Abstract
In an effort to reduce cost and improve quality, health care payers have enacted a number of incentives to motivate providers to focus their efforts on achieving better clinical outcomes and reducing the prevalence and progression of disease. In response to these incentives, providers are entering into new arrangements such as accountable care organizations and patient-centered medical homes to redesign delivery processes and achieve quality and cost objectives. This article reports the results of a study designed to evaluate the impact on cost and quality of care resulting from services provided by Health Diagnostic Laboratory, Inc., a clinical laboratory with a comprehensive care model. The results show that patients who utilized these laboratory services experienced lower total cost of care (23% reduction, P<0.01) and improved lipid profiles during the follow-up period. Total cost reductions were related to cost reductions found in both inpatient and ambulatory care. These findings suggest that accountable care organizations, patient-centered medical homes, and other groups entering shared savings initiatives should consider the potential role ancillary service providers with comprehensive care models can play in the delivery of integrated care. (Population Health Management 2014;17:121–126)
Introduction
O
Other programs, such as the Medicare Shared Savings Program, directly tie improved outcomes to reimbursement. Under the Medicare Shared Savings Program, providers are paid standard Medicare rates and receive bonus payments if total cost of care is below a predetermined benchmark and if quality thresholds are met. As of August 2012, a total of 116 organizations covering more than 4 million individuals had entered into shared savings agreements with the Centers for Medicare and Medicaid Services. 1,2
Private payers are following suit and various initiatives are under way to explore the effectiveness of payment models that combine fee for service, capitation, payment for quality thresholds, and shared savings. More than 8 private insurers, including Aetna, Anthem, and Blue Cross Blue Shield plans in Illinois, Massachusetts, New Jersey, and North Carolina, are exploring such options. 3 In some cases, providers can receive a portion of shared savings but face no penalty if savings are not realized. In other cases, providers pay penalties if actual health care costs are above the total cost of care target. In order to achieve savings goals, providers are transforming the provision of health care services in order to improve the coordination of care, improve the timing and accuracy of decisions, and reconfigure clinical processes to reduce errors. The most common examples of these new delivery models are the patient-centered medical home (PCMH) and the accountable care organization (ACO).
Patient-centered medical homes
A PCMH is a certified primary care practice that provides ongoing evidence that it is capable of providing care reflective of the principles of primary care: (1) an ongoing relationship with a personal physician for comprehensive care; (2) a physician- or nurse practitioner-directed team that is capable of meeting all the patient's needs; (3) a holistic focus; (4) coordinated care across the entire health care system and the patient's community; (5) safe, quality care through evidence-based medicine, effective use of information technology, and continuous improvement; and (6) enhanced access through expanded hours and alternate forms of communication. 4 PCMH initiatives also include payment reform whereby providers receive bonus payments for achieving quality of care thresholds. Thus far, results of PCMH initiatives are mixed. Some studies have found that PCMHs are associated with better outcomes and lower total cost. 5,6 In contrast, other studies have shown that, although PCMHs are effective at improving the patient experience and improving quality of care, there is no statistically significant reduction in total cost of care when compared to control groups receiving care at traditional physician practices. 7
Accountable care organizations
In contrast to the primary care focus of the PCMH, an ACO is formed when there is an arrangement between a payer and provider(s) to offer a complete spectrum of patient care services for a defined population. The ACO may comprise a single organization that is vertically integrated to provide primary, secondary, and tertiary care (eg, Mayo Clinic), or, more commonly, it may comprise a group of providers that collectively offer a complete range of health services. Similar to the PCMH, in order to receive bonus payments the ACO must reduce the total cost of care below a predetermined threshold while simultaneously meeting quality benchmarks. Although ACO initiatives are relatively new, early evidence suggests that most health care organizations struggle to manage populations of patients effectively, and the results of recent ACO initiatives in terms of cost savings are mixed. 8
Changing role of ancillary services
In response to the mixed results achieved by PCMHs and ACOs thus far, new initiatives seek to determine how to configure the composition, roles, and responsibilities of the provider teams that comprise the PCMH and the ACO. Initial PCMH and ACO efforts were focused predominately on improving the communication between health care institutions (eg, hospitals, rehabilitation centers) and on the behaviors of direct health care providers (eg, physicians). More recent initiatives are expressly changing how ancillary health care providers enter the health care delivery process. One example is the Vermont Blueprint for Health. Vermont has spearheaded an effort to proliferate PCMHs that incorporate so-called community health teams (CHT). 9 The role of the CHT is to provide multidisciplinary support for PCMHs and their patients and to integrate PCMHs with community resources. Health Diagnostic Laboratory, Inc. (HDL, Inc.), a clinical laboratory headquartered in Richmond, Virginia, and the subject of this study, is another example of the changing role of ancillary services. In addition to specimen processing and reporting, HDL, Inc. offers a comprehensive care model that links providers, patients, and clinical health consultants (CHCs) to help ensure that the appropriate tests are followed by evidence-based interventions, with subsequent lifestyle changes and counseling to promote wellness.
