Abstract
Background:
Stage 3 grade C periodontitis (S3GCP) has always been a challenge for clinicians. However, it is proposed that the use of lasers in addition to periodontal therapy can result in a more efficient therapy outcome.
Objective:
The aim of this clinical study was to determine the effects of additional application of diode laser (DL, 810 nm ±5) on clinical and microbiological values during Modified Widman Flap (MWF) periodontal surgery in the S3GCP patients.
Methods:
A total of 18 patients were randomly assigned to the test site (MWF + activeDL) and the control site (MFW alone). Clinical parameters and microbial samples were taken preoperatively, and postoperatively at 6 weeks, 3 months, and 6 months. Visual analog scale (VAS), tissue edema (TE), tissue color (TC), and pain medication (PM) consumption, were evaluated postoperatively at 10th day.
Results:
All bacteria were significantly decreased at follow-up times compared with preoperative amounts in both therapy sites (p < 0.05). Bacterial amounts of Treponema denticola, Prevotella intermedia, Tannerella forsythia, and Aggregatibacter actinomycetemcomitans at 6 weeks, Porphyromonas gingivalis and T. denticola at 3 months, T. denticola and A. actinomycetemcomitans at 6 months were statistically lower in the test site than the control site. All clinical parameters significantly improved after MWF surgery at follow-up times compared with preoperative values in intragroup comparisons, but a significant difference was not detected in the intergroup comparison. No differences were found in terms of VAS, PM, TE, and only TC was statistically more pink in the test site than the control site.
Conclusions:
The present study findings suggest that the use of DL together with MWF may have positive effects in the therapy of S3GCP patients by reducing the microbial load. Clinical
Introduction
Stage 3 grade C periodontitis 1 (S3GCP, formerly classified; aggressive periodontitis) is a disease characterized by rapid and severe damage of periodontal tissues that generally affects adolescents and young adults who have a familial predisposition. 2 Gram-negative anaerobes are usually predominant in this disease, such as Porphyromonas gingivalis, Tannerella forsythia, Fusobacterium nucleatum, and especially in localized form, Aggregatibacter actinomycetemcomitans. 3 Treatment of this disease depends on early diagnosis, elimination, and suppression of infected pathogens, and the creation of a suitable environment for long-term maintenance. The main treatment methods of periodontal diseases involve oral hygiene education, cleaning of teeth surfaces, scaling and root planning (SRP), and periodontal surgery. 4 SRP is considered as gold standard therapy for the treatment of periodontitis diseases, including S3GCP. 5
Although subgingival microbial prevalence and levels are significantly reduced by mechanical therapy, periodontopathogens, such as A. actinomycetemcomitans and P. gingivalis can penetrate deep into periodontal tissues by crossing the epithelial barrier and these pathogens may be difficult to remove by nonsurgical periodontal therapy (NSPT) alone. 6 Therefore, local and systemic antibiotics were considered necessary to eliminate pathogenic bacteria from tissues. On the other hand, antimicrobial treatment can cause side effects and the development of bacterial resistance. 7
The treatment of deep pockets with surgical periodontal treatment (SPT) is more advantageous than NSPT as it provides better accessibility with visual instrumentation. However, if pathogens, epithelial remnants, and diseased granulation tissues are not completely removed from the soft tissue wall of the periodontal pocket, it prevents gingival connective tissue attachment to root surface. 8 For these reasons, in the field of periodontology, laser technology is gaining popularity as it offers potential benefits in addition to SPT. 9 It has proved that subgingival calculus was removed efficiently by Er:YAG laser, 10 CO2 laser for root conditioning, 11 and removal of pocket epithelium and control of bacteremia were achieved by ND:YAG 12 and diode laser (DL). 13
Previous literatures 8,14 –16 have demonstrated the impressive safety profile and miscellaneous therapeutic uses of DL, which has wavelength between 808 and 980 nm, in periodontal therapy. There are convincing evidences about: (1) The bactericidal effect of DL on periodontopathogens invading the soft tissue wall of periodontal pockets; 15 (2) The adjunctive role of DL in de-epithelializing the underlining of the mucoperiosteal flap and also facilitating significant bacterial reduction; 8,11 and (3) In soft tissue surgery using laser, better hemostasis, minimal postoperative pain, and sterilization are provided compared with conventional SPT. 17 On the other hand, Chitsazi et al. 18 and Ertugrul et al. 19 did not show additional benefits of the DL application during NSPT in S3GCP patients. Some studies reported positive results both clinically and microbiologically in S3GCP patients. 6,14,16 However, these studies include DL applications during NSPT in S3GCP. To the extent that we could search the literature, we did not find any study reporting the use of DL in addition to SPT in patients with S3GCP.
