Abstract
Objective:
We evaluated the safety and efficacy of an augmented BroadBand Light (BBL™) protocol on the upper and lower eyelids in improving meibomian gland dysfunction (MGD) and/or dry eye disease (DED).
Background:
DED, often associated with MGD, can cause significant morbidity and accounts for 3.54 billion U.S. dollars of health care spending yearly. Intense pulsed light (IPL) has been used to treat MGD DED with some success. BBL therapy, a high-quality IPL machine, shows much promise for decreasing inflammation and redness in rosacea, as well as hyperpigmentation from sun damage.
Methods:
A retrospective medical chart review was performed for MGD DED and/or hyperpigmentation patients who received BBL therapy between January 1, 2015, and February 28, 2020. Inclusion criteria included patients who underwent at least one BBL treatment. Each treatment involved the upper and lower eyelids, as well as cheeks, nose, and face. Each MGD DED subject completed the Ocular Surface Disease Index (OSDI) and underwent pre- and post-treatment standard clinical examinations.
Results:
Forty-seven patients had treatment without significant adverse effects; all patients with MGD DED reported improvement in their dry eye or blepharitis. BBL was determined to be a safe and effective treatment. There were no changes in visual acuity (p = 0.555) and OSDI scores were improved (p = 0.016). There was one case each of mild corneal/conjunctival abrasion, temporary hyperpigmentation, and two of temporary eyelash thinning. Patients with MGD also showed significant improvement in blepharitis and reduced hordeolum frequency after BBL treatment.
Conclusions:
This novel IPL/BBL protocol appears safe and effective for treating dry eye and blepharitis.
Introduction
The prevalence of dry eye in large studies is estimated at 7–33% of the population, with risk factors including but not limited to age, female gender, extreme hot or cold weather, low relative humidity, exposure to video display terminals, history of refractive surgery, contact lens wear, smoking, and medications. 1 Our current understanding of the pathogenesis of these conditions, however, remains poor.
Dry eye disease (DED) is most commonly associated with meibomian gland dysfunction (MGD) and can cause significant morbidity for millions of people, accounting for 3.54 billion U.S. dollars of health care spending. 2,3 Current treatments range from warm compresses and artificial tears to eyelid wipes, meibomian gland expression, systemic treatment, and specialized scleral contact lenses. 4 Unfortunately, they may be limited in efficacy or inconvenient. Treatment has remained largely unchanged for several decades. The meibomian glands are exocrine glands that secrete meibum, the lipid component that stabilizes the ocular tear film. Reduced or thickened meibum quality can lead to clogged and atrophic glands and ultimately MGD and DED.
Intense pulsed light (IPL) has been used to treat DED with some success. 5 –10 This is likely because the heat distribution on the eyelids softens abnormal gland secretions, kills harmful Demodex mites, and reduces inflammatory markers interleukin (IL) 6 and 17 on the ocular surface. 9,11 –13 BroadBand Light (BBL™) therapy, a high-quality IPL device, shows much promise for decreasing inflammation and redness in rosacea, selective targeting of Propionibacterium acnes in acne, as well as reducing hyperpigmentation from sun damage. 14 IPL therapy on the lower eyelids has been shown to significantly improve tear breakup time and patient-perceived dry eye, and has proven to be safe and even more efficacious when performed on both the upper and lower eyelids. 9,10 We sought to explore the safety and efficacy of a novel augmented protocol on the upper and lower eyelids in improving MGD DED.
Methods
This study adhered to the tenets of the Declaration of Helsinki and was approved by the Institutional Review Board of the University of Southern California.
A retrospective medical chart review was performed for MGD DED and/or hyperpigmentation patients who received BBL therapy using the Sciton BroadBand Light™ machine (Palo Alto, CA) between January 1, 2015, and February 28, 2020. Inclusion criteria included adult patients older than the age of 18, who (1) had undergone one to four BBL treatments, typically 1 month apart, (2) had evidence of DED, MGD, 15 ocular rosacea, or hyperpigmentation, and (3) Fitzpatrick skin type 1–5. DED was defined by the Tear Film and Ocular Surface Society Dry Eye Workshop II criteria. 16 MGD was defined as obstructive findings of the gland orifices with impaired meibum expression by slit lamp biomicroscopy, and ocular rosacea was defined by ocular discomfort with findings of telangiectasias near the eyelid margin. We looked at patient characteristics, including age and gender, and characterized patients into the following groups: combined MGD and DED, DED predominant, and MGD predominant.