The HDL, Inc. model
HDL, Inc. offers comprehensive laboratory testing that includes serum biomarkers and genetic screening, enabling physicians to identify “at-risk” patients prior to a disease progressing to the point where an adverse event occurs or complex/invasive procedures are needed. In addition to performing clinical laboratory services, HDL, Inc. offers free supplemental services to providers and patients. Along with the laboratory results, providers receive guidelines for prescribing medication plus diet and exercise recommendations. These guidelines allow providers to develop personalized treatment plans quickly, targeted to patients' risk profiles (eg, heart disease, diabetes, metabolic syndrome). Patients receive a Web-based personal health record and patient portal that enables them to share information electronically with providers of their choosing, and free access to trained CHCs.
CHCs are pivotal to the HDL, Inc. model and their role is to ensure that patients fully understand their conditions and collaboratively develop an action plan to improve their health status. The CHCs include registered dietitians, certified diabetes educators, registered nurses, certified tobacco treatment specialists (trained at the Mayo clinic), and exercise physiologists—all of whom have extensive experience in patient counseling and motivational interviewing. CHCs are fully trained to review the comprehensive HDL, Inc. laboratory report with patients, develop therapeutic lifestyle interventions to reduce disease burden, and help patients achieve optimal health outcomes. CHCs interact with clients in many different ways, including live or telephonic counseling sessions and group classes. Their services are tailored to each patient (eg, individualized diet, stress management, exercise plans for weight control and cardiovascular risk reduction, practical shopping/cooking education).
The motivation behind the deployment of CHCs stems from recent studies showing that, despite well-publicized guidelines on the appropriate management of these diseases and their prevention, risk-reducing practices often are implemented inconsistently and difficult to sustain, especially in high-risk, vulnerable populations. 10 Yet it has been estimated that if all patients received the recommended prevention activities, myocardial infarctions and strokes could be reduced by 63% and 31%, respectively, in the next 30 years. 11 The Diabetes Prevention Program (DPP), a large study of people at high risk for diabetes, also showed that lifestyle intervention to lose weight and increase physical activity reduced the development of type 2 diabetes by 58% during a 3-year period. 8
The objective of the present study was to evaluate the impact of HDL, Inc.'s comprehensive care model on cost and quality of care. Total cost of care was examined as well as the distribution of costs among various types of health services (eg, inpatient services, ambulatory services). Quality of care was measured by frequency of adverse events and improvements in laboratory values associated with lower risk for future adverse events.
Methods
Study design
The study was an observational, retrospective case-control cohort study (complete details of the study design are available from the authors upon request) utilizing administrative pharmacy and medical claims data, and clinical data for a subset of patients. Total cost of care was based on actual medical claims data that were gathered independently and analyzed by HealthCore (Wilmington, DE), a health services research company. The study consisted of 2 cohorts, the HDL cohort and the control cohort.
