Abstract
Background:
Dry eye disease (DED) is a complex ocular surface inflammatory disorder with a multifactorial etiology. Therapies such as intense pulsed light (IPL) and heated eye mask (HEM) have been reported to improve the tear film lipid layer (TFLL) and signs and symptoms of DED.
Methods:
This randomized study aimed to compare the effects of IPL combined with HEM (IPL+HEM) group, IPL group, and control group in participants with evaporative DED. All participants were examined at baseline (D0), day 21 (D21), day 42 (D42), and day 84 (D84) for noninvasive tear breakup time (NITBUT), TFLL, corneal conjunctival staining (CS), meibomian gland quality (MGQ), meibomian gland expressibility (MGEx), and Ocular Surface Disease Index (OSDI).
Results:
The mean age of participants was IPL+HEM: 28.06 ± 3.88 years, IPL: 29.88 ± 4.68 years, and control: 28.52 ± 3.77 years. At D84, significant improvements in TFLL (p < 0.05), noninvasive tear breakup time (NITBUT) (p < 0.05), corneoconjunctival staining (CS) (p < 0.05), MGQ (p < 0.05), MGEx (p < 0.05), and OSDI (p < 0.05) were found in the IPL+HEM and IPL groups, whereas the control group had no significant improvements. Furthermore, ΔTFLL significantly correlated with ΔNITBUT (r = −0.678, p < 0.001), ΔCS (r = 0.321, p < 0.001), ΔMGQ (r = 0.669, p < 0.001), ΔMGEx (r = 0.598, p < 0.001), and ΔOSDI score (r = 0.649, p < 0.001).
Conclusions:
IPL therapy in combination with HEM and IPL therapy only can significantly improve the quality of TFLL and clinically reduce the sign and symptoms of evaporative DED. However, IPL therapy in combination with HEM was found to be more effective than IPL therapy alone.
Introduction
Dry eye disease (DED) is a common ocular disorder and can be grossly categorized as impaired tear production (aqueous-deficient dry eye), excessive tear film evaporation [evaporative dry eye (EDE)], or a combination of both mechanisms. 1,2 DED is a frequent cause of ocular complaints leading patients to seek ophthalmic care, 3,4 and its severity may range from mild, which leads to occasional discomfort to sight-threatening and severely debilitating disease. 5,6 Conventional understanding suggests that the oily component of the tear film assists in maintaining tear stability by reducing tear evaporation and lubrication between the ocular surface and the eyelids during blinking. 2
Recent studies have alluded to the fact that the tear film lipid layer (TFLL) has antimicrobial characteristics that keep the lid margin and ocular surface healthy. 7 TFLL is a mixture of polar lipids (phospholipids) and nonpolar lipids (cholesterol, wax esters, and cholesterol esters) 8 secreted by the meibomian glands, located across the lid margin of the superior and inferior eyelids. 9 Meibomian gland dysfunction (MGD) is documented to be a key component in evaporative DED. 10 The most common mechanism of MGD is gland obstruction caused by increased meibum viscosity, combined with atrophic glands and epithelial hyperkeratinization 11 that leads to stasis and cystic distention of the glands. 12 The main risk factors for MGD include advanced age, extremely dry environment, rosacea, seborrheic dermatitis, and infestation by Demodex. 13 –17 MGD is by far the most prevalent cause of evaporative DED, with a frequency of 5–20% in Western nations and 45–70% in Asian populations. 18 –20
Ocular surface damage in MGD results from a combination of several mechanisms, including increased tear evaporation, hyperosmolarity, secretion of proinflammatory mediators, and increased friction between the eyelids and globe. 21,22 Tear-free baby shampoo helps certain patients and, for more severe instances, meibomian gland expression, massage, and probing. 23 Omega-3 fatty acid supplements improve MGD and dry eye symptoms. 24 Tea tree oil, particularly terpenin-4-ol, treats Demodex mite-related MGD. 25 Topical or systemic antibiotics such as azithromycin or doxycycline may relieve MGD symptoms by modifying the ocular flora and reducing inflammation. 26 –28 Topical N-acetylcysteine and cyclosporine can help dry eye patients. 29,30 Local heating of the eyelid margin area has been the mainstay of MGD treatment for many decades due to its ability to induce liquefaction of the meibum at temperatures closer to 40°C, thus facilitating its expression from the clogged glands. 31 –36
Intense pulsed light (IPL) is a common treatment for face skin rosacea, and Toyos et al. observed that it alleviated dry eye symptoms over a decade ago. 37 Although earlier studies have reported the advantages of IPL treatment on a broad severity spectrum of DED, 32,33,38 –43 none have assessed the combined efficacy of IPL and heated eye mask (HEM) on evaporative DED. Therefore, this study aimed to examine the effectiveness of IPL combined with HEM for evaporative DED.