Patients were continued on their existing dry eye treatments, such as artificial tears, cyclosporine, autologous serum tears, or Prosthetic Replacement of the Ocular Surface Ecosystem (PROSE) lenses, throughout the study to evaluate this treatment as an addition to their baseline regimen. All patients who underwent BBL, and who were given the pre- and postevaluations and surveys, were analyzed and accounted for to reduce selection bias.
Treatment settings
The treatments were performed with the Sciton BBL device. For each BBL treatment, the settings were generally set similarly to the safe start settings for rosacea (Tables 1 and 2), but effectively lowered by a fluence of 1–2 J/cm2 with use of the adaptor if there was no coupling ultrasound gel between crystals. Gel coupling is recommended, however, by placing clear ultrasound gel on top of the rectangular crystal beneath the adaptor. This maximizes the cooling of the sapphire crystals, and thus Table 2 settings below are now preferred and, likely, safer (Table 1). These settings are recommended based upon patient skin type, but if patients had recent sun exposure, or were on sun sensitizing medications such as doxycycline, the treatment was either deferred or settings would be further reduced. A few patients were treated at settings higher than that described in Tables 1 and 2, for example, if they had hordeola, but this is not recommended in patients with dark eyelashes as described below.
If No Gel Coupling Between Rectangular Crystal and Adaptor
Cumulative dose is over 5 min for a treatment area of upper and lower eyelids (∼20 cm2).
With Gel Coupling Between Rectangular Crystal and Adaptor
Cumulative dose is over 5 min for a treatment area of upper and lower eyelids (∼20 cm2).
The patient's face was usually topically anesthetized 30–45 min before the procedure. The facial anesthetic cream was completely removed before treatment since it could turn white when combined with ultrasound gel, limiting the reach of the light therapy. Proparacaine eye drops were then placed in each eye, and gold-plated eye shields with stems were placed on the ocular surface under the eyelids (Fig. 1D). The eyelids were then anesthetized with clear lidocaine hydrochloride ophthalmic gel 3.5%, which was left in place during the treatment. The face, covered generously with clear ultrasound gel, was first treated with the rectangular sapphire crystal (15 × 45 mm) across the cheeks and nose (and for many also the forehead and lower face) with two passes and ∼20% overlap. The 7 mm circular adaptor was then placed on top of the rectangular sapphire crystal, and the upper and lower eyelids were treated (Figs. 1A–D and 2) with ∼20% overlap 2–4 mm away from the lash line for one row and then a second row above that. After two patients with dark lashes were noted by a photograph to have temporary lash thinning (no patient complaint), patients with dark lashes were preferentially treated starting 4 mm away from the lash line, and gel was removed from the eyelashes to prevent coupling of the light therapy onto the lashes. An opaque tongue depressor paddle was also used to block the eyelashes when applying the treatment. This was completed on the right side, then left side, then repeated for a second pass. The settings were increased up to 15 J/cm2 (no longer recommended, see Discussion section), with an adaptor in place, for a few patients with lower Fitzpatrick skin type who had hordeola. Two to three extra shots were placed over sites with hordeola. Each treatment lasted ∼20 min. Those with MGD DED had their meibomian glands expressed at the end of the session.