Using the International Classification of Diseases, Ninth Revision (ICD-9) codes shown in Table 1 as inclusion criteria resulted in at-risk patient cohorts for whom, absent effective intervention, medical complications were more likely to occur in the near term. The ICD-9 codes in Table 1 also represent preexisting conditions for which ongoing medical management likely would benefit from the information included in HDL, Inc. panels. All subjects in this study were between 18 and 64 years old. To be included in the HDL cohort, individual subjects must have undergone at least 2 HDL, Inc. panels between June 1, 2010 and May 31, 2011 (inclusive). The control cohort included subjects who had at least 2 traditional lipid panels between June 1, 2010 and May 31, 2011 (inclusive). Finally, only members of the HDL cohort and control cohort who had 3 years of continuous health plan eligibility during their observation window were retained in the analytic file. The date of the first lipid panel was selected as the index date for each subject. As a result, all subjects had a full 12 months of baseline data and between 13 and 24 months of follow-up data.
BMI, body mass index; ICD-9, International Classification of Diseases, Ninth Revision.
At the first stage of the selection of the control cohort, Nearest Neighbor Propensity Score Matching, with a 0.02 caliper was used to develop a 1:1 match of controls to the HDL cohort during the initial identification of the control cohort members. Matching was based on Deyo-Charlson Co-morbidity Index, state of residence, age, sex, known coronary artery disease, history of ischemic stroke, previous percutaneous coronary intervention, previous coronary artery bypass graft surgery, and the variables listed in Table 1.