Methods
Study design
This randomized study assessed DED patients from January to October 2021 treated at the Dry Eye Clinic, Department of Ophthalmology, He Eye Specialist Hospital, Shenyang, China. The study was conducted according to the guidelines of the Declaration of Helsinki and approved by the institutional review board of the He Eye Specialist Hospital, Shenyang, China (IRB-2019K002.01), approved on January 23, 2019. Written informed consent was obtained from the patients to publish clinical data and any accompanying images. This article includes no identifiable patient information. All subjects gave informed permission before the treatment.
All participants were diagnosed based on the following Asian dry eye diagnostic criteria: (1) the presence of dry eye symptoms; (2) the presence of either a qualitative or quantitative tear film disturbance [Schirmer's test 5 mm or tear breakup time (TBUT) 5 sec]; and (3) the presence of conjunctivocorneal epithelial damage (fluorescein staining score >3 points, rose bengal staining score >3 points, or lissamine green staining score >3 points). 44
This research involved a total of 150 participants (150 eyes) who were diagnosed with dry eye and aged at least 18 years. A custom computer randomization program was used to randomly (1:1:1) allocate participants to three study groups: (1) IPL group, (2) IPL+HEM group, and (3) control group (Fig. 1).
Study design. HEM, heated eye mask; IPL, intense pulsed light; IPL+HEM, intense pulsed light and heated eye mask.
Eligibility requirements: The following were eligibility requirements for all groups: (1) age <18 years, (2) Fitzpatrick skin types I–IV, (3) capable and willing to comply with treatment and follow-up obligations, (4) a determination of severe DED based on (a) the Ocular Surface Disease Index (OSDI; a score of 33 indicates severe DED), (b) a noninvasive tear film breakup time (NITBUT) of <5 sec, or (c) a corneoconjunctival staining (CS) score of <3 points according to the Asian Dry Eye Consensus. 44 Exclusion criteria are as follows: (1) existing ocular trauma, infectious diseases, and recent surgical history; (2) skin defects, pigmentation, moles, scars in the treatment area, and skin cancer; (3) autoimmune diseases and skin allergies; (4) pregnancy or breastfeeding; and (5) Fitzpatrick skin types V or VI.
Objective and subjective examinations
The Keratograph 5M (Oculus, Germany) was used to measure the noninvasive tear breakup time (NITBUT). Interferometry TFLL quality was assessed using DR-1 (Kowa, Nagoya, Japan) 45 and corneal fluorescein staining (CS) tests followed by Asian dry eye diagnostic criteria. 44 Meibomian gland quality (MGQ) and meibomian gland expressibility (MGEx) were measured as mentioned by Korb and Blackie. 46 The Chinese online version of the OSDI questionnaire measured DED symptoms. 47
Treatment
Toyos et al. protocol was used for all treatments, and patients got three sessions spaced by 3 weeks. 41 The IPL system (M22; Lumenis, Yokneam, Israel) with its xenon lamp emits light with a wavelength ranging from 515 to 1200 nm. The device can be adjusted with a 560 nm filter, which allows a range of 11–14 J/cm2 for varied skin tones. On day 0 (D0), day 21 (D21), and day 42 (D42), each patient was administered three sessions of IPL therapy. The HEM used in this investigation was a medically approved disposable air-activated eye mask (Ocuface Medical Co., Ltd., Guangzhou, China) 32,33 (Fig. 1).
Experimental design
IPL+HEM group
Participants underwent IPL treatment with 12 uniformly spaced pulses of light to both eyes on D0, D21, and D42 by a trained physician who was not involved in data collection. During D0, D21, and D42, participants wore opaque goggles, and ultrasound gel was generously applied to their targeted area while wearing opaque goggles. HEM was used for 20 min daily for 84 days. All participants were required to attend their allocated IPL+HEM and IPL therapy session on D0, D21, D42, and day 84 (D84) (Fig. 1).
IPL group
Participants underwent IPL therapy only, with 12 uniformly spaced pulses of light to both eyes on D0, D21, and D42 by a trained physician who was not involved in data collection. During D0, D21, and D42, participants wore opaque goggles, and ultrasound gel was generously applied to their targeted area while wearing opaque goggles (Fig. 1).