Treatment of S3GCP has always been a challenge for clinicians, 18 so the authors stated that the additional application of DL by providing direct vision with SPT in these patients group may reduce the number of periodontal pathogens invading soft tissue wall of periodontal pockets. Thus, it was thought that the use of additional antimicrobial agents could be reduced, and side effects, such as bacterial resistance, could be prevented. Therefore, the aim of this study was to evaluate the effects of DL application during the modified Widman flap (MWF) surgery in patients with S3GCP by identifying the changes on clinical parameters, pathogen bacterial amount (A. actinomycetemcomitans, Treponema denticola, T. forsythia, P. gingivalis, Campylobacter rectus, Prevotella intermedia), in the test (active DL+MWF) and the control (MWF alone) sites during the follow-up periods. In addition to that, SPT is often associated with pain and disturbance regardless of the treatment modality. 20 Therefore, we evaluated the postoperative discomfort and tissue response following the SPT in therapy sites.
Materials and Methods
Study design
The subjects of this study participated from the Zonguldak Bülent Ecevit University, Faculty of Dentistry, Department of Periodontology, Zonguldak, Turkey, between January 2013 and February 2016. The protocol of this randomized control split-mouth study was approved by the Clinical Research Ethics Committee, Zonguldak Bülent Ecevit University, Faculty of Medicine, Zonguldak, Turkey, in line with the Helsinki Declaration of 1975, as revisited in 2000 (protocol ID: 2012-125-30/10). Before starting the study, the nature, potential risks, benefits, and objective of the research were identified to each patient and their signed informed consent form was taken from everyone.
Study population
The status of periodontal disease was diagnosed by the periodontologist (S.B.D.) at the start of the study in line with the clinical and radiographic guidelines in classification 1999. 2 However, as the periodontal disease classification has been revised, the study groups have been reclassified in accordance with the 2017 World Workshop Consensus report on the Classification of Periodontal and Peri-Implant Diseases and Conditions. 1 S3GCP patients with at least six teeth with interdental Probing depth (PD) ≥6 mm and Clinical attachment level (CAL) ≥5, and at least three of these six teeth without first molars and incisors were included in the study. Severe destruction of periodontal support tissue was seen in these patients, which were not compatible with age and plaque levels. Radiographic bone loss/age was used for determination of patients' periodontitis grade. 21 If the bone loss/age% were >1.0, it was recognized as Grade C.
The tooth with the most severe bone loss as a percentage of root length was determined for radiographic bone loss. Systemically healthy patients who have at least 20 teeth were enrolled to this study. A total of 30 participants, who were diagnosed with S3GCP after clinical examination, were included in this study. NSPT phase was performed to these patients. Six weeks after NSPT, clinical periodontal status was reassessed to identify periodontal surgery requirements. A total of 12 people were excluded, 8 of whom did not meet the inclusion criteria, and 4 who did not want to continue study (Fig. 1). Therefore, this study was conducted with 18 S3GCP (mean age: 29.61 ± 3.79 years, 22–35 years of age, gender: 8 females, 10 males) with PD ≥6 mm and CAL ≥5 mm in at least three teeth in both quadrants of the maxilla.
Consort flow diagram.
Study exclusion criteria were as follows: (1) systemic conditions that may affect the development of periodontal disease, (2) smoker (former or current), (3) NSPT during the 6 months preceding the study, (4) prescription of nonsteroidal anti-inflammatory or antibiotics up to 6 months before the study, (5) SPT within 12 months prior the study, (6) postmenopause, pregnancy, and lactation.
Clinical evaluation
All clinical parameters and evaluation of parameters summarized in Table 1. All clinical parameters were evaluated at the follow-up times, and tissue response after removing of sutures postoperatively at 10th day were evaluated by calibrated same investigator (F.Ö.D.), who did not know which treatment was applied to which site.
Clinical Parameters and Evaluation of Parameters
TC, tissue color; TE, tissue edema; VAS, visual analog scale.