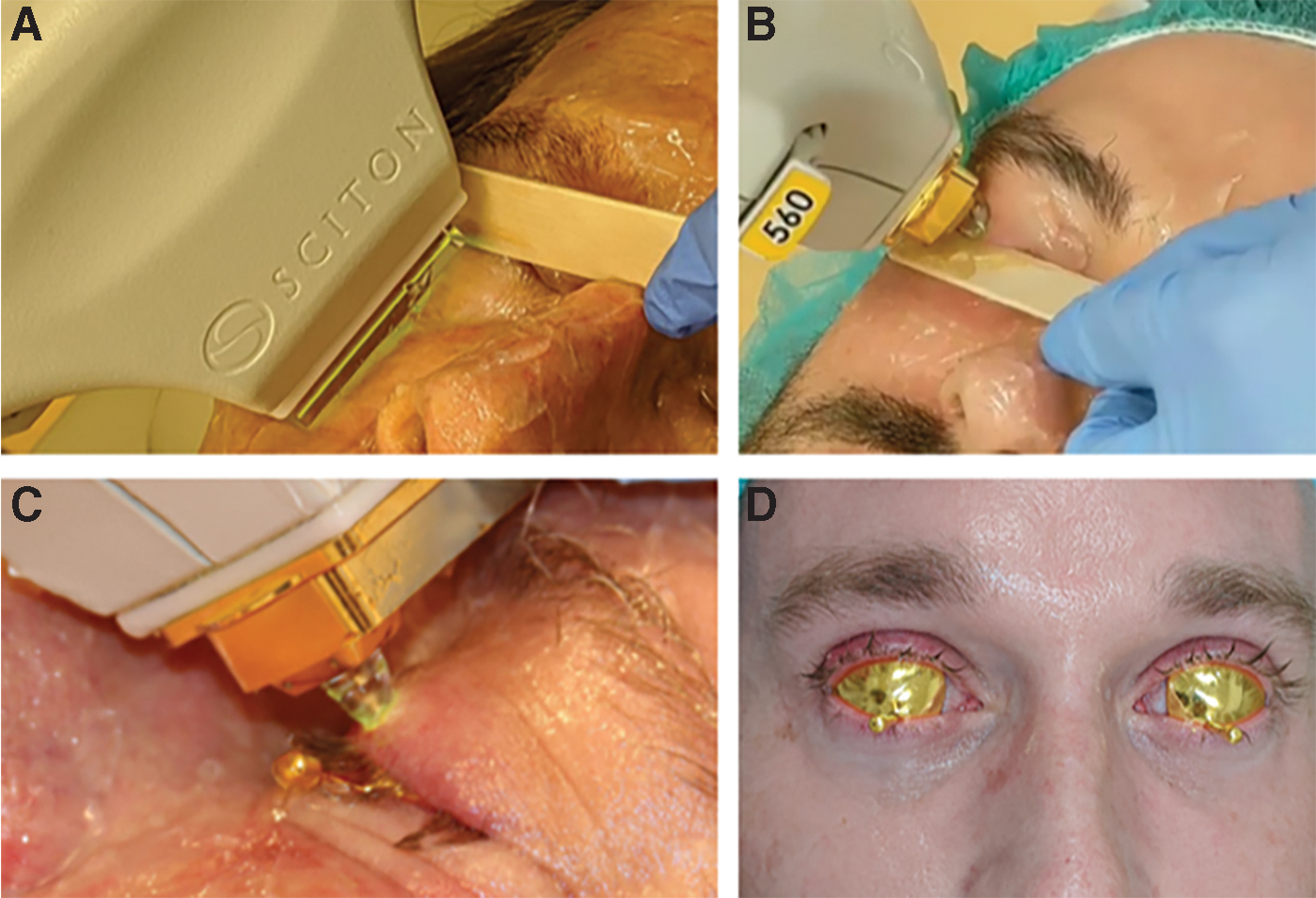
Patients were treated first with two passes on the cheeks and nose from tragus to tragus

Diagram of typical treatment pattern for meibomian gland dysfunction dry eye disease, although many also included the rest of the face to get facial rejuvenation at the same time.
In the MGD DED subset, each subject completed the validated DED questionnaire, Ocular Surface Disease Index (OSDI), and underwent pre- and post-treatment standard DED clinical examinations.
Statistics
Two-tailed t-tests were used to evaluate for statistically significant (p < 0.05) differences in visual acuity and OSDI scores between the patient's pretreatment and post-treatment examinations. Visual acuity was converted to its logMAR equivalent.
Results
Forty-seven patients completed treatment without any significant adverse effects. The mean age was 57 years (range, 28–87) with 63% female. Fifty-nine percent of patients had both MGD and DED (n = 28), 38% of patients had MGD predominant disease (n = 18), and 3% (n = 1) of patients had DED predominant. Patients with MGD and DED predominant were 62.5% female, whereas patients with MGD predominant disease were 75% male. All patients with MGD showed improvement in blepharitis or reduced hordeolum frequency after BBL treatment.
