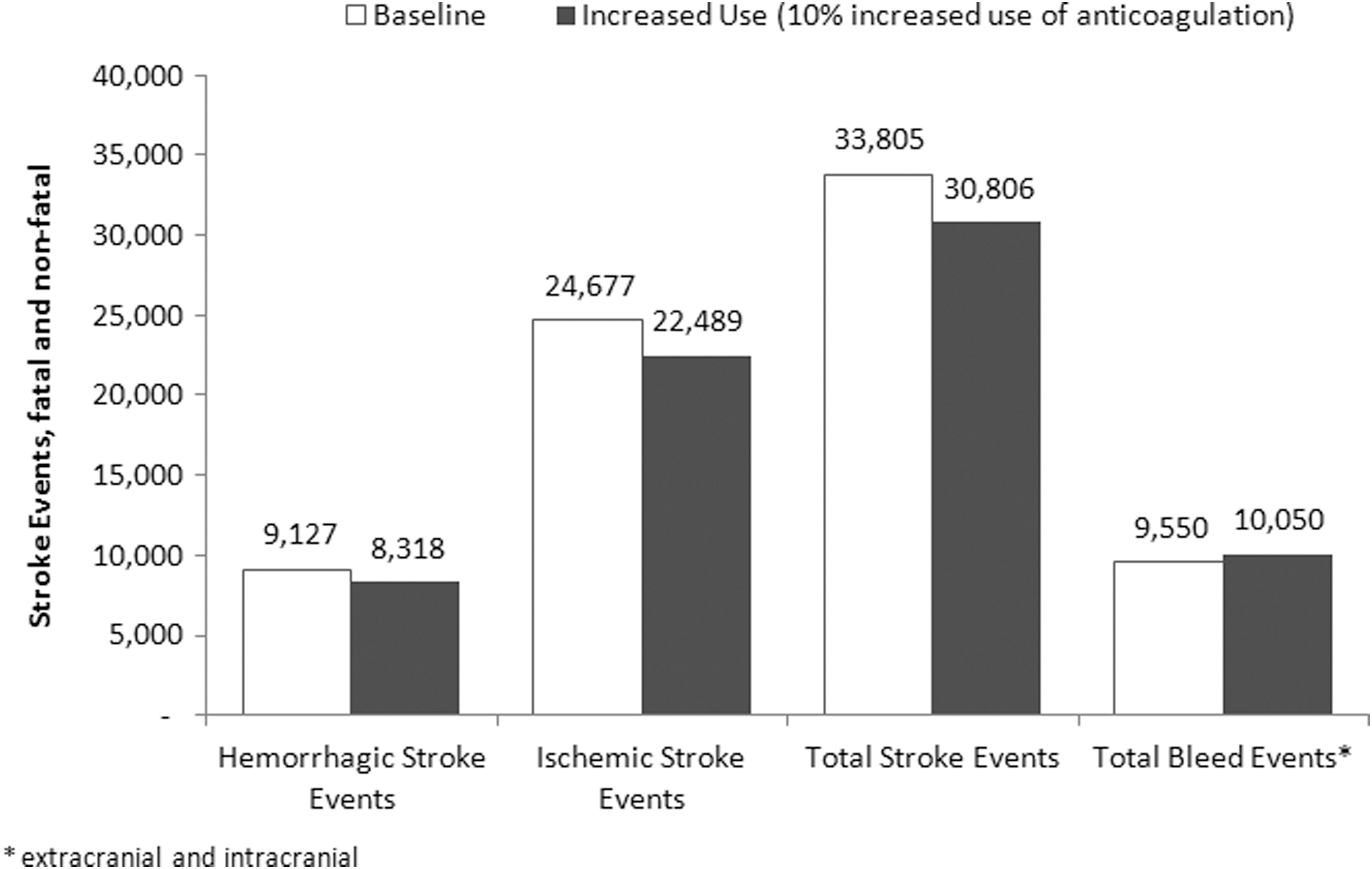
Number of stroke and bleed events with 10% increased use of warfarin (per million eligible patients with atrial fibrillation).