Before starting the study, intraexaminer calibration was performed. Ten randomly selected subjects were used to standardize the researcher. The researcher evaluated all clinical parameters on two occasions 48 h apart. The standardization of the researcher was validated, if two measurements show >90% similarity on the millimeter level. 22
All patients were offered instructions relating to daily plaque control. Depending on the condition of periodontitis patients, NSPT was accomplished by the same investigator (S.B.D.) within 2 weeks, with each appointment lasting ∼45–60 min. Subgingival instrumentation (SRP) was applied by using manual scalers, curettes (Hu-Friedy, Chicago, IL), and ultrasonic instruments (Cavitron select; DENTSPLY, York, PA) on all four quadrants of patients. The maintenance program with repetitive oral hygiene training was applied. Clinical data were recorded again from therapy sites after 6 weeks of SRP.
Microbiological sampling and analysis
Samples were taken the day after clinical evaluation, as blood contamination could occur in probing areas. Before sampling around the teeth, supragingival plaque was carefully removed with a sterile gauze pad and each selected site was dried and isolated with cotton rolls to prevent saliva contamination. The samples were taken from the deepest periodontal pocket of teeth (PD ≥6) in the therapy sites of each patient using sterile paper points (*30) at preoperative and at postoperative 6 weeks, 3 months, and 6 months. Samples were collected from the same teeth throughout the study. The paper point was carefully positioned inside the depth of the pocket from the apical aspect of the pockets and then allowed to be kept in place for 30 sec. 8 Samples isolated with blood and saliva were not included and samples were taken from the relevant site again the next day. The samples taken were placed into sterile Eppendorf tubes and frozen at −80°C until the analysis day.
For DNA isolation, a commercial kit, QIAGEN, the QIAamp DNA Mini Kit (Qiagen Sciences, MD) was used in accordance with the manufacturer's guidelines. Quantity of DNA obtained after isolation were spectroscopically measured by using Qbit (Qiagen) spectroscopically and recorded to confirm the existence of the DNA in the samples. Molecular detection and qualification of the bacteria were determined by using primers (TaqMan), and probes marked 3′-FAM and 5′-TAMRA dyes. The sequences of the primers are given in Table 2.
Bacterial Species, Primer Sequences, and Thermal Cycling
The real-time polymerase chain reaction (PCR) was performed as six individual monoplex reactions with replaceable primer and probe targets (and probe dyes; 5′-FAM- 3′-TAMRA) depending on the target gene combination required [master mix details: Quantitative PCR was performed by using deionized water, MgCl2 (25 mM), enzyme (Tag polymerase)+buffer mixture, 0.5 μM primers (forward–reverse), 0.2 μM probes, and 20 μL PCR mixture and DNA, which was prepared before]. For real-time PCR processes, Rotorgene 6000 (Qiagen, Germany) device was used. Subsequently, the replicated DNA samples were placed in 1.5% agarose gel prepared with 1 × Tris-Asetat-ethylenediaminetetraacetic acid (EDTA) buffer and displayed with a transilluminator using the electrophoresis gel imaging device (Syngene).
Periodontal surgical therapy
All nonsurgical, surgical, and laser therapies were applied by the same expert periodontist (S.B.D.) who was different from an examiner who recorded clinical parameters. Before starting the surgery, treatment sites were randomly divided (by flipping a coin) into test and control sites. The control site was treated with MWF by applying sham DL and the test site was treated with MWF by applying active DL (810 nm ±5; Picasso-AMD) to the inside of the MWF. The MWF technique was applied. 23 Following the application of MWF, hand tools, ultrasound, and rotary instruments were used to eliminate granulation tissue. Additionally, root surfaces were scaled and planned with curettes in both therapy sites. The osseous recontouring and mucogingival surgery were not needed in any patients. A 810 nm DL unit, 400 μm disposable fiberoptic tip at 1 W in continuous wave mode (power density 796 W/cm2) was used to lase the inner surface of the flap from the free gingival margin to the apical aspect in both the labial and palatal test sites. The optical fiber tip was first initialized by activating emmission over a sheet of black paper.