Efficacy of treatment: OSDI and patient satisfaction
Significant differences in OSDI were observed for the MGD DED subset; the average OSDI score before treatment was 37.62 and decreased to an average of 21.27 after treatment (n = 38, p = 0.016, t = 2.70) (Fig. 3). When the MGD predominant patients were excluded (many were treated for hordeola or styes), the mean OSDI improvement was even greater, from 42.14 to 21.41 (p = 0.016, t = 2.78). In those with MGD dominant disease, statistical significance was not achieved because dry eye was not significant at baseline (p = 0.69). The follow-up period for these patients averaged 14 months and ranged from 4 to 44 months. In addition, 100% of patients with MGD DED reported improvement in their dry eye and/or blepharitis symptoms.
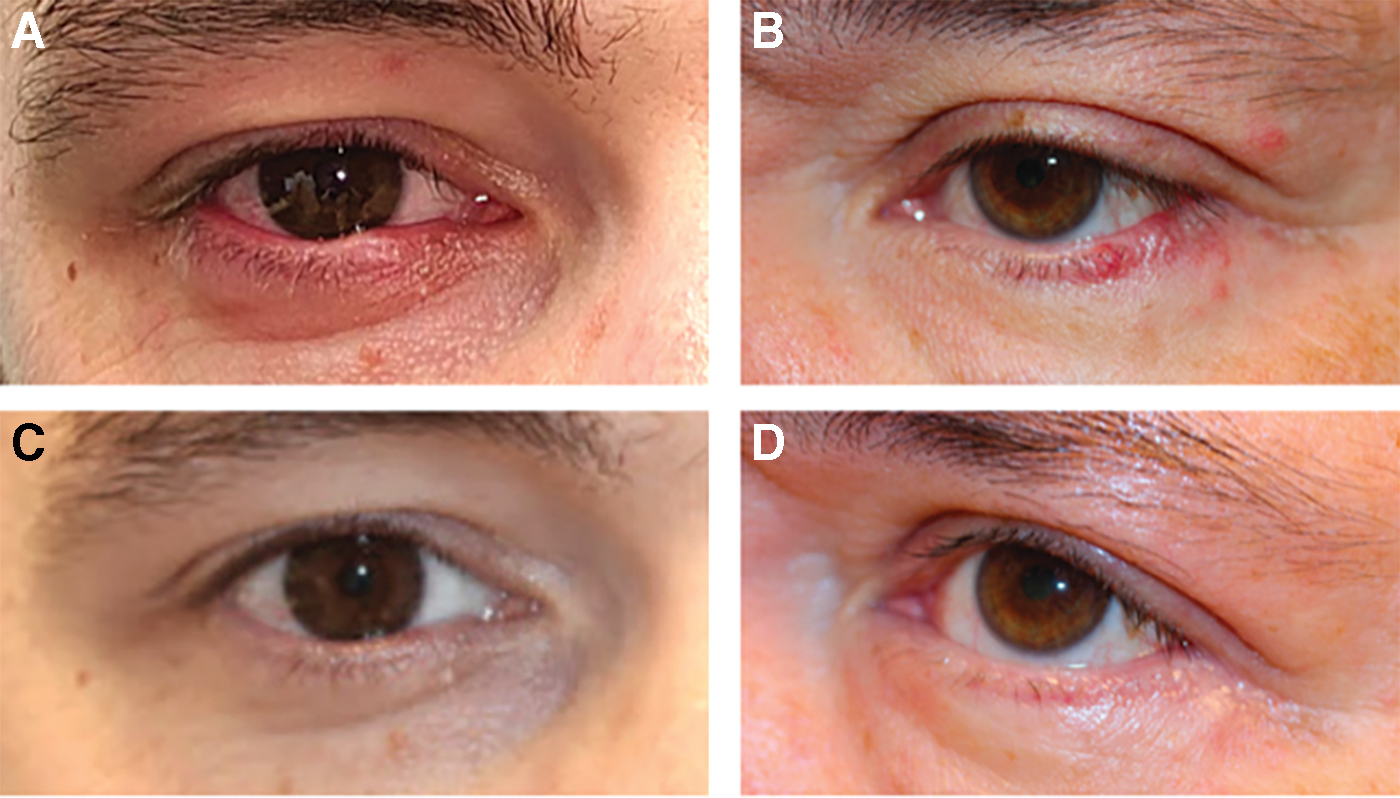
Pretreatment eyelid inflammation
Safety
There was no adverse effect on vision with our augmented protocol; difference in visual acuity was statistically insignificant (p = 0.555). There was one case of presumed mild conjunctival abrasion from tight eyelids and a roughened gold-plated eye shield due to report of irritation the next day that resolved with antibiotic eye ointment within a day. This could also have been an eyelash stuck in the eye as the patient did not feel it was severe enough to come in for an examination.