Results
The final study sample consisted of 443 participants; 229 were followed in the HDL cohort and 214 were followed in the matched control cohort. The average age of both cohorts was 51. The ratio of males was 65% in the HDL cohort and 66% in the matched control cohort. Although there were participants from all geographic regions, 94% were from the southeastern region of the United States (detailed descriptive analyses of the demographic and comorbidity characteristics of the HDL cohort and the control cohort are available from the authors upon request).
Figure 1A shows the study results for average total monthly cost of care during the 2-year follow-up period. Costs shown are average monthly costs per cohort member that have been aggregated over the amount of follow-up time they contributed to the study. During the follow-up period, when medical management included HDL, Inc. services, the average total monthly cost of care was significantly lower by 23% (-$150.00, P<0.01) compared to controls after adjusting for baseline differences in total health care costs. Interestingly, the reduction in total costs was seen despite higher costs observed in the HDL cohort during the baseline year before the first HDL, Inc. test panel. As shown in Figure 1B, total cost of care decreased over time, which suggests the benefit of including HDL, Inc. services accrued over time.
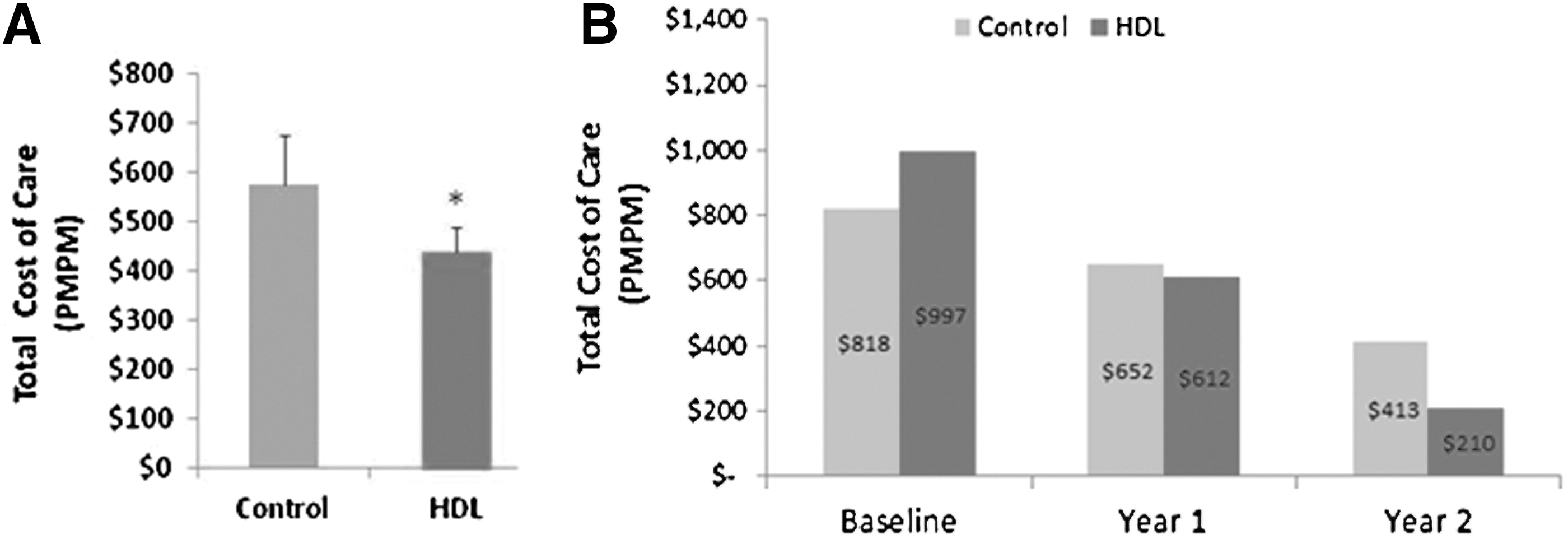
Total monthly cost of care of patients utilizing HDL, Inc. services compared to matched controls. HDL, Health Diagnostic Laboratory; PMPM, per member per month.