To prevent the alveolar bone and root surface from contacting any laser, DL irradiation was applied to the soft tissue flap in parallel paths for 10 sec at a 45° angle. If irradiation time exceeds 10 sec, the laser emission was cutoff for 30 sec. The resulting layer was completely removed with moist gauze before the flap was replaced. A second laser was applied to all surfaces of the flap with the same laser (810 nm ±5) at 0.1 W in continuous mode. All surfaces of the flap (internal and external), exposed bone, and operatively exposed root structures were irradiated at a total dose of 4 J/cm2 per surface. DL application was simulated without pressing the start button in the control site. The MWF was sutured with 3–0 black silk suture (Orhan Boz AS, Ankara, Turkey) in all patients (Fig. 2).
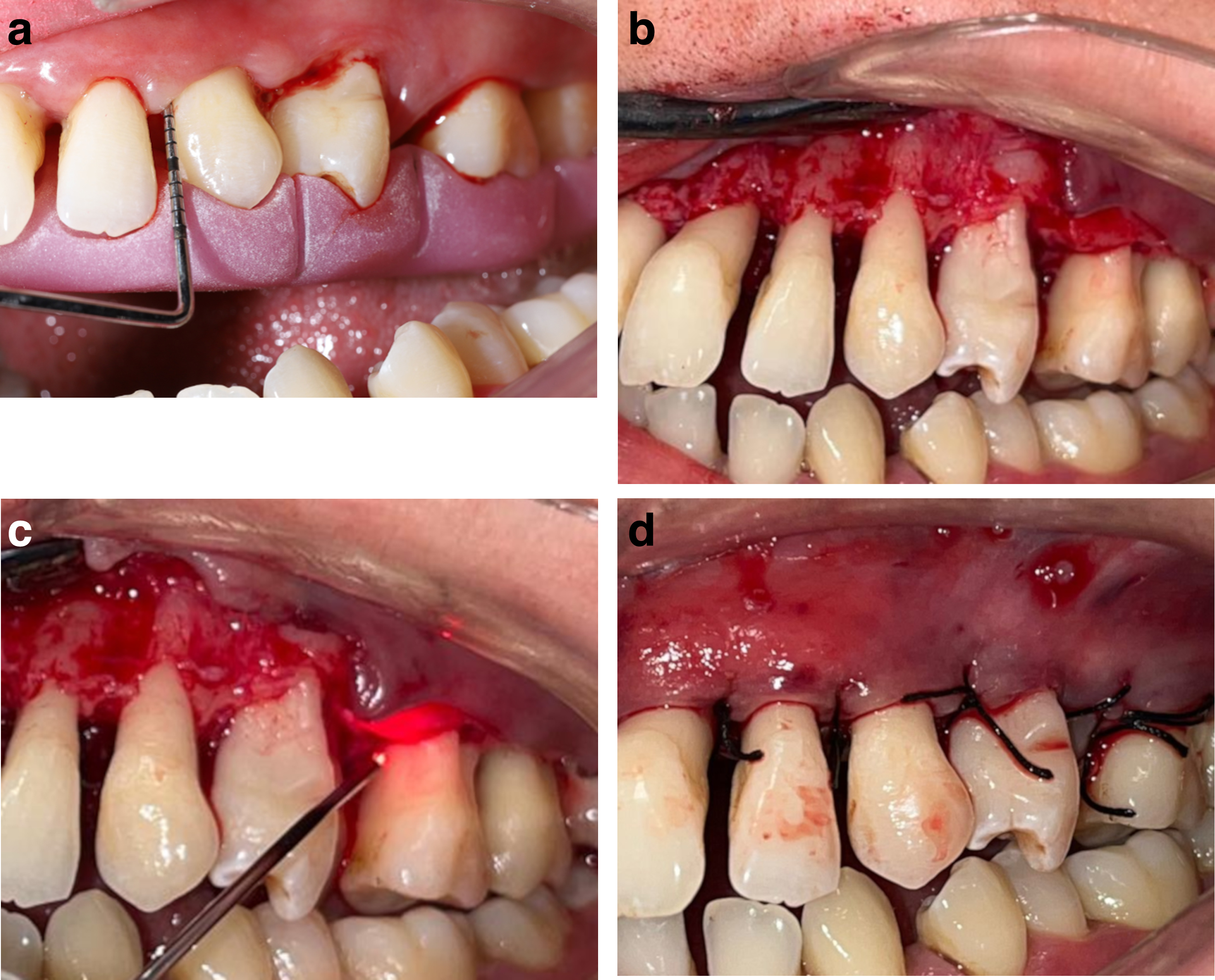
The patients were prescribed 200 mg of ibuprofen and were told they could take it every 8 h if they had pain. They were told that they should not eat hard foods and brush the teeth where surgery was applied. The patients were told that they needed to gargle with chlorhexidine digluconate [0.2%, chlorhexidine digluconate (CHX)] for 1 min twice a day for 10 days after the surgery. The sutures were removed on day 10. After the sutures were removed, the patients began to brush their teeth and oral hygiene training was repeated at each control session.