Control group
Participants in the control group composed of evaporative DED patients and did not receive any dry eye treatment, including IPL or IPL with HEM.
The researcher who collected data at D0 (baseline), D21 (week 3), D42 (week 6), and D84 (week 12) was unaware of the therapeutic assignments of the patients. For the duration of this trial, participants from all the three groups were asked not to use any other dry eye medicines or eye drops, including preservative-free artificial tears.
Statistical analysis
For statistical analysis, Statistical Analysis in Social Sciences (SPSS) for MacOS software was used to analyze the data (version 25; IBM Corp.). The right eye of each participant included in this study was used for the analysis. All data are expressed as the mean ± standard deviation. Pearson's correlation coefficient was used to evaluate the relationship between ΔTFLL and other parameters. A p-value less than or equal to 0.05 was deemed statistically significant. Repeated-measures analysis permitted comparisons across time periods, whereas paired analyses permitted comparisons of pre- and post-treatment data at particular time points. Using the Kolmogorov–Smirnoff test, the normality of variables was determined. Ordinal variables (such as TFLL) and those with non-normal distributions were analyzed by analysis of variance (ANOVA). Before and after therapy, the key end measures for this study were NITBUT, TFLL, CS, and OSDI scores.
Results
The demographic information of the participants is portrayed in Table 1. The mean age of participants (IPL + HEM: 28.06 ± 3.88 years, IPL: 29.88 ± 4.68 years, and control: 28.52 ± 3.77 years) in the three groups was not significantly different (p = 0.079). The duration of their DED and other clinical DED parameters was not significantly different.
Participant Demographics
DE, dry eye; IPL, intense pulsed light; IPL+HEM, intense pulsed light and heated eye mask.
Noninvasive tear breakup time
Significant improvement in NITBUT was observed in the IPL+HEM group from 4.22 ± 0.71 sec at baseline to 9.00 ± 0.93 sec at D84 (p < 0.001), whereas gradual improvement in NITBUT was observed in the IPL group from 4.08 ± 0.59 sec at baseline to 7.62 ± 0.74 sec at D84 (p < 0.001). The control group had a significant deterioration in the NITBUT measurements (baseline: 4.12 ± 0.75 sec, D84: 3.68 ± 3.68 ± 68 sec, p < 1.001). Intergroup comparison of NITBUT found a significant difference between the three groups at D21 (p < 0.001), D42 (p < 0.001), and D84 (p < 0.001). ΔNITBUT (baseline minus D84) was found to be greatest in the IPL+HEM group (−4.79), followed by the IPL group (−3.54) and the control group (0.44) (Table 2). Furthermore, a good negative correlation was found between ΔNITBUT and ΔTFLL (ρ = −0.810, p < 0.01) (Table 3).
Intergroup and With-in Group Comparison of Dry Eye Parameters
p-Value <0.05.
D0, baseline; D21, day 21; D42, day 42; D84, day 84; Δ, baseline minus day 84; CS: corneoconjunctival staining; MGEx, meibomian gland expressibility; MGQ, meibomian gland quality; NITBUT, noninvasive tear breakup time; OSDI, Ocular Surface Disease Index; TFLL, tear film lipid layer.
Correlation with ΔTear Film Lipid Layer
n = 150 eyes.
TFLL score
The TFLL score can range from 1 to 5. A score of 1 represents a “healthy TFLL score,” and a score of 5 represents the “worst TFLL score.” The baseline TFLL score for the IPL+HEM, IPL, and control groups was 3.16 ± 0.68, 3.24 ± 0.43, and 3.10 ± 0.61, respectively (p = 0.488). At D84, the TFLL score for the IPL+HEM and IPL groups significantly improved to 1.14 ± 0.35 (p < 0.001) and 2.22 ± 0.42 (p < 0.001), respectively, whereas the control group had no significant changes at D84 (p = 0.456). ΔTFLL (baseline minus D84) for the IPL+HEM, IPL, and control groups was 2.02, 1.02, and −0.58, respectively (Table 2).