As shown in Figure 2A the overall decrease in average monthly cost of care for the HDL cohort was related to a statistically significant decrease in the cost of treating adverse events requiring inpatient care (-$114.00, P<0.01). The reduction in average monthly cost of inpatient care also corresponded with numerically lower rates of hospitalization and emergency room costs (Fig. 2B).
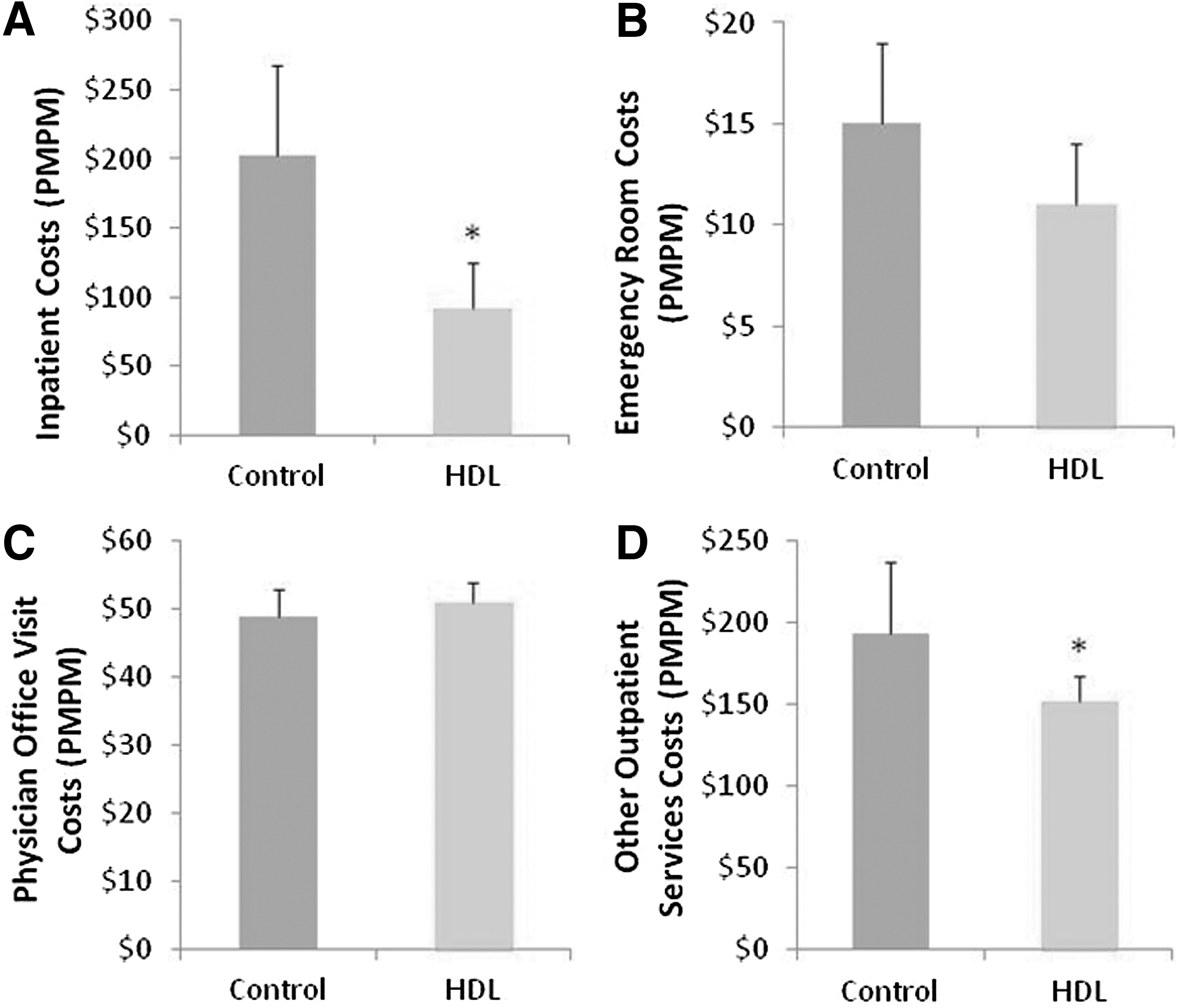
Average monthly cost of acute and ambulatory care for patients utilizing HDL, Inc. compared to matched controls. HDL, Health Diagnostic Laboratory; PMPM, per member per month.
In addition to the reduction in acute care costs, Figures 2C and 2D show that although cost of physician office visits was not significantly higher ($4.00, P=0.331), the HDL cohort experienced a lower cost of all other outpatient services (-$55.00, P=0.02). Other outpatient services represent all ambulatory services not provided by physicians as part of a routine office visit (eg, kidney dialysis, radiological imaging, ambulatory surgical procedures). These cost reductions corresponded with generally lower utilization rates of outpatient services.
Although the relatively small sample size in the present analysis was sufficient to demonstrate significant cost reductions, this study was not appropriately powered to show statistically significant reductions in adverse events. However, numerical trends suggest the HDL cohort experienced fewer total adverse events during the follow-up period. Importantly, diabetes complications (the most frequently observed adverse events tracked in this study) occurred at less than half the rate observed in the control cohort (Hazard Ratio=0.44, P=0.11; Fig. 3).