Statistical analyses
It was determined whether the data showed normal distribution or not using the Shapiro–Wilk test. Comparative analyzes of clinical and microbiological parameters were performed using the Mann–Whitney U nonparametric test after it was determined that the data did not show normal distribution. The Friedman test followed by the Bonferroni-adjusted Wilcoxon signed-rank test was used to compare the differences in parameters at different time points. The bleeding on probing (BOP) percentage was compared using χ 2 analysis. The results were calculated as median with min–max values for every clinical and microbiologic parameters at preoperative, 6-week, 3-month, and 6-month intervals. The statistical software (SPSS, Inc., version 19.0, Chicago, IL) was applied for all tests. A p < 0.05 was considered to be statistically significant.
The resulting variations in PD levels were used to determine the power analysis of the study. However, sample calculation could not be made before the study due to the lack of precise information. Therefore, we calculated based on the pilot study, in which 15 patients participated in each group. We estimated that a sample size of 18 patients in each group would allow for a type II error level of β = 0.20 (80% power) and a type I error level of α = 0.05 (5% probability). A retrospective power was calculated as no sample size calculations could be made beforehand. A power of 99% was found in the posterior power calculation.
Results
Postoperative tissue response results
No side effects were observed in any of the patients due to the treatment. Tissue color (TC) was 2.00 in the control sites and 1.00 in the the test site. There was a statistical difference between the test and the control site in the terms of TC (p = 0.019). The test site recorded more pink than the control site. Tissue edema (TE) was 2.00 and 1.50 in the control and the test site, respectively. TE did not show difference between both therapy sites (p > 0.05) (Table 3).
Descriptive Statistics of the Postoperative Pain and Tissue Response at Control and Test Sites After Diode Laser Application During Periodontal Surgery
Data are expressed as median (range; minimum–maximum).
Significant difference between groups p < 0.05. (Mann–Whitney U nonparametric test).
Postoperative pain discomfort results
The visual analog scale (VAS) was 2.05 and 2.15 in the control and the test sites, respectively. The difference was not significant between the therapy sites (p > 0.05). The pain medication (PM) taken after surgery due to discomfort in the control site was 0.35 and 0.60 in the test site. The consumption of PM did not differ between the therapy sites (p > 0.05) (Table 3).
Clinical results
Clinical results are summarized in Table 4. When the preoperative values were compared with follow-up times, a significant reduction was found in the median values of all clinical parameters (p < 0.05). PD, CAL, BOP, plaque index (PI), and Gingival index (GI) parameters did not show differences in the intragroup and intergroup comparison postoperatively at 6 weeks, 3 months, and 6 months (p > 0.05).
Clinical Variables in Test and Control Sites at Study Time Points and Results of Intra- and Intergroup Comparisons
Data are expressed as median (range; minimum–maximum).
Microbiological results
All microbiological findings are demonstrated in Table 5. The intergroup comparison showed that preoperative amounts of T. forsythia, P. intermedia, T. denticola, P. gingivalis, C. rectus, and A. actinomycetemcomitans did not differ in both therapy sites (p > 0.05). After 6 weeks postoperatively, amounts of T. denticola, T. forsythia, P. intermedia, and A. actinomycetemcomitans were significantly less in the test site than the control site (p < 0.05). The amounts of T. denticola and P. gingivalis were statistically lower in the test site than the control site at 3 months (p < 0.05). Additionally, the amount of T. denticola and A. actinomycetemcomitans was statistically less in the test site than in control site at 6 months (p < 0.05). No differences were found in the amount of C. rectus between the two therapy sites at any time points (p > 0.05).
Total Amount of Periodontal Pathogens in the Samples of the Control Site and Test Site at the Study Time Points and Intra- and Intergroup Comparisons
Data in bold indicate statistically significant.
