Abstract
Background:
Hemoglobin (Hb) has been reported as a prognostic factor in many cancer patients. However, its effect on the prognosis of patients with advanced cancer receiving palliative treatment is still unclear.
Objective:
The aim of this study was to assess whether Hb can be used as a prognostic indicator for patients with advanced cancer receiving palliative treatment.
Methods:
From July 2013 to October 2015, 368 consecutive patients were treated in the Palliative Treatment Center of Shanghai Cancer Center, Fudan University. The data of 105 patients were extended in the follow-up. The cutoff value selected for Hb was 100 g/L.
Results:
The median survival time of patients with low hemoglobin (LHb) was shorter than that of patients with high hemoglobin (HHb; 41 days vs. 84 days). In addition, in the follow-up readmission cohort (n = 105), the median survival time of patients with HHb (HHb → HHb) was 3.44 times longer than that of patients with LHb (HHb → LHb). To sum up, both LHb and descending Hb were identified as independent prognostic factors for poor prognosis.
Conclusions:
In palliative treatment, Hb can be used as a stratification factor to judge poor prognosis in advanced cancer patients.
Introduction
Cancer is a major public health problem, and the number of cancer patients will increase worldwide. It can lead to serious health consequences, especially advanced cancer that poses a great challenge to treatment, and is a common cause of death. 1 With these problems in mind, palliative care has been accepted as an essential part of the overall cancer treatment. 2 In palliative care, clinicians need to predict the life expectancy of patients with advanced cancer because determining the possibility of impending death will help doctors and promote clinical decision making to help patients and their families prepare for the future.3,4
In the past few decades, several prognostic models have been proposed. The most commonly used prognostic model in palliative treatment is clinical prediction of survival, but its inherent nonrepeatability limits its accuracy and clinical application. 5 In our previous studies, we found that the C-reactive protein/albumin (CRP/Alb) ratio, neutrophil count, and percentage affect the prognosis of palliative care patients with advanced cancer,6,7 but we still hoped to find more and easily accessible indicators to predict prognosis, and blood indicators were considered by us.
Hemoglobin (Hb) is widely used in clinical examination. 8 Low hemoglobin (LHb) levels are common in cancer patients, reported to be 30%–77%. 9 Although the number of studies exploring the prognostic role of Hb levels has been increasing, the results are inconsistent and are usually based on small samples. Few studies have reported the effect of LHb on survival in patients with advanced cancer receiving palliative treatment.
Methodology
Data collection and study cohort
Consecutive patients treated at the Palliative Care Unit of Fudan University Shanghai Cancer Center (FUSCC) in Shanghai, China, between July 2013 and October 2015 were retrospectively reviewed. Demographics (age and gender), medical history (comorbidities, smoking status, and family history), tumor-related factors (primary tumor site and tumor stage), nutritional status, and physical status (Eastern Cooperative Oncology Group, ECOG score) were obtained from the medical records of the patients. Hb was measured one to three days before the start of palliative care.
An unintentional weight loss >5% in the previous three months or a food intake below 75% of the normal requirement in the preceding week was considered to be an abnormal nutritional status according to the ESPEN guidelines for nutrition screening. 10 The presence of a concomitant disease was defined as self-reported cardiac disease, hypertension, diabetes, or any cerebrovascular disease.
Two cohorts of patients were identified in our study. In cohort 1, patients with the following inclusion criteria were enrolled: (1) hospitalization for palliative care; (2) the presence of various cancers confirmed by histopathology or at least cytology; (3) availability of pretreatment peripheral blood test results from one to three days before palliative care; and (4) availability of all clinical data. Patients with benign or early stage (I and II) tumors and those with active infectious disease were excluded from the analysis.
In cohort 1, the associations of several potential risk factors with overall survival (OS) were examined and qualified patients with full medical records were enrolled. A patient's OS was defined as hospitalization from the first day of treatment to death or lasting until October 2015. Patients with readmission data were categorized into cohort 2. The effects of change in Hb on OS were evaluated. The last follow-up date was in December 2015.
This study was approved by the Ethics Committee of FUSCC. Informed consent was waived because of the retrospective nature of the study.
Statistical analyses
Data are presented as the mean ± standard deviation (SD) for continuous variables and as totals and frequencies for categorical variables. The distribution of clinicopathological features was tested using the Wilcoxon rank sum test, chi-squared test, or Fisher's exact test, as appropriate. The Kaplan–Meier method was used to plot survival curves, and a log-rank test was conducted to test differences between groups.