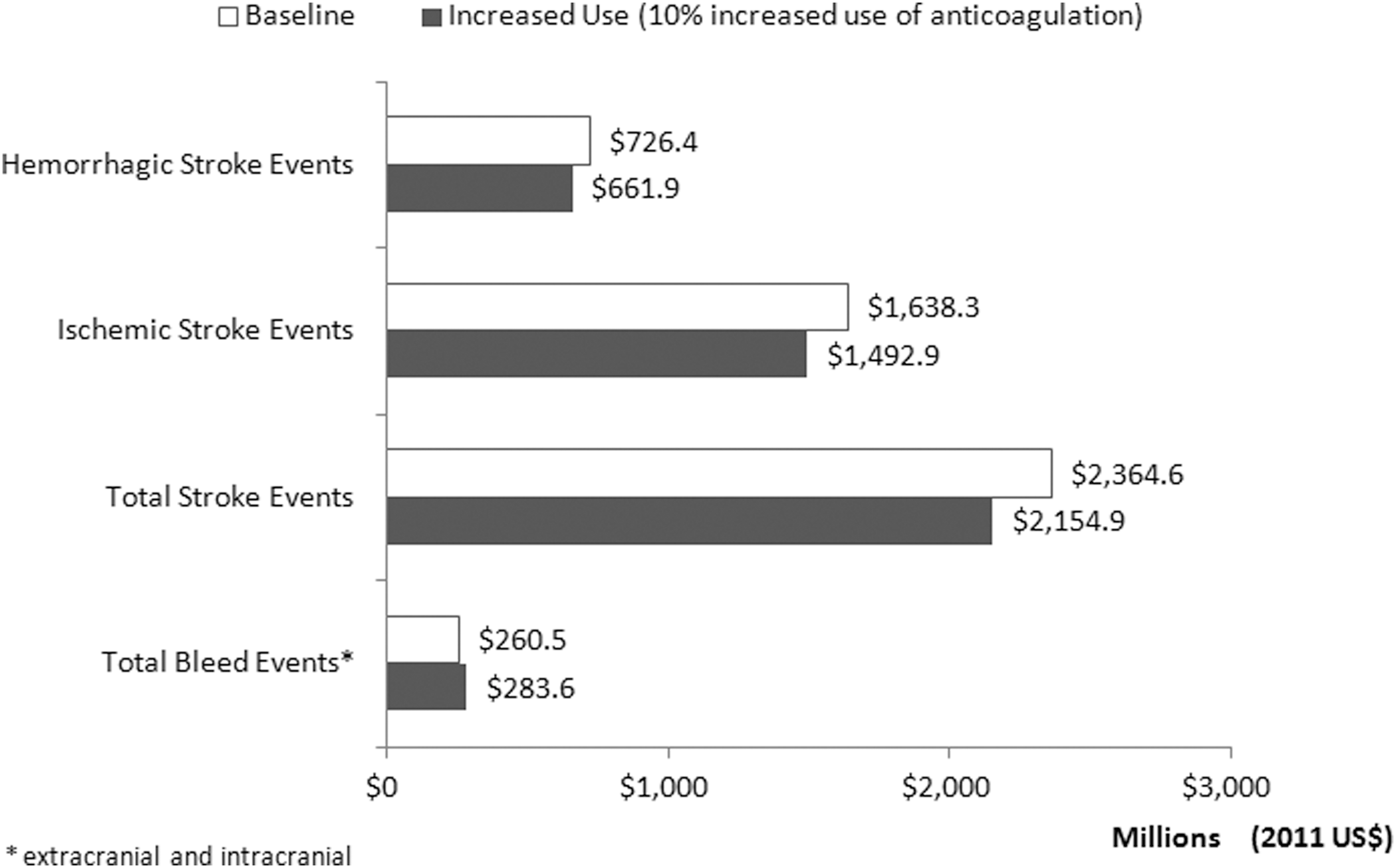
Annual stroke-related and bleed-related costs to Medicare of 10% increased use of anticoagulation (per million eligible patients with atrial fibrillation).
Sensitivity analysis
The baseline model stratifies patient stroke risk using point estimates from the widely used CHADS2 classification scheme. 6 Sensitivity analyses were run to evaluate the impact of lower and higher risk of stroke within the confidence limits of the reported CHADS2 point estimates, holding all other variables constant.
Using the lowest CHADS2 stroke risk estimates (1.2% to 4.5%; Table 1) in the model yielded 25% fewer stroke events (and 23% lower associated costs) compared to baseline. Conversely, use of the highest CHADS2 stroke risk estimates (3.0% to 7.7%; Table 1) yielded 31% more stroke events (and 28% higher associated costs) compared to baseline. The estimated number and cost of bleeding events in these analyses was unchanged (vs. baseline), as was the relative impact of a 10% increased use of anticoagulation on stroke events, stroke fatalities, bleeding events, and total annual costs to Medicare.