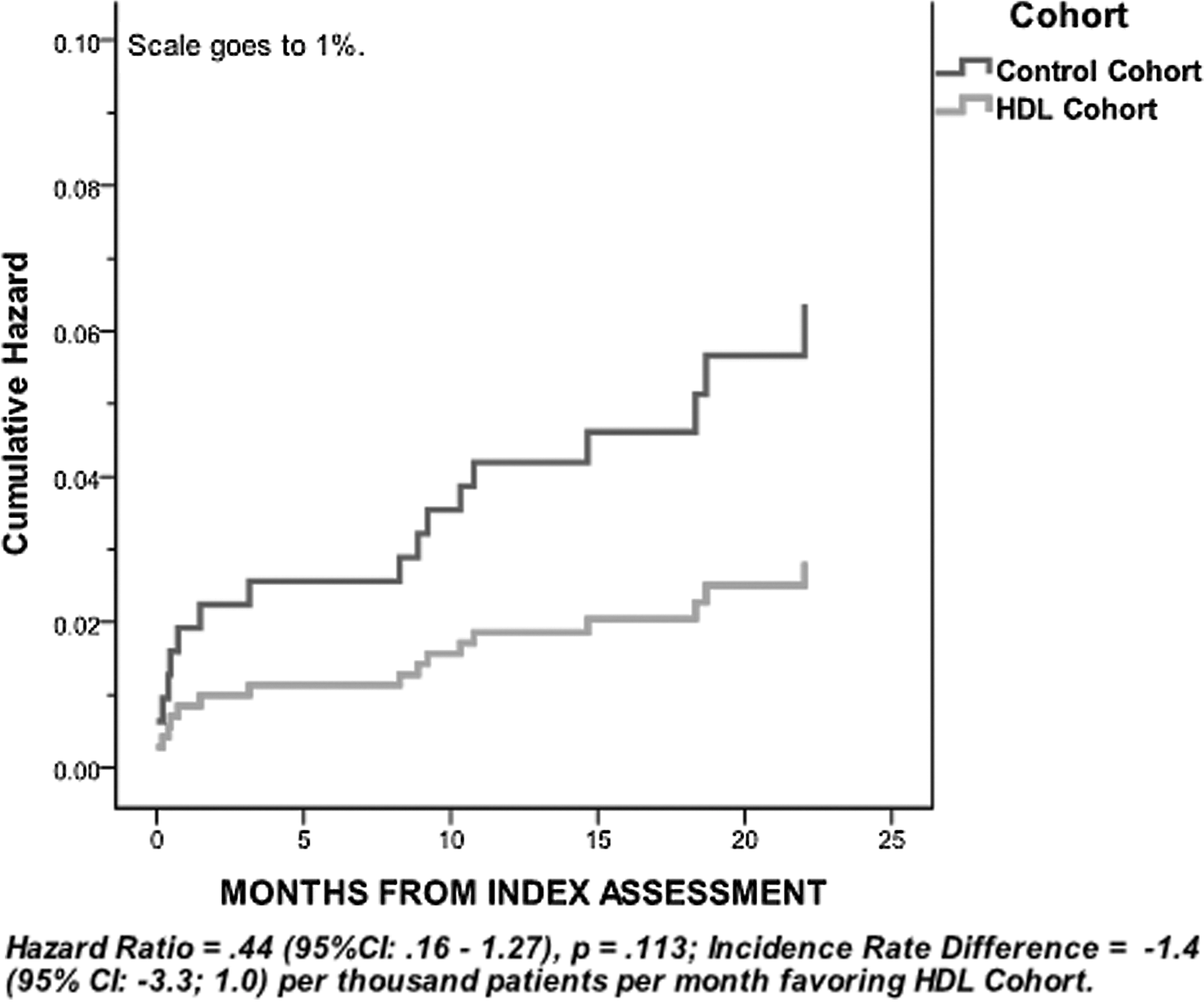
Hazard function for diabetes complications by Cohort.
Furthermore, statistically significantly greater improvements in lipid markers were achieved in the HDL cohort compared to controls (Fig. 4A and 4B). Low-density lipoprotein cholesterol (LDL-C) and triglyceride levels for the control group were obtained from the claims data. By the end of the follow-up period, the average LDL-C level within the HDL cohort had been reduced to below 100 mg/dL, the level recommended by the American Heart Association for those with or at risk for heart disease. Approximately 54% of patients in the HDL cohort achieved serum LDL-C levels below 100 mg/dL compared to 37% in the control group. Furthermore, 15% of the patients in the HDL cohort achieved serum LDL-C levels below 70 mg/dL compared to 10% in the control group. Based on prior medical research, the improvement in serum lipid levels in the HDL cohort suggests those individuals will experience fewer adverse cardiac events and presumably greater cost reductions in the future.