Two patients with dark lashes had temporary focal eyelash thinning: one from treating an internal hordeolum at a higher fluence than in Tables 1 and 2 with extra pulses 9,17 (Fig. 4). Their lash thinning resolved after ∼4–6 weeks, and the patients continued further treatments staying 4 mm away from the lash line, or with lower fluence. This avoided repeat lash loss while still resulting in MGD DED improvement.
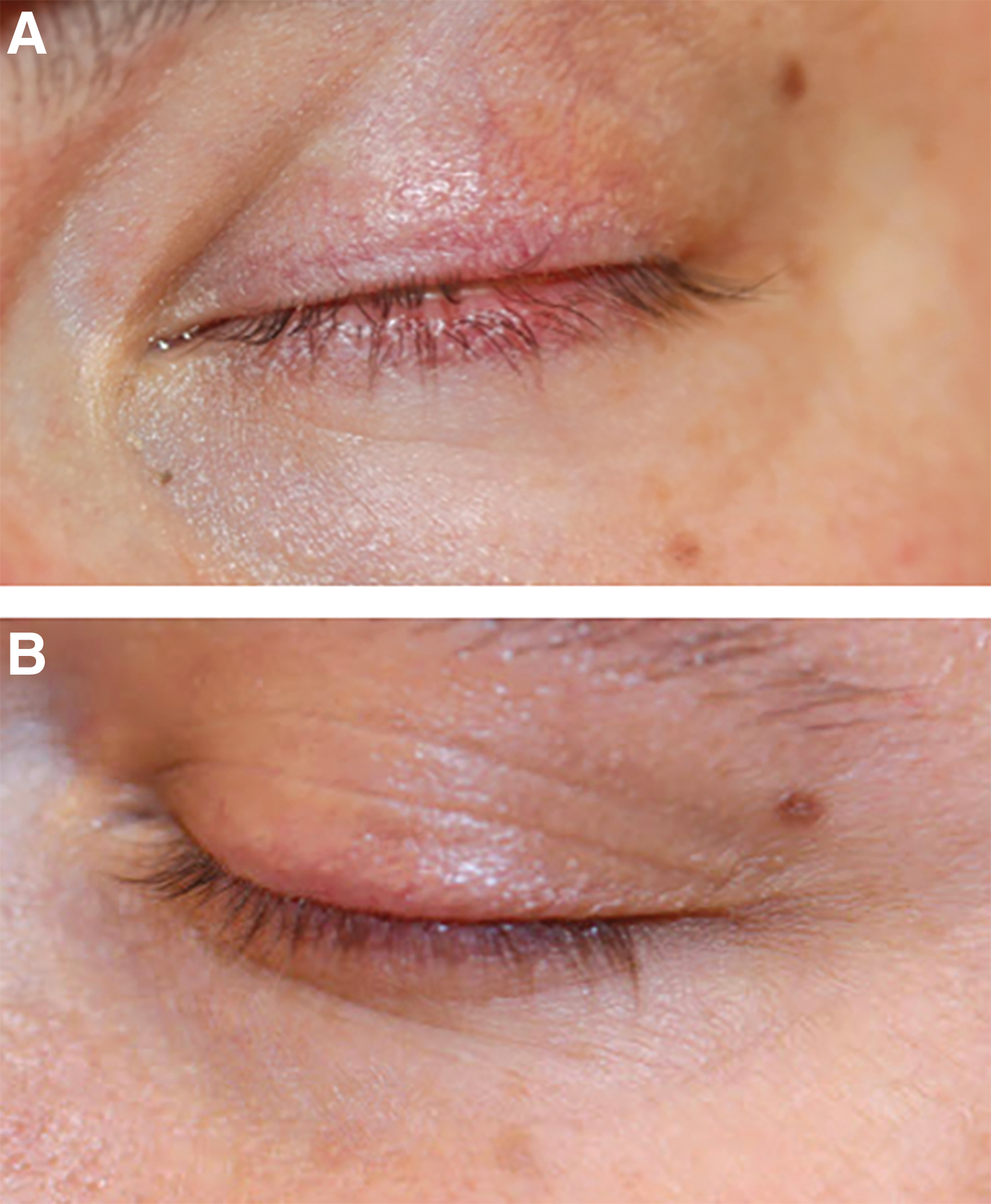
After treating with higher pulses (560 nm filter, fluence at 15 J/cm2, pulse width 20 ms, chill temperature 20°C, 3–6 times in the same spot to treat a chalazion near the dark lashes), this patient had focal lash loss, which was not noticed by the patient
In addition, one more darkly pigmented individual (Fitzpatrick Skin Type 5) developed temporary mild hyperpigmentation that resolved in 1 month (Fig. 5A, B).