Cox proportional hazards models were employed to estimate the magnitude of association between clinicopathological features and OS. X-tile, version 3.6.1 (Yale University, New Haven, CT), was used to determine optimal cutoff values. All tests were two-sided and p-values <0.05 were considered statistically significant. Statistical analyses were performed with SPSS (version 19.0), and R software (version 3.5.3) was used to draw survival curves.
Results
Patient characteristics
A total of 412 patients were admitted to the palliative care unit, and based on the inclusion and exclusion criteria, we got a total of 368 patients in cohort 1. Of the 368 patients, 105 with readmission data were selected for cohort 2.
As shown in Table 1, there were 201 males (54.62%) and 167 females (45.38%) in cohort 1. Most patients had stage IV (92.12%) cancer. In the classification of tumors, gastrointestinal tumors (189, 51.36%) accounted for the largest proportion, while head and neck neoplasms (17, 4.62%) accounted for a relatively small proportion. Most of these patients received the best supportive care (BSC; 275, 74.73%); BSC did not include transfusion in this study. There was no significant difference in the number of patients with ECOG <3 (216, 58.69%) and ECOG ≥3 (152, 41.31%). In terms of nutritional status, patients with poor nutritional status accounted for the vast majority (289, 78.53%).
Comparison of Baseline Clinicopathological Features Based on Hemoglobin in Cohort 1 (N = 368)
HHb: Hb >100 g/L; LHb: Hb ≤100 g/L.
BSC, best supportive care; ECOG, Eastern Cooperative Oncology Group; Hb, hemoglobin; HHb, high hemoglobin; LHb, low hemoglobin; PCR, palliative chemoradiotherapy; SD, standard deviation.
In cohort 2, the number of male (55, 52.38%) and female (50, 47.62%) patients is very close. Patients with stage IV (89, 84.76%) and gastrointestinal tumors (58, 55.24%) accounted for the vast majority. Unlike cohort 1, there were more patients (54, 51.43%) who were managed with palliative chemoradiotherapy (PCR) than (51, 48.57%) with BSC. The patients with ECOG <3 (79, 75.24%) are far more than those with ECOG ≥3 (26, 24.76%). The number of patients with good nutritional status (41, 39.05%) was close to that of patients with poor nutritional status (64, 60.95%) (Table 2).
Comparison of Baseline Clinicopathological Features Based on Hemoglobin in Cohort 2 (N = 105)
Determination of the cutoff value of Hb
In the clinical environment, it is common to convert continuous variables to binary variables. The patients were simply divided into the “high” group and “low” group, which was convenient for diagnosis or prognosis prediction. However, for patients with advanced cancer, the accepted clinical threshold is not available. Using X-tile, based on the minimum p-value algorithm, 11 the optimal cutoff value for Hb was 100 g/L. Therefore, patients with Hb greater than 100 g/L were grouped into the high Hb (HHb) group and patients with Hb less than or equal to 100 g/L were grouped into the LHb group.
Association of Hb and the change in its value with clinicopathological features
The relationship between clinicopathological features and Hb (low vs. high) in cohort 1 is shown in Table 1. There was no difference in age, tumor site, family history, ECOG, nutritional status, or comorbidity between HHb and LHb groups. Palliative treatment and gender were the clinicopathological features significantly associated with Hb.
The characteristics of cohort 2 are shown in Table 2, where patients are divided into two groups: the descending group (change <0) and ascending group (change >0). We found no significant difference in the proportion of patients with specific clinicopathological features between the two groups.
Association of Hb and the change in its value with OS
Patients in cohort 1 were divided into two groups: the HHb group and LHb group. According to the Kaplan–Meier analysis, the survival time of the HHb group was significantly longer than that of the LHb group (median survival time: 84 [95% confidence interval, CI: 61–107] days vs. 41 [95% CI: 31–51] days, p < 0.001) (Fig. 2). The multivariate Cox proportional hazards model showed that tumor stage, ECOG score, palliative treatment, and Hb were independent prognostic factors of OS. After controlling for important confounding variables, Hb (HHb vs. LHb, hazard ratio [HR]: 0.733, 95% CI: 0.558–0.909) was still significantly correlated with OS (Table 3).