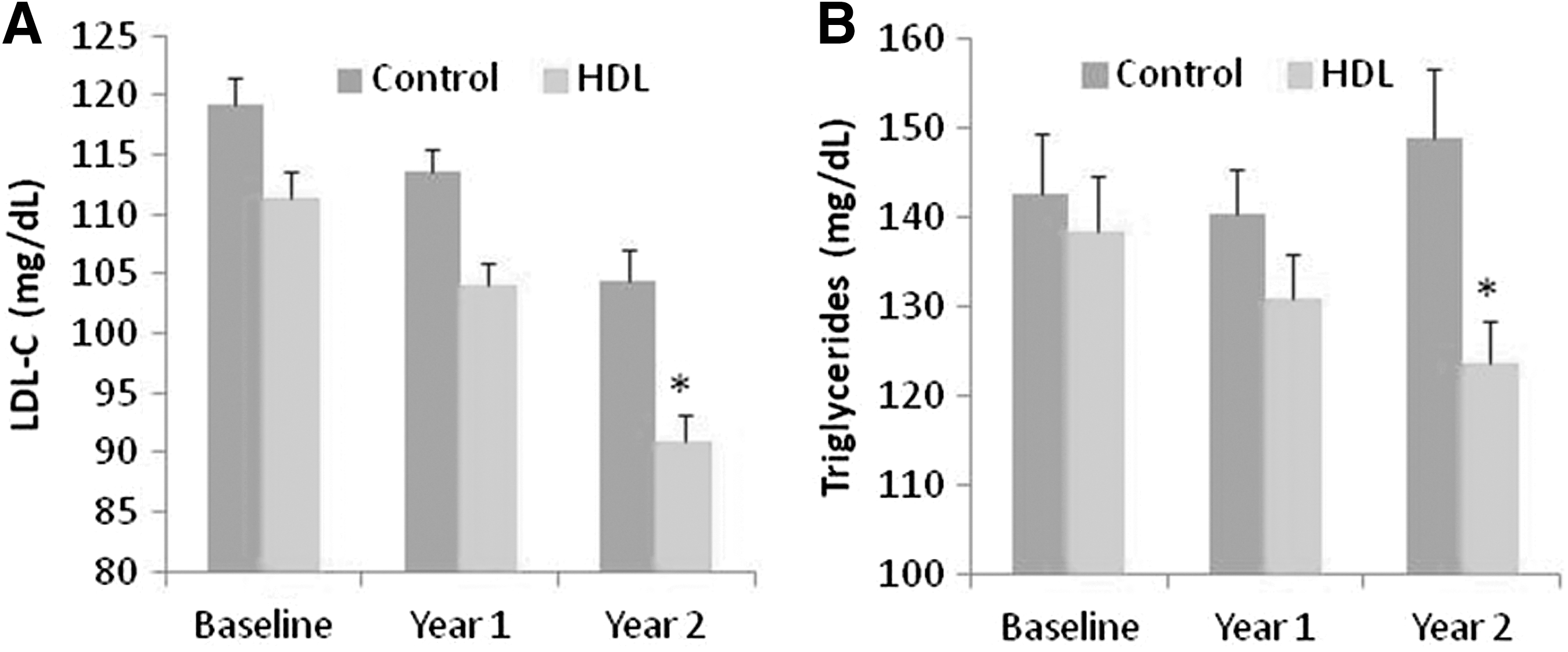
Significantly greater improvements in LDL-C
In addition, although comprehensive laboratory testing results were not available for the control group, significant improvements in several important cardiovascular risk markers including lipoproteins and apolipoproteins were observed in the HDL cohort when baseline values were compared to the last available test (Table 2). 13,14
APO B, apolipoprotein B; APO A1, apolipoprotein A-1; HDL-C, high-density lipoprotein cholesterol; LDL-C, low-density lipoprotein cholesterol; LDL-P, low-density lipoprotein particles; SEM, standard error of mean.