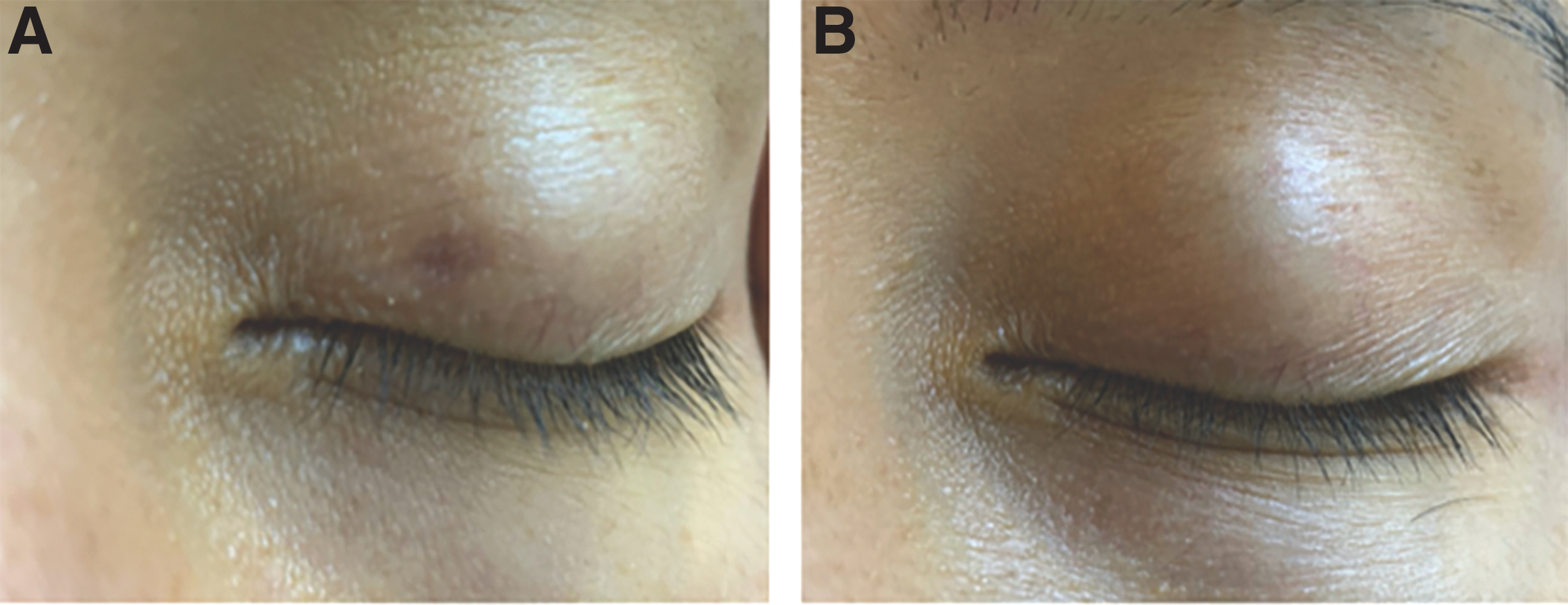
Focal hyperpigmentation 1 day after treatment in a Fitzpatrick type 5 patient using 590 nm filter, fluence 6 J/cm2, pulse width 30 ms, chill temperature 15°C, and 7 mm circle
Discussion
BBL is a safe and effective treatment for DED and MGD with these reported settings. All patients reported significantly improved periocular symptomatology: all OSDI scores were improved from treatment, and all MGD blepharitis symptoms improved. Even the two patients who were noted to have mild temporary lash thinning continued to return for therapy due to their improvement in symptoms. With adjustment of technique and settings as recommended above, no further mild adverse effects have been noted, and patients continue to notice improvement in their DED MGD symptoms and quality of life. The authors now recommend ideally a series of 3 monthly sessions to start, and then twice a year for maintenance. One patient who frequently developed styes and required repeat injections into hordeola or drainage almost on a monthly basis had such improvement after one BBL session to his eyelids, cheek, and nose, that he was symptom-free for a year. Mild recurrence of MGD symptoms 1 year later prompted him to return for his second session, which again gave him lasting improvement in symptoms. BBL treatment as described here provided significant improvement for patients who were symptomatic despite treatment with several other modalities for an extended period of time, highlighting the immense potential for this technology to treat even the most refractory MGD DED.
DED is a multifactorial, destructive, self-sustaining cycle that is characterized by a loss of homeostasis of the tear film, accompanied by ocular symptoms, in which tear film instability and hyperosmolarity, ocular surface inflammation and damage, and neurosensory abnormalities play etiological roles. 16,18 It is hypothesized that IPL might inhibit the inflammatory component of the vicious cycle, which prevents further damage to the ocular surface. 10,19 Several other studies using Toyos' protocol have proven the effectiveness of IPL using the Lumenis M22 technology with a 590 nm filter in reducing dry eye symptoms. 7,10 When other conventional treatments for dry eye have failed, such as topical therapy, warm compresses, or mechanical meibomian expression devices, IPL/BBL can further improve symptoms likely due to its anti-inflammatory properties and possibly by altering the microbiome of the eyelids and ocular surface.