Corneoconjunctival staining score
The CS score can range from 0 to 9. A 0 score represents the “minimum staining score,” and a score of 9 represents the “maximum staining score.” The baseline CS score for the IPL+HEM, IPL, and control groups was 3.20 ± 1.16, 3.16 ± 0.84, and 3.18 ± 0.96, respectively. At D84, the CS score for the IPL+HEM, IPL, and control groups was 2.60 ± 0.73, 3.02 ± 0.74, and 3.24 ± 0.85, respectively (p < 0.001). ΔCS (baseline minus D84) was found to be greatest in the IPL+HEM group (0.60), followed by the IPL group (0.14) and the control group (−0.06) (Table 2). Furthermore, the correlation between ΔCS and ΔTFLL was found to be significant (ρ = 0.321, p < 0.01) (Table 3).
MGQ score
The MGQ score can range from 1 to 4. A score of 1 represents the “best score,” and a score of 4 represents the “worst score.” The baseline MGQ score for the IPL+HEM, IPL, and control groups was 2.54 ± 0.50, 2.62 ± 0.49, and 2.58 ± 0.50, respectively, and at D84, the scores were 1.22 ± 0.42, 1.28 ± 0.45, and 2.98 ± 0.50, respectively. ΔMGQ (baseline minus D84) was found to be greatest in the IPL+HEM group (1.32), followed by the IPL group (1.02) and the control group (−0.52) (Table 2). The correlation between ΔMGQ and ΔTFLL was found to be significant (ρ = 0.688, p < 0.01) (Table 3).
MGEx score
The MGEx score can range from 1 to 4. A score of 1 represents the “best score,” and a score of 4 represents the “worst score.” Significant improvement in MGEx score was observed in the IPL+HEM group from 2.18 ± 0.44 at baseline to 1.12 ± 0.33 at D84, while in comparison, the IPL group baseline MGEx score was 2.12 ± 0.39 at baseline and improved to 1.26 ± 0.44 to at D84 (p < 0.001), whereas the control group had no significant improvement in the MGEx score. ΔMGEx (baseline minus D84) was found to be greatest in the IPL+HEM group (1.06) (Table 2). In addition, the correlation between ΔMGEx and ΔTFLL was found to be significant (ρ = 0.675, p < 0.01) (Table 3).
OSDI score