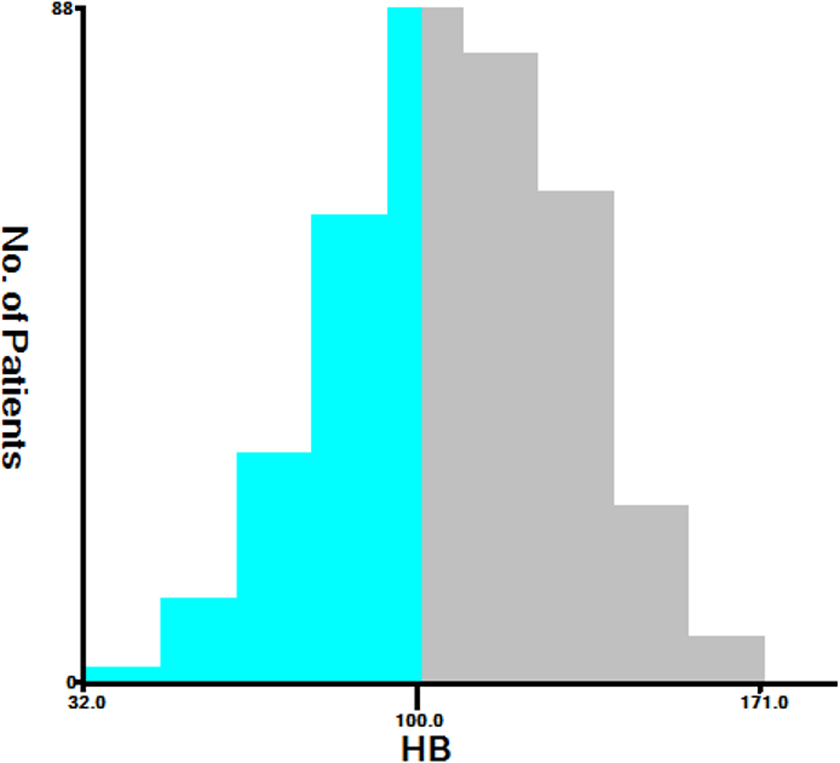
X-tile analysis was performed to determine the optimal cutoff values using the data of cohort 1. The optimal cutoff value for Hb was 100 (χ 2 = 15.85, p = 0.0026). However, for patients with advanced cancer, the accepted clinical threshold is not available. Using X-tile, based on the minimum p-value algorithm, 11 the optimal cutoff value for Hb was 100 g/L. Hb, hemoglobin.

Overall survival of patients under palliative care, stratified by pretreatment Hb (cohort 1). HHb, high hemoglobin (pretreatment >100); LHb, low hemoglobin (pretreatment ≤100).
Multivariate Cox Regression Analysis for Hemoglobin (N = 368)
HHb: pretreatment >100; LHb: pretreatment ≤100.
The potential prognostic factors used for adjustment in the Cox regression model, controlling for gender; age; primary tumor site; tumor stage; ECOG; nutrient status; palliative care; comorbidity; and family history.
CI, confidence interval; HR, hazard ratio.
To verify the prognostic significance of dynamic changes in Hb, we studied cohort 2. According to the change of Hb, patients were divided into the ascending group and descending group. We found that patients who were converted from HHb pretreatment to LHb after the second admission had the worst OS (LHb → LHb median survival time: 109 [95% CI: 49–169] days vs. LHb → HHb median survival time: 104 [95% CI: 0–276] days vs. HHb → LHb median survival time: 68 [95% CI: 45–90] days vs. HHb → HHb median survival time: 234 [95% CI: 168–300] days, p = 0.013) (Fig. 3). The related subgroups were further analyzed.

Overall survival of patients under palliative care, stratified by changes in Hb (cohort 2).
After adjusting for demographic and disease-specific factors, significant results were obtained (Table 4).
Adjusted Hazard Ratios for Overall Survival Stratified by Changes in Hemoglobin in Cohort 2 (N = 105)
HHb: pretreatment >100; LHb: pretreatment ≤100.
The potential prognostic factors used for adjustment in the Cox regression model, controlling for gender; age; primary tumor site; tumor stage; ECOG; nutrient status; palliative care; comorbidity; and family history.
Discussion
Although tremendous progress has been made in the treatment of tumors, the likelihood of treatment failure for patients with advanced cancer remains high. Survival assessment is necessary for risk stratification and further clinical decision making. However, to date, no significant biomarker has been widely used in survival assessment. Almost all hospitals in China test hospitalized patients for Hb. In our study, we evaluated the prognostic impact of Hb on patients with advanced cancer.
At present, there is no study on the dynamic changes of Hb in patients with advanced cancer in a palliative treatment environment. Therefore, this study used the advantage of dynamic change to solve this problem. We established the cutoff value of 100 g/L for Hb. In our study, first, we included all palliative care patients with advanced cancer in cohort 1; second, we divided them into LHb and HHb groups according to the cutoff value of Hb; and third, we compared the survival time of the two groups, and we found that the survival time of the LHb group was shorter than that of the HHb group.