BBL has a reputation of being a high-quality IPL device that is well-established in its treatment of rosacea, hyperpigmentation, and maintenance of the youthfulness of skin. 20 This novel technique of treating both the upper and lower eyelids is now an off-label adaptation for treatment of ocular rosacea (a form of MGD DED). 21 The upper eyelids have more meibomian glands than the lower eyelids, and thus, we examined the safety of adding BBL treatment to the upper eyelids to the more standard cheek- and nose-only treatment. While the MG loss in the upper eyelid and lower eyelid may present differently in MGD, the combination of the two in linear regression analysis contributes to the OSDI score. 22 Our BBL treatments are immediately followed by meibomian gland expression, because IPL when combined with meibomian gland expression (manually or mechanically) may more efficiently and thoroughly express meibomian glands in a way that warm compresses or thermal pulsation technology alone cannot. In a review of eight different treatments for dry eye, meibomian gland expression and thermal pulsation appeared to be effective in treating dry eye symptoms; further, Vegunta et al. showed that patients who failed to improve with thermal pulsation treatment alone improved significantly with IPL treatment. 23,24 The high-intensity red (560–580 nm) to infrared (580–1200 nm) wavelengths of light in our BBL protocol may also be improving the disease by reducing harmful bacteria and Demodex mite infestation. 12,13
This technology, however, has the capacity to harm. 17 Thus, safe settings must be determined, and safety concerns, such as vision, ocular injury, photosensitivity, lash loss, and hyperpigmentation, must be monitored. Out of our group of 47 patients who received treatment, only temporary effects such as eyelash thinning, hyperpigmentation, and presumed mild conjunctival abrasion were observed; the lash thinning was only noted when using higher settings than recommended in this article, and the presumed abrasion was likely from minor trauma related to the corneal shields in the setting of tight eyelids rather than to the BBL treatment itself. Highlighting these minor adverse effects will hopefully help other clinicians avoid such complications; there were no major adverse effects.