The significant improvements in lipid levels also were associated with a statistically significant higher utilization of statins and niacin by the HDL cohort over the course of the study. This suggests that physician prescribing practice was influenced by information contained in the HDL, Inc. panel results. At the same time, total pharmaceutical costs during the follow-up period for the HDL cohort and the control cohort were not statistically significantly different ($135.00 and $114.00 per member per month, respectively, P=0.182).
Because a complete HDL, Inc. panel includes a number of relatively expensive tests, including genetic screening, higher laboratory costs in the HDL cohort were expected. However, total laboratory costs for the HDL cohort and the control cohort during the follow-up period when those panels were performed were not statistically significantly different ($66.00 and $60.00 per member per month, respectively, P=0.241).
Discussion
The reductions in both utilization and cost observed in the present study lend support to HDL, Inc.'s model for patient care, in which advanced diagnostics, aggressive disease state management, and personalized health and wellness advice collectively help to coordinate the delivery of care.
However, further research is needed to understand the mechanisms resulting in these improvements in risk biomarkers and health care costs. For example, it is possible that physicians who are well versed in comprehensive, advanced testing may use HDL, Inc. services more frequently and may treat their high-risk patients more aggressively, and thereby play a moderating role in the outcomes observed. Many of the utilization and outcome metrics considered in this analysis did not yield statistically significant differences between the HDL cohort and the control cohort, possibly because of the small sample size and relatively short follow-up time; hence, future research should evaluate costs and outcomes over a longer time period and across a greater number of patients.
Nevertheless, it is important to note that the improvements in risk factors and reductions in cost observed in the HDL cohort support HDL, Inc.'s model of tightly coupling advanced laboratory services with prevention and disease management programs. Clinical laboratories such as HDL, Inc. are able to offer an increasingly wide array of tools to enhance the effectiveness of these programs. For example, improved risk stratification with a comprehensive panel of biomarkers allows prevention efforts to be appropriately focused most aggressively on those patients most likely to benefit, and identify those who would benefit from treatment who would be missed using traditional approaches. 15 The enhanced utilization of statins observed in the present study is consistent with this intention; the point being that without advanced testing, many such patients would not have been identified as high risk and may not have been treated at all. The ability to predict responses to particular drug treatments more accurately promises to enhance the effectiveness of medical management while reducing adverse events and consequent costs. 16 –18 Tools to access and share historical trends in laboratory markers can quickly support and enhance informational continuity and coordination of care. Perhaps most importantly, engaging patients in this process in a way that is personally meaningful, while educating and empowering them to track and participate in achieving their health goals, has great potential to improve patient compliance. 19
Conclusion
Although additional research is warranted, as providers and payers seek to improve outcomes, lower total cost of care, and improve the wellness of the populations they serve, this study's results suggest that clinical laboratory service providers can evolve to play an important role in the coordination and design of patient care plans that achieve those goals. PCMH and ACO initiatives should consider including clinical laboratory service providers with comprehensive care models, such as HDL, Inc., and should evaluate how other ancillary care providers can evolve to play important roles in the design and delivery of health care services.
Footnotes
Author Disclosure Statement
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Drs. Varvel, Voros, and Thiselton, and Ms. Grami are employees of Health Diagnostic Laboratory, Inc., the subject of this study. Drs. Turner and Barron are employees of HealthCore, Inc., which performed data abstraction, processing, and analysis for this study. Dr. Thompson declared no conflicts of interest. Dr. Thompson received funding for research support from Health Diagnostic Laboratory, Inc. Health Diagnostic Laboratory, Inc. provided funding to HealthCore, Inc. for data extraction, processing, and analysis.