The baseline total OSDI score for the IPL+HEM, IPL, and control groups was 34.20 ± 8.88, 32.30 ± 5.29, and 33.42 ± 9.44, respectively (p = 0.868). At D84, the scores were 16.58 ± 3.91, 22.56 ± 4.60, 36.08 ± 7.83, respectively (p < 0.001). ΔOSDI (baseline minus D84) for the IPL+HEM, IPL, and control groups was 14.34, 8.6, and −2.12, respectively (Table 2). The correlation between ΔOSDI and ΔTFLL was found to be significant (ρ = 0.649, p < 0.01) (Table 3). Furthermore, the correlation between ΔOSDI severity and ΔTFLL was found to be significant (ρ = 0.744, p < 0.01) (Table 3). The frequency of DED severity was assessed using the OSDI questionnaire, and group differences are graphically represented in Fig. 2A–D for the IPL+HEM, IPL, and control groups, respectively. The IPL+HEM group was found to have a better outcome than other groups.
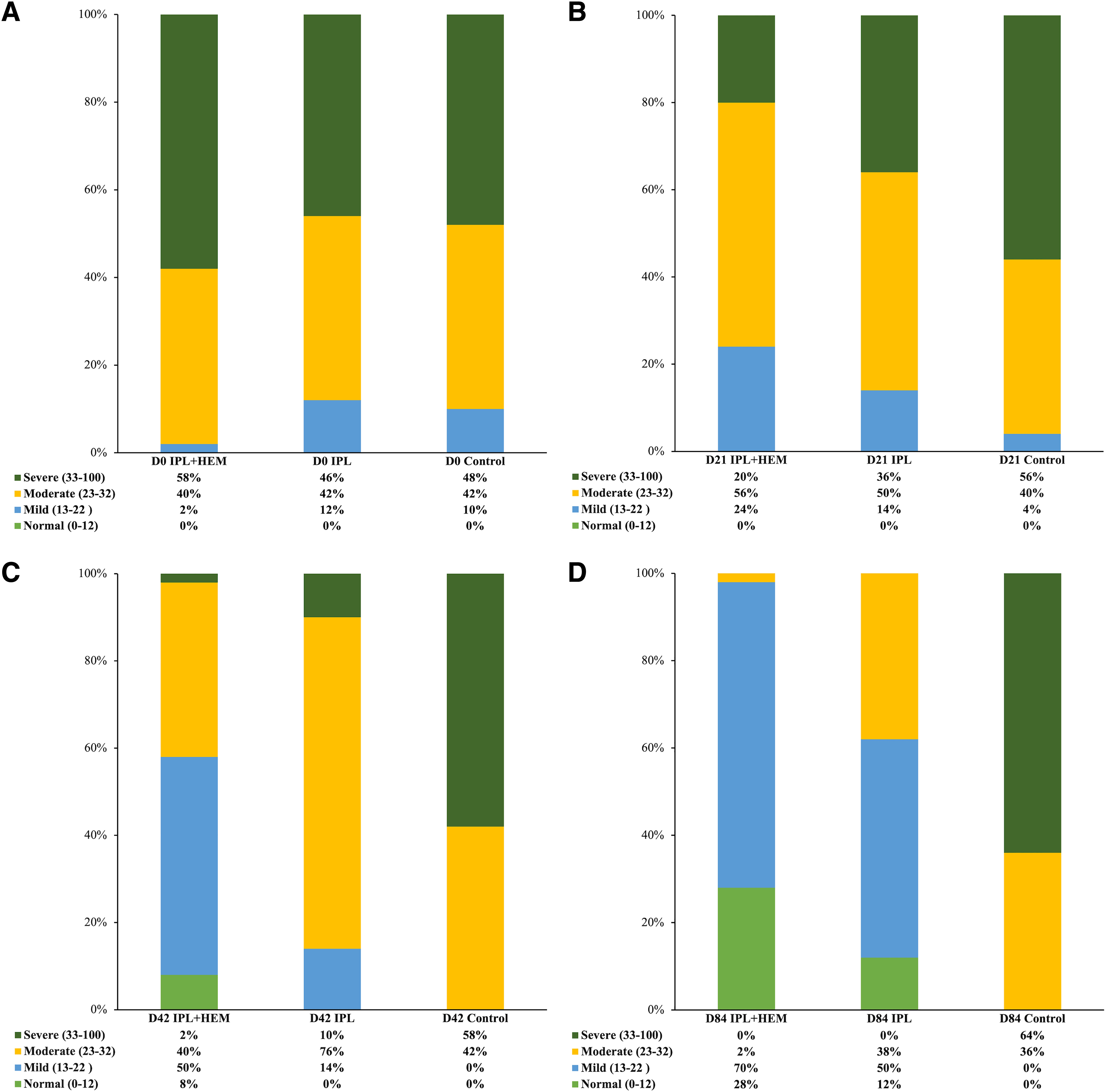
Frequency of OSDI severity among the groups.
Discussion
A deficiency of the TFLL component on the pre-corneal tear leads to exacerbated evaporation of the tears in EDEs. Therefore, in this article, we focus on the effectiveness of combined therapy (IPL+HEM) for improving TFLL in participants suffering from EDE. Our results showed that IPL+HEM therapy was significantly better in improving the TFLL on the pre-corneal tear surface (Table 2) since the change in TFLL score (D0 minus D84), denoted by ΔTFLL, was greatest in the IPL+HEM group, followed by the IPL group and the control group.
Further correlation analysis between ΔTFLL and ΔNITBUT, ΔCS, ΔMGQ, ΔMGEx, and ΔOSDI revealed that all these parameters had significant correlation. These findings are supported by the fact that the TFLL not only supports the tear film homeostasis, but reports suggest that meibum possesses antibacterial properties that maintain the health of the lid margin, 7 therefore enhancing the quality of the TFLL not only minimizes the rate of tear evaporation but also improves the ocular surface health of the individual. 48,49 Furthermore, treatment for EDE and MGD involves boosting the quality and amount of meibum excretion to alleviate symptoms. The efficacy of lipid-containing eye drops 49 –51 and castor oil emulsions 52 in simulating and repairing the TFLL of tears has been shown by research.