Second-admission patients were included in cohort 2. We screened patients in the two groups to divide them into four subgroups to see the effect of dynamic Hb changes on OS, and we found that survival time was shortest when patients went from the HHb group to the LHb group, and patients had the longest survival time if they could have been in the HHb group. In conclusion, as a convenient and intuitive marker, pretreatment and dynamic peripheral Hb showed the ability to predict aggressiveness in patients with advanced cancer.
Ludwig et al. 12 calculated that the incidence of anemia in 11,453 patients with various cancers was 41%. In patients with advanced tumors and those receiving radiotherapy or chemotherapy, the incidence of anemia is as high as 90%. 13 Especially in patients with more aggressive tumors, it may also be related to complex interactions among the immune system, tumor microenvironment, and cancer cells.14,15 In general, cancer-related anemia is caused by a variety of mechanisms, including blood loss, functional iron deficiency, erythropoietin deficiency due to kidney disease, the relationship between cancer and the bone marrow, and other factors. 16
Tumor cells can activate macrophages and promote the secretion of inflammatory factors such as gamma-inF, IL-1, and tumor necrosis factor; these molecules can reduce Hb concentrations by altering the hematopoietic environment,17,18 inhibiting erythropoiesis and erythropoietin, and impairing the erythropoietin response of erythroid progenitor cells.19,20 In addition, bone marrow failure is common in patients with bone metastases and leads to LHb levels. 21
Patients with later tumor stage and greater tumor burden have more inflammatory factors secreted by tumor cells, higher incidence of anemia, and worse prognosis. 22 Hypoxia is an important link in the occurrence and development of malignant tumors, 23 while anemia will aggravate the degree of hypoxia in tumor tissues and affect the stability of chemotherapeutic drugs in the process of diffusion. At the same time, production of oxygen free radicals reduces the damaging effect of chemotherapeutic drugs on the DNA of tumor cells, thus reducing the efficacy of chemotherapy drugs. 24
Tissue oxygenation disorder stimulates the high expression of angiogenic factors, thus promoting the occurrence and development of tumors, reducing the sensitivity of chemotherapy, increasing the probability of blood transfusion after chemotherapy, damaging the function of organs, reducing the quality of life, and increasing postoperative mortality. A meta-analysis involving 60 studies showed that anemia increased the overall risk of death in tumor patients by 65%. 25
Previous studies have shown that anemia may have a certain impact on the biological characteristics and prognosis of tumor patients, may lead to reduced response to chemotherapy in patients with cervical cancer and head and neck malignant tumors, and promote tumor recurrence and metastasis, which is an independent risk factor for poor prognosis in patients. 26 Studies have reported 27 that anemia is not a bad prognostic factor in patients with advanced gastric cancer; however, studies have also confirmed 28 that anemia is a bad prognostic factor in patients with advanced gastric cancer. This may be related to the inconsistent reference criteria used for anemia.
The U.S. National Cancer Institute (NCI) and the World Health Organization (WHO) define their own anemia diagnosis and classification standard, and the Chinese Society of Clinical Oncology (notes) standard is based on clinical practice and the treatment of cancer-related anemia by classification, 29 the severity of different standards in defining the normal range of Hb, and slightly different severity classification for anemia. Therefore, to unify the criteria, we established the threshold value of Hb as a tool to predict the prognosis of patients with advanced cancer.
Although this study provides important insights into the relationship between Hb and the prognosis of patients with advanced cancer, this study still has some limitations. This study is limited by a retrospective single-center design with inherent limitations such as selection bias, possible confounders, and relatively small sample size. Interventions that potentially have an impact on Hb are also factors that we may need to consider. Therefore, larger, prospective multi-institutional studies with more clinical markers are needed to validate the relationship between Hb and poor prognosis.
All in all, we demonstrate for the first time that Hb is a strong independent predictor of shortened survival after palliative treatment in patients with advanced cancer. As an easily accessible and inexpensive biomarker, Hb deserves further research in future as a clinical practice tool for risk stratification and may enable more correct clinical decision making in patients with advanced cancer.
Footnotes
Authors' Contributions
All authors made substantial contributions to one or more of the following: the study conception and design (X.L. and W.Z.); acquisition of data or analysis (X.L., W.Z., C.B., Q.S., M.L., Z.Z., and C.Z.); and interpretation of data (H.L., Z.Z., and J.Z.). X.L. and C.B. drafted the article, and all other authors contributed to revising the article critically for important intellectual content. All authors provided their final approval of the version to be published.
Acknowledgments
The authors are grateful to the participating patients and their families and to all other coinvestigators who contributed to this study.
Funding Information
This work was supported by the National Natural Science Foundation of China (81400810, 81101762, and 11371100).
Author Disclosure Statement
The authors declare no conflicts of interest.