Data are expressed as the median (min–max). p < 0.05 was considered to be statistically significant. Mann–Whitney U test for intergroup comparisons.
Statistically significant difference from preoperative (T0) (Friedman test/the Bonferroni-adjusted Wilcoxon signed-rank test).
Statistically significant difference from 6 weeks (T1) (Friedman test/the Bonferroni-adjusted Wilcoxon signed-rank test).
Statistically significant difference from 3 months (T3) (Friedman test/the Bonferroni-adjusted Wilcoxon signed-rank test).
T0, preoperative; T1, 6th week; T3, 3rd month; T6, 6th month.
The intragroup comparisons showed that amounts of all bacteria were significantly decreased at follow-up times compared with preoperative values in both therapy sites (p < 0.05). The amount of P. gingivalis statistically decreased from 6 weeks to 3 months and from 3 to 6 months in the control site (p < 0.05). P. gingivalis amount decreased at follow-up times in the test site, but no difference was found between at 3 months and at 6 months. The T. denticola amounts were statistically increased from 6 weeks to 3 months and from 3 to 6 months in both therapy sites (p < 0.05). There were no differences in the amount of C. rectus at postoperative follow-up times in the control site (p > 0.05). In the test site, the amounts of C. rectus statistically increased from 6 weeks to 3 months, but its amounts were statistically lower at 6 months than at 3 months (p < 0.05). The P. intermedia amount was statistically increased at 3 months compared with 6 weeks, and statistically decreased at 6 months compared with 3 weeks in both therapy sites (p < 0.05). No differences were reported in the amounts of T. forsythia and A. actinomycetemcomitans at any time points in both therapy sites (p > 0.05).
Correlations
Correlation coefficients are presented in Table 6. When all therapy sites were examined, no significant correlation was found between PI and all bacteria at any time points (p > 0.05).
Sperman's Rank Correlation (r) Among Time Periods with Respect to Plaque Index, Porphyromonas gingivalis, Treponema denticola, Camplyobacter rectus, Prevotella intermedia, Tannerella forsythia, and Aggregatibacter actinomycetemcomitans in All Subjects
No statistically significant difference was reported (p > 0.05).
PI, plaque index.
Discussion
DL application at wavelengths between 800 and 980 nm is properly absorbed by the pigmented tissue, specifically targeting pigmented bacteria and inflamed tissue. 24 Thus, DL helps to completely remove epithelial remnants as well as tissue-invasive perio-pathogens that cannot be removed by the MWF alone. 25,26 To date, there are few investigations on the adjunctive use of DL during SPT in patients with chronic periodontitis, 8,24,26 however, no study has been found in the S3GCP patients. Hence, this study was designed to evaluate the adjunctive use of DL during the SPT in S3GCP patients.
The PI was measured to evaluate the oral hygiene status of the subjects, and the patient's gingival health condition was evaluated by GI. In this study, PI, GI, and BOP showed a significant reduction in both therapy sites at follow-up times compared with preoperative values. However, no difference was recorded between the two therapy sites at any time points. Similarly, Agarwal et al. 8 reported no difference in terms of PI, GI, and BOP in both groups after 6 months. These results showed that there is no additional effect of DL on PI and GI. On the other hand, Lobo and Pol 24 in terms of GI and Karthikeyan et al. 9 in PI and BOP scores demonstrated a significantly greater reduction in the laser-treated group compared with the control group. The important point to know is that it is regular patient education and repeated motivations that are important for maintaining the patient's acquired oral hygiene. 26 Additionally, the reduction in GI can result from the reduction in gingival inflammation seen with effective removal of calculus and diseased granulation tissue. 26
“CAL” being the “gold standard” form assesing the success of periodontal treatment and PD of the pocket is a major factor influencing the long-term stability of the results. 24
Matarese et al. 14 and Kamma et al. 16 reported that SRP+DL therapy produced a significant decrease than SRP alone in the terms of PD and CAL values with reduction of pathogen bacteria in the S3GCP patients. Some studies 8,9,26 demonstrated significantly lower PD and CAL values with reduction of bacteria in the DL+SPT group than in the SPT-alone group in chronic periodontitis patients. Contrary to these results, 14,16 Chitsazi et al. 18 reported no differences in terms of clinical parameters and bacterial colony-forming units (CFUs) in S3GCP patients. On the other hand, other studies showed no differences between the DL+flap surgery and flap surgery alone in terms of CAL and PD. 24,27 Gokhale et al. 28 did not find a difference in the clinical parameters, but there was a statistically significant reduction in CFU of obligate anaerobes in the laser group as compared with the control group. Similarly, in the present study, PD, CAL, and bacterial amounts showed a significant decrease at follow-up periods compared with their preoperative values in both therapy sites, but no difference was reported in terms of PD and CAL between the intergroup comparison.