While some studies hypothesize that the heat energy delivered by IPL liquefies the blocked meibomian glands identified in MGD, hence reducing gland blockage and stimulating the release of meibum, this has not been demonstrated in standard IPL devices, which administer light pulses on the periorbital region around the eyelids. 17,20,33,39,53 Therefore, by adding HEM to IPL therapy, this study successfully achieved liquefaction of the meibum and subsequently improved the TFLL participants in the IPL+HEM group. 32 The findings of this study are similar to the treatment efficacy of the device LipiFlow® (TearScience, Inc., Morrisville, NC, USA), which administers heat (42.5°C) to both inner eyelid surfaces while concurrently applying pulsing pressure to the outer eyelids via an inflated air bladder. 54
The transfer of increased temperature permits unclogging of glandular obstructions as meibum is made more malleable and fluid, which can consequently be efficiently expressed. 38,39,55 As documented by Wu et al., 33 the temperature achieved by the Ocuface Medical HEM in this current study was well over 42.5°C. However, the LipiFlow device simultaneously squeezes the meibomian gland, encouraging the outflow of melted meibum, this mechanical expression was not possible in our study as the HEM is a static device. 56 Nevertheless, our study achieved significant subjective and objective improvements without peristaltic movement for meibum evacuation offered by devices, such as LipiFlow 56,57 and EyePeace®. 58
The rationale behind the combination of HEM that produces heat with IPL is based on the specific functions of each procedure. As mentioned by Farrant. 59 on the TFOS Dry Eye Workshop, describing the recommendations for the management of DED, the former is capable of reaching a therapeutic level of heat that liquefies meibum as “Step-I” (noninvasive method) for managing dry eye. However, “Step-II” (more potent) is the use of IPL [U.S. Food and Drug Administration (FDA), class II surgical device certified], which has been previously demonstrated to improve the function of meibomian glands through various mechanisms, including ablation of blood vessels, fluidification of meibum, and reduction of inflammatory cytokines. 59
Nonpharmacological therapies such as thermal pulsation and IPL technologies have exhibited their efficacy in improving EDE for a prolonged duration, and in some instances, even in one to two sessions, particularly in the case of thermal pulsation. Therefore, with further robust clinical trials, nonpharmacological therapies have the potential to facilitate health care professionals in providing appropriate counseling and therapeutic interventions with less dependence on prolonged pharmacological interventions. 60
The OSDI is a reliable and validated tool for discriminating the severity of DED 61 and found a significant decrease in OSDI score after IPL+HEM and IPL therapies, which agree with previous studies. 40,41,62 –64 Furthermore, the change in total OSDI score (D0 minus D84), denoted by ΔOSDI and ΔOSDI severity score, was greatest in the IPL+HEM group (Table 2), suggesting that the addition of HEM to IPL therapy can lead to significant improvement in patient's symptoms, possibly due to improved blood circulation and symptomatic relief of ocular fatigue.
This study may be criticized because the active control group did not include the HEM-only group. However, Xu et al. 32 have previously demonstrated that signs and symptoms of improvement following the use of HEM in dry eye patients have significantly lower in comparison to the IPL-only group. It is possible that if we include the HEM-only group, then this commitment would have stressed the recruitment for the study. That being said, it is to be noted that the therapeutic effect of IPL and HEM was not under investigation but rather the benefits of combining HEM with IPL. Nevertheless, an independent HEM group will be incorporated into future studies to compare and contrast its effects.
This research has several limitations as it did not include inflammatory indicators and osmolarity parameters, which can assess and monitor dry eye severity. A further problem is that the age range of participants was from 22 to 40 years; therefore, the results cannot be generalized. Future research will concentrate on a larger sample size, and a randomized controlled study to maximize the study's statistical power and cover a broader age range since dry eye is more frequent among the elderly.
Conclusions
In conclusion, the present study showed that IPL with and without HEM improved signs and symptoms of EDE. However, IPL combined with HEM had greater therapeutic efficacy in improving the TFLL in patients with EDE.
Footnotes
Acknowledgments
We appreciate their participation in this research. This research was funded by the He Eye Specialty Hospital in Shenyang, China. The authors have no proprietary interest in any of the products discussed.
Authors' Contributions
All named authors meet the International Committee of Medical Journal Editors (ICMJE) criteria for authorship for this article, take responsibility for the integrity of the work as a whole, and have given their approval for this version to be published. Conceptualization: L.L., S.Y., X.H., and E.E.P. Methodology: J.C., G.Q., Y.Q., Yi.C., M.L., Q.Z., Yu.C., N.G., S.M., J.E.M., S.Y., X.H., and E.E.P. Formal analysis and investigation: L.L., S.Y., X.H., and E.E.P. Writing—original draft preparation: L.L. Writing—review and editing: L.L., S.Y., X.H., and E.E.P. Funding acquisition: S.Y. and X.H. Resources: S.Y. and X.H. Supervision: S.Y., X.H., and E.E.P.
Author Disclosure Statement
No competing financial interests exist.
Funding Information
This study was entirely funded by He Eye Specialist Hospital, Shenyang, China. No support was received for the publication of this article.