Discussion
Patients with AF are at much higher risk of stroke, and stroke-related death than are individuals without AF. This risk is not homogeneous; rather, it varies widely based on patient-specific characteristics such as age, sex, and comorbid conditions. Published guidelines for stroke prevention for patients with AF recommend thromboprophylaxis for patients at moderate-to-high risk of stroke. 11,12 Indeed, there is substantial evidence that antithrombotic prophylaxis decreases the risk of stroke in this population. 2,7,22 This use of thromboprophylaxis must, however, be balanced against the risk of bleeding events, such as intracranial hemorrhage and major extracranial bleeding requiring hospitalization and/or blood transfusion. 20
The present analysis quantifies the opportunity represented by anticoagulation undertreatment in the population of patients with AF. A decision-analytic model was used to estimate the potential economic implications for traditional Medicare resulting from increased use of ASA and warfarin among a hypothetical cohort of moderate- to high-risk AF patients. The model considers the reduced stroke and increased bleeding risk implications of anticoagulation monotherapy as well as reduced stroke-related fatalities. Stroke risk, and stroke risk reduction are adjusted for age, comorbidities, and anticoagulation/antiplatelet status. Annual incremental stroke-related health care costs are drawn from published sources.
The approach uses the CHADS2 scoring algorithm to estimate stroke risk in patients with AF. Substantial, clinically relevant differences exist about how to stratify stroke risk appropriately in AF populations, and alternative scoring schemes may lead to different stroke risk estimates. Indeed, the recently introduced CHA2DS2-VASc schema 23 could improve risk stratification in patients with AF; however, it has yet to be widely adopted in US clinical settings. Scoring algorithm differences also may foster confusion among practicing clinicians and result in the nonuniform use of antithrombotic therapies. 24 Only about half of AF patients in clinical practice settings who are appropriate candidates for stroke prophylaxis receive treatment, despite the availability of effective therapies. 16 This underuse of thromboprophylaxis in moderate- to high-risk AF patients could, if ameliorated, reduce preventable strokes and associated health care costs, morbidity, and mortality.
The present analysis forecasts that a modest 10% increase in the use of thromboprophylaxis would (1) reduce annual stroke/bleed-related dollar costs to Medicare by about 7.1% and (2) reduce stroke-related fatalities by 9%, suggesting that there may be a compelling economic motivation to improve rates of appropriate use of antithrombotic agents in this population. Clinically, though, a 10% increase in the use of warfarin among the currently untreated population of moderate- to high-risk AF patients also would result in a 5% increase in major extracranial and intracranial bleed events. Regardless of any economic benefits, such an increase in bleed events is not to be taken lightly. Clinical trials provide evidence newly emerging oral anticoagulant agents (NOAC) have at least a similar effect on reducing stroke rates compared to warfarin with comparable or improved safety profiles. This may represent an opportunity to further reduce the cost of undertreatment as well as improve clinical outcomes in AF populations. 25 This hypothesis should be evaluated in rigorous models of therapeutic cost-effectiveness once data on the real-world performance of NOAC become available.
Conclusion
A modest 10% increase in the use of antithrombotic monotherapy among patients with AF would (1) reduce stroke/bleed event-related dollar costs to Medicare by 7.1% and (2) reduce stroke-related fatalities by 9% in this cohort, suggesting a potentially compelling economic motivation to improve rates of appropriate thromboprophylaxis. New oral anticoagulation agents offering better balance between the risks of stroke and major bleeds may further improve the clinical and economic outcomes of thromboprophylaxis in this population.
Footnotes
Author Disclosure Statement
Dr. Patel, Ms. Ogden, and Drs. Veerman, Mody, Nelson, and Neil declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Dr. Neil and Ms. Ogden received consulting fees from Janssen Scientific Affairs, LLC for the development of this article. Drs. Patel, Veerman, Mody, and Nelson are employees of Janssen Scientific Affairs, LLC, a subsidiary of Johnson & Johnson (J&J), and are shareholders of J&J stock. This research was supported by Janssen Scientific Affairs, LLC, Raritan, New Jersey.