However, amounts of T. denticola, P. intermedia, T. forsythia, A. actinomycetemcomitans at 6 weeks; P. gingivalis and T. denticola at 3 months; and T. denticola and A. actinomycetemcomitans at 6 months were significantly lower in the test site than the control site. Although the clinical parameters were similar in the two therapy sites, this positive effect may be due to the ability of laser irradiation to destroy bacteria in periodontal pockets. Also, the lower bacterial levels in the test site can be attributed to laser destruction of critical virulence factors found in bacteria. 9 However, the present study found that some periodontal pathogens increased in the follow-up time, amounts of T. denticola and P. intermedia in both therapy sites, and C. rectus in the test site statistically increased from 6 weeks to 3 months. On the other hand, the amount of C. rectus was statistically lower at 6 months than at 3 months in the test site.
However, no correlations were reported between the PI and pathogen bacteria at any time points. Since the bacteria can invade the periodontal soft tissues, these bacteria, which have invaded the connective tissue, act as a reservoir for the proliferation and recolonization of the subgingival plaque content. 6 In addition, the important factor that should not be forgotten is the oral hygiene status of individuals.
There are several randomized controlled clinical trials in humans 27,29,30 analyzing the periodontal healing response using low-level laser therapy (LLLT). While some studies 29,30 had reported that LLLT with DL significantly promoted healing of periodontal tissues following surgical procedures, Jonnalagadda et al. 27 demonstrated that DL did not improve healing. In the present study, TC was recorded as more pink in the test site than in the control site. Although differences in the TC were statistically significant, this may not be clinically significant, and no difference was recorded in terms of TE between the therapy sites. These differences in studies may be due to different types of lasers, variation in application intervals, surgical procedures, and different methods used to evaluate wound healing.
The patient's discomfort or pain perception was assessed by using VAS in the first week, postoperatively. VAS is subjective and highly dependent upon individual experience. 29 Agarwal et al. 8 found that the VAS score was better in the DL+MWF group than in the MWF alone group. Additionally, similar results were reported in studies. 29,31 In disagreement with these studies, some studies 24,27 found that patients experienced more pain in the laser group than in the control group. On the other hand, Heidari et al. 32 reported no significant differences in the VAS values between the laser and the control group after surgery. Similar to Heidari's study, 32 the present study did not find a statistically significant difference in the pain scale and consumption of PM between the therapy sites after SPT. An individual response of the subjects may also be a possible reason for these results. However, it seems that the use of DL when applied during the MWF surgery did not have any harmful effect, did not cause postoperative complications, or delayed healing in this study.
This study includes some limitations, such as small sample size, lack of histological examination, and short follow-up time. Additionally, the treatment of one site of the mouth may affect other sites, especially when microbiological evaluations are required in the split-mouth study.
Conclusions
The current study showed that the use of DL in ajunct to MFW surgery did not provide an advantage in terms of clinical parameters, patient discomfort, and TE, but has an improved effect on TC. Also, DL has been found to be more effective at reducing some specific bacteria harmful to periodontal tissues. Briefly, in view of the good microbiological results, DL application during SPT can be used in addition to SRP in the mangement of S3GCP.
However, studies evaluating the long-term effects of DL as an adjunct to SPT in patients with S3GCP patients are needed.
Footnotes
Authors' Contributions
Ş.B.D. contributed the design, conception, project administration, analysis, data acquisition, interpretation, writing of orginal draft, and critically revised the article; G.A. contributed to interpretation, formal analysis, and data analysis.
Acknowledgments
Authors are grateful to Figen Öngöz Dede, PhD, DDS, for her clinical support, and to Umut Ballı, PhD, DDS, and Fürüzan Köktürk, MD, PhD, for their contributions in statistical analysis.
Author Disclosure Statement
No competing financial interests exist.
Funding Information
This research was financially supported by the Zonguldak Bülent Ecevit University Research Grant (2012-21-008).