Laser parameters were first determined by starting with the safe setting for facial rosacea. To determine if the setting was also safe for the eyelids, an adapter for the eyelids was added without turning up the settings. This effectively turned down the settings by 1–2 J/cm2 fluence. It was determined later that it is better to layer ultrasound jelly between the sapphire crystals to maximize the cooling function. However, this also essentially would keep the fluence at the level indicated on the machine, not lowering it (Tables 1 and 2). To maximize the transmission of light and cooling from the rectangular sapphire crystal to the adaptor sapphire crystal, a layer of ultrasound gel should also be placed on the rectangular sapphire crystal on the base handpiece, beneath the adaptor. Tables 1 and 2 list settings for when gel is in place and when not in place between the crystals to account for whether there is light fully coupling between the crystals. If ultrasound gel is utilized between crystals, then the lower settings from the table with gel coupling may be the safest to use. These setting subtleties may not make a large difference; however, the primary way to avoid temporary lash thinning in a person with dark lashes is to stay 4 mm away from the eyelid margin. Because the meibomian glands extend ∼10 mm up away from the lashes in the upper eyelids, the treatment is still adequate with this safety precaution. Once any temporary lash loss or hyperpigmentation was observed, the settings were adjusted to use the lower end of the fluence range (Tables 1 and 2) for further treatments, and care was taken to remain 4 mm away from the eyelid margin in those particular patients. All patients also have their lashes blocked by a tongue depressor during treatment. With subsequent treatments at the lower fluence and further distance from the eyelashes, no more eyelash thinning was encountered.
Other precautions that may help minimize the low chance of temporary lash thinning include the following: removing gel from the lashes that could couple with the light, and adding a metallic wrap around the sides of the cylinder to maintain treatment solely at the treatment area of the 7 mm round adaptor.
Appropriate technique and use of appropriately placed, well-polished metallic eye shields are extremely important to avoid any long-term complications. Vision changes, corneal damage, and other adverse effects were monitored to determine safety. One has to keep in mind that devices are different and settings should be customized based on the particular technology in use, as well as individual patient characteristics (skin type, sun exposure, light-sensitizing medications, etc.), with full understanding of the tissue effects at various settings/parameters.
Although BBL is effective, the exact mechanisms by which it works remain unknown. Some studies have attempted to elucidate the mechanism by which IPL improves dry eye, including better expressibility of meibum, coagulating abnormal blood vessels, and even corneal nerve tropism. 25 Past studies have shown that chronic anterior blepharitis is associated with heavier colonization of Staphylococcus epidermidis and S. aureus. 26 Further, anti-inflammatory therapies such as corticosteroids and tetracyclines are often used in severe blepharitis and dry eye. Tetracyclines have been shown to decrease IL-1 levels; high IL-1 levels cause higher matrix metalloproteinase (MMP) enzyme production, which is often found in tears of dry eye patients. 27 IPL treatment of DED is associated with significantly decreased levels of IL-4, IL-6, IL-10, IL-17A, and tumor necrosis factor-alpha. 11,28
Limitations to this study include its retrospective nature. We have started adding MMP-9 and lipid layer thickness to our clinical evaluations to further corroborate efficacy, and we look forward to a prospective study where we can compare potentially adding another pass with the 420 nm band pass filter, to see if this additional pass would be helpful. Further studies are needed to reveal more about the mechanism underlying the efficacy of BBL therapy. Future studies are planned prospectively investigating the impact of BBL treatment on multiple different ocular inflammatory markers in the tear film and changes in the lipid layer and ocular surface microbiome.
Conclusions and Summary
IPL has shown much promise in the treatment of refractory MGD DED. Research has shown that with the development of careful protocols for eye and eyelash protection and adaptors for the eyelids, treating both the upper and lower eyelids can be not only safe but also more effective. We investigated the safety and efficacy of a high-quality IPL system, BBL, on the upper and lower eyelids as well as face, and have developed a novel protocol for dry eye and blepharitis treatment that is safe and significantly improves the quality of life for many patients.
Footnotes
Author Disclosure Statement
No competing financial interest exists.
Funding Information
This study was not sponsored by the industry. Unrestricted grant to the Department of Ophthalmology from Research to Prevent Blindness, New York, NY.
